Abstract
Haploidentical hematopoietic stem cell transplantation (haplo-HSCT) produces similar survival outcomes as HLA-matched sibling donor allogeneic HCST in younger patients with acquired severe aplastic anemia (SAA). This study reported a 29-years-old man with SAA and intracranial hemorrhage who underwent haplo-HSCT with a modified BU/CY + ATG conditioning regimen. Neutrophil and platelet engraftment were both achieved on day 14 after HSCT. The patient developed grade IV acute graft-versus-host disease (aGVHD) on day 20 and acquired cytomegalovirus (CMV) and Epstein–Barr virus (EBV) infections on day 47. After the failure of methylprednisolone, basiliximab, ruxolitinib, and antiviral treatment, the patient was diagnosed with steroid-resistant grade IV aGVHD and refractory CMV and EBV infections. We performed fecal microbiota transplantation and infused CMV- and EBV-specific cytotoxic T lymphocytes. After that the stool volume and frequency gradually decreased, and viral DNA was undetectable on day 80. This report provides helpful clinical experience for treating steroid-resistant aGVHD and refractory viral infections.
Keywords
Background
Acquired severe aplastic anemia (SAA) is a life-threatening disorder, and allogeneic hematopoietic stem cell transplantation (allo-HSCT) is an available curative treatment. 1 Allo-HSCT using cells from an HLA-matched sibling donor (MSD) is the standard frontline therapy for patients younger than 50 years. 2 Recently, related haploidentical HSCT (haplo-HSCT) has been reported to produce similar survival rates as MSD allo-HSCT in younger patients, making it a possible alternative for patients with SAA lacking an MSD. 3 However, delayed immune recovery, a high incidence of infections, refractory graft-versus-host disease (GVHD), and transplant-related mortality are major drawbacks to the universal clinical use of haplo-HSCT in patients with SAA. In this study, we present the case of a man with SAA and intracranial hemorrhage who lacked an MSD. The patient was successfully treated by fecal microbiota transplantation (FMT) and the infusion of cytotoxic T lymphocyte (CTL) specific for cytomegalovirus (CMV) and Epstein–Barr virus (EBV) after experiencing steroid-resistant intestinal acute GVHD (aGVHD) and refractory CMV and EBV infections complicated by haplo-HSCT.
Case presentation
A 29-years-old man was admitted to our center because of fatigue for half a month as well as black stool, bleeding gums, and mild headache for 1 day. The physical examination indicated sporadic petechiae throughout the skin, and no enlargement of the superficial lymph nodes, the liver, or the spleen. He presented with pancytopenia (white blood cell [WBC] count, 1.43 × 109/L; neutrophil count, 0.13 × 109/L; hemoglobin [Hb] level, 49 g/L; platelet [PLT] count, 1 × 109/L), hematopoietic elements were greatly reduced, and the marrow space was replaced with adipose tissue. Computed tomography of the brain revealed acute hemorrhage in the right cerebellum, basal ganglia, and semioval center. After excluding congenital aplastic anemia, he was diagnosed with acquired SAA and intracranial hemorrhage.
Because of the lack of an MSD, the patient underwent haplo-HSCT with a modified BU/CY + ATG conditioning regimen as described by the consensus statement from the Chinese Society of Hematology. 2 Hematopoietic stem cell sources included granulocyte colony-stimulating factor-primed bone marrow and peripheral blood samples from his father (mononuclear cells, 13.07 × 108/kg; CD34+ cells, 4.44 × 106/kg), and cyclosporine A, mycophenolate, and methotrexate were used for GVHD prophylaxis. 4 Neutrophil and platelet engraftment were both achieved on day 14. On day 20, the patient presented with fever, a maculopapular rash covering 60% of his body without bullae or desquamation, and increased total serum bilirubin levels (3.4 mg/dL) accompanied by nausea, severe abdominal pain, and diarrhea (>2000 mL/day), and he was diagnosed with grade IV aGVHD, skin stage 3, liver stage 2, GI stage 4. After using methylprednisolone (2 mg/kg, 80 mg/day × 7 days; 1.5 mg/kg, 60 mg/day × 7 days) as the first-line anti-GVHD drug, his symptoms including fever, skin rash, abdominal pain, and bilirubin elevation were improved, but the stool volume and frequency were unchanged. We further eliminated other etiologies causing diarrhea such as infection, colitis, intestinal flora disturbance, and intestinal thrombotic microangiopathy. Therefore, he was diagnosed with steroid-resistant aGVHD. Basiliximab (20 mg on days 38, 46, and 53) and ruxolitinib (10 mg, twice a day) were administered as the second-line regimen for 2 weeks, but his diarrhea did not improve. Furthermore, CMV DNA (9.95 × 103 IU/mL in peripheral blood and 1.15 × 104 IU/mL in pharyngeal swab; less than 2.0 × 102 IU/mL was defined as normal) and EBV DNA (4.51 × 107 IU/mL in pharyngeal swab; less than 2.0 × 102 IU/mL was defined as normal) were detected on day 47. After treatment with ganciclovir and foscarnet sodium, this patient experienced severe granulocytopenia and thrombocytopenia (WBC, 1.28 × 109/L; PLT, 38 × 109/L), but CMV DNA (4.67 × 103 IU/mL, peripheral blood) and EBV DNA (1.52 × 105 IU/mL, pharyngeal swab) were still detected on day 54. After reducing the immunosuppressive drug dose, we performed FMT on day 54 after stopping antibiotics 24 hours before transplantation and a 6-hour fast. Frozen fecal microbiota was obtained from a healthy young volunteer who passed the screening for transmissible diseases. The fecal bacteria liquid was delivered (120 mL, three times, every other day every other day for three times) via a nasogastric tube. After two rounds of fecal bacteria solution infusions, the number of daily bowel movements was reduced from six (1800 mL) to two (550 mL), followed by a gradual decline to one per day. At the same time, we obtained CMV- and EBV-specific CTL (day 62, 173.42 × 107; day 80, 194.14 × 107) from his HSC donor, and CMV and EBV DNA became undetectable on day 80. Adverse events including fever, chills, and gastrointestinal discomfort were not observed during FMT, and GVHD did not recur after the CTL infusions. On day 95, the patient was discharged after blood cell count recovery was confirmed (WBC, 4.57 × 109/L; Hb, 71 g/L; PLT, 95 × 109/L), and his cerebral hemorrhage was completely absorbed. To date, the patient has remained alive without failure for 43 months. The treatment course is presented in Figures 1 and 2.
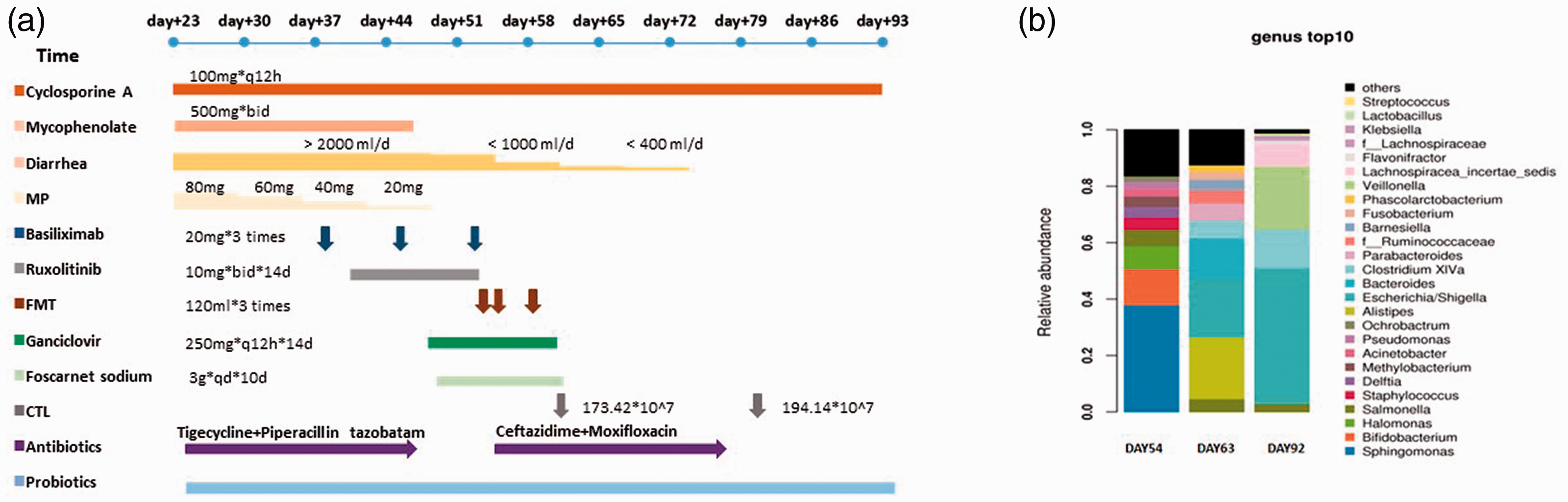
(a) Total clinical course of our patient. (b) Dominant microbes were Sphingomonas on day 54; Bacteroides, Alistipes, and Escherichia on day 63; and Escherichia, Veillonella, and Clostridium on day 92 before and after FMT.

(a) Dynamic changes of CMV DNA copy numbers in peripheral blood and pharyngeal swab samples. (b) Dynamic changes of EBV DNA copy numbers in pharyngeal swab samples. (c) Changes of body weight in this patient before and after fecal microbiota transplantation.
Written informed consent was obtained from the patient for all treatments and the publication of this report.
Discussion
The association between intestinal bacterial imbalance and the severity of GVHD has attracted increasing attention. Conditioning-induced mucosal barrier injury, T-cell migration, and systemic inflammation could cause the development of aGVHD and alter the microbiota homeostasis after allo-HSCT. 5 In a retrospective analysis, a low fecal flora diversity was associated with an increased mortality rate (52%) compared with a high diversity (8%). 6 Other studies also revealed that some flora such as Firmicutes, Proteobacteria, and Escherichia coli appeared to increase GVHD, whereas Lactobacillus reduces the risk of acute GVHD. 7 One group reported that four patients with refractory GVHD who underwent FMT could be weaned off steroids, and the side effects were controllable. 8 The findings of our case were consistent with these reports, as diarrhea symptoms gradually improved after two rounds of FMT and steroid therapy was successfully discontinued. The final dominant genera were Veillonella, Alistipes, Escherichia, Clostridium, and Bacteroides instead of the previously dominant genus Sphingomonas (Figure 1b). Moreover, no severe adverse event was observed during or after FMT.
The use of ATG and anti-GVHD treatment might delay the recovery of specific T-cell immunity and lead to CMV and EBV infection, which is one of the most fatal complications after allo-HSCT. The adoptive transfer of CMV- and EBV-specific T-cells could reconstitute virus-specific T-cell immunity. In this case, the patient exhibited insufficient control of CMV and EBV reactivation despite anti-viral treatment including ganciclovir and foscarnet sodium, whereas the viral burden was significantly alleviated after CMV- and EBV-specific T cell infusion, indicating that CMV- and EBV-specific CTL could be a suitable choice for refractory CMV and EBV infections.
Does FMT have an effect on CMV infection? No research on this subject has been reported. Emerging evidence suggests that the resident gastrointestinal microbiota plays an influential role in shaping anti-viral defenses and modulating the outcome of viral infections. 9 One study indicated that early Staphylococcus aureus colonization decreased the time to CMV acquisition. 10 Increased colonization by butyrate-producing microbiota might be correlated with recurrent CMV reactivation and viral persistence in non-SPF animals. Concurrently, butyrate plays an important role in host physiology by inducing anti-inflammatory responses, thereby controlling excessive viral replication. 9 Whereas butyrate can reactivate latent viral expression, butyrate-mediated anti-inflammatory effects may drive the balance between pro- and anti-inflammatory mechanisms during subclinical viral infections. As described previously, the microbiota can limit viral infection, promote viral infection, or have no effect. The microbiota might also exert direct or indirect effects on viral infection. For example, the microbiota (or microbial products) can directly interact with viral particles to alter infectivity or responses. Conversely, the microbiota may indirectly affect viral infection by inducing immune priming. 11 In fact, the microbiota promotes immune homeostasis by driving both pro- and anti-inflammatory T-cell responses. In addition, commensal bacteria contribute to the production of IgA and anti-microbial peptides. 12 It is possible and perhaps likely that the microbiota influences each virus through multiple mechanisms.
In summary, this case report provides helpful clinical experience concerning the application of FMT and CMV- and EBV-specific CTL infusion. However, the efficacy and safety of this strategy require further evaluations in larger cohorts for confirmation.
Footnotes
Acknowledgement
We thank the patient for providing informed consent for publication of this report and the related images.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable requests.
Ethics approval and consent to participate
The study was reviewed and approved by the First Affiliated Hospital of Nanchang University. Written informed consent was obtained from the patient for all treatments.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and the accompanying images.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was financially supported by grants from the National Natural Science Foundation of China (81960043), the Natural Science Foundation of Jiangxi province (20192ACB20030), and the Traditional Chinese Medicine Research Project of Jiangxi province (2019A159).
