Abstract
Introduction:
Bortezomib has been reported to favourably impact the outcomes of t(4;14) and del(17p) in multiple myeloma (MM), but its impact on gain 1q (+1q) is unknown.
Methods:
To address this, 250 patients treated with bortezomib-based induction were analysed. All myeloma samples had fluorescence in situ hybridization (FISH) performed on CD138-sorted bone marrow aspirate, and plasma cells were analysed using DNA probes specific for the following chromosomal aberrations: del(13q14), del(17p), t(14;16), t(4;14), and +1q. Presence of +1q was defined as the presence of at least three copies of 1q21 at the cut off level of 20% of bone marrow plasma cells.
Results:
+1q identified in 167 (66.8%) and associated with t(4;14) and high lactate dehydrogenase (LDH). +1q was not associated with response rate but shorter event-free survival (EFS) (median EFS 35 vs 55 months, p = 0.05) and overall survival (OS) (median OS 74 vs 168 months, p = 0.00025). Copy number and clone size did not impact survival. Multivariate analysis showed +1q was an independent adverse factor for OS together with International Staging System (ISS)3, high LDH, del(17p) and t(4;14). When a risk score of 1 was assigned to each independent adverse factor, OS was shortened incrementally by a risk score from 0 to 4. Post-relapse/progression survival was inferior in those with +1q (median 60 vs 118 months, p = 0.000316). Autologous stem cell transplantation (ASCT) improved OS for those with +1q (median OS 96 vs 49 months, p = 0.000069).
Conclusion:
+1q is an adverse factor for OS in MM uniformly treated with bortezomib-based induction but was partially mitigated by ASCT. A risk scoring system comprising +1q, LDH, high-risk FISH, and ISS is a potential tool for risk stratification in MM.
Introduction
Multiple myeloma (MM) is a genetically heterogeneous malignancy of plasma cells characterized by clinical outcomes and results in variable patient outcomes. 1 Cytogenetic abnormality detected by fluorescence in situ hybridization (FISH) is a powerful prognostic marker in newly diagnosed MM (NDMM). Incorporation of elevated lactate dehydrogenase (LDH) and International Myeloma Working Group (IMWG)-defined high-risk FISH alterations, 2 including t(4;14), t(14;16), or del(17p), improved the International Staging System (ISS) resulting in the Revised International Staging System (R-ISS) 3 but notably one of the major prognostic factors gain 1q21 was not incorporated into that system.
Gain in chromosome 1q (+1q) is a common cytogenetic abnormality in MM occurring in 35–40% of patients.4–10 The frequency increases from smouldering myeloma to relapsed disease, 5 consistent with it being important in disease progression. Several studies have identified the poor prognostic impact of carrying this marker5–13 but some of the data on the significance of copy number and clone size remains conflicting,7–10 which was further confounded by the use of variable induction regimens with and without the incorporation of proteasome inhibitor therapy which is known to have a beneficial impact on patients with some high-risk (HR) FISH abnormalities.5–8,11,14,15 The impact is further obscured by incomplete data sets lacking the ISS in some studies5–9,11 or lack of knowledge of the use of autologous stem cell transplant (ASCT)5–8,11 or LDH.7,8,11 Importantly, bortezomib has been shown to have a different set of side effects in patients of Chinese origin which might impact both the dose administered and its impact on different risk factors.
In this study, we have evaluated the impact of +1q on treatment response and survival in a cohort of patients with complete clinical demographics and biochemical data, who had uniformly received a bortezomib-based triplet induction therapy with either bortezomib/thalidomide/dexamethasone (VTD) or bortezomib/cyclophosphamide/dexamethasone (VCD).
Methods
Newly diagnosed symptomatic MM patients treated in seven haematology centres, including Queen Mary Hospital, Princess Margaret Hospital, Tuen Mun Hospital, Pamela Youde Nethersole Eastern Hospital, United Christian Hospital, Tseung Kwan O Hospital and Queen Elizabeth Hospital, from January 2006 to July 2020 in Hong Kong were included in this retrospective study. Patients were included if they had FISH on pre-treatment bone marrow specimen performed. All MM samples had FISH performed on CD 138-sorted bone marrow aspirate, hence the purity of myeloma cells is more than 90%. Plasma cells were analysed using DNA probes specific for the following chromosomal aberrations: del(13q14), del(17p), t(14;16) and t(4;14). Gains of 1q21 were assessed using a 339 kb probe (MetaSystems, Germany) at the 1q21-22 region which includes the entire CKS1B gene. A total of 100–200 interphase nuclei were analysed. The presence of +1q was defined as the presence of at least three copies of 1q21 at the cut-off level of 20%, that is, +1q in at least 20% of bone marrow plasma cells (BMPCs). Low clone size was defined as the presence of at least three copies of 1q21 in 20–49% BMPCs, and high clone size in ⩾50%.
Demographics and disease characteristics at the time of diagnosis, as well as treatment and transplant information were retrieved from the electronic medical records with approval from the Institutional Review Board of the University of Hong Kong. Patients were classified by the ISS stage, while treatment response was evaluated using the IMWG Uniform Response Criteria. An event was defined as primary refractory disease, relapse from complete response (CR), progression (⩾25% increase in M protein from nadir or ⩾25% difference between involved serum-free light chains from best response on two consecutive tests four weeks apart), development of extra-medullary plasmacytomas/new bone lesions, or death due to any cause. Autologous stem cell transplant (ASCT) was generally offered to all consenting myeloma patients aged ⩽65 years, with due consideration of their performance status or comorbidities. The reporting of this study conforms to the STROBE statement. 16
Statistical analysis
Statistical analysis was conducted using IBM SPSS Statistics, version 26. The overall survival (OS) was calculated from the date of diagnosis till the date of death or last follow-up. Event-free survival (EFS) was measured from the date of diagnosis to the date of progression, relapse, death, or the last follow-up. Post-relapse/progression survival was measured from the date of progression or relapse to the date of death or last follow-up. The survival curves for OS, EFS, and post-relapse/progression survival were plotted using the Kaplan–Meier method, compared by the log rank test. Chi-square or Fisher’s exact test was utilized to assess the association between categorical variables, with a confidence interval (CI) of 95%. Multivariate Cox regression analysis was performed to analyse the impact on OS of risk factors which are significant in the univariate analysis, including ISS, elevated LDH, del(17p), t(4;14), +1q. Only two patients had t(14;16) and hence were not included in the multivariate analysis. A p-value of <0.05 was considered statistically significant and all p-values are two-sided.
Results
Baseline characteristics
Complete cytogenetic data at diagnosis was available for 250 patients including +1q that were included in the study (Table 1). The median duration of follow-up was 37 months, median age 62 years (range: 37–98), 64 (26.2%) were ISS1, 75 (30.7%) ISS2, and 105 (43%) ISS3. Patients with ISS3 had significantly inferior OS compared with ISS1/2 (median OS 62 months vs 119 months, p = 0.001).
Baseline characteristics of patients with +1q and without +1q.

ISS, International Staging System; LDH, lactate dehydrogenase; ULN, upper limit of normal.
IgG was the major isotype (n = 104, 50.5%). One hundred and twelve (53.3%) patients received ASCT, 205 (90.7%) received bortezomib-based induction, the majority of which was triplet-based induction, including VTD (n = 162; 84.4%), VCD (n = 16; 8.3%), VRD (bortezomib/lenalidomide/dexamethasone) (n = 3; 1.6%), VMP (bortezomib/melphalan/prednisolone) (n = 6; 3.1%), PAD (bortezomib/doxorubicin/dexamethasone) (n = 3; 1.5%), and VD (bortezomib/dexamethasone) (n = 2; 1.1%). A total of 167 patients (66.8%) had +1q. There was an association between +1q with elevated LDH (p = 0.025) and t(4;14) (p = 0.048), but no association between +1q and ISS3 (p = 0.128) or other high-risk FISH (p = 0.245).
Response rate and survival outcome
Patients with or without +1q had comparable CR rate (40.4% vs 59.6%, p = 0.587). By comparison with patients lacking +1q, patients with +1q had shorter EFS, (median 35 months vs 55 months, p = 0.05), and inferior OS (median 74 months vs 168, p = 0.00025) (Figure 1). Concurrent +1q with IMWG high-risk FISH (t(4;14), del(17p) or t(14;16)) was associated with worse OS (median 52 months vs 119 months, p = 0.001) than those with +1q only. One of the ‘double hit’ myeloma groups, defined by Walker et al. (>3 copies of 1q21 with ISS3) 13 had inferior OS (median 48 months vs 119 months, p = 0.000083) compared with the non-double hit myeloma. Patients with +1q had inferior post-relapse/progression survival compared to those without +1q (median 60 months vs median 118 months, p = 0.000316) (Figure 2).
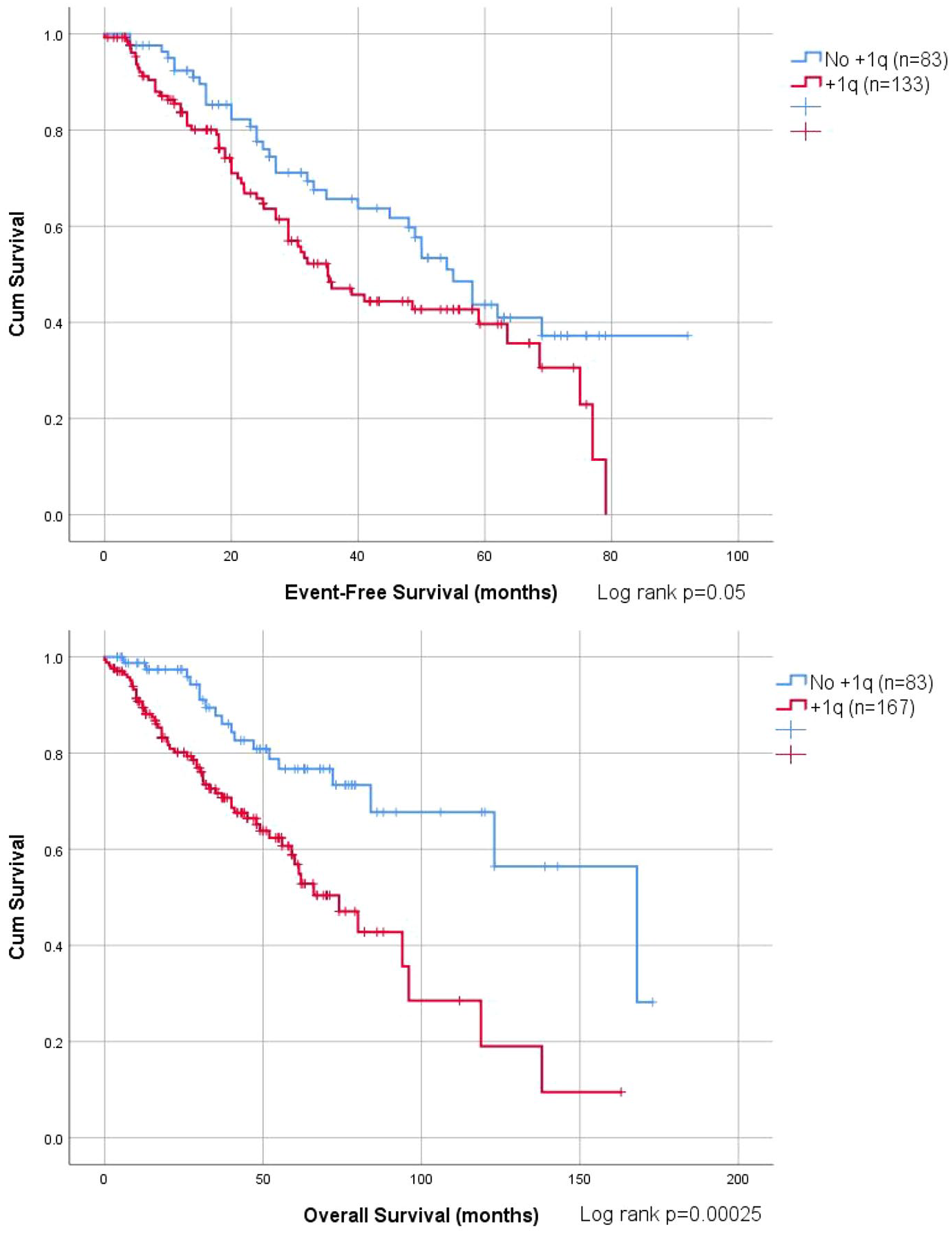
OS and EFS among MM patients with +1q (red) and without +1q (blue).

Post-relapse/progression survival among +1q (red) and without +1q (blue).
Univariate and multivariate analysis are shown, Table 2. In univariate analysis, ISS3, elevated LDH, del(17p), t(4;14), and +1q were identified as risk factors associated with inferior OS. On multivariate analysis, +1q retained its negative impact on OS (p = 0.037, HR 1.840, 95% CI 1.036–3.268). Other factors that were significantly associated with adverse OS on multivariate analysis included ISS3 (p = 0.04, HR 1.395, 95% CI 1.015–1.917), high LDH (p = 0.046, HR 1.696, 95% CI 1.009–2.851), del(17p) (p = 0.001, HR 2.73, 95% CI 1.479–5.036) and t(4;14) (p = 0.018, HR 1.929, 95% CI 1.118–3.328).
Multivariate analysis (Number of observations used = 221. t(14;16) was not included in the model due to small case number).

ISS, International Staging System; LDH, lactate dehydrogenase; ULN, upper limit of normal.
Impact of copy number at 1q and clone size
Among the cohort of 167 patients with +1q, 75 (44.9%) had three copies of 1q and 92 (55.1%) had >3 copies. There was no difference in EFS (median 39 months vs 35 months, p = 0.853) and OS (median 74 months vs 61 months, p = 0.937) among those with >3 copies or three copies of 1q (Figure 3).

OS and EFS comparing without +1q (blue), amp 1q (green), and gain 1q (red).
Among the 167 patients with +1q, 111 (66.4%) patients had +1q in ⩾50% and 56 (33.6%) had +1q in <50% BMPCs. Among patients with +1q, those with <50% had comparable EFS (median not reached vs 32 months, p = 0.51) and OS (median not reached vs 62 months, p = 0.95) than those with clone size ⩾50%. Compared with those with normal 1q copy number, patients with a +1q in <50% BMPCs had shorter OS (median not reached vs 168 months, p = 0.034) but not EFS (median 39 months vs 55 months, p = 0.494) while those with +1q in ⩾50% BMPCs had significant shortening of both EFS (median 32 months vs 55 months, p = 0.029) and OS (median 62 months vs 168 months, p = 0.000112).
ASCT on +1q
ASCT was performed on 112 patients (53.3%), of which 56 (43.1%) patients had +1q. Seventy-four patients with +1q did not receive ASCT. Among patients with +1q, ASCT was associated with significantly longer OS (median 96 months vs 49 months, p = 0.000069) than those who did not receive ASCT. Among patients who received ASCT, those without +1q had a trend to superior OS than those with +1q (median 96 months vs 168 months, p = 0.086) (Figure 4).

OS according to +1q and ASCT status (without +1q and ASCT: blue, +1q and ASCT: red, and +1q and no ASCT: green).
Prognostic scoring system incorporating +1q, ISS, IMWG-defined HR FISH and LDH
Based on the prognostic parameters identified in this study, including ISS, LDH, +1q, and IMWG-defined high-risk FISH, a model for OS was developed. In this, one point was assigned for each risk factor generating a range of score from 0 to 4. The median OS for patients with no risk factors was not reached, 94 months for 1–2 risk factors and 48 months for 3–4 risk factors (Figure 5). Patients with 0 risk factor had significantly better OS (p = 0.005, HR 3.22 CI 1.36–7.64) compared to those with 1–2 risk factors. Patients with 1–2 risk factors also had significantly better OS (p = 0.014 HR 1.86 CI 1.13–3.09) compared to those with 3–4 risk factors (Table 3).
OS according to risk factor score.

CI, confidence interval; HR, hazard ratio; OS, overall survival.

OS according to risk score 0 (blue), 1–2 (red) and 3–4 (green).
Discussion
The data presented here shows that +1q is common at diagnosis in Chinese MM patients and is associated with inferior OS, and, to a lesser extent, EFS. The inferior OS was predominantly due to a significantly shorter post-relapse/progression survival, indicating the presence of more resistant disease after disease relapse/progression. However, the copy number and clone size did not impact EFS and OS. Nonetheless, ASCT could partially mitigate the adverse impact of +1q on survival. The data showed that in this series of cases treated with bortezomib, the treatment was well tolerated, and there was no excess of pulmonary toxicity, yet cases with gain 1q21 had a consistently poor prognosis.
These findings are consistent with recent publications on the adverse impact of +1q in MM which show inferior OS in patients with +1q.5–13,17,18 This study supports a number of unique points. First, we showed that +1q was present in 66.8% of the series and is the highest percentage identified to date and may be a consistent feature of Chinese patients and another study identified +1q in 47.8% Chinese patients at diagnosis. 7 Second, the size of the clone exerts important prognostic effects with the adverse prognosis more pronounced in the ⩾50% group. This observation suggests an overall greater contribution towards poorer OS from those with a larger clone, and perhaps a more intermediate risk in those with <50% clone size. Furthermore, we showed that the adverse impact of +1q could be partially mitigated by the use of ASCT.9,10
The assignment of adverse prognosis can be enhanced using the known associations of +1q. One prior study showed a greater adverse impact on prognosis of >3 copies (amplification (amp) 1q) than 3 copies (gain 1q) on progression free survival in the presence HR FISH. 9 The adverse impact of amp 1q was not overcome by proteasome inhibitor-based induction regimen followed by ASCT in prospective trials.19–22 One of the ‘double hit’ groups defined as >3 copies of 1q21 together with ISS3, was shown to severely impact survival defining an ultra-high-risk group. 13 Our data showed comparable OS and EFS in patients with gain or amp 1q of 1q21 similar to some previous reports.7,8,10 The effect of clone size on prognosis is a more complex issue. The cut off for +1q positivity is variable, and there is no universal definition for high or low clone sizes.7,8,10,17 Among studies using the same definition of high (⩾50%) and low clone size (20–49%) including ours herin7,17 that addressed the effect of clone size, there was no significant impact of clone size on survival.
Adverse cytogenetic subgroups include t(4;14), t(14;16), or del(17p) 23 and have been incorporated into the R-ISS. 1 This staging system could be improved by adding additional prognostic FISH data including 1q21 gain.24,25 An increasing number of abnormalities have been associated with adverse prognosis and we confirm this observation in our data set of Chinese patients.12,25 As the number of cases in our study were small, our preliminary data herein calls for international collaboration to verify the utility of our scoring system. Recently, the Intergroupe Francophone du Myelome identified six statistically relevant cytogenetic abnormalities: del(17p), del(1p32), gain 1q, t(4;14), trisomy 21, and trisomy 5, of which trisomy 5 was protective. A weighted cytogenetic scoring system with three prognostic groups were proposed, including low risk (score ⩽ 0), intermediate risk (0–⩽1), and high risk (>1). Therefore, additional cytogenetic alterations, including +1q, appeared important apart from the IMWG-defined high-risk cytogenetics. 26
Despite being retrospective in nature, the case series had uniform induction with a bortezomib-based regimen majority comprising of bortezomib-based triplet, and prolonged follow-up. Notably, our data set is a representative cohort of Chinese MM patients as indicated by the expected survival impact of ISS.
In summary, +1q is an independent adverse risk factor for OS in MM among patients receiving a uniform bortezomib-based induction, attributed by a shorter post-relapse/progression survival, hence more resistant disease. However, the adverse impact of +1q could be partially mitigated by the use of ASCT. Finally, addition of +1q may enable development of a new risk-scoring system.
Supplemental Material
sj-docx-1-tah-10.1177_20406207221082043 – Supplemental material for The impact of bortezomib-based induction in newly diagnosed multiple myeloma with chromosome 1q21 gain
Supplemental material, sj-docx-1-tah-10.1177_20406207221082043 for The impact of bortezomib-based induction in newly diagnosed multiple myeloma with chromosome 1q21 gain by Hoi Ki Karen Tang, Chi Yeung Fung, Gareth J. Morgan, Shaji Kumar, Lisa Siu, Ho Wan Alvin Ip, Sze Fai Yip, Ka Ngai Harry Lau, Chi Kuen Lau, Harold Lee, Kwan Hung Leung, Bonnie Kho, Howard Wong, Cheong Ngai, Yu Yan Hwang, Joycelyn Sim, Yok Lam Kwong and Chor Sang Chim in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The authors thank Hong Kong Society of Myeloma for facilitating inter-hospital collaboration.
Author contributions
Conflict of interest statement
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics statement
The retrospective study was approved by the Institutional Review Broad of the University of Hong Kong/Hospital Authority Hong Kong West Cluster (HKU/HA HKW IRB, IRB Reference Number: UW 20-600).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
