Abstract
Background:
The outcome of patients with myelodysplastic syndrome (MDS) and chronic myelomonocytic leukemia (CMML) post clofarabine is unknown.
Methods:
We reviewed 109 patients with MDS or CMML with a median age of 67 years, treated with a clofarabine-based chemotherapy as frontline (n = 38) or salvage (n = 71) therapy. A total of 58 (53%) patients received salvage therapy after clofarabine failure: 13 allogeneic stem cell transplant (ASCT), 18 high-dose cytarabine-containing regimen, 10 hypomethylating agents and 17 investigational treatments.
Results:
Eight patients achieved complete remission (CR) and three had stable disease for an overall response rate of 19%. With a median follow-up of 3 months from clofarabine failure, 12 patients (11%) remained alive, 5 remain in CR, 4 of them after ASCT. The median overall survival post clofarabine failure was 4 months with a 1-year survival rate of 23%.
Conclusions:
This outcome is predictable, with patients with high-risk disease at the time of clofarabine failure having the worse survival. To date, patients with MDS continue to have a short survival after failure of all available therapies. Ultimately, patients who are candidates for additional treatments should be offered novel approaches. In conclusion, the outcome of patients with MDS and CMML post clofarabine failure is poor. The pattern is similar to patients with MDS post hypomethylating agent failure and predictable using University of Texas M. D. Anderson Cancer Center global scoring system.
Introduction
Hypomethylating agents (HMAs) are the standard of care for patients with myelodysplastic syndromes (MDS). However, the duration of response tends to be short and the majority of patients do ultimately acquire resistance [Kantarjian et al. 2006; Fenaux et al. 2009; Garcia-Manero and Fenaux, 2011]. Hence, new approaches are needed for such patients.
Clofarabine is a rationally designed, second-generation purine nucleoside analog [Xie and Plunkett, 1995; Lotfi et al. 1999]. Clofarabine was synthesized based on the experience with the earlier deoxyadenosine analogs fludarabine and cladribine. It was designed as a hybrid molecule to overcome the limitations and incorporate the best qualities of fludarabine and cladribine, both of which are used for the treatment of hematologic malignancies. Clofarabine has a chloro-group at the 2-position of adenine; its chemical structure is more closely related to cladribine than to fludarabine. Halogenation at the 2-position of adenine renders this class of compounds resistant to intracellular degradation by the enzyme adenosine deaminase. Substitution of a fluorine at the C-2′-position of the arabinofuranosyl moiety of clofarabine increases its stability in gastric acid and decreases its susceptibility to phosphorolytic cleavage by the bacterial enzyme Escherichia coli purine nucleoside phosphorylase in the gastrointestinal tract, both of which may lead to enhanced oral bioavailability. It is active in acute lymphoblastic leukemia [Pui and Jeha, 2005]. It also has significant activity in patients with acute myeloid leukemia (AML) and MDS. An increasing number of clinical trials have incorporated clofarabine in the treatment of newly diagnosed and relapsed/refractory adult and elderly MDS patients [Faderl et al. 2008, 2012a]. Despite encouraging results, responses are short lived and most patients fail treatment and/or experience disease relapse. The outcomes of patients failing clofarabine are largely unknown and constitute the focus of this analysis.
Patients and methods
A total of 109 consecutive patients (88 MDS, 21 CMML) who failed clofarabine between June 2001 and May 2012 were reviewed: 36 patients (35%) had received frontline clofarabine and 71 patients (65%) salvage therapy; 96 patients (88%) had received single-agent clofarabine and 13 patients (12%) a clofarabine-containing combination. On the clofarabine monotherapy study, patients were adaptively randomized to 1 of 2 dose schedules: 15 mg/m2 versus 30 mg/m2 intravenous (IV) over 1 hour daily for 5 consecutive days [Faderl et al. 2012a]. During the consolidation, clofarabine was administered at 30 mg/m2 daily for 3 consecutive days (days 1–3). In the combination study, clofarabine was administered as a 1 hour IV infusion at 30 mg/m2 daily for 5 days on days 1–5 and cytarabine was added at 20 mg/m2 by subcutaneous injection daily for 14 days on days 1–14 during induction [Faderl et al. 2008]. During consolidation, 20 mg/m2 cytarabine was added by subcutaneous injection daily for 7 consecutive days (days 1–7). Courses were repeated every 4–8 weeks depending on resolution of toxicities and marrow recovery. Therapy was permitted for a maximum of 12 courses, unless patients had no response after ≥3 courses, progressed, or relapsed on therapy. Treatment schemas for the clofarabine monotherapy or combinations have been detailed previously [Faderl et al. 2008, 2012a]. All patients provided written informed consent according to institutional guidelines. Inclusion and exclusion criteria have been reported earlier [Faderl et al. 2008, 2012a]. The studies were approved by the institutional review and were conducted in accordance with the principles of the Declaration of Helsinki.
Responses to clofarabine and subsequent therapies were coded according to the modified International Working Group Criteria [Cheson et al. 2006]. Patients were categorized for MDS risk at the initiation and failure of clofarabine according to the International Prognostic System Score (IPSS) and the University of Texas M. D. Anderson Cancer Center (MDACC) model [Greenberg et al. 1997; Kantarjian et al. 2008]. Survival curves were constructed using the Kaplan–Meier method, and the curves were compared using the log-rank test.
Results and discussion
Patients’ characteristics are summarized in Table 1 and Figure 1. A total of 22 patients (20%) had failed 2 or more MDS directed therapies prior to initiation of clofarabine and 59 patients (54%) had failed HMA before initiation of a clofarabine-containing salvage therapy. Overall, 21 patients (19%) achieved a complete response (CR) with clofarabine, 8 (7%) achieved a CR with incomplete platelet recovery (CRp), 1 (1%) a marrow CR, and 1 (1%) had hematologic improvement for an overall response rate (ORR) of 28%. Of the 38 patients who received frontline clofarabine treatment, 15 (40%) achieved a CR, 3 (8%) CRp, 1 marrow CR (3%), and 1 hematologic improvement (3%), for an ORR of 54%. Of the 71 patients who received salvage clofarabine treatment, 6 (8%) achieved CR and 5 (7%) CRp, for an ORR of 15%. Response rates were 54%, 14%, 18%, and 20% among patients who received no, one, two, or three or more prior treatments, respectively. Specifically, ORR was 15% among the 59 patients who had received a prior HMA (Table 2). Duration of therapy was not significantly different between frontline and salvage settings. Median duration of clofarabine therapy was 13 weeks (range 5–197 weeks) for patients receiving clofarabine as first-line therapy and 10 weeks (range 1–79 weeks) for patients receiving clofarabine as salvage therapy (p = 0.08)
Patients characteristics.

MDS, myelodysplastic syndrome; RA, refractory anemia; RAEB, refractory anemia with excess blasts; RCMD, refractory cytopenias with multilineage dysplasia; RAEBT, refractory anemia with excess blasts in transformation; RARS, refractory anemia with ringed sideroblasts; CMML, chronic myelomonocytic leukemia; IPSS, International Prognostic Scoring System; HMA, hypomethylating agent; Xme, chromosomes. MDACC: MD Anderson Cancer Center.

Summary of patients outcomes after clofarabine failure.
At time of clofarabine failure, 14 (13%) had progressed to AML. A total of 58 patients received salvage therapy after clofarabine failure. Only 11 patients (19%) responded, 8 patients with CR and 3 patients with stable disease. One out of 18 patients (5%) responded to high-dose cytarabine (HDAC), 7 of 13 patients (54%) to ASCT, 1 of 10 patients (10%) to HMA, and 2 of 17 (12%) patients to investigational agents. Median duration of response was not reached (range 0–40 months). Within a median follow up of 3 months from clofarabine failure, 12 patients (11%) remained alive, 5 remain in CR, 4 of them after ASCT (Figure 1). The median OS post clofarabine failure was 4 months with a 1-year survival rate of 23%. Overall survival (OS) and event-free survival (EFS) were significantly better for patients undergoing ASCT after clofarabine failure (p = 0.02 and 0.01, respectively; see Figures 2 and 3). OS and EFS were independent of disease status at time of clofarabine failure (MDS versus AML). The MDACC score was predictive of OS when applied at the initiation of clofarabine therapy (p = 0.00229; see Figure 4) as well as at the time of failure (p = 0.00035; see Figure 5).

Overall survival post clofarabine failure (by type of therapy).

Event-free survival post clofarabine failure (by type of therapy).
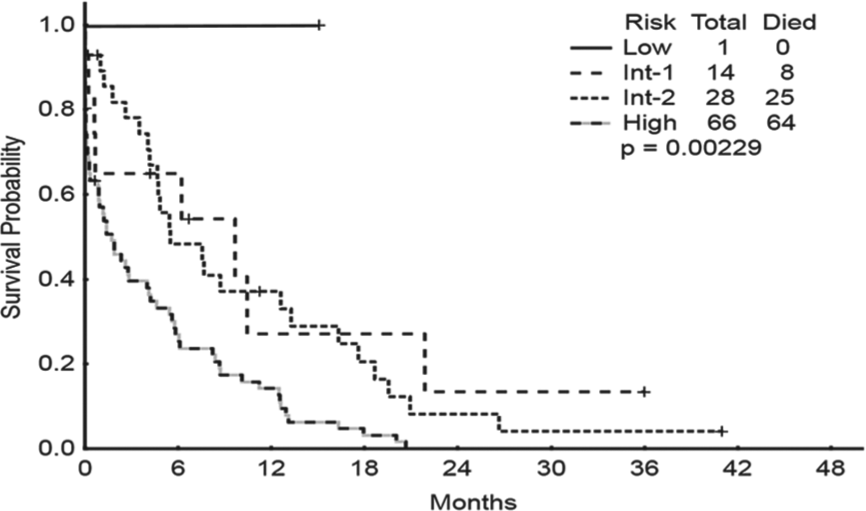
Overall survival from initiation of clofarabine therapy (by MDACC model risk categories).

Overall survival from time of clofarabine failure (by MDACC score risk categories).
To the best of the authors’ knowledge, the current study is the first to provide survival estimates for patients with MDS and CMML after clofarabine failure. Based on our findings, the prognosis after clofarabine failure is poor, with a median survival of 4.3 months. There was no difference in outcome noted between patients whose disease transformed into AML and those in whom it did not. Response rates to salvage chemotherapy after clofarabine failure were low. Patients appeared to have better outcomes when ASCT was feasible. However, there seemed to be no benefit for high-intensity chemotherapy over investigational agents when ASCT was not performed. In addition, our present study validates the utility of the MDACC model as a powerful dynamic tool in the prediction of outcomes of patients with MDS patients at different time points of treatment.
We have recently reviewed our experience with patients with MDS after failure of decitabine therapy [Jabbour et al. 2010]. In that analysis, with a median follow up of 21 months, the median survival after decitabine failure was only 4.3 months and the estimated 12-month survival rate was 28%. Thus, patients who do not respond to HMA and those who relapse or progress after an initial response have a dismal prognosis. These patients often have a resistant-disease phenotype and generalized deconditioning at time of failure. This pattern of HMA failure seems to be similar to the one observed with patients failing clofarabine, whether they had received HMA therapy prior to clofarabine or not.
The paucity of active agents in this setting creates a challenging situation and an opportunity for further research. Moreover, the sequence in which HMA and clofarabine therapies are best used remains to be determined. Combinations alternating HMA and clofarabine are being tested in elderly patients with MDS and AML [ClinicalTrials.gov identifier: NCT00778375; Faderl et al. 2012b]. To date, patients with MDS continue to have a short survival after failure of all available therapies. Ultimately, patients who are candidates for additional treatments should be offered novel approaches.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors have no competing financial interests to declare.
