Abstract
Loncastuximab tesirine-lpyl (ADC Therapeutics) is an anti-CD19 antibody-drug conjugate which consists of anti-CD19 antibody and cytotoxic alkylating agent, SG3199. Data from preclinical in vitro and animal studies demonstrated its selectivity and efficacy. The phase I LOTIS-1 study for relapsed, refractory B-cell non-Hodgkin lymphoma (NHL) demonstrated efficacy and a tolerable safety profile, with major adverse effects being neutropenia, thrombocytopenia, elevated liver enzymes, and fluid accumulation. Based on pharmacokinetics analysis in this study, a dose of 150 μg/kg every 3 weeks for cycles 1 and 2 followed by 75 μg/kg every 3 weeks until disease progression or intolerability was chosen for the phase II LOTIS-2 study. This study recruited relapsed, refractory diffuse large B-cell lymphoma and confirmed similar safety profile. Overall response rate was 48.6% (24.1% complete response), and overall survival was 9.9 months. Due to its safety and efficacy reported in the above trials, loncastuximab tesirine was recently approved by the US Food and Drug Administration for the treatment of relapsed, refractory diffuse large B-cell lymphoma. Several clinical trials are ongoing to assess its safety and efficacy in NHL in various clinical settings.
Keywords
Background
Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of B-cell non-Hodgkin lymphoma (NHL), occurring in almost 30% of all cases of NHL and an estimated 25,000 new cases annually in the United States. 1 Standard treatment with rituximab in addition to cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) results in cure in about 60–65% of cases. 2 However, almost 10–15% of patients treated with R-CHOP have primary refractory disease which is defined as an incomplete response or a relapse within 6 months after treatment.3,4 In addition, 20–25% relapse, which is defined as appearance of any new lesion after a complete response (CR), typically occurring within the first 2 years.
Despite progress in the upfront treatment of DLBCL, the treatment options in patients with relapsed/refractory DLBCL (r/r DLBCL) are limited. Outcomes particularly in patients with refractory disease remain poor, with an overall survival (OS) of about 6 months in these patients. 5 In patients who are eligible for transplant, treatment includes high-dose chemotherapy followed by autologous hematopoietic cell transplant (auto-HCT). However, only about half of these patients have a response to initial salvage therapy, and the overall cure rate is about 35–45% after auto-HCT.6,7
The treatment in patients who are transplant ineligible, those who do not respond to salvage therapy, and those who relapse after auto-HCT is challenging. These options include either single agent or multiagent low-dose chemotherapy with a palliative intent.8–11 Although patients with first-line refractory disease or early relapse after chemoimmunotherapy have a higher risk of treatment failure and mortality within the first 9 months, auto-HCT has shown an overall good and durable response in patients with refractory or relapsed disease. 12 Chimeric antigen receptor (CAR) T-cell therapy, which was recently approved, has shown durable responses and has revolutionized the treatment of r/r DLBCL. 13 However, it is associated with life-threatening toxicities and is only available in large academic centers, making it a less than ideal option for all patients. In addition, compared with CAR T-cell therapy, auto-HCT provides a durable disease control, better OS, and a lower risk of relapse in DLBCL patients who only achieve a partial response (PR) after first line or salvage therapy.14,15
More recently, there has been tremendous progress in the treatment in r/r DLBCL resulting in the approval of multiple novel therapies. One class of drug which has gained prominence is the antibody-drug conjugates (ADCs). These drugs target specific antigens without causing the significant on-target off-tumor toxicity. Generally, they consist of an antibody directed to a particular cell marker and a small molecule cytotoxic payload. One of the first ADCs was BR-96 doxorubicin which demonstrated significant antitumor activity against human breast cancer cell lines in the experimental murine models. 16
Notable agents which have been approved recently in r/r DLBCL include polatuzumab vedotin-piiq in combination with bendamustine and rituximab in June 2019, selinexor in June 2020, tafasitamab-cxix in combination with lenalidomide in July 2020, and most recently loncastuximab tesirine-lpyl (Lonca) in April 2021.17–20 With the recent approval of these anti-CD19 agents, the review of its mechanism of action, efficacy, and possible side effects is timely and useful.
Introduction to loncastuximab tesirine
Structure and mechanism of action
Lonca is an ADC which consists of the humanized antibody directed against CD19 and tesirine. The tesirine part is made of cathepsin-cleavable valine-alanine linker and the warhead a pyrrolobenzodiazepine (PBD) dimer called SG3199 21 (Figure 1). PBD dimers act as a DNA minor groove interstream crosslinking agent. 22 This class of warheads has shown superiority over others due to its ability to target low copy number antigens and tumor-initiating cells and to exploit low drug–antibody ratios (DARs) and has thus shown activity against multidrug-resistant cancers.23,24

Structure of loncastuximab tesirine.
Given its expression in B-cell malignancies including leukemias and NHLs, CD19 is an attractive target for these ADCs. By using CD19 as a target, Lonca is able to deliver the highly cytotoxic PBD to B-cells. This drug is then internalized and undergoes lysosomal degradation. SG3199 binds in the DNA minor groove and forms highly cytotoxic DNA inter-strand crosslinks, preventing the separation of opposing strands and thus resulting in cell death 25 (Figure 2).

Mechanism of action of loncastuximab tesirine.
Pharmacokinetics and pharmacodynamics
Lonca is given intravenously and is therefore readily absorbed in the circulation with an average volume of distribution of about 7.11 liters. The monoclonal antibody portion of the drug is expected to be metabolized into smaller peptides by catabolic pathways while the cytotoxin portion, SG3199, may be in part metabolized by CYP3A4/5. However, it is not considered to be a potent inhibitor of the human cytochrome P450 isozymes. Although the exact excretory pathways in humans are not known, this drug is not expected to be excreted by kidneys.
Preclinical studies
Zammarchi et al. 26 performed in vitro and in vivo studies in animals. Both in in vitro and in vivo studies, Lonca exhibited a dose-dependent targeting of human CD19 expressing cells. As demonstrated by the in vitro studies, Lonca was specifically bound, internalized, and trafficked to lysosomes in these cells. The SG3199 warhead was then released by cleavage of Val-Ala dipeptide linker and formed crosslinks with the DNA strands where it persisted for about 36 h.
In vivo studies done on rats and cynomolgus monkeys demonstrated similar pharmacokinetics (PK) of Lonca. The half-life for total and PBD-conjugated antibody was found to be 9.9 and 10.4 days after a 1.5 mg/kg single dose administration to rats. Similarly, the half-life was 14.1 and 13.7 days after a dose of 0.6 mg/kg of the drug was given to cynomolgus monkeys on days 1 and 22. The drug also showed excellent stability and safety profile in both rat and monkey studies.
In terms of pharmacology of PBD dimer SG3199 itself, the SG3199 was found to be highly protein bound in rats, cynomolgus monkeys, and humans in a preclinical study by Hartley et al. 25 It was found to be poorly metabolized and rapidly excreted, mostly via feces with only minor urinary excretion.
Clinical studies
Pharmacokinetics and pharmacodynamics data are available in two phase I studies as well as phase II study in patients with B-cell malignancies.
Phase I study-LOTIS-1. In the LOTIS-1 phase I trial, PK data were available in 161 patients for PBD-conjugated antibody, 160 patients for total antibody, and 37 patients for free warhead SG3199. 27 The maximum concentration (Cmax) achieved during cycle 1 was 2995 μg/L and that during cycle 2 was 3155 μg/L. Area under the curve (AUC) was 15,245–22,823 μgxday/L. At a dose of 150 µg/kg, the mean half-life increased from 4.46 days in cycle 1 to 9.77 days in cycle 2, indicating accumulation of drug in the body. Substantial variability was seen for total antibody, PBD-conjugated antibody dose, and SG3199 due to different CD19 levels between different patients. Although 5 out of 183 and 1 out of 183 patients had pre- and post-dose anti-drug antibodies (ADA), the titers were very low showing low immunogenicity of Lonca.
Phase II study-LOTIS-2. The PK data from the LOTIS-2 study were analyzed for cycles 1, 2, and 3. The Cmax for conjugated antibody, total antibody, and SG1399 after cycle 1 (150 μg/kg dose) were 2430, 3267, and 0.04 ng/mL respectively. The half-life was 8.85 days after the first dose and 15.2 days after the second dose for PBD-conjugated antibody and 8.66 days after first dose and 20.9 days after the second dose for total antibody. The accumulation of drug after cycle 3 (dose 75 µg/kg) was lower than in cycles 1 and 2. Although high variability was seen with SG3199, the variability for total and PBD-conjugated antibody was moderate. No post-ADAs were detected in any patient.
Efficacy of loncastuximab tesirine
Phase I trial: LOTIS-1
The multicenter phase I study was dose escalation and dose expansion study, which enrolled 88 patients in the initial report and 183 patients in the final analysis, among which 75% had DLBCL.27,28 The median age was 63 years, 62% were men, and patients had received a median of three lines of therapy including 23% with prior hematopoietic stem cell transplant (HSCT) and 1.6% with prior CAR T-cell therapy (Table 1).
Basic characteristics of patients enrolled in the LOTIS-1 and LOTIS trials.
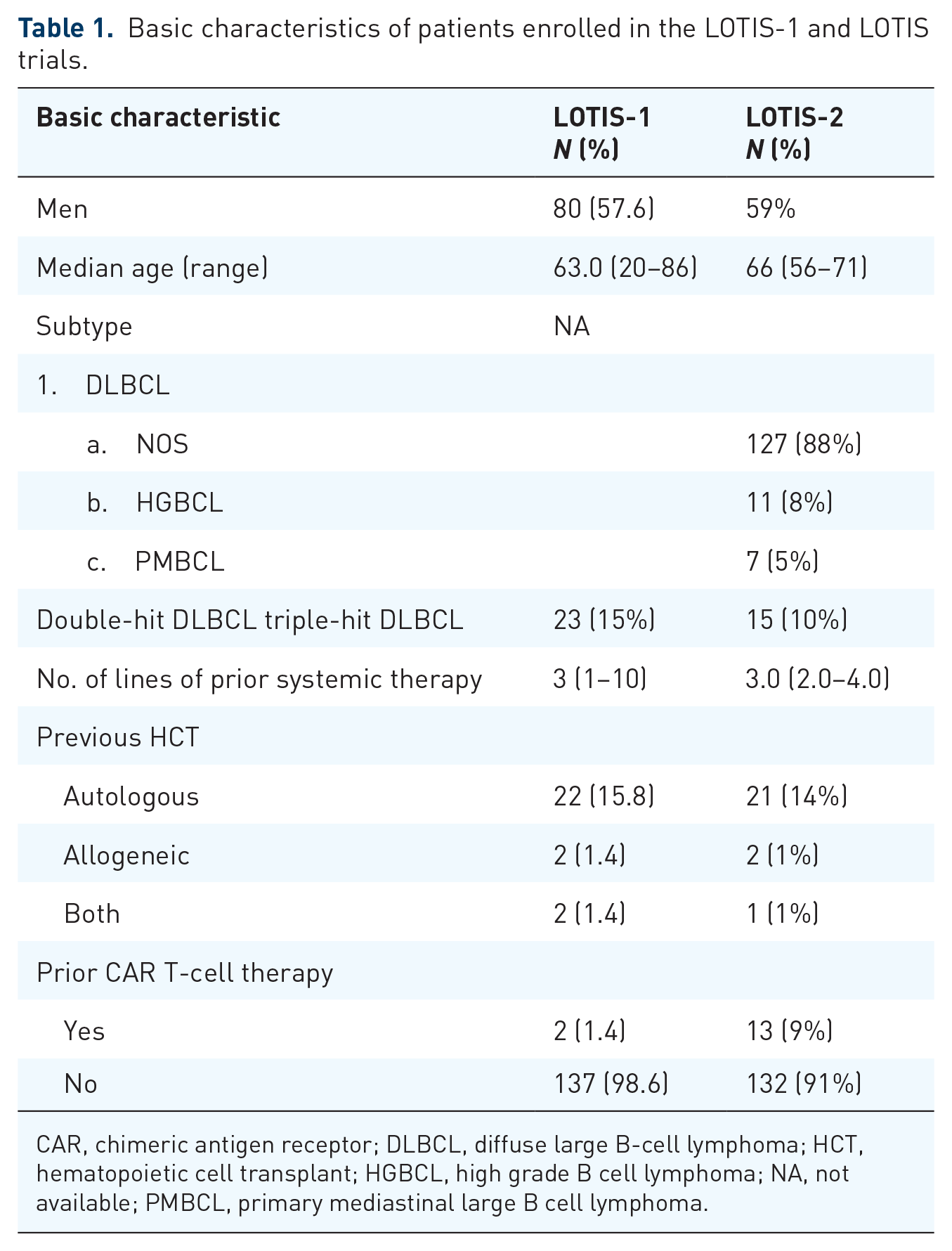
CAR, chimeric antigen receptor; DLBCL, diffuse large B-cell lymphoma; HCT, hematopoietic cell transplant; HGBCL, high grade B cell lymphoma; NA, not available; PMBCL, primary mediastinal large B cell lymphoma.
In the dose escalation phase (part I) of the trial, Lonca dose was escalated from 15 to 200 μg/kg IV every 3 weeks in the 3 + 3 design. Part II (dose expansion portion) utilized the doses of 120 and 150 µg/kg, selected based on antitumor activity and tolerability seen during part I. The overall response rate (ORR) in patients with DLBCL, mantle cell lymphoma, and follicular lymphoma was 42.3%, 46.7%, and 78.6%, respectively. The median progression-free survival (PFS) was 3.1 months in all patients with B-NHL and 2.8 months in patients with DLBCL, whereas the median OS was 8.3 months in all patients and 7.5 months in patients with DLBCL (Table 2).
Overall response rates, duration of response, and survival of patients treated with loncastuximab tesirine.

B-NHL, B cell non Hodgkin lymphoma; CR, complete response; DLBCL, diffuse large B-cell lymphoma; DOR, duration of response; ORR, overall response rate; OS, overall survival; PFS, progression-free survival.
DOR for dose levels ⩾120 μg/kg.
Phase II trial: LOTIS-2
The subsequent pivotal phase II trial of Lonca, a multicenter open-label single-arm trial, was recently published. This trial enrolled 145 patients with R/R DLBCL from the United States and Europe. These patients received Lonca at 150 μg/kg for two cycles (21-day cycle) followed by 75 μg/kg which was then continued for up to 1 year or until disease relapse or progression, death, unacceptable toxicity, pregnancy, deviation from study protocol, or patient, investigator, or sponsor decision. The median age of patients was 66 years, 59% were men, 43% had received at least two previous lines of therapy, 14% had an auto-HCT, 1% had allo-HCT, and 9% had previous CAR T-cell in the past (Table 1).
The ORR in patients who received at least one dose of Lonca was 48.3% and complete response rate (CRR) was 24.1%, whereas 15% had stable and 21% had progressive disease. Median time to response was 41 days and the median duration of response (DOR) was 10.3 months. The median PFS and OS were 4.9 and 9.9 months, respectively (Table 3).
ALT, alanine transaminase; GGT, gamma-glutamyl transferase.
Dosing: 150 μg/kg IV every 3 weeks for two doses followed by 75 μg/kg IV every 3 weeks thereafter.
More recently, in the follow-up analysis of LOTIS-2, the updated analysis yielded similar overall results with DOR, PFS, and OS of 12.6, 4.9, and 9.5 months, respectively. The ORR for all patients, patients aged 75 years or older, and patients with double/triple hit DLBCL was 48.3%, 52.4%, and 33.3%, respectively. 29
Spira et al. 30 also reported health-related quality of life (HRQoL), symptoms, and tolerability in patients receiving Lonca for relapsed or refractory DLBCL in the LOTIS-2 study. This was done by checking patient-reported outcomes through EQ-5D and FACT-Lym at baseline, day 1 of each cycle, and the last day of treatment visits. The EQ VAS overall health score improved while the FACT-Lym score remained stable during the treatment with no differences reported in elderly compared with the general population. These findings further suggest that this Lonca has manageable toxicity profile even in the elderly population.
Safety of loncastuximab tesirine
Phase I trial: LOTIS-1
In the phase I trial, the safety of the drug was assessed in all 183 patients. 28 In part 1, dose escalation part of the study, patients received median of two doses and a median dose of 129.9 μg/kg for a median duration of 64 days. Four out of these 183 patients experienced dose-limiting toxicities including grade 4 thrombocytopenia in one patient receiving 120 μg/kg and two patients receiving 200 μg/kg, and grade 3 febrile neutropenia in one patient receiving 150 μg/kg dose.
In the subsequent safety analysis, 98% of patients had at least one treatment-emergent adverse event (TEAE) and 46.4% had at least one serious TEAE with 19.1% being fatal and 19% leading to treatment discontinuation. 27 These adverse effects were more common at doses above 200 μg/kg. Among these, the most common were hematologic adverse effects including grade 3 or above thrombocytopenia (26.7%), neutropenia (39.7%), febrile neutropenia (5.5%), and lymphopenia (6.6%). Other adverse effects in order of their incidence included fatigue, nausea, peripheral edema, and increase in γ-glutamyltransferase (GGT). Rash especially in sun-exposed areas was seen in 24.6% of patients. The most common serious TEAE included febrile neutropenia (5.5%), pyrexia (3.8%), pleural effusion (3.8%), dyspnea (3.3%), sepsis (2.7%), and abdominal pain (2.2%). Among fatal adverse effects, six were considered treatment related, all of which were infections.
Based on these adverse effects, premedication with dexamethasone was introduced in the part II of the study which led to a decrease in incidence of these TEAE as described in more detail below.
Phase II trial: LOTIS-2
Based on the results of LOTIS-1 trial, several steps were taken in phase II to decrease the TEAEs. First, the dose of 150 μg/kg of the drug was chosen to be given every 3 weeks for two cycles followed by 75 μg/kg every 3 weeks. The dose was based on adjusted ideal body weight in patients with BMI ⩾35 kg/m2. In addition, patients were premedicated with dexamethasone, treated with spironolactone in case of edema/pleural effusions, and dose reductions as well as treatment delays were permitted per study protocol. Median duration of treatment was 45 days, median average weight-adjusted dose per cycle was 113.5 μg/kg, and median number of cycles was 3. TEAEs were reported in 99% of patients. The most common side effects were hematologic including thrombocytopenia (33%), anemia (26%), neutropenia (50%), and febrile neutropenia (3%). Other TEAE included infusion reaction, fatigue, nausea, peripheral edema, rash, elevated liver enzymes, and electrolyte imbalance. Serious TEAEs were reported in 39% of 145 patients. 15% had TEAEs possibly related to Lonca, including febrile neutropenia (3%), anemia (1%), pleural effusion (1%), and non-cardiac chest pain (1%) (Table 4).
Ongoing clinical trials for loncastuximab.

DLBCL, Diffuse large B-cell lymphoma; MCL, mantle cell lymphoma; NHL, non-Hodgkin lymphoma.
There were 77 deaths (53%), the majority of which (68%) were from progressive disease. 6% of patients had fatal TEAE, while 16% died after the adverse event reporting period. The five fatal outcomes, which included sepsis, small bowel perforation, septic shock, pneumonia, and acute kidney injury, were considered unrelated or unlikely to be related to Lonca. Three patients were reported to have fatal TEAEs (DLBCL, hemoptysis, and disease progression); however, cause of death was recorded as disease progression. Two had non-treatment-related fatal outcomes but were considered by the investigator as possibly related to the drug: one with acute respiratory distress syndrome (ARDS), likely related to pulmonary infection, occurring 31 days after last dose of study and second interstitial lung disease which occurred 63 days after the last dose and was suspected to be due to disease progression.
CAR T-cell therapy after loncastuximab tesirine
Since CAR T-cell therapy and Lonca target the same CD19 agent on the malignant B-cells, there is particular interest in this area. Most CD19-directed CAR T-cell therapy studies have frequently excluded patients previously treated with CD19 targeting immunotherapies. We reported a series of 14 DLBCL patients who relapsed or progressed after Lonca and were subsequently treated with CD19 CAR T-cell therapy. 31 Six out of the 14 patients had a complete response while 1 patient had a partial response.
In addition, in the follow-up analysis of the LOTIS-2 trial, 15 patients received CD19-directed CAR T-cell therapy upon progression after Lonca. The ORR in these patients was 46.7%. Eleven patients in the LOTIS-2 study received HCT as consolidation after Lonca response. 32 Interestingly, 13 patients who progressed after CAR T-cell therapy received Lonca in the LOTIS-2 trial and the ORR was 46.2% in these patients. 20 Whereas commercial anti-CD19 CAR T-cells bind the FMC63 epitope of CD19, the target of Lonca is the RB4 epitope of CD19.26,33 Point mutations in CD19 of malignant B-cells can be found in patients who relapse following commercial anti-CD19 CAR T-cell therapy targeting FMC63, leading to impaired antitumor efficacy. 33 The targeting of a different epitope of CD19 could at least partially explain the efficacy of Lonca after anti-CD19 CAR T-cell therapy failure.
In addition, Locke et al. 34 recently demonstrated improved event-free survival (EFS) with axicabtagene ciloleucel (axi-cel) compared with standard of care (SOC) (8.3 versus 2.0 months, respectively) when used as a second-line therapy for DLBCL. Similar results were reported by Kamdar et al. 35 with lisocabtagene maraleucel (liso-cel) when used as second-line therapy, with median EFS of 10.1 months compared with 2.3 months with SOC. Of note, in the analysis by Bishop et al., 36 the EFS was not significantly different between SOC and tisagenlecleucel. Based on these studies, it is plausible that soon CAR-T will move to second line. The role of CD19-directed ADCs including Lonca in terms of sequencing is not clear given paucity of data but lonca could potentially be a relevant agent in CD19+ relapse after CAR T-cell therapy.
Future directions
There are several ongoing trials for Lonca to assess it efficacy and safety in various clinical scenarios.
LOTIS-3 trial
This is a phase I/II study to evaluate the safety and efficacy of Lonca with Ibrutinib in patients with DLBCL and MCL. The phase I is a 3 + 3 dose escalation portion followed by phase II which will use the dose of the drug determined in phase I. All patients will get 560 mg daily ibrutinib. The trial started recruiting 1 December 2018 and is expected to be completed by 14 April 2023 (NCT03684694).
LOTIS-5 trial
This is a phase III randomized trial to assess efficacy of Lonca combined with rituximab (Lonca-R) compared with standard Rituximab, Gemcitabine, and oxaliplatin (R-GemOx) in patients with R/R DLBCL. This is also a two-part trial: part 1 will consist of a nonrandomized safety run-in period to evaluate Lonca-R safety followed by part 2 which will randomize patients to Lonca-R versus R-GemOx. The trial started recruiting 16 September 2020 and is expected to be completed by 30 June 2028 (NCT04384484).
LOTIS-6 trial
This is a phase II randomized study to assess the efficacy and safety of Lonca versus idelalisib in patients with R/R follicular lymphoma. This study started on 30 October 2021 and is expected to complete on 30 December 2024 (NCT04699461).
LOTIS-7 trial
This is a phase Ib nonrandomized trial to evaluate the safety and antitumor activity of Lonca in combination with various other anticancer agents in patients with R/R B-cell NHL consisting of dose escalation and dose expansion parts. The other drugs include gemcitabine, lenalidomide, polatuzumab vedotin, and umbralisib. It is estimated to start in December 2021 and end in February 2026 (NCT04970901).
LOTIS-8 trial
This is a phase Ib, open-label, multicenter study to assess the safety and tolerability of Lonca in combination with R-CHOP in patients with previously untreated DLBCL. It consists of two parts: dose escalation part followed by dose expansion part. It is estimated to start in December 2021 and end in May 2027 (NCT04974996).
Conclusion
Lonca has proven to be a promising treatment for R/R DLBCL which is both efficacious and safe in this patient population. Its role in frontline setting, in combination with other chemoimmunotherapies, is yet to be evaluated. Also, its role in other NHL is also being investigated as mentioned in the above ongoing and future trials. The follow-up data for LOTIS-2 has shown durable responses including in high-risk groups. Further trials and follow-up study will help better appraise its role in both frontline as well as relapsed refractory setting.
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mehdi Hamadani reports: Consultancy: Janssen R &D (2015); Incyte Corporation (2021); ADC Therapeutics (2021); Pharmacyclics (2018), Omeros (2021), Verastem (2019), TeneoBio (2019), MorphoSys (2021), Kite (2021), Genmab (2021), SeaGen (2021), and Gamida Cell (2021). Speaker’s Bureau: Sanofi Genzyme (2021), AstraZeneca (2021), and BeiGene (2021).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

