Abstract
Background:
Myelodysplastic syndrome (MDS) is associated with persistent immune activation. High mobility group box-1 (HMGB1) is a ubiquitous, functionally diverse, non-histone intranuclear protein. During acute and chronic inflammatory states, HMGB1 is actively released by inflammatory cells, further amplifying the inflammatory response. A role in MDS and other hypoplastic bone marrow (BM) disorders is incompletely understood.
Objectives:
The objective of the study is to evaluate whether circulating HMGB1 is elevated in patients with MDS and other BM failure syndromes [namely, aplastic anemia (AA) and paroxysmal nocturnal hemoglobinuria (PNH)].
Design:
This is a observational, cross-sectional, single-center, exploratory study.
Methods:
We evaluated circulating concentrations of HMGB1, interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α in patients with MDS and age-matched hematologically healthy controls as well as patients with AA and PNH.
Results:
We included 66 patients with MDS and 65 age-matched controls as well as 44 patients with other BM failures (AA = 27, PNH = 17). Circulating levels of HMGB1 were higher in patients with MDS [median, 4.9 ng/ml; interquartile range (IQR): 2.3–8.1] than in AA (median, 2.6 ng/ml; IQR: 1.7–3.7), PNH (median, 1.7 ng/ml; IQR: 0.9–2.5), and age-matched healthy individuals (median, 1.9 ng/ml; IQR: 0.9–2.5) (p = 0.0001). We observed higher concentrations of HMGB1 in the very low/low-risk MDS patients than in the intermediate/high/very high-risk ones (p = 0.046). Finally, in comparison with patients with AA, those with hypocellular MDS (h-MDS) had significantly higher levels of circulating HMGB1 (n = 14; median concentration, 5.6 ng/ml, IQR: 2.8–7.3; p = 0.006). We determined a circulating HMGB1 value of 4.095 ng/ml as a diagnostic cutoff differentiator between h-MDS and AA.
Conclusion:
These observations indicate that circulating HMGB1 is increased in patients with MDS. HMGB1 (but not IL-1β or TNF-α) differentiated between MDS and other BM failures, suggesting that HMGB1 may be mechanistically involved in MDS and a druggable target to decrease inflammation in MDS.
Introduction
Myelodysplastic syndrome (MDS) is a heterogeneous group of hematopoietic neoplastic illnesses characterized by bone marrow failure, myeloid dysplasia, ineffective hematopoiesis, and an increased risk of acute myeloid leukemia (AML). Among their heterogeneous features, MDS progression and survival rate differ according to risk stratification: low-risk patients progress slowly and usually die of cardiovascular disease or complications from bone marrow failure; in contrast, high-risk patients are likelier to progress to AML. The annual incidence of MDS is 3–4/100,000 persons, increasing with age. In people older than 60 years of age, MDS has an annual incidence of 7–35/100,000.1,2
Genetic factors are critical for the development of MDS, including mutations in genes related to epigenetic regulation of DNA, chromatin homeostasis, RNA splicing, signal transduction, and DNA repair; 3 mutations are found in 78–89% of MDS patients.4,5 In parallel, immune dysfunction is critical in the origin and progression of MDS; overexpression of immunity-related genes has been reported in 50% of patients with MDS. 6 Mutations or overexpression of Toll-like receptors (TLRs), including TLR1, TLR2, TLR4, and TLR6, 7 have been described in the progenitor cell of MDS. Ineffective hematopoiesis in MDS is amplified by cytokines – including interferon (INF)-γ, tumor necrosis factor (TNF)-α, Fas ligand (FasL), transforming growth factor (TGF)-β, interleukin (IL)-1β, and nitric oxide synthase released by stromal cells, macrophages, and CD34+ cells.
HMGB1 is a nuclear protein involved in chromatin folding, transcription, and signaling; in inflammatory states, HMGB1 can be passively shed by necrotic cells or actively released by mononuclear cells, further amplifying inflammation. Aberrant inflammatory signaling induces apoptosis, NLRP-3 inflammasome activation, and pyroptosis of bone marrow (BM) progenitors.8,9 In experimental sepsis, HMGB1 is necessary and sufficient to induce inflammatory anemia, suggesting that HMGB1 may interfere with steady-state hematopoiesis in other inflammatory states, including MDS. 10 Untangling the effect of inflammatory mediators in the pathogenesis of MDS may result in novel therapeutic strategies for an, as yet, incurable illness.
The role of HMGB1 in the pathobiology of MDS is incompletely understood. In the BM of patients with MDS, a threefold increase in the levels of HMGB1 has been observed; in this niche, HMGB1 contributes to the failure of macrophage clearance, leading to apoptosis, inflammatory cytokine release, and BM inflammation. 11 HMGB1 is a key driver of inflammatory anemia in experimental sepsis, suggesting that in MDS it may also interfere with erythropoiesis. CD34+ cells derived from MDS patients overexpress HMGB1; sivelestat, an inhibitor for both HMGB1 and neutrophil elastase, in combination with azacitidine, reduces in vitro the expansion of aberrant MDS (but not healthy) colony-forming units (CFU). Inhibition of HMGB1 also reduces the in vitro expression of TLR2, TLR4, TLR6, and TLR9 as well as NF-κB. 12 We hypothesized that HMGB1 is involved in the inflammatory response leading to – or perpetuating – the inflammatory milieu required for MDS. We also wondered whether circulating HMGB1 would differ between MDS and other non-inflammatory hypoplastic BM conditions.
Our main objective was to evaluate whether circulating HMGB1 is elevated in patients with MDS, and if so, whether it can help differentiate MDS from two BM failure syndromes: aplastic anemia (AA) and paroxysmal nocturnal hemoglobinuria (PNH).
Methods
This is an observational, cross-sectional, single-center, exploratory study conducted at Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán, in Mexico City, Mexico. We included adult (⩾18 years) patients with the diagnosis of MDS according to the World Health Organization (WHO) 2016 criteria as well as patients with AA or PNH as bone marrow failure comparators. As controls, we included hematologically healthy volunteers adjusted to the cases by sex and age (Table 1). Among cases and controls, we excluded those with medical conditions that increase HMGB1, including epilepsy, acute or chronic infection, autoimmune disorders, congestive heart failure, acute myocardial infarction, stage 5 chronic renal failure, acute liver disease, and active cancer. The study was approved by the Institutional Research and Ethics Boards (protocol identifier: NER-2514-18-19-1). We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
Baseline characteristics of patients with MDS and hematologically healthy controls.

IPSS-R, 2012 Revised International Prognostic Scoring System; IQR, interquartile range; LRIPSS, Lower International Prognostic Scoring System; MDS, myelodysplastic syndrome; MDS-EB, MDS with excess blasts; MDS-5q, MDS with 5q deletion; MDS-MLD, MDS with multilineage dysplasia; MDS-RS, MDS with ring sideroblasts; MDS-SLD, MDS with single lineage dysplasia; WHO, World Health Organization.
We calculated a sample size of 64 participants per group based on an estimated institutional population of patients with MDS of ~500 and estimated a confidence interval of 95% and a margin of error of 5%. However, as an exploratory study with no previous data on circulating HMGB1 in patients with MDS, we were open to recalculating the sample size according to preliminary results. After signing informed consent, peripheral blood was obtained using EDTA-coated tubes; contents were centrifuged to separate plasma, and samples were stored at –80°C until analysis. Concentrations of plasma cytokines (IL-6, IL-1β, TNF-α) were analyzed in duplicates using multiplex kits (Meso Scale Diagnostics, Rockville, MD, USA), while HMGB1 was measured by ELISA (Shino-Test, Tokyo, Japan) according to the manufacturer’s instructions.14,15
Statistical analysis
Qualitative and nominal variables were described in terms of proportions; quantitative variables were described by median and quartiles. We used Mann–Whitney or Kruskal–Wallis tests to evaluate differences in the concentrations of HMGB1 and other cytokines between patients with MDS, AA, and PNH and controls. Spearman correlation test was used to evaluate correlations between variables. For post hoc data analysis, we used the Scheffé method. We considered a value of p ⩽ 0.05 as significant. For data analysis, we used STATA version 14 (StataCorp, College Station, TX, USA).
Results
We included 66 patients with MDS, 27 with AA,17 with PNH, and 65 participants without hematological pathology as controls. Baseline clinical and laboratory characteristics of MDS subjects, including 2016 WHO classification, IPSS-R stratification, BM cellularity, and MDS-specific treatment, are represented in Table 1; baseline characteristics of control subjects, including past medical history, are represented also in Table 1.
Circulating HMGB1 was significantly higher in patients with MDS than in those with AA (p = 0.002), PNH (p < 0.001), and controls (p < 0.001) using Scheffé’s method (Figure 1; Table 2). Circulating levels of IL-1β were also different between the MDS and control groups. There was a positive correlation between HMGB1 and IL-1β (r = 0.43, p = 0.0002). Levels of IL-1β, however, were not different between MDS and either AA or PNH, suggesting that IL-1β has little relevance as a biomarker between BM failure syndromes (Table 2).
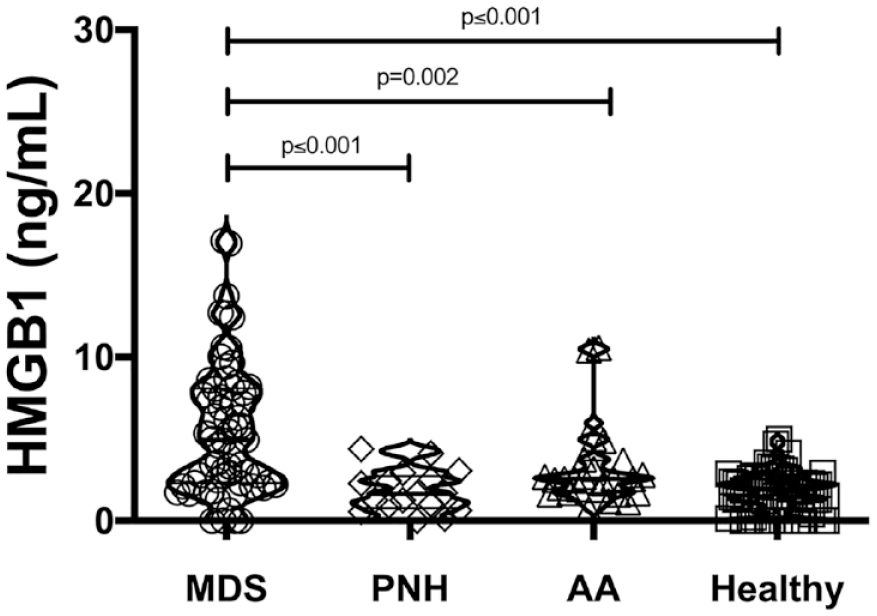
Circulating concentrations of HMGB1 are increased in MDS, but not in other bone marrow failure syndromes. HMGB1 was determined in plasma from patients with myelodysplastic syndrome (MDS, n = 66), paroxysmal nocturnal hemoglobinuria (PNH, n = 17), aplastic anemia (AA, n = 27), as well as controls (n = 64). Statistical differences, shown as p-value, were determined using the Kruskal–Wallis test with post hoc analysis using the Scheffé method.
Concentrations of HMGB1 and other inflammatory mediators in bone marrow failure syndromes.

All concentrations are expressed as median (IQR). Kruskal–Wallis (one-way ANOVA on ranks), p < 0.05.
AA, aplastic anemia; ANOVA, analysis of variance; HMGB1, high mobility group box–1; IL, interleukin; IQR, interquartile range; MDS, myelodysplastic syndrome; PNH, paroxysmal nocturnal hemoglobinuria; TNF, tumor necrosis factor.
Hypoplastic MDS (h-MDS) is a subset of MDS that is sometimes difficult to differentiate from AA on clinical grounds and is associated with more frequent karyotype abnormalities, including trisomy 8 and 1q, and 20q deletion. 16 There is no circulating biomarker that reliably differentiates between h-MDS and AA. We, therefore, evaluated plasmatic concentrations of HMGB1 in patients with h-MDS (n = 14) versus AA (n = 27), observing higher concentrations in h-MDS (5.6 ng/ml, IQR: 2.8–7.3 versus 2.6 ng/ml, IQR: 1.7–3.7, respectively; p = 0.006) (Figure 2), suggesting that circulating concentrations of HMGB1 can help in the differential diagnosis between those two conditions. We then evaluated the ability of HMGB1 to classify between h-MDS and AA. For that, we plotted a receiver operating characteristic (ROC) curve and used the Youden index 17 to determine a cutoff value of HMGB1, determining that a value of 4.095 ng/ml [area under the curve, 0.752; 95% confidence interval (CI), 0.58–0.91; p = 0.0016] has a sensitivity of 84% and specificity of 64%, with a positive predictive value of 81% and a negative predictive value of 69%.

Circulating HMGB1 concentrations differ between hypocellular MDS and AA. HMGB1 was determined in plasma from patients with hypocellular myelodysplastic syndrome (MDS, n = 14) or aplastic anemia (AA, n = 27). Statistical differences, shown as p value, were determined using the Mann–Whitney test.
Circulating levels of IL-1β were higher in those with h-MDS than in AA (0.4 pg/ml, IQR: 0.3–0.5 versus 0.2 pg/ml, IQR: 0.2–0.3, respectively; p = 0.036).
IL-6 and TNF-α are inflammatory cytokines closely related to sepsis severity, but with no known role in MDS. Circulating levels of IL-6 were similar among the different BM syndromes evaluated. Circulating TNF-α was significantly higher among controls in comparison with all BM failures, but we did not observe differences between the BM failure syndromes (Table 2).
Finally, we evaluated whether circulating concentrations of HMGB1 correlate with MDS risk stratification. Indeed, very low and low-risk patients had higher plasma concentrations of HMGB1 than those in the intermediate, high, and very high-risk groups (Table 3); moreover, MDS patients who had not been treated had higher HMGB1 concentrations than those already treated (p = 0.044). We did not observe differences in circulating HMGB1 when comparing treated versus untreated according to IPSS-R stratification, either those in the low-risk (7.14, IQR: 4.60–8.67 versus 5.33, IQR: 2.35–8.12, respectively; p = 0.311) or those in the high-risk strata (3.44, IQR: 2.30–9.66 versus 2.77, IQR: 1.89–3.82, respectively; p = 0.130). We observed the same when analyzing circulating IL-1β among those who had been treated versus those who had not (p = 0.135). We then evaluated circulating HMGB1 in the h-MDS subset according to treatment (six treated versus eight untreated); we did not observe differences (p = 0.196), but the study was underpowered for such analysis.
Concentrations of HMGB1 in MDS according to risk stratification, treatment, and BM cellularity.

BM, bone marrow; HMGB1, high mobility group box–1; IPSS-R, 2012 Revised International Prognostic Scoring System; IQR, interquartile range; MDS, myelodysplastic syndrome.
No differences in HMGB1 levels were observed in MDS patients with or without concomitant autoimmune disorders.
Discussion
Here we show that circulating HMGB1 is elevated in patients with MDS in contrast to those with AA or PNH, two hyporegenerative syndromes with overlapping clinical and laboratory features, supporting a role for persistent HMGB1-mediated low-grade inflammation in MDS and suggesting that HMGB1 may have diagnostic potential in the differentiation between BM failure syndromes.
In MDS, an inflammatory microenvironment gives a survival advantage to the mutated clones over the rest of the normal compartment, leading to ineffective hematopoiesis manifested by anemia, thrombocytopenia, and neutropenia as well as an increased risk of transformation to AML over time. 9
TLRs are involved in the development of the hematopoietic stem cell, also playing a deleterious role under inflammatory conditions.9,11
HMGB1 is the prototype of damage-associated molecular patterns (DAMPs), endogenous molecules that, in the wrong location or concentration within the body, may produce or amplify an inflammatory response. HMGB1 signals through several receptors, including TLRs, CXCR4, and RAGE.18 –20 HMGB1 also binds to myeloid differentiation factor 2 (MD2) 21 and activates NLRP3 inflammasome, 22 resulting in the pyroptosis-induced demise of hematopoietic cells. 8 Autophagy plays a critical role in normal hematopoiesis;23,24 HMGB1 can interfere with autophagy, hypothetically inducing or amplifying ineffective hematopoiesis and transformation to AML. 25
HMGB1 can activate the NLRP3 inflammasome, resulting in increased production of IL-1β. 26 Experimentally, IL-1β and HMGB1 can act synergistically to amplify inflammation.27,28 HMGB1 and IL-1β were significantly higher in MDS than in controls and there was a correlation between both inflammatory mediators. Among the evaluated groups, only HMGB1 levels differentiated between MDS and other BM failure syndromes. A subanalysis of h-MDS indicated that circulating HMGB1 and IL-1β levels are significantly increased when compared with patients with AA and both may be relevant as circulating biomarkers in the differential diagnosis of h-MDS and AA.
CD34 + cells from patients with MDS overexpress TLR2, TLR4, and TLR9.12,29 The aberrant signaling of TLRs negatively impacts the function of hematopoietic stem cells.12,30,31 For instance, stimulation of BM-derived CD34 + cells with TLR2 agonists causes ineffective erythropoiesis. 32 It is therefore feasible that HMGB1-induced or HMGB1-sustained alterations in TLR function may lead to disruption in hematopoiesis.
The role of HMGB1 in MDS is incompletely understood. Expression of HMGB1 is two to threefold higher in CD34+ cell lines obtained from MDS patients than in those derived from healthy umbilical cords or BM. Blocking the effect of HMGB1 via siRNA or its release with sivelestat (an inhibitor of neutrophil elastase) results in decreased expansion of myelodysplastic stem cells (while sparing healthy ones) and aberrant overexpression of TLRs. 12 In that context, that suggests that HMGB1 may induce inflammation not only by binding to but also by inducing its receptors, leading to a cycle of inflammation that may induce or facilitate the clonal expansion of myelodysplastic stem cells. Our finding of increased circulating HMGB1 in MDS – but not in other BM failure syndromes – further suggests that HMGB1 is involved in the immunopathogenesis of MDS, and accordingly, that it may be a therapeutic target in MDS.
HMGB1 is also linked to inflammation-mediated dyserythropoiesis. 10 Our findings suggest that HMGB1 plays a pathogenic role in MDS, probably by inducing the production, maturation, or release of other cytokines, resulting in alterations in the hematopoietic stem cell.
Interestingly, circulating HMGB1 was higher among untreated MDS patients than among those who had already been treated. Our findings also suggest that HMGB1 is involved in the origin or perpetuation of the inflammatory milieu required for MDS clonal survival. However, as a pilot study, our study was not designed or powered to understand whether any specific treatment results in a faster or more pronounced reduction of circulating HMGB1.
IPSS-R relies on specific characteristics (blood parameters, BM blasts, cytogenetics) to stratify patients into five risk categories that can be further subdivided into lower- and higher-risk MDS. Interestingly, we observed that lower risk MDS patients had higher plasmatic levels of HMGB1 than higher risk patients; this can be explained in part because the inflammatory burden is higher in patients with low-risk MDS, resulting in the active release by immune cells, as well as passive shedding by necrotic and apoptotic cells, of HMGB1.
Finally, while karyotypic and morphologic features are undoubtedly helpful, differentiating hypocellular MDS from AA may be difficult. 33 Interestingly, we observed higher circulating concentrations of HMGB1 in patients with hypocellular MDS than in patients with AA. Hence, HMGB1 may be helpful in the differential diagnosis between both entities.
Circulating levels of TNF-α were surprisingly more elevated in hematologically healthy controls than among participants with any of the evaluated BM failure syndromes. While puzzling, this can be explained in part because the control group was adjusted for age and lacked hematological disease; however, in an aging control group, pathologies known to increase the inflammatory milieu were frequent, including hypertension, diabetes mellitus, or dyslipidemia, something observed already by others. 34
Our article has some limitations. We relied on a relatively small sample size derived from a single-center cohort in one Institute in Mexico City and the study is cross-sectional; therefore, it may be possible that other biological factors related to cytokine production and release were not taken into consideration. Also, as a source of bias, therapeutic interventions used may have changed the rate of apoptosis, ineffective myelopoiesis, and inflammation. Ideally, to understand the role of HMGB1 in the pathobiology of MDS, our results need to be validated longitudinally starting at diagnosis (i.e. before any therapeutic intervention has occurred), particularly among patients in different IPSS-R risk strata. Moreover, the proposed mechanisms are – at this moment – purely hypothetical; therefore, several questions need to be addressed further, including which receptors are targeted by HMGB1 in hematopoietic stem cells, as well as the signaling pathways involved in MDS. Whether or not HMGB1 may help in predicting the prognosis of MDS needs to be further addressed. Finally, our failure to detect between-group differences in several cytokines could be the effect of sample size and not necessarily indicate a lack of mechanistic connection.
In conclusion, our study shows that HGMB1 may play a mechanistic role in MDS. If our results are independently replicated, circulating HMGB1 could be yet another tool in the differentiation between hypocellular MDS and AA.
Supplemental Material
sj-docx-1-tah-10.1177_20406207221125990 – Supplemental material for Circulating HMGB1 is increased in myelodysplastic syndrome but not in other bone marrow failure syndromes: proof-of-concept cross-sectional study
Supplemental material, sj-docx-1-tah-10.1177_20406207221125990 for Circulating HMGB1 is increased in myelodysplastic syndrome but not in other bone marrow failure syndromes: proof-of-concept cross-sectional study by Elia Apodaca-Chávez, Roberta Demichelis-Gómez, Adriana Rosas-López, Nancy R. Mejía-Domínguez, Isabela Galvan-López, Meghan Addorosio, Kevin J. Tracey and Sergio Iván Valdés-Ferrer in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
