Abstract
The methodologies and applications of ex vivo expansion of umbilical cord blood cells represent a significant research domain, primarily owing to the distinctive characteristics of these cells and their prospective uses in regenerative medicine and hematopoietic stem cell transplantation. We searched PubMed and Web of Science using the terms “umbilical cord blood cells,” “cytokine,” “haematopoietic cells,” “umbilical cord mesenchymal stem cells,” “in vitro expansion,” and “microenvironment,” and the selected literature was organized and analyzed. We present a narrative review of the mechanisms by which umbilical cord mesenchymal stem cells enhance the in vitro expansion of umbilical cord blood-derived hematopoietic stem cells, focusing on three primary aspects: secretion of diverse cytokines to replicate the natural hematopoietic microenvironment, transmission of critical signals via direct cell-to-cell contact, and exertion of immunomodulatory effects to alleviate environmental stress. Although these processes can significantly promote the proliferation and survival of hematopoietic stem cells, the challenge of concurrently preserving the long-term stemness of these cells in an in vitro environment remains a critical issue for future research.
Keywords
Introduction
Umbilical cord (UC) mesenchymal stem cells (MSCs) considerably advance the proliferation of umbilical cord blood (UCB)-derived hematopoietic stem cells (HSCs), owing to their beneficial biological characteristics, varied cytokine profiles, and enhanced regenerative capacities. We composed this review utilizing the Narrative Review Evaluation Scale (SANRA) 1 as a guideline. The comprehensive relationship between UC-MSCs and UCB-HSCs creates a complex system, encompassing cytokine signaling, matrix interaction, intercellular communication, and extracellular transfer of regenerative signals (Figure 1). The critical role of UC-MSCs in the in vitro proliferation of hematopoietic progenitor cells underscores their potential for clinical application in regenerative medicine and hematological treatment. Continued investigation into the optimization of coculture settings and the elucidation of pertinent molecular pathways is essential for translating these discoveries into viable treatment solutions. This review summarizes pertinent findings to enhance the understanding of the clinical significance of in vitro expansion of UCB-HSCs.
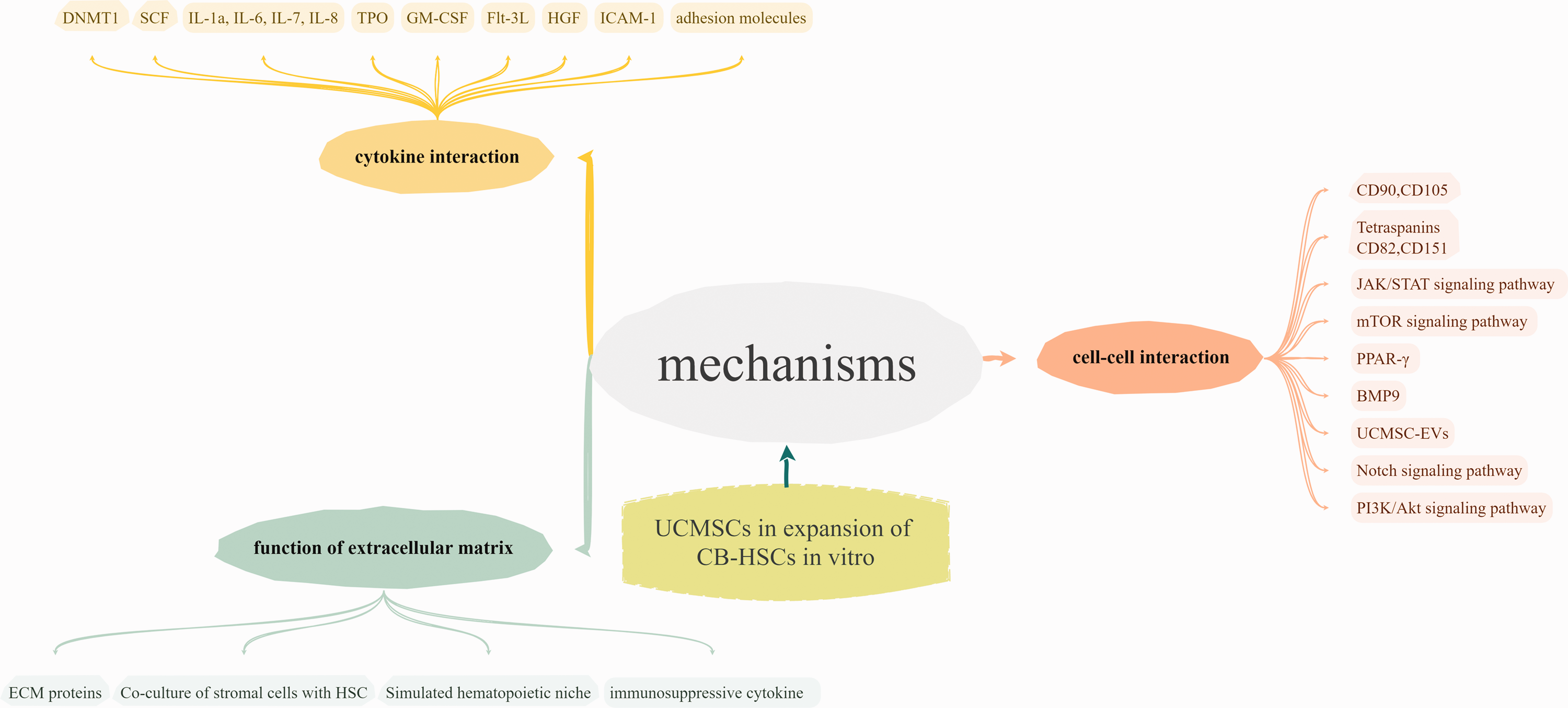
Flowchart of potential mechanisms of UC-MSCs in the expansion of UCB-HSCs in vitro. UCMSC-EVs: extracellular vesicles of umbilical cord mesenchymal stem cells; ECM: extracellular matrix; HSCs: hematopoietic stem cells; UC-MSCs: umbilical cord mesenchymal stem cells; UCB-HSCs: umbilical cord blood-derived hematopoietic stem cells; DNMT1: DNA methyltransferase 1; SCF: stem cell factor; IL: interleukin; TPO: thrombopoietin; GM-CSF: granulocyte-macrophage colony-stimulating factor; FLT3L: FMS-related tyrosine kinase 3 ligand; CD: cluster of differentiation; PPAR-γ: peroxisome proliferator-activated receptor-γ; BMP9: bone morphogenetic protein 9; HGF: hepatocyte growth factor; ICAM-1: intercellular cell adhesion molecule-1.
Search strategy
A MeSH word search and a free search were used in the literature search. The search terms “umbilical cord blood cells” or “microenvironment,” “umbilical cord mesenchymal stem cells,” “haematopoietic cells,” “in vitro expansion,” and “cytokine” were all part of the retrieval approach. English-language articles published between 1 January 2005 and 31 December 2024 were included in the search. We searched the Web of Science, PubMed, and Google Scholar databases. Retrieved articles with Journal Citation Reports impact factor ≤3.0 or those unrelated to the topic were manually removed. The literature review steps included determining research topics, collecting relevant literature, classifying the literature, extracting key information, conducting comparative analysis and synthesis, and finally forming a literature review.
Biological properties of UC-MSCs
Human UC-MSCs originate from the mesoderm and possess capabilities for self-renewal and multidifferentiation as well as the ability to secrete numerous bioactive substances.2,3 They can be extracted from several regions of the UC, including cord glue, perivascular zones, and umbilical veins. Research indicates that MSCs derived from Wharton’s jelly exhibit markedly superior growth capacity and viability compared with those extracted from other regions of the UC. 4 Distinct cell types exhibit variations in surface marker expression; for instance, UC-MSCs characteristically express markers such as CD90, CD73, and CD105 while demonstrating minimal-to-absent expression of hematopoietic markers (CD34 and CD45), thereby further substantiating their mesenchymal ancestry.5,6 UC-MSCs can differentiate into many cell types, including adipocytes, chondrocytes, and osteocytes. 7 Research indicates that UC-MSCs can undergo a biphasic adipogenic differentiation phase, initially transforming into precursor cells before maturing into adipocytes. 8 A study demonstrated that UC-MSCs can modulate the activity of dendritic cells and natural killer cells, promoting a balanced immune response. 9 They often multiply more rapidly than adult-derived MSCs, such as those from bone marrow or adipose tissue, leading to greater yields in vitro, which is essential for therapeutic applications. 10 Furthermore, UC-MSCs demonstrate distinct immunological characteristics. Their immunological immaturity facilitates a diminished immune response, enhancing graft acceptance in allogeneic transplantation and lowering the risk of rejection. 11 Paracrine substances released by these cells are believed to considerably influence treatment efficacy by modulating inflammation and facilitating tissue repair. 12
Research progress on the in vitro expansion of UCB-HSCs
Significance of expansion of UCB-HSCs in vitro
UCB is recognized for its rich supply of hematopoietic stem and progenitor cells, which are essential for addressing numerous hematological cancers and genetic disorders. The restricted quantity of these cells in a single UCB unit presents a considerable obstacle for transplantation, rendering ex vivo multiplication essential to optimize their therapeutic efficacy.
A primary advantage of in vitro expansion of UCB-HSCs is the ability to surmount the constraints associated with the limited quantity of cells that can usually be derived from a single cord blood unit. Augmenting the quantity of viable stem cells enhances the probability of successful implantation post-transplantation. 13 Recent studies have indicated that new culture techniques employing multiple cytokines and growth factors can markedly enhance the proliferation of UCB-HSCs while reducing their differentiation into mature blood cells. 14 These methods have shown that particular combinations of growth factors, including stem cell factor (SCF), FMS-like tyrosine kinase 3 ligand (Flt-3L), and thrombopoietin (TPO), can efficiently augment the UCB-HSC population, thereby enhancing outcomes in the context of clinical HSC transplantation.14,15 A recent study by Guo et al. 16 highlighted that modulating metabolic pathways, particularly glycolysis, might improve the differentiation of hematopoietic lineages from HSCs during in vitro growth. This metabolic approach enables the development of more potent therapeutic stem cell units for transplantation. The use of UC-MSCs for the in vitro expansion of UCB-HSCs does not present ethical concerns, as these cells are generally considered waste from embryonic–extraembryonic tissues. 17
Traditional external expansion methods and limitations
The conventional in vitro development of UCB-HSCs primarily requires culture methodologies that frequently incorporate cytokine supplementation and coculture systems. Nonetheless, although conventional techniques can attain a degree of UCB-HSC expansion, they possess considerable restrictions that impact the quality and quantity of the expanded cells. Standard growth techniques may augment cell quantity, but they frequently fail to attain the requisite threshold for therapeutic use, particularly for adult patients who require elevated cell dosages for successful transplantation. In vitro cultivation can lead to the risk of differentiation into nonhematopoietic lineages, potentially reducing the stem cell population. Cytokines are employed to inhibit differentiation, but the equilibrium between sustaining an undifferentiated state and encouraging cell proliferation is precarious, with conventional techniques frequently resulting in premature lineage commitment. Extended culture durations, even under optimal conditions, may alter the functional properties of UCB-HSCs, including diminished homing capability and transplantation potential.
The mechanism of UC-MSCs in the expansion of UCB-HSCs in vitro
Cytokine interaction
The cytokines generated by UC-MSCs significantly influence the in vitro proliferation of UCB-HSCs. This connection facilitates the proliferation, survival, and functionality of UCB-HSCs, augmenting their therapeutic potential. The equilibrium between self-renewal and differentiation is crucial for sustaining a sufficient stem cell reservoir. Cytokines interact with specific receptors on UCB-HSCs, activating intracellular signaling pathways that facilitate cell proliferation and suppress differentiation. Ahmadnejad et al. 18 established that the expression of DNA methyltransferase 1 is correlated with the proliferation of HSCs when they are cocultured with UC-MSCs. This indicates that the interaction between UC-MSCs and cytokines augments the self-renewal capacity of UCB-HSCs. Cytokines have been demonstrated to extend the lifespan of hematopoietic progenitor cells in vitro, thereby substantiating the concept that the cytokine milieu is crucial for preserving the properties of stem cells during the culture phase. UC-MSCs foster an environment that mitigates the early differentiation of UCB-HSCs. In cultures devoid of UC-MSCs, HSCs exhibit accelerated differentiation into adult lineage cells, leading to a reduction in the population of progenitor cells. This dynamic pertains to the signaling pathway initiated by cytokine interactions, which is crucial in preserving the undifferentiated status of UCB-HSCs inside the coculture system. De Lima et al. 19 demonstrated that the coculture of UC-MSCs and UCB-HSCs resulted in substantial enhancement of the survival and proliferation of hematopoietic progenitor cells, primarily attributable to the cytokine support provided by the UC-MSCs. This demonstrates the significance of the paracrine signaling system in facilitating the proliferation of hematopoietic cells. Fajardo-Orduña et al. 20 revealed that the integration of UC-MSCs with a specific cytokine cocktail might significantly enhance the proliferation of UCB-HSCs, leading to an increased stem cell population for prospective therapeutic applications. Human Wharton’s jelly stem cells (hWJSCs) frequently exhibit elevated levels of interleukins (ILs) (IL-1a, IL-6, IL-7, and IL-8), SCF, hepatocyte growth factor, and intercellular cell adhesion molecule-1, suggesting their potential role in HSC proliferation. 21 Allogeneic hWJSCs offer a promising platform for the ex vivo expansion of a significant number of UCB CD34+ cells while maintaining their primitive characteristics. 22
The cytokine interaction of UC-MSCs is crucial for the in vitro expansion of UCB-HSCs. Improving our understanding of these interactions offers a viable approach to refine the practical implementation of stem cell treatment in hematological disorders.
Cell–cell interaction
A crucial element of cell–cell interaction is the expression of surface-specific markers on UC-MSCs, which facilitates adhesion to UCB-HSCs. UC-MSCs often display markers such CD90 and CD105, which are crucial for cell adhesion and signal transduction processes that preserve the properties of HSCs. These interactions facilitate the proliferation and undifferentiation of CD34+ hematopoietic progenitor cells during coculture. 23
Cocultures of mesenchymal cells can replicate essential hematopoietic niche attributes and function via paracrine and contact-dependent processes. 19 UC-MSC-mediated signaling improves the survival of UCB-HSCs and directs their lineage selectivity. Numerous studies have demonstrated that soluble substances derived from MSCs can significantly influence the proliferation of HSCs by enhancing their self-renewal capacity and inhibiting unwarranted differentiation. 24 The dynamic interaction between UC-MSCs and UCB-HSCs facilitates the integration of gene regulation and niche signaling, highlighting the significance of paracrine interactions in regulating hematopoiesis. Li et al. 25 demonstrated that MSCs significantly influence the implantation potential of HSCs by modulating the hematopoietic microenvironment, highlighting a paracrine signaling mechanism that improves the survival and metabolic support of HSCs. Recent studies have investigated how hypoxia preconditioning enhances the paracrine action of UC-MSCs. Niazi et al. 26 demonstrated that hypoxia-pretreated UC-MSC-derived exosomes enhance the proliferation of CD133+ UCB-HSCs by activating the Notch signaling pathway, thereby highlighting the intricacy of the paracrine interactions involved. This underscores the opportunity to refine UC-MSC culture settings to improve the effectiveness of UCB-HSC growth techniques.
The paracrine signaling of UC-MSCs not only encompasses the provision of growth factors but also facilitates cell-to-cell contacts via extracellular exosomes. Exosomes secreted by UC-MSCs encompass a diverse array of physiologically active chemicals, including microRNAs (miRNAs) and proteins, which can interact with the receptors on target cells to enhance the activity and proliferation of HSCs. Research indicates that exosomes generated from MSCs enhance the proliferation and viability of HSCs by modulating the AKT signaling pathway. 27 Moreover, the constituents of exosomes may contribute to the regulation of the milieu around HSCs. Exosomes originating from MSCs might augment contact between the HSC and its microenvironment by upregulating surface adhesion molecules, thereby improving its survival rate.28,29 This paracrine pathway facilitated by exosomes enhances the self-renewal and developmental potential of HSCs. 30
Cytokines are essential for activating multiple signaling pathways, notably the Janus kinase-signal transducer and activator of transcription (JAK–STAT) pathway, which increases the self-renewal capacity of UCB-HSCs and maintains their functionality in the hematopoietic compartment. The JAK–STAT pathway regulates HSC behaviors by mediating the effects of extracellular signals, including inflammatory cytokines such as TPO and ILs. The activation of the JAK–STAT pathway results in the phosphorylation of STAT proteins, which are responsible for transcribing genes that facilitate the proliferation, survival, and self-renewal of HSCs. 31 Recent studies indicate that, alongside JAK–STAT signaling, mTOR signaling—activated by various hematopoietic growth factor receptors—is crucial for HSC function and responds to cytokines such as TPO. The inhibition of mTOR negatively impacts HSC self-renewal during regenerative conditions, highlighting its essential role in maintaining stem cell function amid various cytokines. 32 Guo et al. 16 indicated that the antagonism of the nuclear hormone receptor peroxisome proliferator-activated receptor-γ facilitated the proliferation of phenotypic and functional subsets of human UCB-HSCs and bone marrow hematopoietic stem and progenitor cells (HSPCs) in vitro. The intercellular connection between UC-MSCs and UCB-HSCs influences the differentiation route. The environment offered by UC-MSCs can modify the lineage orientation of UCB-HSCs, enabling the regulation of differentiation while facilitating expansion. 20 This equilibrium is crucial to guarantee the availability of sufficient stem cells, enabling their maturation into diverse blood cell lineages as required.
The interaction of cell surface proteins is of equal significance. The four transmembrane protein superfamilies, CD82 and CD151, are involved in cell-to-cell signal transduction and cell recognition. The expression of these proteins can facilitate the interaction between MSCs and HSCs, thereby augmenting the functional proliferation of HSCs. Bone morphogenetic protein-9 can modulate the differentiation of MSCs via unique signaling pathways, significantly influencing the regulation of the HSC microenvironment.
The intercellular contact between UC-MSCs and UCB-HSCs is crucial for efficiently growing hematopoietic progenitor cells in vitro. The advancement of research in characterizing and optimizing cell interactions can yield significant insights for enhancing treatment efficacy and advancing regenerative medicine applications.
Function of the extracellular matrix (ECM)
The ECM consists of many proteins and other chemicals that establish a supportive milieu and influence cellular behaviors such as adhesion, migration, proliferation, and differentiation. The interaction of UC-MSCs with the ECM and modification of the ECM during the expansion of UCB-HSCs is crucial for optimizing stem cell treatment. Fernández-Pernas et al. 33 indicated that UC-MSCs facilitate these connections by synthesizing specific ECM proteins that enhance cell adhesion and stability throughout the culture, leading to sustained proliferation and maintenance of stem cell properties. The regenerative capacity of UC-MSCs is partially attributed to their capability to dynamically remodel the ECM, thereby modifying the milieu to support the proliferation and sustenance of stem cells. Furthermore, the findings of Nirmal and Nair 34 substantiate the notion that particular signaling pathways triggered in UC-MSCs can augment the deposition of ECM components vital for facilitating cartilage creation and may also be advantageous for hematopoietic expansion. The interplay between the ECM and UC-MSC paracrine signals can further augment the therapeutic efficacy of expanded UCB-HSCs.
The hematopoietic microenvironment, abundant in the ECM, offers a crucial habitat for HSCs. The findings presented by Song et al. 35 underscore this notion and highlight the benefits of coculturing stromal cells and HSCs, as they collaboratively enhance the composition and functionality of the ECM, thereby facilitating the growth and proliferation of hematopoietic cells. A primary function of UC-MSCs in facilitating UCB-HSC multiplication is their ability to replicate the physiological conditions of the hematopoietic niche. Research indicates that UC-MSCs can replicate essential signals in the hematopoietic milieu in vivo that are frequently diminished in conventional suspension culture methods. Furthermore, UC-MSCs have demonstrated the capacity to modulate the immune response through the secretion of immunosuppressive substances, thereby fostering an environment conducive to the proliferation of HSCs without eliciting an unfavorable immunological response. 36
By enhancing the physiological conditions of the ECM and utilizing the paracrine signals from UC-MSCs, researchers can optimize the yield and therapeutic efficacy of UCB-HSCs for clinical applications in regenerative medicine.
Research progress on the assistive role of UC-MSCs in the expansion of UCB-HSCs in vitro
Establishment of a cocultivation system
UC-MSCs significantly contribute to the proliferation of UCB-HSCs via multiple mechanisms, notably through the secretion of cytokines and growth factors in a coculture setting. Jing et al. 37 demonstrated that the surface of mesenchymal stromal cells serves as the primary site for the proliferation of HSCs. In contrast, the area beneath the mesenchymal stromal cell layer appears to replicate the stem cell niche for less mature cells. The interaction between SDF-1 and CXCR4, along with integrin-mediated cell adhesion, is crucial for the distribution of HSCs within the coculture system. This may have implications for future clinical applications. Branco et al. 38 compared two clinically relevant expansion platforms for HSPCs by optimizing the mixtures of three cytokines: SCF, Flt-3L, and TPO. Results indicated that the HSPC coculture system with bone marrow MSCs surpassed the feeder-free system, exhibiting superior maintenance of HSPC marker expression and multilineage differentiation potential. This cytokine mixture may have a comparable influence on UC-MSCs.
Three-dimensional cultivation method
Three-dimensional culture techniques can more accurately simulate the intricate interactions between stem cells and their environment, which cannot be frequently achieved by typical two-dimensional culture methods. Conversely, three-dimensional culture techniques can enhance pertinent cell–cell and cell–matrix interactions, more effectively preserving the stemness and differentiation potential of UC-MSCs and UCB-HSCs.
Conclusion
This review encompasses the recent research progress on the mechanism of action of UC-MSCs in the in vitro expansion of UCB-HSCs: (a) secreting various cytokines to replicate the natural hematopoietic microenvironment; (b) transmitting essential signals via direct cell–cell interaction; and (c) exerting immune regulatory functions to alleviate environmental stress. Nonetheless, although several studies have indicated that UC-MSCs can enhance in vitro expansion through the secretion of bioactive molecules, the efficiency and controllability of this expansion have not yet met the criteria for clinical application. The phenotypic alterations of UC-MSCs are significant determinants of their functional constraints. UC-MSCs may display varying biological properties under distinct culture circumstances, including the expression of stem cell markers and their proliferation and differentiation ability. This variation may impact its stability and efficacy in therapeutic applications. This review determined the influence of the microenvironment on the proliferation of UCB-HSCs and correlated it with therapeutic applications in recent years, compared with existing research on Wharton’s jelly mesenchymal stromal cells as feeder layers to expand stem cells. 39 This review had some limitations. It does not summarize and analyze the external factors that may affect the expansion of UCB-HSCs in vitro, such as temperature, light, and chemicals, which needs to be verified by further animal research and clinical trials. In summary, while UC-MSCs exhibit potential, their capacity to enhance the proliferation of UCB-HSCs in vitro remains constrained, necessitating further investigation into optimization techniques to address these limitations. These findings enhance our comprehension of in vitro cell expansion and offer prospective therapeutic concepts for clinical applications.
Footnotes
Acknowledgments
I sincerely thank my mentor Li Xu for the selfless help and support provided throughout the process of writing the manuscript. We appreciate the support of the Department of Hematology of the Anhui No.2 Provincial People’s Hospital. We used QuillBot Premium and Grammarly Premium to improve our language quality.
Author contributions
Conceptualization: Zhuo-yang Bian
Data curation: Zhuo-yang Bian
Writing–original draft: Zhuo-yang Bian
Writing–review & editing: Li Xu, Zhuo-yang Bian
Consent for publication
None.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
This article does not contain any experiments involving human participants or animals.
Funding
This work was supported by the Scientific Research Project of the Education Department of Anhui Province (2023AH053386).
