Abstract
Background:
Haploidentical hematopoietic stem cell transplantation (haplo-HSCT) based on granulocyte colony-stimulating factor plus anti-thymocyte regimens (‘Beijing Protocol’) provides a salvage treatment for patients of acquired severe aplastic anemia (SAA) in China. However, graft-versus-host disease (GVHD) is a major impediment of haplo-HSCT due to human leukocyte antigen disparity. Recently, haplo-HSCT combined with umbilical cord blood (UCB) (haplo-cord HSCT) is performed in clinical trials to potentially reduce the risk of severe GVHD. Nevertheless, studies comparing GVHD in pediatric patients receiving haplo and haplo-cord HSCT for SAA are limited.
Objective:
The objective of this study was to investigate the impact of UCB co-infusion on GVHD in pediatric patients receiving haplo-HSCT for SAA.
Design:
We conducted a retrospective study of 91 consecutive SAA children undergoing haploidentical transplantation based on the ‘Beijing Protocol’ with or without co-infusion of UCB in our center.
Methods:
All patients received uniform non-myeloablative conditioning and GVHD prophylaxis. We compared baseline characteristics and transplant outcomes between the haplo (n = 35) and haplo-cord (n = 56) recipients.
Results:
All 91 patients achieved hematopoietic recovery from haploidentical donors, with a higher incidence of peri-engraftment syndrome observed with the haplo-cord group as compared with the haplo group (75.0% versus 48.6%, p = 0.029). Notably, the haplo-cord group showed a lower incidence of II–IV acute GVHD (aGVHD) than the haplo group (16.1% versus 42.9%, p = 0.002). Observed incidences of chronic GVHD (cGVHD) and moderate to severe cGVHD in the haplo-cord group were also lower than that in the haplo group (25.6% versus 51.3%, p = 0.019; 16.2% versus 41.3%, p = 0.016, respectively). Haplo-cord HSCT was identified as the only factor associated with a lower incidence of II–IV aGVHD and cGVHD in multivariate analysis. However, no differences were observed between the two groups for infections and survival outcomes.
Conclusion:
Our data indicated that co-infusion of UCB in ‘Beijing Protocol’-based haplo-HSCT may be effective for reducing the risk of severe GVHD in SAA children.
Keywords
Introduction
Acquired severe aplastic anemia (SAA) is a life-threatening hematological disorder characterized by bone marrow failure that results in pancytopenia. 1 Allogeneic hematopoietic stem cell transplantation (allo-HSCT) from a human leukocyte antigen (HLA)-matched related donor (MRD) is a recommended treatment for patients with SAA, 2 with long-term survival rates over 80%.3,4 However, only about one third of patients have access to an MRD. Over the past two decades, the development of haploidentical HSCT (haplo-HSCT) has provided a feasible salvage treatment for SAA patients in urgent need of allo-HSCT. 5 In China, the ‘Beijing Protocol’ of haplo-HSCT, which is based on the granulocyte colony-stimulating factor (G-CSF) plus anti-thymocyte (ATG) regimens, has been performed in nearly 98% of transplant centers with encouraging results. 6 For SAA recipients receiving this treatment modality, hematopoietic recovery and long-term survival have been comparable to those undergoing HSCT from MRD. 7 Nevertheless, relatively high risk of graft-versus-host disease (GVHD) is an obstacle preventing further applications of this haplo-HSCT protocol due to HLA disparity.8,9
Several strategies to prevent GVHD of haplo-HSCT, such as optimizing ATG dose in conditioning 10 and clinical investigations of post-transplant cyclophosphamide (PT-Cy), have been developed in recent years. 11 However, ATG is associated with delayed immune recovery and increased cytomegalovirus (CMV) and Epstein–Barr virus (EBV) infections. 12 Although the use of PT-Cy is highly effective in preventing GVHD, the incidence of acute GVHD (aGVHD) in haplo recipients was still inferior to MRD recipients using homogeneous prophylaxis with PT-Cy. 13 Moreover, the toxic effects caused by cyclophosphamide increase the risk of hemorrhagic cystitis and myocardial damage.14,15 Thus, a novel strategy to reduce the GVHD risk is warranted as a supplement to current GVHD prophylaxis for haplo-HSCT.
Umbilical cord blood transplantation (UCBT) is increasingly being used for patients of hematologic diseases 16 and reportedly has lower risks of aGVHD and chronic GVHD (cGVHD) in children and adults.17–19 Particularly, human umbilical cord blood (UCB) is characterized by lower numbers of mature or primed T cells that play a predominant role in inducing GVHD, compared with peripheral blood grafts.20,21 In addition, UCB has been reported to be a rich and rapidly accessible source of several immunomodulatory cells, such as regulatory T cells (Tregs) and mesenchymal stromal cells (MSCs), both of which have been shown to control severe GVHD.22–24 In recent years, haplo-HSCT combined with a third-party UCB unit (haplo-cord HSCT) has been used in clinical trials.25,26 Previous reports of this haplo-cord HSCT were mainly restricted to adult patients with hematological malignancies; the impact of this transplant protocol on GVHD was controversial.26–28 However, whether this haplo-cord HSCT protocol is associated with GVHD in pediatric patients with SAA remains unknown.
Hence, based on the UCB characteristics, we hypothesized that co-infusion of a third-party UCB unit with the haploidentical grafts might reduce the risk of GVHD for pediatric patients with SAA. To evaluate the outcomes of haplo-HSCT with or without co-infusion of UCB in SAA children, we retrospectively analyzed 91 consecutive SAA children undergoing haplo-HSCT based on the ‘Beijing Protocol’ in our center.
Methods
Patients
Between 15 April 2015 and 15 July 2021, consecutive patients with SAA who underwent haplo-HSCT based on the ‘Beijing Protocol’ 6 at the Children’s Hospital of Soochow University were retrospectively analyzed. Patients with congenital bone marrow failure (BMF) syndromes (Fanconi anemia, Diamond-Blackfan anemia, Shwachman-Diamond syndrome, and dyskeratosis congenita) were excluded clinically and by laboratory assays, including DNA analysis by targeted next-generation sequencing. 29 The detected genes are listed in Supplementary Table 1. All patients enrolled in this study were diagnosed with acquired SAA or acquired very severe aplastic anemia (VSAA) based on the guidelines. 30 Patients receiving haplo-HSCT combined with a UCB unit were selected for the haplo-cord group. As a comparison, patients receiving haplo-HSCT without UCB co-infusion were selected for the haplo group. The decision on whether to use the UCB co-infusion or no co-infusion protocol was made based on physician’s discretion. The last follow-up date was 30 October 2021. Clinical data and laboratory assay results were collected from outpatient and inpatient medical history, which were further confirmed by two attending doctors responsible for managing these patients. This study was approved by the Institutional Review Board at the Children’ Hospital of Soochow University (No. 2022CS025). The Institutional Review Board agreed to waive the requirement for informed consent for patients enrolled in this retrospective study, and we have de-identified all patient details. This study was conducted in accordance with the Declaration of Helsinki. No commercial sponsor was involved in the study. The reporting of this study conforms to the STROBE statement. 31
HLA typing and donor selection
We routinely detected the HLA-A, HLA-B, HLA-C, HLA-DRB1, and HLA-DQB1 typing of the recipients and related donors at high resolution; the HLA-A, HLA-B, and HLA-DRB1 typing of the UCB units were detected at low resolution. Haploidentical donors were chosen based on gender, age, HLA typing, health conditions, and willingness to donate, while the UCB units were selected on the basis of HLA typing and cell doses determined before freezing. For UCB collection, one unit with HLA typing of at least 4/6 was selected from cord blood banks for each recipient, and HLA matching took precedence over cell doses. When the level of HLA matching was the same, the richest UCB units were chosen. Donor-specific antibodies (DSA) were tested using the Luminex platform for all patients before conditioning regimens. For patients with DSA ⩾ 2000 mean fluorescence intensity, solutions included donor alteration, plasma change, rituximab, and immunoglobulin (Supplementary Figure 1).
Conditioning regimen and GVHD prophylaxis
All patients were treated with the uniform non-myeloablative (NMA) conditioning regimen based on busulfan (Bu)/cyclophosphamide (Cy). As shown in Figure 1, the components included fludarabine: 45 mg/m2/day intravenously (i.v.) on days −7 to −4; Bu: 3.2 mg/kg/day i.v. on days −7 to −6; Cy: 40 mg/kg/day i.v. on days −5 to −2; rabbit anti-human thymocyte immunoglobulin (thymoglobulin, Genzyme, Polyclonals S.A.S, Lyon, France): 2.5 mg/kg/day i.v. on days −5 to −2. GVHD prophylaxis consisted of cyclosporin A (CSA) or tacrolimus (FK-506) and a short course of methotrexate (MTX) plus mycophenolate mofetil (MMF). CSA or FK-506 was administered orally at a dose of 6–10 mg/kg/day or 0.1–0.3 mg/kg/day on day −1. Dosages were adjusted to recipient blood levels and ranged from 200 to 250 ng/mL CSA and 10 to 15 ng/mL FK-506. Upon maintaining the target concentration for 6–9 months after HSCT, the calcineurin inhibitors were gradually tapered and withdrawn completely over the following 3–6 months in the absence of GVHD. When GVHD occurred, CSA or FK-506 was maintained, and its dose was adjusted to therapeutic concentration. Oral MTX were given at 15 mg/m2 on day +1 and 10 mg/m2 on days +3, +6, and +11. MMF was administered orally (15–20 mg/kg/day) from days −1 to + 30 and a half dose was subsequently administered for 15 days. The first-line treatment for aGVHD was intravenously administration of 1–2 mg/kg/day methylprednisolone, while the second-line treatment for steroid-refractory aGVHD consisted of CD25 monoclonal antibody and ruxolitinib. Treatment of cGVHD included corticosteroids, ruxolitinib, and mesenchymal stromal cells.
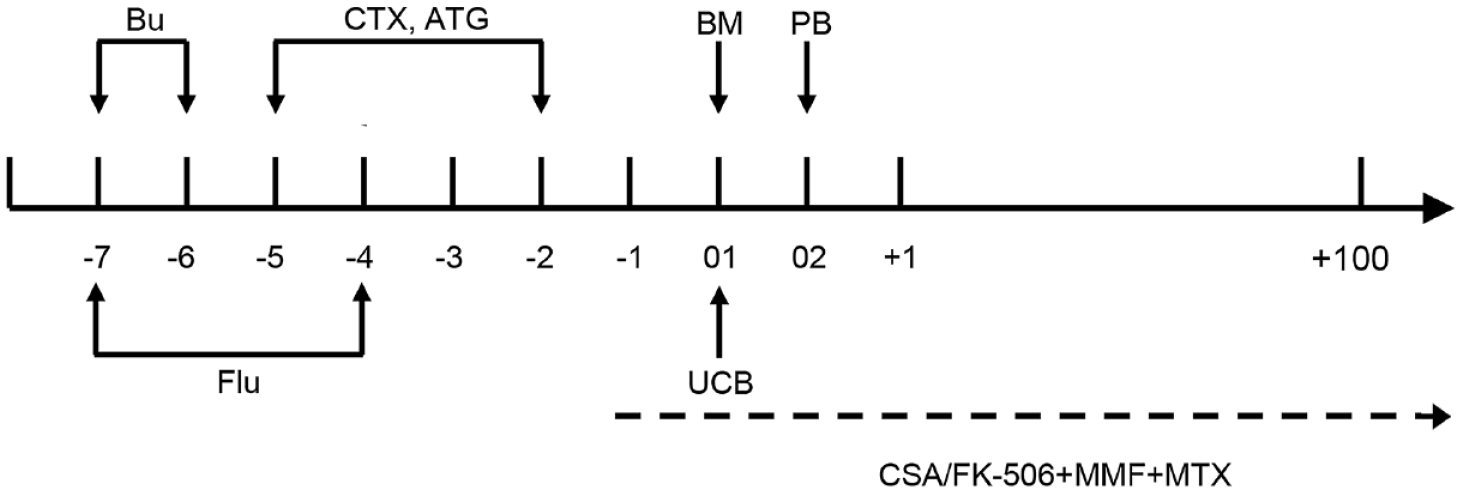
Schematic diagram of transplantation procedures. Non-myeloablative conditioning regimens were as follows: fludarabine (Flu) (45 mg/m2/day on days −7 to −4), busulfan (Bu) (3.2 mg/kg/day on days −7 to −6), cyclophosphamide (Cy) (40 mg/kg/day on days −5 to −2); rabbit anti-human thymocyte immunoglobulin (ATG) (2.5 mg/kg/day on days −5 to −2).
Stem cell collection and infusion
To mobilize stem cells, recombinant human G-CSF (rhG-CSF) were subcutaneously injected into donors on days −4 to 0 (5 μg/kg/day). Recipients were infused with haploidentical grafts on 2 consecutive days (designated as ‘Day 01’ and ‘Day 02’). On Day 01, the BM of haploidentical donors (HID) were infused into patients, while PB of HID were infused into recipients on Day 02 (Figure 1). The UCB unit for individual patients in the haplo-cord group came from the cord blood banks of Shanghai, Shandong, or Guangdong Province in China, and was infused into each recipient 8 hours before the BM infusion (Figure 1).
Supportive care and post-transplantation surveillance
Patients were hospitalized in their respective laminar airflow room until hematopoietic recovery. During the conditioning and immunosuppressive period, prophylactic antibiotics (meropenem or vancomycin), antifungals (posaconazole, voriconazole or caspofungin), and antivirals (ganciclovir and acyclovir) were administered. rhG-CSF was subcutaneously administered at a dose of 5 μg/kg/day from day +6 until the neutrophil count was >1 × 106/L. Patients with hemoglobin < 60 g/L or platelet count < 20 × 109/L were transfused with irradiated and leuko-depleted blood products. Copies of viral genomes in peripheral blood were detected weekly after HSCT using real-time quantitative polymerase chain reaction. The chimerism of donor cells was evaluated by the multiplex fluorescent analysis of short tandem repeat in peripheral whole blood of recipients from the onset of neutrophil engraftment. Patients who tested positive for CMV viremia were given ganciclovir as preemptive therapy, while patients with EBV viremia were treated with upfront rituximab. For patients developing hemorrhagic cystitis (HC), BK and adeno virus DNA in plasma and urine were tested weekly from the onset of HC.
Definitions
Neutrophil engraftment was defined as a neutrophil count of >0.5 × 109/L for 3 consecutive days. Platelet engraftment was defined as a platelet count exceeding 20 × 109/L for 7 consecutive days without transfusion. Primary graft failure (GF) was defined as the failure of neutrophil engraftment at day +28 after HSCT, while secondary GF was defined as the development of neutrophil count < 0.5 × 109/L occurring after the initial engraftment. Full donor chimerism (FDC) was defined as the presence of >95% haploidentical donor cells post-transplantation. Peri-engraftment syndrome (peri-ES) was defined according to the previously described criteria. 32 Moreover, diagnosis and grading of aGVHD and cGVHD were performed based on the standard criteria.33,34 Steroid-refractory aGVHD was defined as progression of aGVHD severity after 3 days of steroid treatment or no improvement after 5 days of steroid treatment. 35 Patients who survived >30 days post-HSCT with successful engraftment were evaluated for aGVHD analysis, whereas patients who survived >100 days from transplant without GF were selected for cGVHD analysis. Viral DNA ⩾ 500 copies/mL in plasma was defined as viremia for CMV and EBV. Overall survival (OS) was defined as the time from transplant to death from any cause. Failure-free survival (FFS) was defined as survival with response. Death, primary or secondary GF, and relapse were considered as treatment failures. GVHD-free, failure-free survival (GFFS) was defined as survival without grades III–IV aGVHD, moderate to severe cGVHD, and treatment failures. Transplantation-related mortality (TRM) was defined as death without relapse of previous disease.
Statistical considerations
Descriptive analysis was performed to summarize the differences in transplant outcomes observed between patients undergoing haplo and haplo-cord HSCT. Between the two groups, differences among the continuous and categorical variables were compared using the Mann–Whitney U test and chi-square test, respectively. Analysis of survival outcomes was performed using the Kaplan–Meier method, and differences were compared by log-rank test. The cumulative incidences were estimated for hematopoietic recovery, peri-ES, GVHD, and CMV and EBV viremia in the competing risk model, with early death and GF as competing risks. For significantly different outcomes between the haplo and haplo-cord groups, univariate and multivariate analysis, in which continuous variables were converted to categorical variables based on the median values, were conducted to determine whether transplant type (haplo or haplo-cord) was the independent influencing factor of the outcomes. In multivariate analysis, variables with p ⩽ 0.10 in the univariate analysis were considered in the Cox proportional regression model. All statistical analysis was performed using IBM SPSS Statistics 25 (IBM, Armonk, NY, USA), while the cumulative incidence and survival curves were generated using GraphPad Prism 8 (GraphPad software, San Diego, USA). A significance level of 0.05 was used for all analyses.
Results
Basic characteristics
A total of 110 patients were diagnosed with aplastic anemia, including 19 patients with inherited BMF syndromes. The 91 patients with acquired SAA were consecutively selected for the study. Characteristics of patients and grafts were listed in Table 1. Of these, 35 patients (38.5%) were assigned to the haplo group and 56 patients (61.5%) were assigned to the haplo-cord group. The median age of the cohort was 7 years (range, 1–16). Half of the recipients were male (n = 46, 50.6%), and the sex match of female to male accounted for 17.6% (n = 16) of the cohort. For SAA treatment before allo-HSCT, 15 (16.5%) patients failed to respond to standard immunosuppressive therapy (ATG + CSA) and 53 (58.2%) patients did not respond to CSA or ATG + stanozolol or testosterone + corticosteroids treatments. The remaining 23 (25.3%) patients received stanozolol or testosterone + corticosteroids treatments and failed. The median interval from SAA diagnosis to HSCT was 86 days (range, 23–252). In the haplo-cord group, the HLA matches of UCB units were mostly 5/6 (n = 35, 62.5%), and the median CD34+ cell and mononuclear cell (MNC) dose of UCB units were 1.34 × 105/kg (range, 0.15–7.95×105) and 3.76 × 107/kg (range, 0.38–7.88×107), respectively. To prevent UCB engraftment, the CD34+ cell and MNC doses in haploidentical grafts were approximately one order of magnitude greater than those of the corresponding UCB units, as shown in Table 1. Notably, no significant differences between the two groups were observed in the baseline information, but the haplo group had a trend of higher cell doses from haploidentical grafts than that in the haplo-cord group (Table 1).
Baseline characteristics of patients and grafts.
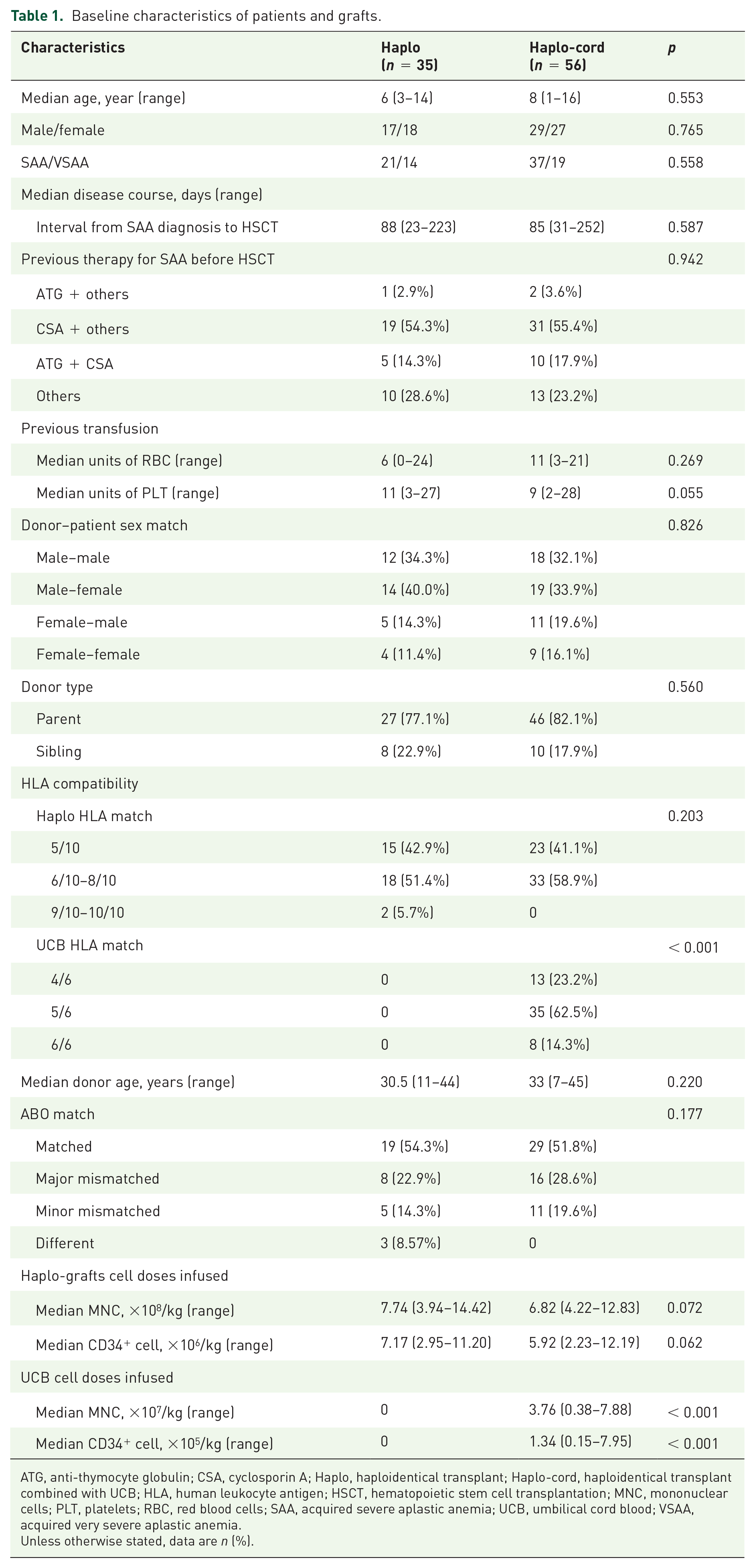
ATG, anti-thymocyte globulin; CSA, cyclosporin A; Haplo, haploidentical transplant; Haplo-cord, haploidentical transplant combined with UCB; HLA, human leukocyte antigen; HSCT, hematopoietic stem cell transplantation; MNC, mononuclear cells; PLT, platelets; RBC, red blood cells; SAA, acquired severe aplastic anemia; UCB, umbilical cord blood; VSAA, acquired very severe aplastic anemia.
Unless otherwise stated, data are n (%).
Engraftment
All patients achieved hematopoietic recovery from haploidentical donors. For the haplo and haplo-cord groups, the median time to neutrophil engraftment was 12 days (range, 10–20) and 11 days (range, 8–20), respectively, with a 30-day cumulative incidence of neutrophil engraftment of 100% for both groups (p = 0.292, Figure 2(a)). Notably, the median time to platelet engraftment for the haplo group was 11.5 days (range, 8–32), which was observed to be faster than that of the haplo-cord group (14.5 days, range, 6–46). The 30-day cumulative incidences of platelet engraftment were 97.1% and 98.2% for the haplo and haplo-cord groups, respectively (p = 0.028, Figure 2(b)). Multivariate analysis suggested that the haplo or haplo-cord transplant type was not associated with the time to platelet engraftment, while a low CD34+ cell dose in haploidentical grafts was correlated with delayed platelet recovery [hazard ratio (HR), 2.495; 95% confidence interval (CI), 1.570–3.964; p < 0.001, Supplementary Tables 2 and 3]. Stable FDC was achieved and sustained in 95.6% of patients (87/91); 5.7% of the haplo group (2/35) and 3.6% of the the haplo-cord group (2/56) underwent secondary GF on days +95, +38, +262, and +74, respectively. Of these, three patients underwent a second transplant from the original haploidentical donors, and 1 patient refused further therapy and died of severe infection of the central nervous system (CNS) on day +222. Among the three patients receiving second transplants, one died of severe infection on day +51 after the second transplant, whereas the remaining two recipients survived to date.

Kaplan–Meier plots showing hematopoietic recovery and peri-engraftment syndrome of the haplo and haplo-cord groups. (a) Cumulative incidences of neutrophil engraftment; (b) cumulative incidences of platelet engraftment; (c) cumulative incidences of peri-engraftment syndrome.
In addition, 17 (48.6%) patients in the haplo group and 42 (75.0%) patients in the haplo-cord group developed peri-ES post-HSCT at a median of 11 days (range, 8–17). We observed that the haplo-cord HSCT was associated with a higher 20-day cumulative incidence of peri-ES, compared with haplo-HSCT in univariate analysis (75.0% ± 5.8% versus 48.6% ± 8.4%, p = 0.029, Figure 2(c), Supplementary Table 2). Multivariate analysis identified haplo-cord HSCT as the only significant factor associated with a higher incidence of peri-ES (HR, 1.767; 95% CI, 1.002–3.115; p = 0.049; Supplementary Table 4). Among the 57 patients with peri-ES, 3 patients in the haplo group and 4 patients in the haplo-cord group ultimately recovered from persistent fever and skin rash with corticosteroid treatment, and the remaining patients recovered without drug therapy. The median length of peri-ES duration was 3 days (range, 1–6).
aGVHD
Twenty-four (26.4%) patients developed II–IV aGVHD at a median of 27 days (range, 14–71), including 13 (14.3%) with grade II, 10 (11.0%) with grade III, and 1 (1.1%) with grade IV. Remarkably, the haplo group showed a higher incidence of II–IV aGVHD at 100 days than that of the haplo-cord group (42.9% ± 8.4% versus 16.1% ± 4.9%, p = 0.002, Figure 3(a)). With respect to target organs involved in aGVHD, the 100-day cumulative incidence of II–IV cutaneous aGVHD was higher in the haplo group than that in the haplo-cord group (48.9% ± 8.5% versus 14.3% ± 4.7%, p < 0.001, Figure 3(b)). Four patients in the haplo group and seven patients in the haplo-cord group developed II–IV aGVHD of the gastrointestinal tract, with 100-day cumulative incidences of 11.4% ± 5.4% and 12.5% ± 4.4% (p = 0.921, Figure 3(c)), respectively. Only two children in the haplo group (5.7%) developed II–IV aGVHD of the liver. In addition, 14.3% of the haplo group (5 of 35) and 7.1% of the haplo-cord group (4 of 56) developed steroid-refractory aGVHD (p = 0.298). By univariate and multivariate analysis, haplo-cord HSCT was identified as the only significant factor associated with a lower incidence of II–IV aGVHD (HR, 0.335; 95% CI, 0.143–0.784; p = 0.012, Tables 2 and 3).

Kaplan–Meier plots showing acute graft-versus-host disease (aGVHD) of the haplo and haplo-cord groups. (a) Cumulative incidences of II–IV aGVHD; (b) cumulative incidences of II–IV cutaneous aGVHD; (c) cumulative incidences of II–IV aGVHD of the gastrointestinal tract.
Univariate analysis of factors associated with II–IV aGVHD and cGVHD.
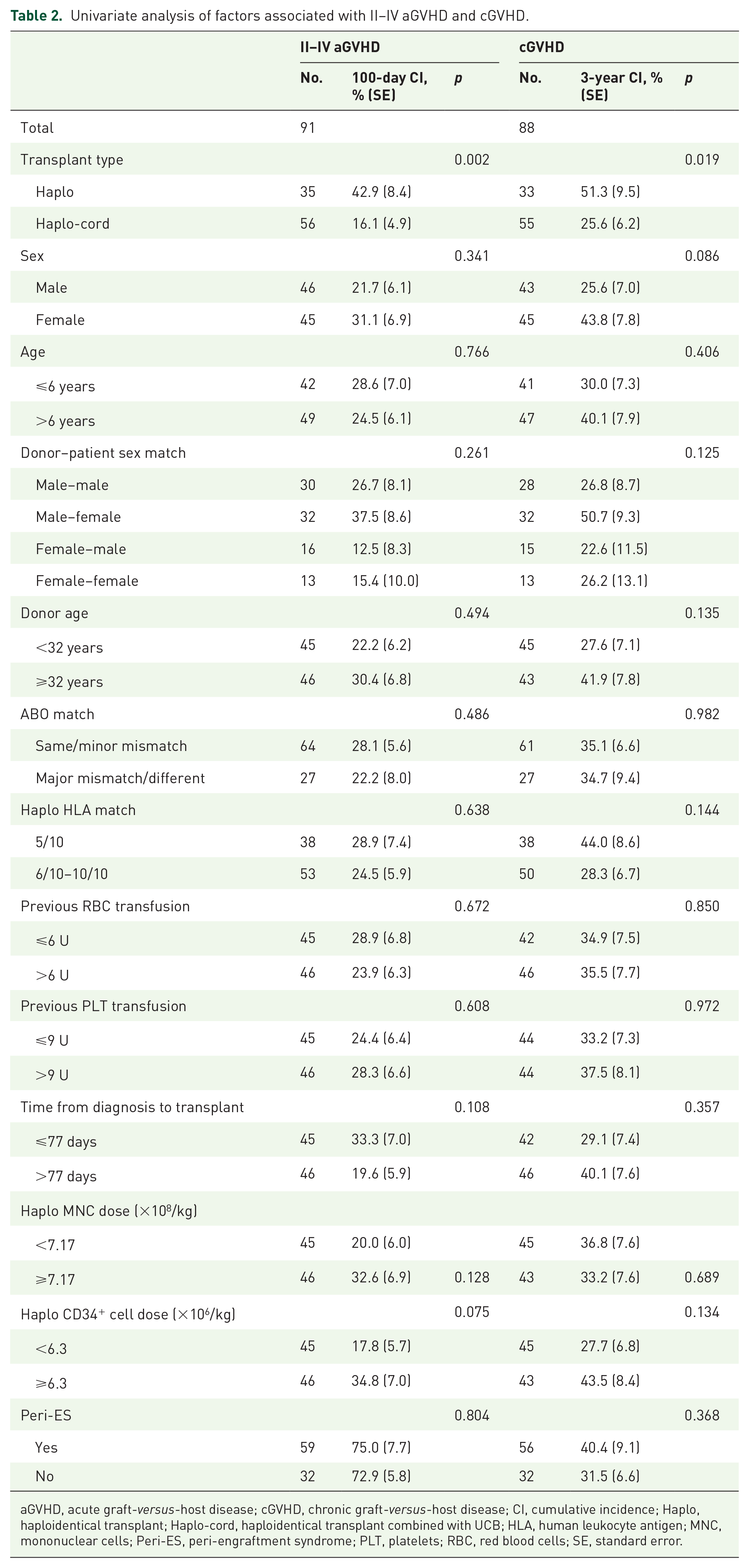
aGVHD, acute graft-versus-host disease; cGVHD, chronic graft-versus-host disease; CI, cumulative incidence; Haplo, haploidentical transplant; Haplo-cord, haploidentical transplant combined with UCB; HLA, human leukocyte antigen; MNC, mononuclear cells; Peri-ES, peri-engraftment syndrome; PLT, platelets; RBC, red blood cells; SE, standard error.
Multivariate analysis of factors associated with II–IV aGVHD.

aGVHD, acute graft-versus-host disease; Haplo, haploidentical transplant; Haplo-cord, haploidentical transplant combined with UCB.
cGVHD
Three patients occurring second GF before day +100 post-transplantation were excluded from cGVHD assessment. Among the 88 patients evaluable for analysis of cGVHD, 28 patients developed cGVHD at a median of 153 days (range, 101–405), with a 3-year cumulative incidence of cGVHD of 36.2% ± 5.5%. The haplo group had a higher 3-year cumulative incidence of cGVHD than haplo-cord group (51.3% ± 9.5% versus 25.6% ± 6.2%, p = 0.019, Figure 4(a)). Moreover, a higher incidence of moderate to severe cGVHD were observed in the haplo group than in the haplo-cord group (41.3% ± 9.9% versus 16.2% ± 5.3%, p = 0.016, Figure 4(b)). By univariate and multivariate analysis, haplo-cord HSCT was identified as the only variable associated with a lower incidence of cGVHD (HR, 0.429; 95% CI, 0.203–0.907; p = 0.027; Tables 2 and 4).

Kaplan–Meier plots showing chronic graft-versus-host disease (cGVHD) of the haplo and haplo-cord groups. (a) Cumulative incidences of cGVHD; (b) cumulative incidence of moderate to severe cGVHD.
Multivariate analysis of factors associated with cGVHD.

cGVHD, chronic graft-versus-host disease; Haplo, haploidentical transplant; Haplo-cord, haploidentical transplant combined with UCB.
Infectious complications
Within the first 100 days after HSCT, the most common infection was CMV viremia, which occurred in 82.4% (n = 75) of our cohort at a median of 24 days (range, 7–95). Cumulative incidences of CMV viremia at 100 days after HSCT were similar between the haplo and haplo-cord groups (80.0% ± 6.8% versus 83.9% ± 4.9%, p = 0.201, Figure 5(a)), with no child developing CMV disease. Forty-nine patients (53.9%) had EBV viremia at a median of 27 days (range, 11–79), with no patient developing EBV-associated post-transplantation lymphoproliferative disease (PTLD). The 100-day cumulative incidences of EBV viremia were comparable between the haplo and haplo-cord groups (57.1% ± 8.4% versus 51.8% ± 6.7%, p = 0.960, Figure 5(b)). Of the two patients who were infected with human herpes virus on days +31 and +9, respectively, one patient developed viral encephalitis and died of cerebral hemorrhage, while the other eventually recovered after acyclovir administration. In addition, 8 cases (22.9%) in the haplo group and 11 cases (19.6%) in the haplo-cord group developed hemorrhagic cystitis (HC) (p = 0.714), at a median of 23 days (range, 1–59). Of the 19 patients with HC, 7 in the haplo group (7/8) and 10 in the haplo-cord group (10/11) tested positive for BK virus. Ongoing infection occurred in two patients with bacterial blood stream infections after HSCT [1 in the haplo group (2.9%) and 1 in the halplo-cord group (1.8%), respectively], including one developing bacterial pneumonia and the other developing bacterial infection of CNS. No cases in this study had fungal infections.

Kaplan–Meier plots showing the cytomegalovirus (CMV) and Epstein–Barr virus (EBV) viremia of the haplo and haplo-cord groups. (a) Cumulative incidences of CMV viremia; (b) cumulative incidences of EBV viremia.
Survival outcomes
The last follow-up date was 30 October 2021. Within a median follow-up of 23.17 months (range, 3.37–79.63), 2 of the 35 children in the haplo group and 3 of the 56 children in haplo-cord group died of TRM. The haplo and haplo-cord groups had similar estimated 3-year OS [92.8% ± 4.9% versus 94.3% ± 3.2%, p = 0.846, Figure 6(a)] and FFS [90.4% ± 5.4% versus 92.5% ± 3.6%, p = 0.722, Figure 6(b)]. Causes of death for the haplo group included one cerebral hemorrhage and one bacterial infection of the lungs. Causes of death for the haplo-cord group included one obstructive bronchiolitis developed from cGVHD, one bacterial infection of CNS, and one capillary leakage syndrome. In addition, the haplo-cord group showed a better GFFS than the haplo group [68.2% ± 6.4% versus 52.3% ± 9.1%, p = 0.158, Figure 6(c)].
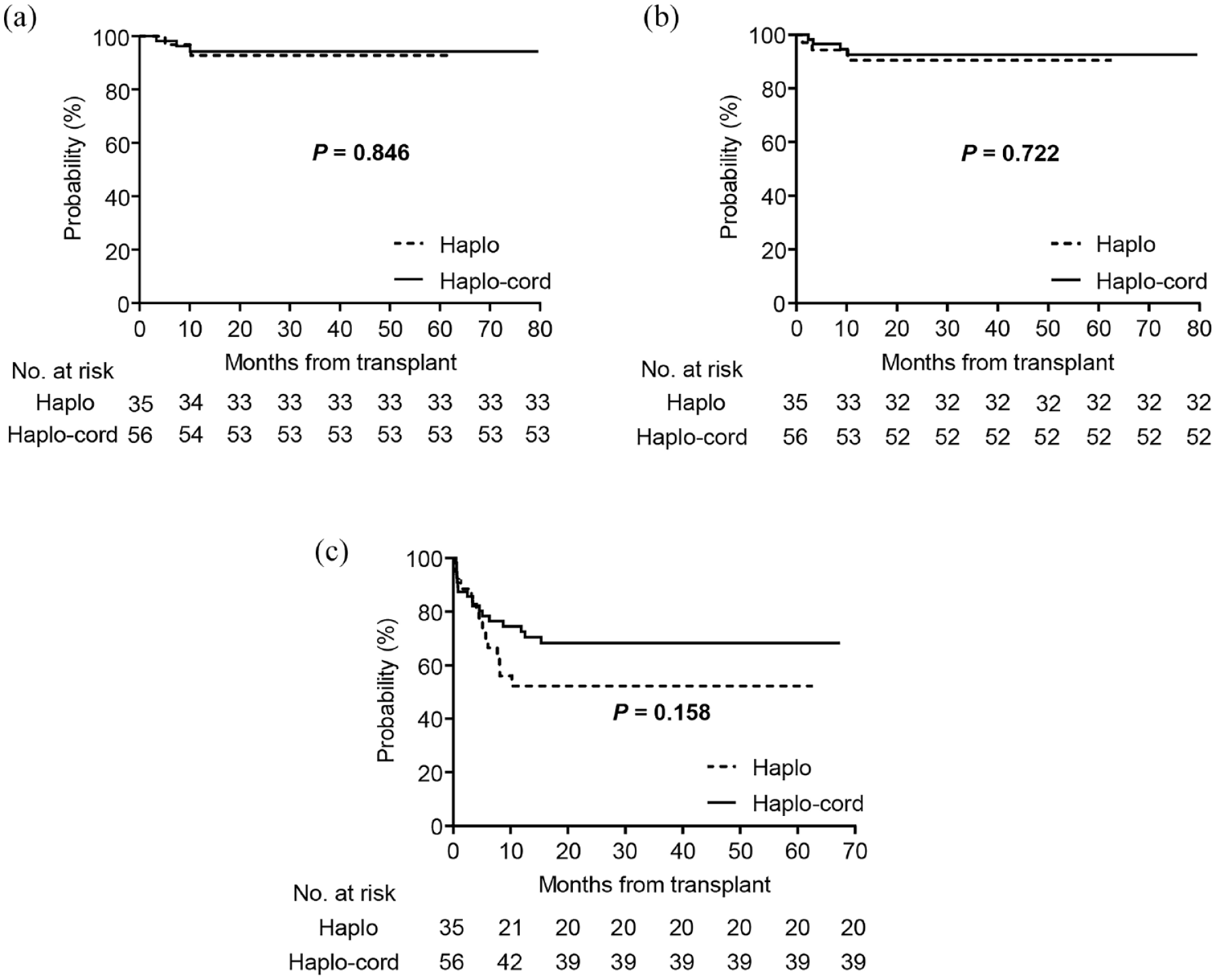
Kaplan–Meier plots showing survival outcomes of the haplo and haplo-cord groups. (a) Overall survival; (b) failure-free survival; (c) graft-versus-host disease-free, failure-free survival.
Discussion
In this study, we treated 91 children of SAA by haplo-HSCT based on the ‘Beijing Protocol’ with or without co-infusion of a third-party UCB unit. Basic characteristics and transplant outcomes between the haplo and haplo-cord groups were compared, in which we primarily found that the haplo-cord group had lower incidences of II–IV aGVHD and cGVHD.
With improved conditioning regimens and supportive care, haplo-HSCT has provided a feasible salvage treatment for patients with malignant and non-malignant hematological diseases over the past decade.8,36,37 In China, the transplant modality of the ‘Beijing Protocol’, developed by Huang’s group, has accounted for 51.0% of allo-HSCT for SAA patients up to 2019, 7 with reported GF rates of less than 5% and 3-year survival rates over 80% from different centers.9,38,39 However, there is still a relatively high GVHD risk for large numbers of patients undergoing haplo-HSCT.8,9 Researchers have investigated multiple strategies to prevent GVHD for haplo-HSCT, including the use of G-CSF-primed grafts and treatment with high dose PT-Cy. 6 In recent years, several studies observed a low incidence of severe GVHD in UCBT,18,19 indicating that UCB has potential immune properties for controlling GVHD. In recent years, the combination of haplo-HSCT and a third-party UCB unit has been developed based on the ‘Beijing Protocol’ to optimize potential advantages of UCB in reducing the risk of severe GVHD. Remarkably, a few studies have reported the superior incidence of GVHD in patients receiving haplo-cord HSCT for hematological malignancies.25,26 However, studies comparing outcomes of haplo and haplo-cord HSCT in SAA children remain limited.
This study suggested that the haplo-cord group had a superior incidence of II–IV aGVHD, cGVHD, and moderate to severe cGVHD to haplo group. In contrast to our findings, a comparative study using a similar transplant protocol to treat 139 patients with refractory acute leukemia failed to observe a difference in incidences of aGVHD between haplo and haplo-cord groups, 28 with higher incidence of II–IV aGVHD than those observed in our study. On the other hand, another single-arm study by Liu et al. 38 investigating 146 adult patients who received haplo-cord HSCT for SAA suggested incidence rates of II–IV aGVHD and cGVHD that were similar to those found in our results; however, haplo-HSCT was not evaluated in this study. We inferred that different conditioning regimens might be responsible for these observations. Unlike NMA conditioning regimens for SAA patients in this study and in Liu’s study, studies by Wang et al. used myeloablative conditioning based on Bu or total body irradiation for enrolled patients with hematological malignancies, which is a widely known risk factor for severe GVHD. 20 The potential effect of UCB grafts on controlling aGVHD might be mitigated by the high-intensity conditioning regimens. The aGVHD advantage in the haplo-cord group was primarily restricted to II–IV cutaneous aGVHD in this study. We failed to observe differences between the haplo and haplo-cord groups in aGVHD of the gastrointestinal tract and liver, which might be attributed to the low incidences (II–IV gastrointestinal aGVHD: 11/91, II–IV liver aGVHD: 2/91). It is acknowledged that skin is the most frequently affected organ in aGVHD, 20 and this was also suggested in this study. Cases of steroid-refractory aGVHD were also limited in this study (nine cases), which precluded a formal analysis for investigating the differences between the haplo and haplo-cord groups. The mechanism underlying the advantages of inhibiting GVHD in the haplo-cord group might lie in enriched immune regulatory cells contained in UCB, such as Treg and MSC. 23 Tolar et al. 23 discovered that a distinct CD4+ CD25bright subset of relatively high frequency existed in UCB compared with PB, which had a skewing toward a Treg suppressor cell phenotype to inhibit GVHD. Moreover, a few studies have shown the abilities of MSC to inhibit T and B cell proliferation and to suppress dendritic cells.40,41 The three cell types were crucial in pathogenesis of acute and chronic GVHD.20,42 Therefore, co-infusion of a UCB unit might reduce the risk of severe GVHD for haplo-HSCT recipients, as found in this study. However, given that corresponding studies in pediatric patients were few, more research investigating SAA children receiving this HSCT protocol is needed to confirm this observation.
Haploidentical grafts were used as predominant source of hematopoietic stem cells in this study, and all patients initially achieved FDC from haploidentical donors. The possible reason for haploidentical engraftment in the haplo-cord group might be the higher doses of MNC and CD34+ cells in haploidentical grafts than those in UCB units, which contributed to a stronger engraftment ability. The haplo-cord group had slower platelet recovery, which might be attributed to the lower CD34+ cell dose from haploidentical grafts in this cohort than that in the haplo group. Multivariate analysis identified CD34+ cell dose as the only significant factor influencing platelet recovery, which is broadly in line with other reports.43,44 Hence, an adequate dose of CD34+ cells from haploidentical donors may be warranted to accelerate platelet recovery for SAA children receiving haplo-cord HSCT. While GF often occurred in SAA patients receiving allo-HSCT, 45 no primary GF was observed in all patients. Secondary GF rates were low in both the haplo (2/35) and haplo-cord (2/56) groups, which is similar to the latest findings. 38 Although co-infusion of UCB with haplo-HSCT might induce an immunological graft-versus-graft effect, 46 this may not result in GF because of the relatively low cell doses in UCB units. Another probable explanation for the encouraging engraftment rates is the G-CSF primed haploidentical grafts and the high doses of ATG used, which induced immune tolerance and successful hematopoietic recovery.6,47
Pre-engraftment syndrome, a type of peri-ES, 32 is a common complication after UCBT. 48 In this study, we observed a higher incidence of peri-ES in the haplo-cord group than in the haplo group. The association between haplo-cord HSCT and peri-ES was not reported, and the mechanism underlying the association observed in our study remains unknown. In a previous study investigating the cytokine profiles of cord blood samples from 224 infants, Takahashi et al. 49 demonstrated that granulocyte-macrophage colony-stimulating-factor (GM-CSF) serum levels were high in cord blood. In a recent study on pre-ES, Jin’s team revealed that GM-CSF produced by cord blood-derived inflammatory monocytes drives pre-ES pathology in UCBT. 50 Therefore, a possible reason for the peri-ES susceptibility observed in the haplo-cord group might be attributed to the potent GM-CSF-producing monocytes remaining in UCB grafts. Despite its high incidence, children suffering from peri-ES recovered ultimately following hematopoietic recovery and/or corticosteroid therapy.
CMV and EBV viremia were the most common infectious complications at 100 days after haplo-HSCT for SAA patients in our study, which is broadly in line with reports from other centers.9,39 We failed to observe a difference in viral infection between haplo and haplo-cord groups, consistent with the aforementioned study of Wang et al. 28 However, the incidences of CMV and EBV viremia in Wang’s study were lower than those in our study. The possible reason for this difference may be due to distinct ATG dosages in conditioning. From day −5 to −2, the total ATG dose in our patients was 10 mg/kg, twice that of Wang’s recipients (5 mg/kg); a high dose of ATG was associated with delayed immune reconstitution, resulting in inferior antiviral immune response.51,52 Although naive T lymphocytes contained in UCB were shown to contribute to antigen-specific T-lymphocyte immunity generation early after HSCT, patients in the haplo-cord group predisposed to a high risk of viremia similar to haplo group recipients. Delayed immune reconstitution caused by high doses of ATG, HLA disparity 53 and limited numbers of naive T cells based on the minor UCB units might be responsible for the comparable viremia incidences between the two groups. An optimized ATG dose and a UCB cell dose, together with modified virus prevention for haplo-HSCT, may potentially improve this outcome. Despite the relatively high viremia incidence in this study, no children developed CMV disease and EBV-associated PTLD due to intensive surveillance performed weekly and preemptive treatment with ganciclovir and rituximab for serologically positive patients.
Remarkable progress has been made in haploidentical transplantation based on the ‘Beijing Protocol’ for SAA patients in the recent years, with satisfactory OS and FFS indicated in several studies. Xu et al. 8 reported 3-year OS, FFS, and GFFS of 89.0%, 86.8%, and 80.8%, respectively, for SAA patients of haplo-HSCT in a multicenter prospective study. Lu et al. 39 showed similar survival outcomes in the haplo group (80.3% for OS; 76.4% for FFS; 79.0% for GFFS) in a retrospective study comparing transplantation from HID and matched unrelated donors. Our data also showed a satisfactory 3-year OS and FFS in the haplo and haplo-cord groups (92.8% and 94.3% for OS; 89.4% and 92.3% for FFS). Comparable survival outcomes were observed between the haplo and haplo-cord groups, potentially indicating that the co-infusion of a UCB unit with haplo-HSCT based on the ‘Beijing Protocol’ at least yielded a non-inferior impact on the long-term survival of SAA children, compared with those undergoing haplo-HSCT without UCB. Nevertheless, the haplo-cord group had a superior 3-year GFFS compared with the haplo group, which may be attributed to less severe aGVHD and moderate to severe cGVHD in the haplo-cord group. Still, this observation requires further investigation in larger scale multicenter research.
Several limitations existed in our study. This is a retrospective study with a limited sample size. Although we found no difference in incidences of II–IV aGVHD of the gastrointestinal tract and liver as well as steroid-refractory aGVHD between the haplo and haplo-cord groups, the potential difference may be revealed in a multicenter study with a larger sample of pediatric patients with SAA. Such future research would also provide sufficient evidence to confirm the potential impact of UCB co-transplantation in haplo-HSCT on GVHD and survival outcomes. In addition, the UCB units used for the haplo-cord group differed in HLA mismatch and cell doses in this study, indicating that certain heterogeneity existed in patients undergoing haplo-cord HSCT. A relatively uniform transplant protocol describing specific cell doses and detailed HLA match might solve this problem.
Conclusion
In summary, our study provided compelling results of descriptive analysis suggesting that co-infusion of a third-party UCB unit with haplo-HSCT might be associated with a lower risk of II–IV aGVHD and cGVHD. Although this co-infusion protocol may come with a higher risk of peri-ES, the survival outcomes may not be negatively affected. Despite the limited sample size, this study is the first real-world comparative study to investigate the impact of haplo-cord HSCT on GVHD in SAA children, which may stimulate further research of haplo-cord HSCT in pediatric patients with SAA and hematologic malignancies, as well as the potential association with GVHD. A well-designed prospective study of multicenters with large sample sizes is warranted to confirm these encouraging results in GVHD of the ‘Beijing Protocol’-based haplo-cord HSCT. Moreover, basic research to reveal the mechanisms underlying the impact of UCB co-infusion on these transplant outcomes in haplo-HSCT is still needed.
Supplemental Material
sj-docx-1-tah-10.1177_20406207221134409 – Supplemental material for Association between haploidentical hematopoietic stem cell transplantation combined with an umbilical cord blood unit and graft-versus-host disease in pediatric patients with acquired severe aplastic anemia
Supplemental material, sj-docx-1-tah-10.1177_20406207221134409 for Association between haploidentical hematopoietic stem cell transplantation combined with an umbilical cord blood unit and graft-versus-host disease in pediatric patients with acquired severe aplastic anemia by Di Yao, Yuanyuan Tian, Jie Li, Bohan Li, Jun Lu, Jing Ling, Defei Zheng, Yanhua Yao, Peifang Xiao, Lijun Meng and Shaoyan Hu in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
