Abstract
Background:
Patients with relapsed/refractory multiple myeloma (RRMM) usually have dismal prognostic outcomes. Venetoclax, a selective inhibitor of antiapoptotic protein B-cell lymphoma-2 (BCL-2), demonstrates antimyeloma activity in plasma cells with t(11;14) or high BCL-2 expression.
Objectives:
This meta-analysis aimed to investigate the efficacy and safety of venetoclax-based therapy in RRMM.
Design:
This is a meta-analysis study.
Data Sources and Methods:
PubMed, Embase, and Cochrane were searched for studies published up to 20 December 2021. Overall response rate (ORR), rate of very good partial response or better (≧VGPR), and complete response (CR) rate were pooled with the random-effects model. Safety was evaluated by the incidences of grade ≧3 adverse events. Subgroup analysis and meta-regression were performed to identify the causes of heterogeneities. All the analyses were conducted by STATA 15.0 software.
Results:
A total of 14 studies with 713 patients were included for analysis. The pooled ORR, rate of ≧VGPR, and CR for all patients were 59% [95% confidence interval (CI) = 45–71%], 38% (95% CI = 26–51%), and 17% (95% CI = 10–26%), respectively. The median progression-free survival (PFS) ranged from 2.0 months to not reached (NR), and the median overall survival (OS) ranged from 12.0 months to NR. Meta-regression showed that patients treated with more drugs combined or less heavily pretreated had higher response rates. Patients with t(11;14) had superior ORR [relative risk (RR) = 1.47, 95% CI = 1.05–2.07], ≧VGPR (RR = 1.71, 95% CI = 1.12–2.60), CR (RR = 1.86, 95% CI = 1.34–2.57), PFS [hazard ratio (HR) = 0.47, 95% CI = 0.30–0.65], and OS (HR = 0.30, 95% CI = 0.08–0.52) compared with patients without t(11;14). Most grade ≧3 adverse events were hematologic, gastrointestinal, and infectious related and were manageable.
Conclusion:
Venetoclax-based therapy is an effective and safe option for RRMM patients, especially those with t(11;14).
Introduction
Multiple myeloma (MM) is a plasma cell disorder characterized by the proliferation of clonal plasma cells and the secretion of monoclonal proteins. In the past decade, the emergence of new antimyeloma agents – such as proteasome inhibitors (PIs), immune-modulatory drugs (IMiDs), and monoclonal antibodies – has substantially improved the clinical outcomes of MM patients. MM remains incurable, however. Most patients will eventually relapse and become refractory to these antimyeloma drugs. 1 This makes novel agents with new mechanisms of action become a promising strategy to treat relapsed/refractory multiple myeloma (RRMM).
Venetoclax is a selective inhibitor of the antiapoptotic protein B-cell lymphoma-2 (BCL-2). It exhibits antitumor activities against a variety of hematologic malignancies, including acute myeloid leukemia, non-Hodgkin lymphoma, and chronic lymphatic leukemia, and has received approval to treat these diseases.2,3 About 20% of MM patients harbor t(11;14) translocation, and they demonstrate overexpression of BCL-2.4,5 Preclinical studies indicate that venetoclax is capable of inducing apoptosis in plasma cell lines dependent on BCL-2 for survival.6,7 For these reasons, although venetoclax has not been approved by the Food and Drug Administration (FDA) for the treatment of MM, a growing number of studies are investigating its efficacy and safety as an alternative antitumor strategy to treat RRMM, especially in patients with t(11;14). Most of these studies, however, are not randomized, and the results are heterogeneous. It is necessary to better understand the efficacy and safety of venetoclax for the treatment of RRMM and to generate reliable benchmarks for comparison. In this study, we performed a systemic review and meta-analysis to objectively assess the response rates, survival outcomes, and safety profiles in RRMM patients treated with venetoclax-based regimens. We will also evaluate whether patients with t(11;14) benefit more from venetoclax-based therapy.
Methods
Search strategy and selection criteria
This study was conducted under the guidance of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guideline and Cochrane handbook (see Supplementary file 1). 8 We performed literature searches on PubMed, Embase, and Cochrane Library for records about RRMM patients treated with venetoclax-based regimens published up to 20 December 2021. The detailed search strategy is available in Supplementary file 2. We also searched for gray literature manually through the reference lists of the relevant articles.
Studies on RRMM patients treated with venetoclax-based regimens were considered for inclusion. For records from the same cohort, those with the most updated and detailed information were included for analysis. Case reports, cohort studies with less than 10 patients, or studies with insufficient patient information were excluded. Two investigators (L.X. and S.L.) independently conducted the literature search and assessed the eligibility of all identified citations. Disagreements were resolved by consensus in consultation with the third investigator (X.X.).
Study outcomes and data extraction
The efficacy outcomes included response rates and survival outcomes. Response rates consisted of complete response (CR) rate, rate of very good partial response or better (≧VGPR), and overall response rate (ORR), defined as CR + VGPR + partial response (PR), according to the International Myeloma Working Group (IMWG) guideline. 9 Progression-free survival (PFS) and overall survival (OS) were survival outcomes of interest. Safety endpoints included treatment-related grade 3 or greater adverse events.
We extracted the following key characteristics from the included trials: the name of the first author, publication year, number of the patients, median age, median prior lines of therapies (PLOTs), type of venetoclax-based regimens, venetoclax daily dose, duration of follow-up, response rates, survival outcomes, and safety endpoints. Two reviewers (L.X. and S.L.) extracted the data independently, and disagreements were resolved by consensus.
Statistical analysis
We assessed the quality of the included studies using the modified Downs and Black checklist, which has a total score of 28. Two reviewers (L.X. and S.L.) assessed the study quality independently, and disagreements were resolved by consensus. Studies with scores of 24–28, 19–23, 14–18, and ≧13 were considered excellent, good, fair, and poor, respectively.10,11
Response rates and 95% confidence intervals (CIs) were pooled using the random-effects models. We also performed subgroup analyses and meta-regression based on drug combinations (monotherapy, doublet, triplet, quadruplet, or mixed), median PLOT (≧3, 4–5, or ≧6), study types (prospective or retrospective), and median venetoclax daily dose (<800 mg or ≧800 mg).
To compare the efficacy of venetoclax-based regimens between patients with and without t(11;14), pooled relative risks (RRs) and hazard ratios (HRs) were used to evaluate response rates and survival outcomes, respectively. As the HRs and the corresponding 95% CIs could not be directly extracted from the included articles, we extracted data from the Kaplan–Meier survival curve in the papers and used the log-rank test to obtain them. 12 The random-effects model was used for the pooled RRs and HRs.
We used the Q test and I2 statistic to evaluate the between-study heterogeneity. I2 values of 0–25%, 25–75%, and >75% were defined as low, moderate, and high degrees of heterogeneity. 13 Publication bias was assessed by the visual inspection of the funnel plot and Egger’s test. To evaluate the stability of the results, sensitivity analysis was performed by removing one study at a time and repeating the meta-analysis. All the analyses were conducted using STATA 15.0 software with packages metan, metaprop, metareg, metafunnel, metainf, and metabias. p value < 0.05 was considered to have statistical significance.
Results
Study selection and characteristics
We identified 744 records from the initial search and kept 14 studies with a total of 16 records for final analysis.14–29 Figure 1 demonstrates the detailed study selection processes. A study from Bahils et al. 22 contained two patient groups that received different treatment regimens, and another survey from Kaufman et al. 24 included two groups of patients with different basic characteristics. Therefore, we analyzed them separately. A controlled arm of a randomized controlled trial from Kumar et al. 21 did not receive venetoclax-based regimens and was not included for analysis in this study.

Flow diagram of the search results.
The detailed information about the included studies is listed in Table 1. There were six prospective studies and eight retrospective studies. The median duration of follow-up of the included studies ranged from 3.3 to 45.6 months. A total of 713 RRMM patients were included, 308 of whom were t(11;14) positive, and 377 were t(11;14) negative. A total of 64% of the patients underwent autologous stem cell transplantation (ASCT) prior to venetoclax therapy. A total of 37% and 49% of the patients were refractory to bortezomib and lenalidomide, respectively. The age of the patients ranged from 31- to 92-year-old, and the number of PLOT varied from 1 to 19.
Baseline characteristics of the included studies.
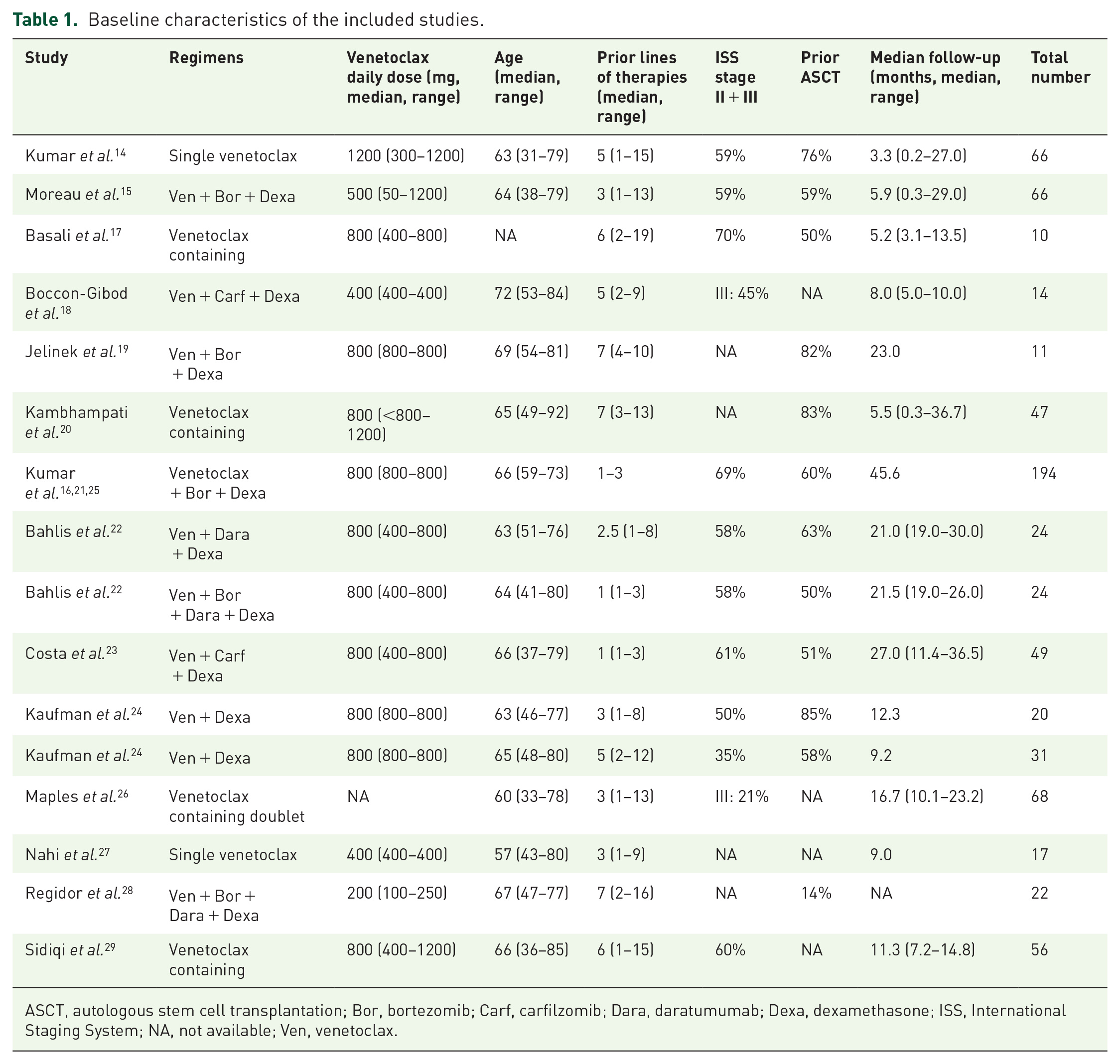
ASCT, autologous stem cell transplantation; Bor, bortezomib; Carf, carfilzomib; Dara, daratumumab; Dexa, dexamethasone; ISS, International Staging System; NA, not available; Ven, venetoclax.
We used the modified Downs and Black checklist to assess the quality of the included studies. The median total score was 20 (range = 15–26). Most studies had good to excellent quality, but four retrospective studies with a small sample size had fair quality. Detailed information about quality assessment is provided in Supplementary file 3.
Efficacy
Response rates
ORR. All 14 studies reported the ORR, rate of ≧VGPR, and CR, except one study that did not report the CR rate. The pooled ORR for all studies combined was 59% (95% CI = 45–71%, I2 = 91.18%, p < 0.001) (see Figure 2(a)). There was considerable heterogeneity among studies, so we performed subgroup analysis and meta-regression. The pooled ORR for patients with venetoclax monotherapy, doublet, triplet, and quadruplet therapies was 24% (95% CI = 15–34%), 61% (95% CI = 46–75%), 71% (95% CI = 54–85%), and 70% (95% CI = 56–83%), respectively. Meta-regression showed that regimens with more drugs combined were associated with a higher ORR (p = 0.034). For studies that included patients with a median PLOT ≧3, 4–5, and ≧6, the pooled ORR was 76% (95% CI = 65–85%), 34% (95% CI = 16–54%), and 42% (95% CI = 33–51%), respectively. Meta-regression indicated that patients with less PLOT correlated with a higher possibility of achieving ORR (p = 0.004). No significant difference, however, was identified regarding study types and median venetoclax daily dose (see Figure 3).

Forest plots for pooled response rates of venetoclax-based therapies in relapsed/refractory multiple myeloma. The pooled response rates for all patients, patients with t(11;14), and non-t(11;14) are shown in panel (a), (b) and (c), respectively.

Pooled ORR in subgroups defined by treatment, PLOT, study types, and venetoclax daily dose.
The pooled ORR for patients with t(11;14) was 71% (95% CI = 59–82%, I2 = 76.03%, p < 0.001) (see Figure 2(b)). Meta-regression showed that there was statistical significant difference in drug combinations (p = 0.017), but not in PLOTs, study types, and median venetoclax daily dose (see Figure 3). For patients without t(11;14), the pooled ORR was 45% (95% CI = 23–69%, I2 = 94.02%, p < 0.001) (see Figure 2(c)). Non-t(11;14) patients receiving less PLOTs were associated with a higher ORR (p = 0.004) (see Figure 3). Patients with t(11;14) harbored higher ORR (RR = 1.47, 95% CI = 1.05–2.07, I2 = 82.50%, p < 0.0001) compared with those without t(11;14) (see Figure 4(a)).

Forest plots for RR (a) of ORR, ≧VGPR, and CR, as well as for HR (b) of PFS and OS in patients with t(11;14) versus non-t(11;14).
Rate of ≧VGPR. The pooled rate of ≧VGPR for all patients was 38% (95% CI = 26–51%, I2 = 90.59%, p < 0.001) (see Figure 2(a)). The earlier initiation of venetoclax-based therapy was associated with a higher rate of ≧VGPR [median PLOT ≧3 versus 4–5 versus ≧6: 56% (95% CI = 41–70%) versus 26% (95% CI = 12–44%) versus 18% (95% CI = 11–27%), p = 0.002]. The pooled rate of ≧VGPR for patients with venetoclax monotherapy, doublet, triplet, and quadruplet therapies was 14% (95% CI = 7–23%), 41% (95% CI = 30–52%), 51% (95% CI = 34–69%), and 44% (95% CI = 29–59%), respectively. There was a tendency of increased rate of ≧VGPR among patients with more drugs combined, but this did not reach statistical significance (p = 0.089). Meta-regression also demonstrated a significant difference regarding study types [prospective versus retrospective studies: 54% (95% CI = 35–71%) versus 23% (95% CI = 13–35%), p = 0.043], but not median venetoclax daily dose (see Supplementary file 4).
The rate of ≧VGPR in patients with and without t(11;14) was 51% (95% CI = 35–67%, I2 = 84.75%, p < 0.001) and 22% (95% CI = 5–44%, I2 = 92.44%, p < 0.001), respectively (see Figure 2(b) and (c)). Patients with t(11;14) were associated with a higher rate of ≧VGPR (RR = 1.71, 95% CI = 1.12–2.60, I2 = 58.10%, p = 0.014) (see Figure 4(a)).
Rate of CR. The pooled rate of CR for all patients was 17% (95% CI = 10–25%, I2 = 82.19%, p < 0.001) (see Figure 2(a)). The less number of prior therapies was associated with a higher rate of CR [median PLOT ≧3 versus 4–5 versus ≧6: 30% (95% CI = 21–41%) versus 7% (95% CI = 2–13%) versus 7% (95% CI = 1–17%), p = 0.037]. Meta-regression showed no significant differences regarding drug combinations, study types, and median venetoclax daily dose (see Supplementary file 4).
The rate of CR in patients with and without t(11;14) was 23% (95% CI = 12–36%, I2 = 78.31%, p < 0.001) and 11% (95% CI = 3–23%, I2 = 78.84%, p < 0.001), respectively (see Figure 2(b) and (c)). t(11;14)-positive patients were associated with a higher rate of CR (RR = 1.86, 95% CI = 1.34–2.57, I2 = 0.00%, p = 0.864) (see Figure 4(a)).
Survival
PFS. Eight studies with 495 patients reported PFS (see Table 2). The median PFS in individual studies of all patients ranged from 2.0 months to not reached (NR). Studies with less median PLOTs seemed to have longer PFS. The median PFS ranged from 2.0 to 5.8 months in studies with a median PLOT of ≧6, while for studies with a median PLOT ≧3, the median PFS reached more than 14.1 months.
Survival outcomes of RRMM patients treated with venetoclax-based therapies.

CI, confidence interval; NA, not available; NE, not estimable; NR, not reached; OS, overall survival; PFS, progression-free survival; RRMM, relapsed/refractory multiple myeloma.
Not reach for patients with <6 prior lines of therapies and 16.2 months for ≧6 prior lines of therapies.
The median PFS in individual studies of patients with t(11;14) and without t(11;14) ranged from 5.8 months to NR and 1.0 to 22.8 months, respectively. Patients with t(11;14) were associated with a longer PFS (HR = 0.47, 95% CI = 0.30–0.65, I2 = 0.00%, p = 0.650) (see Figure 4(b)).
OS. Seven studies with a total of 446 patients reported OS (see Table 2). The median OS in individual studies of all patients ranged from 12.0 months to NR. Studies with less median PLOTs seemed to have longer OS. The median OS ranged from 12.0 to 28.4 months in studies with a median PLOT ≧6. In studies with a median PLOT ≧3, the median OS was NR.
The median OS in studies of patients with t(11;14) was NR. For patients without t(11;14), however, the median OS ranged from 9.3 to 46.4 months. Patients with t(11;14) were associated with a longer OS (HR = 0.30, 95% CI = 0.08–0.52, I2 = 0.00%, p = 0.764) (see Figure 4(b)).
Safety
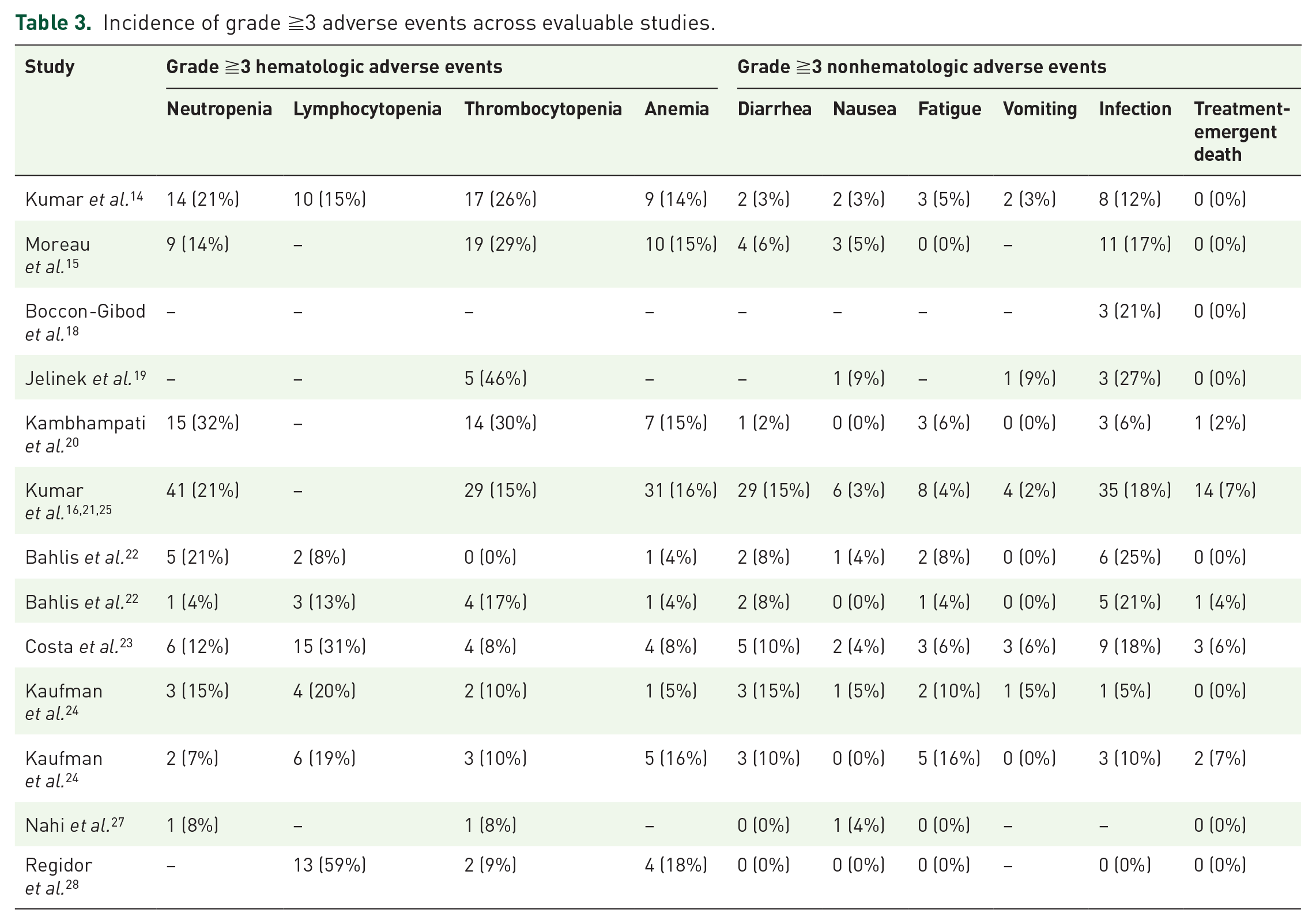
A total of 11 studies with a total of 585 patients reported adverse events related to venetoclax-based treatments (see Table 3). The most common grade 3 or greater hematological adverse events were neutropenia (4–32%), lymphocytopenia (8–59%), thrombocytopenia (0–46%), and anemia (4–18%). The common grade ≧3 nonhematological adverse events included diarrhea (0–15%), nausea (0–5%), fatigue (0–16%), and vomiting (0–9%). The average rate of grade ≧3 infection was 18% (range = 0–27%). There were 21 treatment-emergent deaths, about two-thirds of which were infection-related.
Incidence of grade ≧3 adverse events across evaluable studies.
Publication bias and sensitivity analysis
No publication bias was identified in accordance with the visual inspection of the funnel plots and results of Egger’s tests (see Supplementary file 5). Sensitivity analysis showed that the overall results were stable.
Discussion
At present, MM remains an incurable disease, and most patients will inevitably relapse eventually. A recent real-world study reported that for double-class and triple-class refractory patients, the median OS is only 22.2 and 11.6 months, respectively. 30 The heterogeneity of the disease makes the treatment of MM challenging. With multiple drugs and regimens available, there are still no biomarkers, such as genetic abnormalities and specific proteins, to guide the selection of therapies in the management of MM. To our knowledge, this study is the first systemic review and meta-analysis that evaluates the safety and efficacy of venetoclax-based therapy in RRMM patients. We demonstrated that venetoclax-based therapy could provide promising efficacy with a tolerable safety profile in RRMM patients, especially those with t(11;14). Therefore, venetoclax would probably become the first targeted therapy in MM.
When used as a single agent in heavily pretreated patients, venetoclax generated a pooled ORR of 24% for all patients and 38% for patients with t(11;14). The response rate is comparable with the other novel agents, such as daratumumab monotherapy (29–36%), carfilzomib monotherapy (24%), pomalidomide + dexamethasone (35%), and selinexor + dexamethasone (21–26%).31–36
When venetoclax was combined with other drugs, such as dexamethasone, PI, IMiD, and daratumumab, the response rates could significantly improve. The pooled ORR for doublet and triplet therapies was 61% and 70%, respectively. This possibly resulted from the synergistic activities existing in some combinations. For example, dexamethasone can increase venetoclax sensitivity by shifting Bim, a pro-apoptotic activator, binding toward BCL-2. 37 Proteasome inhibitors, such as bortezomib, can downregulate myeloid cell leukemia-1 (MCL-1) activity through the upregulation of NOXA, turning the survival of MM cells from MCL-1 dependent into BCL-2 dependent. 38 Other antimyeloma drugs may also act by other mechanisms to increase the activity of venetoclax. It remains to be determined which combination harbors the greatest efficacy.
This study found statistically significant differences regarding PLOTs when taking all the patients together. Patients with less PLOTs were associated with higher response rates. Although there seemed to be a lower ORR in t(11;14) patients with more PLOTs, however, it did not reach statistical significance. It may partially result from the limited number of included studies. On the other hand, it may indicate that heavily pretreated patients with t(11;14) can still benefit from venetoclax-based therapy. This study did not identify statistical differences in groups with different median venetoclax daily doses. The possible causes included the limited number of the studies and the fact that there were variations in the doses in individual studies, as a proportion of the studies contained dose-expansion cohorts.
t(11;14) is considered a standard-risk factor in MM based on the evidence before the emergence of current therapies, but recent studies indicate that the survival outcomes in patients with t(11;14) are inferior to other standard-risk patients. It is particularly true in t(11;14) MM with high-risk cytogenetic abnormalities coexisted, comprising about one-third of t(11;14) patients.39–41 Patients with t(11;14) are associated with an elevation of BCL-2 and a biological pattern similar to B cell. 7 For this reason, venetoclax, a BCL-2 inhibitor, becomes a possible option for patients with t(11;14). Our meta-analysis shows that patients with t(11;14) had better response rates and survival outcomes than patients without this cytogenetic lesion. This phenomenon can also be observed in t(11;14) patients with coexisted high-risk cytogenetic abnormalities.2,22,24,26,42,43 Given the promising results of venetoclax, early detection of t(11;14) is necessary to guide the selection of treatments in this subgroup of patients. In addition, other biomarkers, such as BCL-2 expression level and BCL-2/MCL-1 ratio, also harbor significant predictive value for the response to venetoclax. 4 Combining these biomarker approaches may further promote the identification of patients who are more likely to benefit from venetoclax.
This study shows that venetoclax had a tolerable safety profile. Most of the adverse events were manageable. There were 21 treatment-emergent deaths, most of which were infection-related. The exact mechanism underlying life-threatening infections is not well understood, but there are some possible explanations. Treatment-induced immunosuppression, lack of response, and disease progression may make some patients more susceptible to infections. Therefore, it is necessary to take precautions during treatment and identify the patient group that could benefit from venetoclax.
This study had several limitations. First, this study was based on a relatively small number of patients. Second, most of the included studies were single-armed, which limited the direct comparison between regimens with and without venetoclax. In addition, there were considerable heterogeneities among studies, the main possible causes of which included patient population, study design, venetoclax doses, drug combinations, and PLOTs. Although we have performed subgroup analysis, results should be interpreted cautiously in this meta-analysis. Furthermore, due to the limited number of the included studies, we did not evaluate the impact of other markers, such as BCL-2 expression level and BCL-2/MCL-1 ratio, on patients treated with venetoclax-based regimens. Further studies are required to identify markers that can select the most suitable patients.
Despite these limitations, this study demonstrated that the venetoclax-based treatment was a safe and potent option for RRMM patients, especially those with t(11;14). Efforts should be made for the early identification of this cytogenetic abnormality to guide treatment selections. More studies are needed to compare the efficacy between regimens with and without venetoclax directly, and to explore other drug combinations and the use of venetoclax in the earlier course of the disease.
Supplemental Material
sj-docx-1-tah-10.1177_20406207231155028 – Supplemental material for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207231155028 for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis by Linchun Xu, Shaoze Lin, Xueyang Xing and Yongzhong Su in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207231155028 – Supplemental material for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis
Supplemental material, sj-docx-2-tah-10.1177_20406207231155028 for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis by Linchun Xu, Shaoze Lin, Xueyang Xing and Yongzhong Su in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-3-tah-10.1177_20406207231155028 – Supplemental material for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis
Supplemental material, sj-docx-3-tah-10.1177_20406207231155028 for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis by Linchun Xu, Shaoze Lin, Xueyang Xing and Yongzhong Su in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-6-tah-10.1177_20406207231155028 – Supplemental material for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis
Supplemental material, sj-docx-6-tah-10.1177_20406207231155028 for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis by Linchun Xu, Shaoze Lin, Xueyang Xing and Yongzhong Su in Therapeutic Advances in Hematology
Supplemental Material
sj-jpg-4-tah-10.1177_20406207231155028 – Supplemental material for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis
Supplemental material, sj-jpg-4-tah-10.1177_20406207231155028 for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis by Linchun Xu, Shaoze Lin, Xueyang Xing and Yongzhong Su in Therapeutic Advances in Hematology
Supplemental Material
sj-jpg-5-tah-10.1177_20406207231155028 – Supplemental material for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis
Supplemental material, sj-jpg-5-tah-10.1177_20406207231155028 for Efficacy and safety of venetoclax-based regimens for the treatment of relapsed/refractory multiple myeloma: a systemic review and meta-analysis by Linchun Xu, Shaoze Lin, Xueyang Xing and Yongzhong Su in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
