Abstract
Purpose:
Graft-versus-host disease (GVHD) is an important complication after human leukocyte antigen (HLA) haploidentical donor (HID) hematopoietic stem cell transplantation (HSCT), which may lead to poor prognosis. Our study intends to identify the efficacy and safety of mesenchymal stem cells (MSCs) for multidrug-resistant (MDR)-GVHD after HID HSCT.
Methods:
MDR-GVHD was referring to GVHD remaining no response to at least two types of therapy, and hUCB-MSCs were given at the dose of (1.0–2.0) × 106/kg once a week.
Results:
A total of 21 patients were enrolled in this retrospective study (acute GVHD (aGVHD): n = 14, chronic GVHD (cGVHD): n = 7). The median dose of MSCs was 1.2 × 106 cells/kg (range, 0.8–1.8 × 106) cells/kg, and the median numbers of infusion were 2 (range, 1–7) and 3 (range, 2–12) for MDR-aGVHD and MDR-cGVHD patients, respectively. In MDR-aGVHD patients, the overall response rate (ORR) was 57.1%, including 50.0% complete response (CR) and 7.1% partial response (PR), and the median time to response was 49.5 days (range, 16–118) days. The 2-year probability of overall survival after MSCs was 64.3%. Five patients (35.7%) developed infections after MSCs, and no obvious hematologic toxicities were observed. Five MDR-aGVHD patients died after MSCs treatments because of GVHD progression (n = 1), severe infection (bacterial central nervous system infection: n = 1; fungal pneumonia: n = 2), and poor graft function (n = 1). In MDR-cGVHD patients, three patients (42.9%) achieved PR after MSCs and the median time to response was 56 days (22–84) days. The ORRs for moderate and severe cGVHD were 50.0% and 33.3%, respectively. Four MDR-cGVHD patients died after MSCs treatments because of GVHD progression (n = 2), severe fungal pneumonia (n = 1), and relapse (n = 1).
Conclusion:
MSCs treatment may be safe and effective for MDR-GVHD after HID HSCT.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is the most effective treatment for malignant hematologic diseases. 1 Graft-versus-host disease (GVHD) is a major complication and an important cause of non-relapse mortality after allo-HSCT. Although several methods had been used to induce immune tolerance, 2 there are still 40–50% and 30–70% of allo-HSCT recipients suffering from grade II to IV acute GVHD (aGVHD) and chronic GVHD (cGVHD), respectively.3–5
Corticosteroid is the first-line treatment for GVHD, but its response rate can only reach about 50% of patients with severe GVHD.6,7 There is no clear consensus on the best options for second- and third-line treatments of aGVHD and cGVHD yet. 8 In patients with steroid-refractory GVHD (SR-GVHD), 17–72% of patients remain unresponsive despite the availability of various second-line agents.7,9–11 There are few studies on salvage therapy for second-line treatment resistance (i.e. multidrug-resistant (MDR)-GVHD).
Mesenchymal stromal cells (MSCs) are a group of non-hematopoietic progenitor cells derived from the mesoderm of embryonic development with multispectral differentiation potential, which can be isolated from bone marrow, umbilical cord, adipose tissue, and so on. 12 MSCs have anti-inflammatory and tissue repair capabilities. In addition, MSCs can regulate T and B lymphocyte proliferation, activation and maturation, induce regulatory T lymphocyte production, and have regulatory functions on both the intrinsic and adaptive immune responses. Tissue repair and immunomodulatory functions of MSCs are the critical theoretical basis for their use in the treatment of GVHD. 13
There is a large amount of clinical data on MSCs in the treatment of GVHD in patients receiving identical sibling donors (ISDs) or unrelated donors (URDs) HSCT.14–19 With the development of HSCT, the haploidentical donors (HIDs) have become one of the most important alternative donors which could achieve similar clinical outcomes to ISDs and URDs.20–22 However, it is well-known that the incidence and severity of GVHD is higher23–25 and some post-transplant complications, such as viral reactivation and poor graft function (PGF), are more common in HID HSCT compared with those receiving ISD and URD HSCT.26–28 Although the study of Le Blanc et al. 29 included 18 HID HSCT recipients and the study of Cetin et al. 30 included 4 HID HSCT recipients, they did not identify the value of MSCs treatment in the specific population of HID HSCT recipients. Thus far, few studies had identified the efficacy and safety of MSCs treatment for GVHD in HID HSCT recipients, and the efficacy of MSCs in patients receiving HID HSCT remains unclear, particularly for those with MDR-GVHD.
Therefore, in this study, we aimed to identify the efficacy and safety of MSCs for the treatment of MDR-GVHD in HID HSCT recipients.
Method
Patients
Consecutive patients who received MSCs as salvage therapy for MDR-GVHD after HID HSCT 31 at the Peking University Institute of Hematology (PUIH) from 2016 to 2020 were enrolled in this retrospective study. The last follow-up visit was on October 30, 2020. The study protocol was approved by the institutional review board of Peking University People’s Hospital (Beijing, November 2016) and was conducted in accordance with the Declaration of Helsinki. The reporting of this study conformed to the STROBE statement. 32 Informed consent was obtained by all patients or their guardians.
Transplant regimen
For patients with hematologic malignancies, the preconditioning regimen mainly included cytarabine, busulfex, cyclophosphamide, simustine, and rabbit anti-thymocyte globulin (ATG).2,22,24 For patients with severe aplastic anemia, the preconditioning regimen mainly included busulfex, cyclophosphamide, fludarabine, and ATG. 33 We used cyclosporine A (CSA), mycophenolate mofetil (MMF), and methotrexate (MTX) to prevent GVHD. 34 As it was reported by Chen et al. 35 that the co-infusion of an unrelated cord blood unit may potentially improve the engraftment of HID HSCT, two patients with relatively higher donor-specific anti-human leukocyte antigen (HLA) antibodies levels (2000 ⩽ median fluorescent intensity ⩽ 10,000) received umbilical cord blood in their graft in this study. Particularly, for the patients receiving second transplantation for PGF, the mainly conditioning regimen included fludarabine and cyclophosphamide, 36 and GVHD prevention consisted of basixilimab 20 mg on days −1 and +4, plus CSA and MMF (Supplementary Methods). For the patients who had minimal residual disease or experienced relapse after allo-HSCT, modified donor lymphocyte infusion (DLI) was given according to our protocols (Supplementary Methods). 37
The diagnosis and treatment of GVHD
The diagnosis and grading of aGVHD was based on the international consensus criteria.38,39 When aGVHD was identified, corticosteroid and the optimized level of CSA applied first. 40 If the patients with SR-aGVHD, second-line treatments included basiliximab, MMF, tacrolimus, and ruxolitinib. The use of these drugs was mostly based on the competence and experience of each physician.
The diagnosis and grading of cGVHD was based on the consensus criteria of the National Institutes of Health (NIH).41,42 For patients with moderate or severe cGVHD, we first used the corticosteroid and/or CSA. 43 Second-line treatments include mercaptopurine (6-MP), MMF, ruxolitinib, basiliximab, and imatinib. The use of these drugs was also mostly based on the competence and experience of each physician.
Definition of MDR-GVHD
MDR-GVHD was defined as no improvement in GVHD after more than 2 types of treatments, regardless of GVHD prophylaxis. Patients with MDR-GVHD were eligible for MSCs treatment if they met the following criteria: (1) aGVHD progressed within 1 week or did not respond by 2 weeks after the start of second-line therapies or (2) cGVHD progressed within 4 weeks or did not respond by 8 weeks after the start of second-line therapies. 44
MSCs treatment
The human umbilical cord blood MSCs (hUCB-MSCs) were obtained from Beijing Engineering Lab for Cell Therapy (Beijing, China; aGVHD: n = 12; cGVHD: n = 5) and Beijing Cord Blood Bank (Beijing, China; aGVHD: n = 2; cGVHD: n = 2). The dose of MSCs was chosen based on the previous researches.29,45–47 Moreover, the hUCB-MSCs were mainly isolated from Wharton’s Jelly and perivascular tissues in the umbilical cord with informed consent of the mothers. The umbilical cord segments (about 10 cm) were cut longitudinally and minced into 3 cm sections. They were cultured in the serum-free culture system after dissecting into 1–3 mm3 pieces. The serum-free culture system included the UltraGRO™-Advanced (HPCFDCRL50, Helios Bioscience, America), which used platelet lysate. The cells were expanded until they reached subconfluence (80%) with changing the medium every week. The culture medium was poured out and the culture flask was washed with 0.9% normal saline twice, 1.5 mL of trypsin (0.125%) was added into the flask for digestion until the cells became round and suspended then 3 mL of medium was added to stop digestion. The cell suspension was centrifuged and cleaned then replated in the flask in the same culture conditions for passages. Cells were passaged until P5 and then the supernatant was tested for sterility and a small number of cells were taken for phenotypic testing. If cells passed all the tests, they would be conserved for clinical use. 48 It required to meet all the following criteria for clinical use: (1) all hUCB-MSCs were required to be within the 5th generation or less with the cell count of 5 × 107 (100 mL) and were spindle-shaped or fibroblast-like in vitro culture; (2) molecular marker detection of hUCB-MSCs requires CD34+/HLA-DR+/CD45+ ⩽ 2%; and CD44+/CD73+/CD90+/CD105+ ⩾ 95% (Beijing Cord Blood Bank) or CD73+/CD90+/CD105+ ⩾ 95% (Beijing Engineering Lab for Cell Therapy), (3) no viral, fungal, bacterial, or mycoplasma contamination, endotoxin ⩽ 0.5 EU/mL; (4) time from discharge to completion of infusion < 18 h, live cell ratio ⩾ 90% before infusion. Patients with a clinical diagnosis of MDR-GVHD were given hUCB-MSCs at the dose of (1.0–2.0) × 106/kg, once a week for 2–6 weeks.
The response evaluation of MDR-GVHD
Responses were evaluated every day after the first MSCs infusion. The definitions of response to GVHD treatment were based on the consensus criteria.49–51 The overall response rate (ORR) was defined as complete response (CR) plus partial response (PR).
Statistical analysis
Continuous variables were compared using the Mann–Whitney U-test; categorical variables were compared using χ 2 and Fisher’s exact tests. Survival was estimated by the Kaplan–Meier method. Univariable and multivariable Cox regression analysis were used to estimate hazard ratios (HRs) for clinical outcomes. The factors associated with the clinical outcomes with p < 0.1 by univariable analysis were included in the multivariable analysis (Supplementary Methods). The p values were two-sided, and p < 0.05 was considered to be statistically significant. The SPSS 20 (SPSS Inc./IBM, Armonk, NY, USA) and the R software package (version 4.0.0; http://www.r-project.org) were used for data analysis.
Result
Patient characteristics
A total of 21 patients were included in this study (Table 1, Figure 1, Supplementary Table 1 and 2). The median time of aGVHD (n = 14) and cGVHD (n = 7) diagnosis was at 21 days (range, 11–699) days and at 326 days (range, 185–940) days after HSCT, respectively.
Patient characteristics of aGVHD and cGVHD.

aGVHD, acute graft-versus-host disease; cGVHD, chronic graft-versus-host disease; HLA, human leukocyte antigen; HSCT, hematopoietic stem cell transplantation.
Minor ABO mismatched indicated that donor possessed isohemagglutinins against recipient red cells, including the following blood group combinations: O (donor) into A, B, or AB (recipient), and A or B (donor) into AB (recipient). Major ABO mismatched indicated that recipient possessed isohemagglutinins against donor red cells, including the following blood group combinations: A, B, or AB (donor) into O (recipient), and AB (donor) into A or B (recipient). Major–minor mismatched indicated that both donor and recipient possessed isohemagglutinins to each other; A into B and vice versa.

Response. Swimmer plot displaying all patients who received MSCs treatments for MDR-GVHD.
The characteristics of aGVHD before MSCs treatment were shown in Table 2, and the median time from aGVHD occurrence to MSCs treatment was 22.5 days (range, 7–142 days). Two patients and 12 patients received MSCs treatments because of aGVHD progression and resistance to other second-line therapy, respectively. The characteristics of cGVHD before MSCs treatment were shown in Table 3, and the median time from cGVHD occurrence to MSCs treatment was 102 days (range, 19–456 days). Two patients and 5 patients received MSCs treatments because of cGVHD progression and resistance to other second-line therapies, respectively. One and 2 patients experienced aGVHD and cGVHD, respectively, after modified DLI.
Characteristics of aGVHD before MSC treatment.
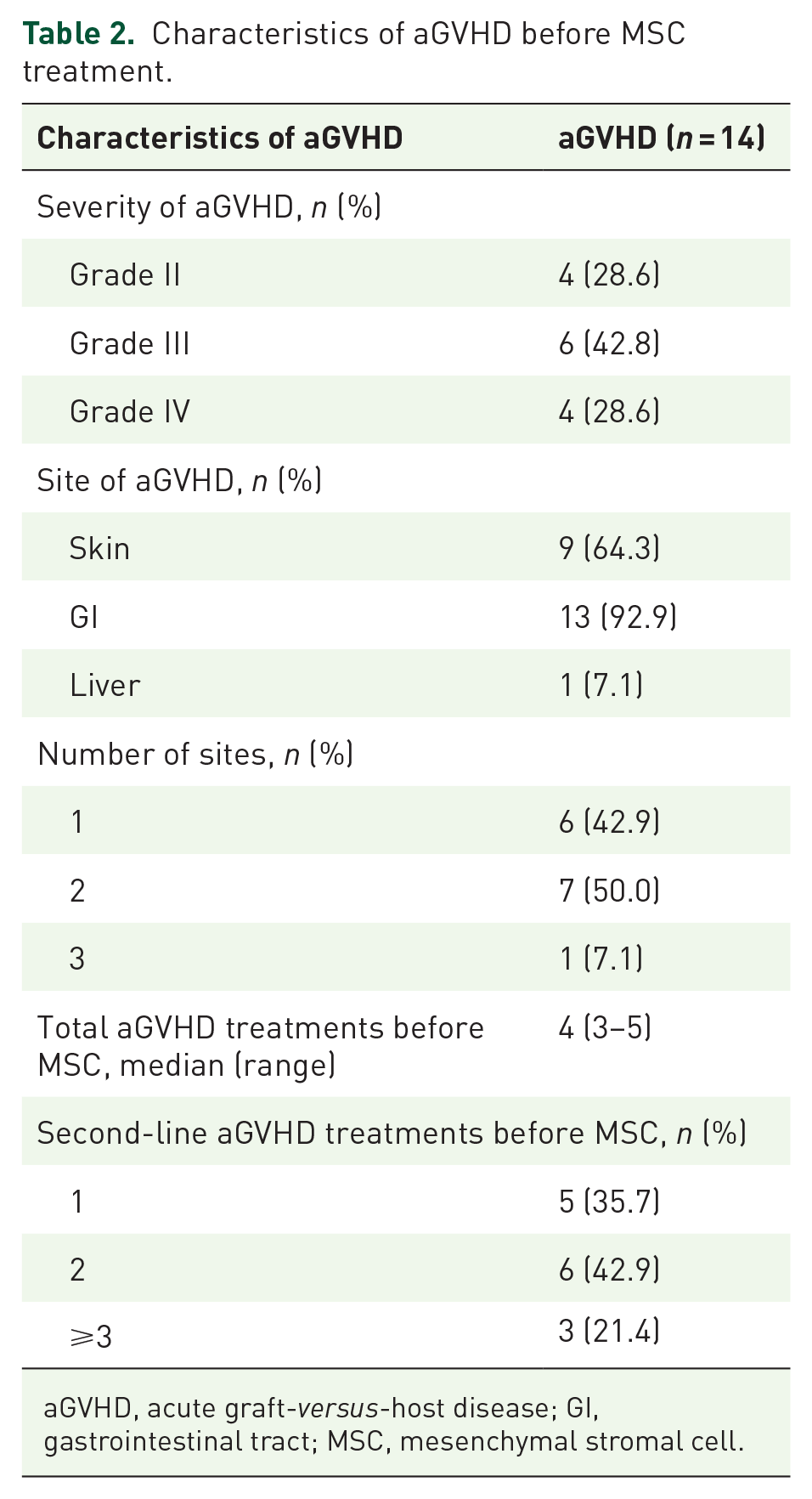
aGVHD, acute graft-versus-host disease; GI, gastrointestinal tract; MSC, mesenchymal stromal cell.
Characteristics of cGVHD before MSC treatment.
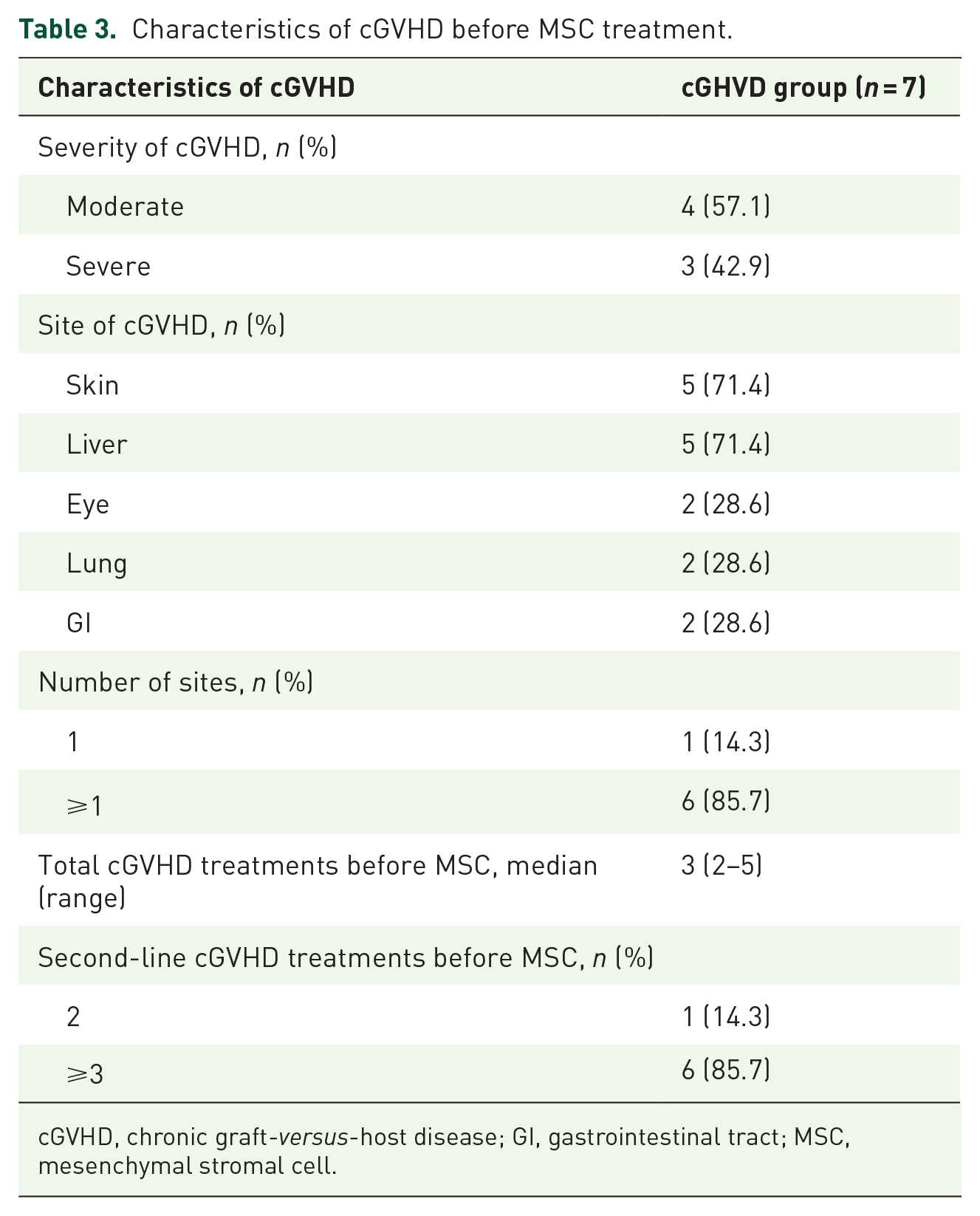
cGVHD, chronic graft-versus-host disease; GI, gastrointestinal tract; MSC, mesenchymal stromal cell.
The median dose of MSCs was 1.2 × 106 cells/kg (range, 0.8–1.8 × 106) cells/kg, and the median numbers of infusion were 2 (range, 1–7) and 3 (range, 2–12) for MDR-aGVHD and MDR-cGVHD patients, respectively.
Response
aGVHD
The ORRs at 28 days and at any time were 21.4% and 57.1%, respectively. The median time of response was 49.5 days (range,16–118) days. The ORRs at any time were 100% (4/4) and 40% (4/10) for grades II and III–IV aGVHD, respectively (Figure 2(a)). The ORRs at any time were 66.7% (6/9), 61.5% (8/13), and 100% (1/1) for skin, gastrointestinal (GI), and liver aGVHD, respectively (Figure 2(b)). The ORRs were 40.0% (2/5) and 66.7% (6/9) for patients with 1 and 2–3 organs involvement, respectively (Figure 2(c)). The ORRs were 80.0% (4/5) and 44.4% (4/9) for patients with 2 and ⩾3 treatments prior or in concomitance to MSCs treatment, respectively (Figure 2(d)). Three patients experienced cGVHD occurrence after MSCs treatment and were all controlled without reuse of MSCs infusion.
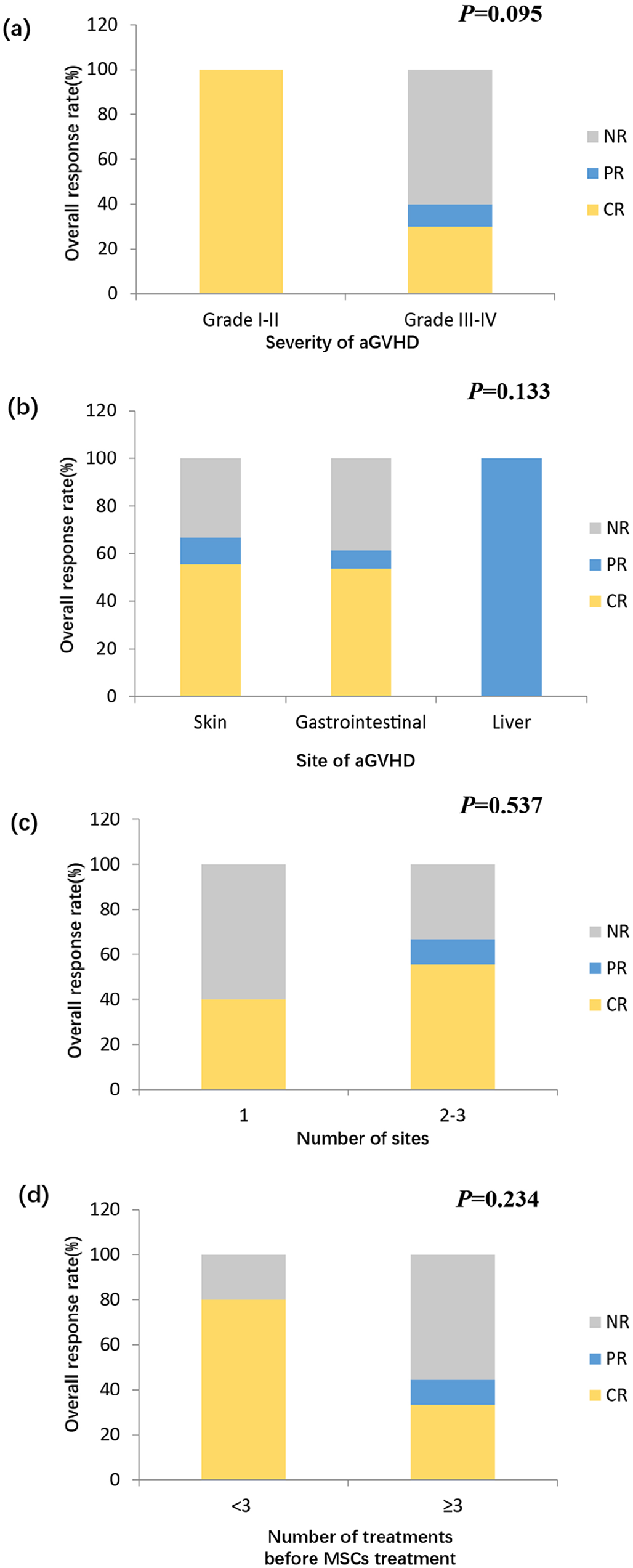
Response rate of MDR-aGVHD patients after MSCs treatment. Patients are grouped by (a) severity of aGVHD; (b) site of aGVHD; (c) number of involved sites; (d) number of treatments before MSCs treatment.
cGVHD
The ORRs at 28 days and at the time of final follow-up were 14.3% (1/7) and 42.9% (3/7), respectively. The median time of response was 56 days (range, 22–84) days. The ORRs of moderate and severe cGVHD were 50.0% (2/4) and 33.3% (1/3), respectively (Figure 3(a)). The ORRs for involved skin, GI, eyes, liver, and lung cGVHD were 40% (2/5), 50% (1/2), 50% (1/2), 40% (2/5), and 0% (0/2), respectively (Figure 3(b)). The ORR of 1–2 involved organs (75%, 3/4) seemed to be higher than that of more than two involved organs (0%, 0/3; Figure 3(c)). The ORRs were 0.0% (0/1) and 50.0% (3/6) for patients with 2 and ⩾3 treatments prior or in concomitance to MSCs treatment, respectively (Figure 3(d)).

Response rate of MDR-cGVHD patients after MSCs treatment. Patients are grouped by (a) severity of cGVHD; (b) site of cGVHD; (c) number of involved sites; and (d) number of treatments before MSCs treatment.
Toxicities
No acute allergic reactions such as fever, rash, and acute laryngeal edema, occurred during or shortly after the infusion of hUCB-MSCs. Neither did infusion-related liver or kidney injury happen. No hematological toxicity was observed after MSCs treatment. No hUCB-MSCs-related tumors were observed. The infections events were showed as follows.
Infections events of aGVHD patients
Infection occurred in 5 (35.7%) patients, three had one type of infection and two had two types of infections (cytomegaloviremia plus central bacterial infection: 1; cytomegaloviremia plus Epstein-Barr viremia: 1). The incidence of cytomegaloviremia was 28.6% (4/14) with a median time of 32.5 days (range, 14–105) days after MSCs treatment. None of them experienced cytomegalovirus (CMV) disease. The rates of Epstein-Barr viremia, bacterial sepsis, and bacterial central infection after MSCs treatment were 7.1% (1/14), 7.1% (1/14), and 7.1% (1/14), respectively.
Infections events of cGVHD patients
One case (14.3%) of bacterial sepsis and bacterial pneumonia occurred at 16 days after MSCs treatment, and one case (14.3%) experienced fungal pneumonia at 49 days after MSCs treatment.
Long-term outcomes
Survival of aGVHD patients
Causes of death after MSCs treatment included GVHD progression (n = 1), severe infection (bacterial central nervous system infection: n = 1; fungal pneumonia: n = 2), and PGF (n = 1). The median follow-up time after HSCT and MSCs treatment was 598 days (range, 93–1270) days and 345.5 days (range, 25–1162) days, respectively. The 2-year probability of OS after MSCs treatment was 64.3%, which was 87.5% and 33.3%, respectively for ORR and non-ORR groups (p = 0.031; Figure 4(a) and (b)).

Overall survival of MDR-aGVHD and MDR-cGVHD patients after MSCs treatment. (a) the OS of MDR-aGVHD patients; (b) the OS of the ORR and non-ORR groups of MDR-aGVHD patients; (c) the OS of MDR-cGVHD patients; (d) the correlation analysis of the total MSCs dose and OS.
Survival of cGVHD patients
The median follow-up time after HSCT and MSCs treatment was 501 days (range, 302–2584) days and 114 days (range, 15–1704) days, respectively. The 2-year probability of OS was 42.9% (Figure 4(c)), and causes of death after MSCs treatment included GVHD progression (n = 2), severe fungal pneumonia (n = 1), and relapse (n = 1).
The correlation analysis for the total dose of MSCs and OS
The correlation analysis of the total dose of MSCs treatment and OS was conducted, and we observed that the total dose of MSCs was positively correlated with OS (r = 0.69, p = 0.0006, Figure 4(d)).
Univariable and multivariable analysis for ORR and OS
None of variables was significantly associated with ORR in the univariable analysis (data not shown). In the univariable analysis, the donor/recipient gender matched and GVHD severity were associated with poorer OS (Supplementary Table 3). However, none of variables was significantly associated with OS in multivariable analysis.
Discussion
According to our results, the ORRs at any time were 57.1% and 42.9%, respectively, for MDR-aGVHD and MDR-cGVHD patients after HID HSCT. This was the first study to identify the efficacy and safety of MSCs treatment in MDR-GVHD patients after HID HSCT.
In the studies enrolled patients most of who received ISD or URD HSCT, the meta-analysis of Hashmi et al. 52 showed that the ORR of MSCs treatment for SR-aGVHD was 72% (241/336), and the CR rate was 28% (25/336). Another meta-analysis included 13 studies with 301 patients showed that the ORR of SR-aGVHD patients receiving MSCs treatment was 68.1%. 53 In this study, the ORR at any time of MSCs treatment was 57.1% for MDR-aGVHD, which was similar to the previous studies.
Weng et al. 54 used MSCs to treat 19 ISD HSCT recipients with SR-cGVHD. Four and 10 patients achieved CR and PR, respectively, and the ORR was 73.3%. Similarly, Introna et al. 55 used MSCs to treat 40 ISD and URD HSCT recipients with SR-cGVHD and the ORR and CR rate was 67.5% and 27.5%, respectively. The meta-analysis of MSCs treatment for SR-cGVHD patients showed that the ORR and CR rate was 67% and 47%, respectively. 56 In this study, seven cGVHD patients received MSCs treatment and the ORRs at any time was 42.9% (3/7). The ORR was relatively low, which suggested that the MDR-cGVHD after HID HSCT might be more difficult to control than that of patients receiving ISD or URD HSCT.23–25
Infection events were common in other second-line treatments for SR-GVHD, for example, the infection rates of patients receiving basiliximab, ATG, MMF, infliximab, rituximab, and alemtuzumab were 59.6–74%, 67%, 23–67%, 81%, 15–66%, and 77.8–100%, respectively.9–11,57–64 In our study, we also observed that infection was the most common toxicity after MSCs treatment. Among 14 patients with MDR-aGVHD, 5 (35.7%) developed infections and the incidence of cytomegaloviremia was 28.6%. Among seven patients with MDR-cGVHD, two (28.6%) developed bacterial infections. In addition, we observed patients died of severe fungal infection (n = 3) or bacterial infection (n = 1) after MSCs treatments. Nevertheless, there was a high background rate of infectious events in SR-GVHD patients, 65 and infection events might not be attributed solely to MSCs treatment. Thus, the risk of infection of HID HSCT recipients receiving MSCs treatments was acceptable.
Compared with hUCB-MSCs, MSCs isolated from bone marrow are used more commonly.66,67 The ORR and OS of BM-MSCs treatment for SR-aGVHD patients were 60–70.9% and 62–80%, respectively.29,68–71 The ORR and OS of BM-MSCs treatment for SR-cGVHD patients were 87–100% and 78.3–100%, respectively.72,73 In this study, the ORR and OS of hUCB-MSCs treatment for MDR-aGVHD patients were 57% and 64.3%, respectively. The ORR and OS of hUCB-MSCs treatment for MDR-cGVHD patients were 42.9% and 42.1%, respectively. Thus, the results between hUCB-MSCs and BM-MSCs seemed to be similar in aGVHD patients; however, our results of hUCB-MSCs treatment seemed to be worse than BM-MSCs treatment in cGVHD patients. Considering that few patients with SR-cGVHD following HID HSCT were enrolled in the trials using BM-MSCs, the efficacy between hUCB-MSCs and BM-MSCs should be further identified in these patients.
Our study has several limitations. First, this was a retrospective study with a small sample size, which might affect the accuracy of our results. Second, the number of particularly involved organs (e.g. lung cGVHD) was small, and the efficacy of MSCs treatment on these organs needs to be further investigated. Third, our study used MSCs as an add-on immunosuppressive treatment for MDR-GVHD patients; however, most of the studies identifying MSCs treatment in GVHD used MSCs in concomitance to other second-line treatments.29,68–73 The protocols for SR-GVHD treatments prior to MSCs treatment were not uniform, but we observed that the former treatments seemed not to influence the ORR of MSCs treatment. Fourth, although ruxolitinib had been proved to be significantly better than best available therapy in SR-aGVHD and SR-cGVHD, only 13 patients receiving MSCs treatment were enrolled in REACH2 trial, 74 and MSCs were not included in the control group in REACH3 trial. 75 Thus, we could further compare the efficacy and safety between ruxolitinib and MSCs treatment in patients with MDR-GVHD in future. Finally, the median follow-up time after MSCs treatment was relatively short, and its long-term efficacy in HID HSCT needed to be further determined.
Conclusion
In conclusion, MSC may be a safe and effective therapy for MDR-GVHD patients after HID HSCT. It still needs to be confirmed by prospective large-scale registry trials in future.
Supplemental Material
sj-docx-1-tah-10.1177_20406207211072838 – Supplemental material for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation
Supplemental material, sj-docx-1-tah-10.1177_20406207211072838 for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation by Meng-Zhu Shen, Xin-Xin Liu, Zhi-Yuan Qiu, Lan-Ping Xu, Xiao-Hui Zhang, Yu Wang, Chen-Hua Yan, Huan Chen, Yu-Hong Chen, Wei Han, Feng-Rong Wang, Jing-Zhi Wang, Si-Ning Liu, Kai-Yan Liu, Xiao-Jun Huang and Xiao-Dong Mo in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207211072838 – Supplemental material for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation
Supplemental material, sj-docx-2-tah-10.1177_20406207211072838 for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation by Meng-Zhu Shen, Xin-Xin Liu, Zhi-Yuan Qiu, Lan-Ping Xu, Xiao-Hui Zhang, Yu Wang, Chen-Hua Yan, Huan Chen, Yu-Hong Chen, Wei Han, Feng-Rong Wang, Jing-Zhi Wang, Si-Ning Liu, Kai-Yan Liu, Xiao-Jun Huang and Xiao-Dong Mo in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-3-tah-10.1177_20406207211072838 – Supplemental material for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation
Supplemental material, sj-docx-3-tah-10.1177_20406207211072838 for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation by Meng-Zhu Shen, Xin-Xin Liu, Zhi-Yuan Qiu, Lan-Ping Xu, Xiao-Hui Zhang, Yu Wang, Chen-Hua Yan, Huan Chen, Yu-Hong Chen, Wei Han, Feng-Rong Wang, Jing-Zhi Wang, Si-Ning Liu, Kai-Yan Liu, Xiao-Jun Huang and Xiao-Dong Mo in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-4-tah-10.1177_20406207211072838 – Supplemental material for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation
Supplemental material, sj-docx-4-tah-10.1177_20406207211072838 for Efficacy and safety of mesenchymal stem cells treatment for multidrug-resistant graft-versus-host disease after haploidentical allogeneic hematopoietic stem cell transplantation by Meng-Zhu Shen, Xin-Xin Liu, Zhi-Yuan Qiu, Lan-Ping Xu, Xiao-Hui Zhang, Yu Wang, Chen-Hua Yan, Huan Chen, Yu-Hong Chen, Wei Han, Feng-Rong Wang, Jing-Zhi Wang, Si-Ning Liu, Kai-Yan Liu, Xiao-Jun Huang and Xiao-Dong Mo in Therapeutic Advances in Hematology
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Program of the National Natural Science Foundation of China (grant no. 82170208), the Foundation for Innovative Research Groups of the National Natural Science Foundation of China (grant no. 81621001), the CAMS Innovation Fund for Medical Sciences (CIFMS) (grant no. 2019-I2M-5-034), the Key Program of the National Natural Science Foundation of China (grant no.81930004), and the Fundamental Research Funds for the Central Universities.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
