Abstract
Introduction:
Donor lymphocyte infusion (DLI) is one of the effective options for post-transplant disease control of myelodysplastic syndrome (MDS). Its success or failure depends on the induction of antitumor immune reactions, durability of clinical responses, and severity of unwanted toxicities mainly from graft-versus-host disease (GVHD).
Methods:
By analyzing 61 patients receiving DLI for post-transplant MDS relapse, we assessed treatment outcomes and affecting factors, especially focusing on the level of relapse (hematological, molecular, and imminent relapse).
Results:
The response rate (42.1%, 36.4%, 72.7%), and overall survival (OS) at 2 years (27.8%, 45.5%, 70.1%) were different for each relapse level with imminent relapse group showing the most promising results. For OS, response to DLI or pre-DLI chemotherapy, and time to relapse were independent prognostic factors. Meanwhile, post-DLI GVHD and time to relapse were independently predictive for DLI response; post-DLI GVHD was predictive for DLI response, but not for OS, suggesting a potential detrimental impact of GVHD on survival. The incidence of GVHD and GVHD-related deaths were 37.7% and 10.0%, respectively, and CD3+ cell doses triggering GVHD tended to be lower in cases with haploidentical donor or imminent relapse.
Conclusion:
Despite being limited by small number of cases and its retrospective nature, this study again demonstrated the therapeutic effects of DLI in relapsed MDS, and that earlier detection and intervention at lower level relapse might possibly be associated with better results. Furthermore, we propose that tailored cell dosing schedule based on relapse level and donor source may be helpful in minimizing fatal GVHD.
Keywords
Introduction
Myelodysplastic syndrome (MDS) is a heterogeneous group of clonal hematopoietic stem cell disorders characterized by ineffective hematopoiesis in bone marrow, peripheral blood cytopenia, and variable risk of progression to acute myeloid leukemia (AML). 1 Although several medications, such as azacitidine, decitabine, lenalidomide, and luspatercept, have proven to offer benefit to a subset of MDS patients in recent decades,2–5 none of them have demonstrated a curative potential. Allogeneic hematopoietic stem cell transplantation (allo-SCT) is still an only curative option for patients with MDS6–8 and has been increasingly performed even in elderly patients mainly due to reduced transplant toxicities, which resulted from development of conditioning regimens having reduced intensity or toxicity and advances in graft-versus-host disease (GVHD) prophylaxis and antimicrobial agent.9–12 While there is improvement in treatment-related mortality (TRM), relapse after treatment with allo-SCT presents a major challenge; approximately 30–40% of patients experience relapse resulting in dismal outcomes of allo-SCT treatment.13–15
There have been several studies on therapeutic interventions for post-transplant MDS relapse,13,15– 19 including intensive chemotherapy, hypomethylating agents (HMAs), and immunotherapy involving donor lymphocyte infusion (DLI) and second allo-SCT. These treatment options have been variably employed alone or in combination based on the general approach for relapsed hematological maligances.20–22 Until now, no consensus has been reached on selection, combination, and sequence of each treatment modality for post-transplant MDS relapse.
Since its promising results via enhanced graft-versus-leukemic (GVL) effect in chronic myeloid leukemia,23–25 DLI has been employed in other hematologic malignancies and currently constitutes backbone of treatment of MDS relapsing after SCT.13,16,18,19 However, information on DLI specific for MDS is limited; most of the previous studies included both MDS and de novo AML, and many investigators focused on the efficacy of other therapeutics such as HMA, rather than DLI.13,15,18,19 Furthermore, a study focusing on DLI tried in-depth analysis, 16 but the information provided seems to be outdated. Therefore, there is need for more DLI data specific to MDS not just because of its scarcity but also to help reflect on the changes in transplantation process over time, such as increasing use of human leukocyte antigen (HLA) mismatched donors and early detection of lower level relapse via monitoring of minimal (measurable) residual disease (MRD). This prompted us to reexamine the efficacy and toxicities of DLI and attempted to identify prognostic factors for DLI response and survival.
In this study, we analyzed 61 consecutive MDS and secondary AML patients receiving DLI for the control of post-SCT relapse. The objective of the current work was to gain insights into DLI outcomes and relevant factors in these patients. In addition, we took a closer look at DLI outcomes in association with type of relapse and donors, with an aim to build a basic idea for individualized risk-adaptive DLI strategies.
Methods
Patient selection
Adult patients (age ⩾ 18 years) diagnosed according to 2016 World Health Organization (WHO) criteria, and receiving allo-SCT for MDS and related diseases at the Seoul St. Mary’s Hematology Hospital between November 2009 and February 2019 were screened, and all the patients receiving DLI for post-transplant relapse were selected. A total of 61 patients were selected and their median age at the time of transplantation was 48 years (range, 20–68). Diagnosis according to 2016 WHO criteria before SCT were MDS in 40 (65.6%), chronic myelomonocytic leukemia in 7 (11.5%), and secondary AML from MDS in 14 cases (22.9%). Among them, 13 patients (21.3%) had poor or very poor karyotype based upon Revised International Prognostic Scoring System (IPSS-R). Before relapse, 19 (31.1%) and 7 (11.5%) patients had acute (aGVHD) and chronic GVHD (cGVHD), respectively, in which aGVHD of overall grade 2 or more and cGVHD of moderate or severe severity occurred in 14.8% and 4.9%, respectively. Characteristics of patients, diseases, and transplantation are listed in Table 1. Detailed comparison of the baseline characteristics according to relapse level before DLI is demonstrated in Supplementary Table 1. This study was approved by the Institutional Review Board and was conducted according to the Declaration of Helsinki. The reporting of this study conforms to the STROBE statement. 26
Disease, transplantation, and relapse characteristics (n = 61).

Very low risk, low risk, and intermediate risk by IPSS-R.
High and very high risk by IPSS-R.
ATG, anti-thyomocyte globulin; CMML, chronic myelomonocytic leukemia; GVHD, graft-versus-host disease; HLA, human leukocyte antigen; IPSS-R, Revised International Prognostic Scoring System; MDS, myelodysplastic syndrome; MDS-EB-1, MDS with excess blasts-1; MDS-EB-2, MDS with excess blasts-2; MDS-MLD, MDS with multilineage dysplasia; NIH, National Institutes of Health.
Types of relapse and their definitions
Relapses were categorized based on relapse level as hematological relapse (HemRel), molecular relapse (MolRel), and imminent relapse (ImmRel), and each was defined by the following criteria; HemRel: increase of blast ⩾ 5% in bone marrow, appearance of blasts in peripheral blood, extramedullary involvement, or reappearance of dysplastic features meeting the criteria of MDS diagnosis:13,19,27 MolRel: reappearance of disease-specific chromosomal aberrations by conventional karyotyping13,19 or WT1 transcript level > 250 copies/104 ABL after consecutive measurements without evidence of HemRel,28,29 ImmRel: loss of full donor chimerism (⩽95%) accompanied with the occurrence of cytopenias which was not associated with GVHD, infection, or drug toxicities.6,13,30,31 Donor chimerism was measured through DNA short tandem repeats analysis32–34 and WT1 transcript levels from bone marrow samples were determined by real-time quantitative polymerase chain reaction (PCR) using the WT1 ProfileQuant kit from Ipsogen (Marseille, France).35,36
Treatment strategies according to relapse types
All patients had stopped taking an immunosuppressive agent if they were still on the treatment at the time of relapse, thereafter, therapeutic options were chosen according to the type of relapse and the availability of donor lymphocytes. In case of HemRel or MolRel, chemotherapy followed by DLI was considered, while for ImmRel, treatment with DLI was first considered. If chemotherapy was indicated, HMA was considered first whenever available, but intensive chemotherapy was considered for patients with post-transplant AML. Second allo-SCT was also a treatment option and decided by the treating physician’s discretion and a patient’s choice. DLI schedule adopted escalating-dose scheme (first DLI 1 × 106 → second DLI 1 × 107 → third DLI 5 × 107 or higher CD3+ T cells/kg) at 1- to 2-month intervals without GVHD prophylaxis. If GVHD occurred, next DLI dose schedule was stopped and GVHD was managed using standard protocol.
Response and survival assessment following DLI
Response to DLI was assessed in accordance with previous studies,13,18,19,30 but with some modifications. And it was defined differently depending on the type of relapse as follows. For HemRel, achieving of any of following was considered to be responsive; (1) marrow CR or CR using the 2006 International Working Group criteria 37 if the patients had blasts ⩾ 5% in bone marrow at relapse, (2) disappearance of extramedullary involvement, and (3) disappearance of specific disease relapse features that meet the criteria of MDS diagnosis. For MolRel, disappearance of chromosomal aberrations or normalization of WT1 level to <250 copies/104 ABL was considered as a response. In case of ImmRel, restoration of chimerism to >95% and hematologic reconstitution were considered as a response. When DLI was given to those who had responded to pre-DLI chemotherapy, maintenance of preexisting response for >6 months after DLI 13 was defined as response to DLI. When the delayed response during overlapped treatment with HMA and DLI occurred in association with newly developed GVHD or persisted more than 6 months, it was regarded as DLI response.
Overall survival (OS) was analyzed as the final outcome of DLI, for which, an event was death from any cause, with any patients alive censored at the last follow-up or at the time of second allo-SCT, and was accounted for from the starting date of the initial DLI. TRM caused by DLI was only defined in patients whose disease was not evident at the time of death.
Statistics
The data were analyzed based on information available as of June 2020. The frequencies and distribution of clinical features involving disease, transplant, relapse, and DLI characteristics were demonstrated using descriptive statistics. Differences between groups were calculated using the chi-square test or Fisher’s exact test for categorical variables and a two-sample t-test or Mann–Whitney U test for continuous variables, and the p value was corrected by Bonferroni’s method when multiple testing was indicated. If the comparison between more than two groups was performed, one-way analysis of variance (ANOVA) or Kruskal–Wallis test was used for the continuous variables. The OS was estimated by the Kaplan–Meier method, and compared by the log-rank test. Cumulative incidence of GVHD was analyzed in a competing risk framework by using the cumulative incidence of competing events. Univariate and multivariate logistics were performed in assessment of affecting factors for DLI response, and Cox proportional-hazard regression models were used for identifying risk factors for OS. In this process, occurrence of GVHD after DLI was treated as a time-varying covariate. Possible variables affecting the post-DLI outcomes were screened in the univariate analysis first, and any variable significant at the level of <0.1 (p value) was put in the multivariate analysis. Two-sided p values less than 0.05 were considered statistically significant. All statistical analyses were conducted using IBM SPSS statistics version 25 and EZR software version 1.40.
Results
Characteristics of relapse and DLI
Relapse occurred at a median time of 7.2 months after allo-SCT (range, 0.4–80.7), where the initial relapse type was HemRel, MolRel, and ImmRel in 33 (54.1%), 16 (26.2%), and 12 (19.7%) patients, respectively, and each 5 and 1 of MolRel and ImmRel progressed to HemRel before DLI (Table 2). A total of 127 cycles of DLI were given, and patients received a median of 2 cycles (range, 1–4 cycles), and the sum of CD3+ cells per patient ranged from 1.0 to 200.0 × 106 cells/kg with a median dose of 21 × 106 cells/kg. In 27 patients, DLI was the only treatment for the post-SCT relapse (44.3%), whereas remaining 34 patients received HMA (n = 21, 34.4%) or intensive chemotherapy (n = 13, 21.3%) prior to DLI. Twenty-one cases (34.4%) showed no response to pre-DLI chemotherapy and they were grouped as ‘refractoriness’, while the remaining 40 patients (65.6%) were ‘no refractoriness’.
DLI characteristics and outcomes.

Bold value indicates statistically significant value (two-sided p<.05).
Response information was not available in one patient whose death occurred before DLI response assessment.
Statistical difference of DLI response duration by relapse type.
Statistical difference of OS by relapse type.
DLI, donor lymphocyte infusion; HemRel, hematological relapse; ImmRel, imminent relapse; MolRel, molecular relapse; OS, overall survival.
Response to DLI and related factors
Among 60 patients evaluable for response assessment after excluding one early death, 28 (46.7%) achieved response to DLI, while 32 (53.3%) failed. The characteristics of relapse type, pre-DLI chemotherapy, and their refractoriness were not different between DLI responders and non-responders (Table 2). For DLI responders, a median of 2.5 cycles (range, 1–3) and of 11 × 106/kg (range, 1–111 × 106/kg) CD3+ cells were given, and the median duration of response was 15.5 months (range, 0.4–52.6). Response rate in HemRel, MolRel, and ImmRel was 42.1%, 36.4%, and 72.7% (p = 0.451), respectively, and response duration according to relapse type was 8.4 versus 16.4 versus 22.8 months (p = 0.098). In univariate analysis, the worst WHO diagnosis and karyotype before transplantation, time to relapse, and GVHD occurrence after DLI were factors that determined DLI response (Table 3). Multivariate analysis to identify independent factors showed that poor/very poor karyotype had a trend toward worse response (p = 0.057, hazard ratio (HR) = 0.045 (95% confidence interval (CI) 0.002–1.092)), while longer time interval between SCT and relapse and the occurrence of post-DLI GVHD increased the chances of achieving DLI response (p = 0.039, HR = 1.049 (95% CI 1.002–1.097); p = 0.006, HR = 14.10 (95% CI 2.137–92.91), respectively). As the most powerful predictor for DLI response, association of post-DLI GVHD with DLI response was as follows: 17 out of 22 patients (77.3%) with GVHD attained response whereas 11 out of 38 patients (28.9%) without GVHD responded to DLI (p < 0.001).
Prognostic factors for DLI response and survival: Univariate and multivariate analysis.

Bold value indicates statistically significant value (two-sided p<.05).
GVHD occurrence after DLI was treated as a time-varying covariate.
AML, acute myeloid leukemia; CI, confidence interval; CMML, chronic myelomonocytic leukemia; DLI, donor lymphocyte infusion; GVHD, graft-versus-host disease; HLA, human leukocyte antigen; HR, hazard ratio; IPSS-R, Revised International Prognostic Scoring System; MAC, myeloablative conditioning; MDS, myelodysplastic syndrome; OR, odds ratio; RIC, reduced intensity conditioning; SCT, stem cell transplantation.
Survival and affecting factors
With a median follow-up period of 26.7 months for survivors, OS rate at 2 years were 38.5%, and median OS was 12.0 months (Figure 1(a), Table 2). The median OS differed by relapse type before DLI: the median OS of 7.0 months, 21.0 months, and not reached in patients with HemRel, MolRel, and ImmRel (p = 0.023, Figure 1(b)). Table 3 shows the factors affecting OS. Longer duration from allo-SCT to relapse was independently advantageous for OS (p = 0.029, HR = 0.972 (95% CI 0.948–0.997)), and response achievement to DLI was favorable for longer survival (p = 0.001, HR = 0.170 (95% CI 0.060–0.486)). In contrast, treatment refractoriness to pre-DLI chemotherapy was found to be an independent prognostic factor for poor survival rates (p = 0.011, HR = 3.083 (95% CI 1.298–7.321)). Differences in OS according to these independent factors are shown in Figure 1(c) and (d). Subgroup analysis showed that favorable effects of DLI response on survival was evident regardless of response/refractoriness to pre-DLI chemotherapy (Supplementary Figure 1).

(a) Overall survival after donor lymphocyte infusion (DLI) in overall patients. Comparison of overall survival according to (b) relapse type, (c) pre-DLI chemotherapy response, and (d) DLI response.
Toxicities of DLI and related factors
Post-DLI GVHD was observed in 23 of the 61 patients (37.7%) (Table 4). Overall, the acute and chronic GVHD after DLI was observed in 24.6% and 14.8% of patients, respectively, and 1 patient developed both acute and chronic GVHD. All cases of aGVHD (n = 15) manifested as grade 2 or more, and 6 out of 9 cGVHD cases (66.7%) were moderate or severe by National Institutes of Health (NIH) consensus criteria. Onset of GVHD was noticed after a median of two cycles of DLI (range, 1–3) and the median dose of CD3+ cells before GVHD was 15.0 × 106/kg (range, 1–200 × 106/kg). The median interval between the last DLI and onset of GVHD was approximately 31.0 days (range, 6–108 days).
DLI-induced GVHD and related factors.

Bold value indicates statistically significant value (two-sided p<.05).
One patient experienced both aGVHD and cGHVD at a distance of time (total 24 cases of GVHD in 23 patients).
Treatment-related mortality (TRM) caused by DLI was only defined in patients whose disease was not evident at the time of death, and death before response assessment (n = 1) was not included for calculation.
DLI, donor lymphocyte infusion; GVHD, graft-versus-host disease; HemRel, hematological relapse; ImmRel, imminent relapse; MolRel, molecular relapse.
The cumulative incidence of GVHD over time appeared to be higher in patients who received DLI from haploidentical donors than in patients with matched sibling donor or unrelated donor (p = 0.071) (Figure 2). Furthermore, CD3+ cell doses triggering GVHD tended to be different according to the type of donor and the type of relapse although the differences were not statistically significant. Median CD3+ cells of 46.0 × 106/kg, 57.5 × 106/kg, and 6.0 × 106/kg doses were administered in patients with matched related, unrelated, and haploidentical donor, before the occurrence of post-DLI GVHD (Table 4) (p = 0.219). The median CD3+ doses inducing GVHD by type of relapse were as follows: 45.5 × 106/kg, 26.0 × 106/kg, and 11.0 × 106/kg in HemRel, MolRel, and ImmRel, respectively (p = 0.752). Time to occurrence of GVHD following the last DLI did not differ with relapse type. When focusing on donor-recipient sex, female donor to male recipient cases were associated with increased risk of GVHD development after DLI (p = 0.012).
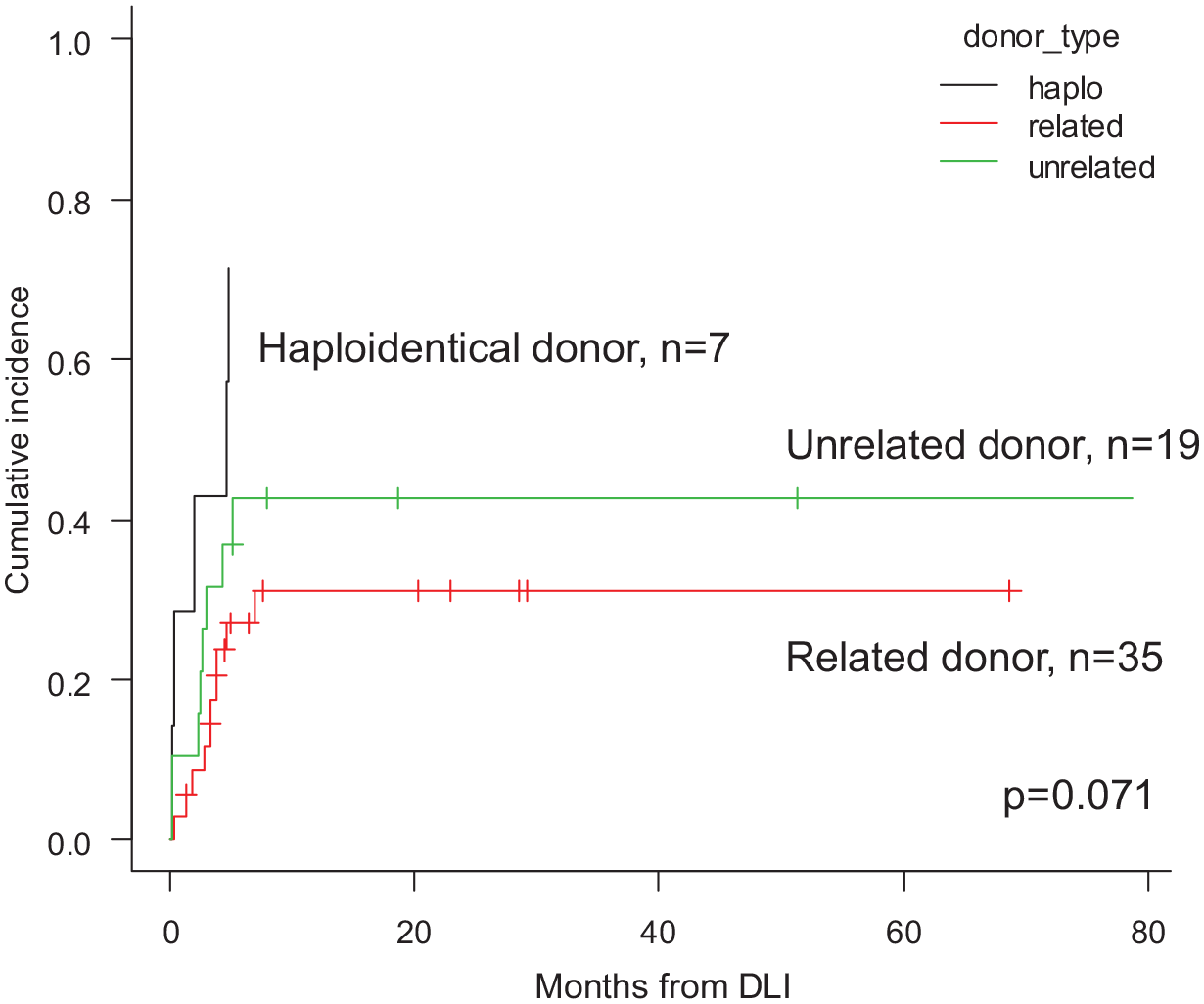
Cumulative incidence of post-DLI graft-versus-host disease (GVHD) according to donor type. (GVHD indicates either acute or chronic GVHD after DLI, whatever comes first).
Out of the 60 patients assessable for disease status at the last follow-up, there were 7 cases (11.7%) of DLI-related TRM; one death occurred due to thrombotic microangiopathy of uncertain cause after DLI, while six deaths were caused by GVHD with or without subsequent infection.
Discussion
In this study, we retrospectively analyzed DLI outcomes in relapsed MDS and related diseases. The response rate was 46.7%, and OS at 2 years was 38.5%. Favorable risk karyotype, longer relapse interval after allo-SCT, and GVHD occurrence after DLI were favorable factors for response achievement. As for post-DLI survival, longer relapse interval after allo-SCT, no refractoriness to pre-DLI chemotherapy, and response to DLI significantly contributed to better survival rates. The incidence of GVHD after DLI was 37.7%, and GVHD-related death was observed in 10.0% of the patients. CD3+ cell doses triggering GVHD were lowest in cases with haploidentical donor or ImmRel.
In line with our results, the importance of karyotype as an attributing factor for DLI response was previously demonstrated in a study comprising 11 MDS and secondary AML patients where all the 6 patients with complex karyotype did not respond to DLI. 38 The significant impact of interval between SCT and relapse on post-SCT treatment response and survival has already been revealed in prior studies as well,18,19 although they did not always include DLI as a post-SCT treatment. The burden of disease at relapse as distinguished by HemRel, MolRel, and ImmRel in our study is also important in discriminating post-DLI outcomes. Patients receiving DLI at ImmRel were more likely to respond, and survival of these patients was much better. Similarly, the importance of disease burden has been shown in prior studies, and MDS rather than AML either at relapse or before SCT was related to better survival and higher probability to respond to post-SCT relapse treatment.13,18 In addition, Krishnamurthy and colleagues 16 showed that the estimated OS at 5 years was 80% versus 40% in each, when DLI was used preemptively versus therapeutically for post-transplant relapse among the patients with AML and MDS. This implies that the stringent monitoring of disease status and earlier intervention at lower-level relapse is essential in improving the outcomes of post-SCT relapse.
For earlier detection of relapse, risk-adapted monitoring depending on identified risk factors per patient39–42 would be more effective. Currently, our institutional strategies for identifying relapse-prone cases are based on our own prognostic scoring system for SCT in MDS 42 and bone marrow WT1 level at 1 month post-SCT, 36 and patients with high risk are regularly monitored for their WT1 levels every 3 months. In this study, we used WT1 levels as an indicative marker for lower level relapse, and incorporated its value when defining molecular relapse. Even though current recommendation do not recommend WT1 for assessment of MRD 43 and newer MRD detection technologies including digital PCR and next generation sequencing are emerging,44–47 it still has the advantage of having an ELN-certified assay with a reproducible and validated cut-off of 250 copies for bone marrow and 50 copies for peripheral blood, 35 to discriminate normal and WT1 overexpression. Moreover, the prognostic impact of WT1 in MDS and AML was quiet consistent across different study groups,36,48– 51 and several investigators used WT1 level as a trigger for making a prompt decision to start preemptive or prophylactic therapy.28,29 Accordingly, we suggest that it may serve as one of the alternatives unless there is a better way to detect post-SCT relapse at an earlier time point, especially in MDS.
Regarding GVHD after DLI, there were more patients experiencing GVHD among the DLI responders than among the non-responders, and in addition, GVHD was found to be an independent prognosticator for DLI response. However, the favorable impact of GVHD on DLI response was offset in several patients, suggesting detrimental impact of GVHD on survival. Actually, there were 6 (21.4%) GVHD-related deaths among the 28 DLI responders, and a total of 9 deaths (14.8%) among the 61 patients were directly linked to GVHD occurrence regardless of their disease status at the last follow-up (n = 6, death without disease; n = 2, death with disease; n = 1, death at unknown disease state). These findings indicated that the beneficial impact of GVL effect can be maximized by preventing unwanted GVHD, which might be particularly important in the setting of lower-level relapse. 52
In this regard, we attempted to find hints to minimize fatal GVHD with closer look at the type of donor and type of relapse of this cohort. In the aspects of donor types, we observed that the GVHD cases tended to be more frequently noticed and the median CD3+ doses inducing GVHD seemed to be lower in patients receiving DLI from haploidentical donors than in the other patients. In addition, from a view of relapse level, we observed that CD3+ cell doses inducing GVHD were relatively fewer in a case of ImmRel compared with those in HemRel on uniformed escalating-dose DLI scheme, even though our interpretations were substantially limited by a few patients with haploidentical donor and lack of statistical power. From these findings, although not sufficient to conclude, we propose that there could be a need for a different DLI strategy based on the type of donors and relapse as such to preferentially adopt higher CD3+ cell dose scheme for HemRel and the HLA matched cases and vice versa when ImmRel occurs and the HLA disparity is substantial such as in the case of DLI from haploidentical donor. 53 Given that many strategies to separate GVHD from GVL effects have been failed and it would not be easy in real practice, this study may provide a small clue regarding the dosing schedule according to the type of relapse and donors. In addition, our recent work that demonstrates the importance of activation status of dendritic cells upon GVHD and GVL effect rather than number of cells of DLI may provide guidance in separating them by optimizing infusion time of DLI after HMA. 54
Finally, for answering the question whether to proceed to DLI would have any value or not if the patients did not respond to pre-DLI chemotherapy, we separately analyzed post-DLI survival among patients with ‘refractoriness’ and ‘no refractoriness’ to pre-DLI chemotherapy. Based on our results, even cases having refractoriness to prior chemotherapy had a chance of responding to DLI and DLI responders showed significantly better OS, which suggest that immunotherapy such as DLI or second allo-SCT should be considered regardless of drug sensitivity.
We acknowledged that there were several limitations in the present study. One of the major weaknesses was retrospective nature of this study. Therefore, monitoring for relapse was not always consistent and the timing of response assessment after intervention varied with individuals as well. In addition, a small sample size, including only a few patients who received allo-SCT and DLI from haploidentical donors, potentially led a weakened statistical power and thus made it hard to draw solid conclusion regarding different DLI strategies based on donor types. Regarding the mechanisms of DLI response, the role of mismatched HLA loss or dysregulation of immune-related pathway including MHC class II down regulation in leukemia relapse,55–58 which could also possibly influence on DLI response, was not analyzed in this study but needs to be elucidated in the future. Nevertheless, our presents study certainly had its own strengths; we met the objective of the study by exclusively focusing on DLI rather than other chemotherapeutics such as HMA, in the setting of post-SCT relapse, and the analysis was specifically planned for patients with MDS rather than de novo AML; previous information of DLI had been drawn from mixed cohorts of AML and MDS, where de novo AML comprised majority. In addition, in the present study, patients were treated with the same strategy at a single institution, which indicates homogeneity of this cohort.
In summary, this study again showed that DLI can be used as salvage or preemptive option for post-transplant relapse in patients with MDS and secondary AML from MDS. The survival benefit of DLI could possibly be maximized with earlier detection of relapse at lower disease burden; and an effort not to induce fatal GVHD after DLI is critically required, for which to adopt an individualized DLI strategy based on relapse level and donor source seems to be reasonable. However, due to the limitations from a few patients’ number and an insufficient statistical power, further analysis in a larger cohort as well as prospective studies are necessary to confirm our findings and to achieve a better understanding of DLI in MDS patients.
Supplemental Material
sj-pptx-1-tah-10.1177_20406207211043748 – Supplemental material for Differential effects of donor lymphocyte infusion upon treatment response and GVHD according to relapse level and donor sources in patients with myelodysplastic syndrome
Supplemental material, sj-pptx-1-tah-10.1177_20406207211043748 for Differential effects of donor lymphocyte infusion upon treatment response and GVHD according to relapse level and donor sources in patients with myelodysplastic syndrome by Silvia Park, Tong Yoon Kim, Jong Hyuk Lee, Joon yeop Lee, Gi June Min, Sung Soo Park, Seung-Ah Yahng, Seung-Hwan Shin, Jae-Ho Yoon, Sung-Eun Lee, Byung Sik Cho, Ki-Seong Eom, Seok Lee, Hee-Je Kim, Chang-Ki Min, Jong Wook Lee and Yoo-Jin Kim in Therapeutic Advances in Hematology
Footnotes
Author contributions
YJK conceived the idea and designed the study. SP collected and analyzed data, and SP and YJK wrote the main manuscript text. SP prepared figures and tables including supplementary materials. Remaining authors contributed by providing study material, and all authors reviewed the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2019R1C1C1008917). This study was also supported by Research Fund of Seoul St. Mary’s Hospital, The Catholic University of Korea (ZC18SISI0523).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
