Abstract
While survival rates in paediatric acute lymphoblastic leukaemia (ALL) nowadays exceed 90%, systemic ALL relapse, especially after haemopoietic stem cell transplantation (HSCT), is associated with a poor outcome. As there is currently no standardized treatment for this situation, individualized treatment is often pursued. Exemplified by two clinical scenarios, the aim of this article is to highlight the challenge for treating physicians to find a customized treatment strategy integrating the role of conventional chemotherapy, immunotherapeutic approaches and second allogeneic HSCT. Case 1 describes a 2-year-old girl with an early isolated bone marrow relapse of an infant KMT2A-rearranged B-cell precursor ALL after allogeneic HSCT. After bridging chemotherapy and lymphodepleting chemotherapy, chimeric antigen receptor (CAR) T-cells (tisagenlecleucel) were administered for remission induction, followed by a second HSCT from the 9/10 human leukocyte antigen (HLA)-matched mother. Case 2 describes a 16-year-old girl with a late, isolated bone marrow relapse of B-cell precursor ALL after allogeneic HSCT who experienced severe treatment toxicities including stage IV renal insufficiency. After dose-reduced bridging chemotherapy, CAR T-cells (tisagenlecleucel) were administered for remission induction despite a CD19- clone without prior lymphodepletion due to enhanced persisting toxicity. This was followed by a second allogeneic HSCT from the haploidentical mother. While patient 2 relapsed around Day + 180 after the second HSCT, patient 1 is still in complete remission >360 days after the second HSCT. Both cases demonstrate the challenges associated with systemic ALL relapse after first allogeneic HSCT, including chemotherapy-resistant disease and persisting organ damage inflicted by previous therapy. Immunotherapeutic approaches, such as CAR T-cells, can induce remission and enable a second allogeneic HSCT. However, optimal therapy for systemic ALL relapse after first HSCT remains to be defined.
Keywords
Introduction
Survival rates in paediatric acute lymphoblastic leukaemia (ALL) nowadays exceed 90%. 1 However, relapsed ALL is associated with a salvage rate of only 55% 2 and is, thus, a leading cause of cancer-related death in children and adolescents.3,4 Early systemic relapses – occurring within the first 12 months after allogeneic haematopoietic stem cell transplantation (HSCT) – have a poor prognosis, with a median survival of 7.4 months 5 and a long-term survival of only 20–30%.6,7 Relapse therapy can be challenging in these patients, as physicians are often confronted with chemotherapy-resistant disease and the burden of acute and long-term toxicity of previous anti-leukaemic treatments. 8 Novel immunotherapeutic approaches, such as blinatumomab, inotuzumab ozogamicin (CMC-544) or chimeric antigen receptor (CAR) T-cell therapy (e.g. tisagenlecleucel), may improve outcomes. 5 CAR T-cell therapy by itself has the potential to induce durable remissions, while antibody-based therapies are usually followed by cellular therapy of any kind to achieve long-term survival. Tisagenlecleucel has been studied primarily as a standalone therapy but also in combination with immune checkpoint inhibitors to augment the response to the CAR T-cells.9–11 Yet, some patients might also benefit from a subsequent allogeneic HSCT.
While current literature mostly focuses on targeted therapies with regard to safety and efficacy and not in deep on the combined therapy elements as a whole, individual patient-related aspects often cannot be considered properly. Patients with pre-existing morbidities are often excluded from phase I/II clinical trials. Furthermore, technical issues during apheresis in low-weight infants might render the production of tisagenlecleucel for remission induction in infant ALL difficult. To the best of our knowledge, only few cases have been reported so far.12,13 Herein, we discuss the challenge of developing customized treatment approaches integrating the role of conventional chemotherapy, immunotherapeutic approaches and second allogeneic HSCT for systemic relapses after a first allogeneic HSCT, exemplified by two different clinical scenarios.
Description of case 1
The first patient we present is a 2-year-old girl with an early, isolated bone marrow (BM) relapse of B-cell precursor (BCP)-ALL after allogeneic HSCT (Figure 1 and Table 1). The girl was diagnosed with an KMT2A-rearranged BCP-ALL (KMT2A-AFF1 gene fusion positive) at the age of 7 weeks. She received treatment according to the Interfant-2006 protocol for high-risk patients (Clinicaltrials.gov registration number: NCT00550992) 14 and underwent an allogeneic HSCT in first complete remission (CR1). At that time, she was negative for minimal residual disease (MRD) as measured by polymerase chain reaction (PCR) of immunoglobulin/T-cell receptor and KMT2A rearrangements. During front-line therapy, the patient had considerable treatment-related toxicity, including an episode of respiratory failure that required mechanical ventilation, repeated septicaemias and hepatopathy with pronounced hyperbilirubinaemia. After chemoconditioning with fludarabine, treosulfan, and thiotepa, the girl received a BM graft from a human leukocyte antigen (HLA)-matched unrelated donor, matched at 10 out of 10 loci. Graft-versus-host disease (GvHD) prophylaxis consisted of anti-thymocyte globulin, cyclosporine A and methotrexate. Further serious infectious complications occurred in the pre-engraftment phase, including an extensive soft tissue infection at the implantation site of her percutaneous endoscopic gastrostomy tube. This required broad-spectrum antibiotics and granulocyte transfusions. Neutrophil engraftment in the peripheral blood (PB) was documented on Day + 19. BM puncture on Day + 26 revealed molecular remission (MRD negative by both immunoglobulin/T-cell receptor- and KMT2A-based targets) with full donor chimerism.
Patient and disease-related data for both cases.

6-MP: 6-mercaptopurine; ALL: acute lymphoblastic leukaemia; ATG: anti-thymocyte globulin; BCP: B-cell precursor; CAR: chimeric antigen receptor; CNS: central nervous system; CPM: cyclophosphamide; CRS: cytokine release syndrome; CSA: cyclosporine A; FORUM: For Omitting Radiation Under Majority; GvHD: graft-versus-host disease; HLA: human leukocyte antigen; HSCT: haematopoietic stem cell transplantation; MRD: minimal residual disease; MTX: methotrexate; MUD: matched unrelated donor; PEG: percutaneous endoscopic gastrostomy; TBI: total body irradiation; VP-16: etoposide.

Timeline of case 1.
The patient had a CD19+ (>99% CD19 expression), isolated BM relapse on Day + 180 after allogeneic HSCT. After having a multidisciplinary leukaemia and cellular therapy board session, we opted for tisagenlecleucel as remission induction therapy followed by allogeneic HSCT. Given the patient’s body weight (10 kg), apheresis was performed via the AMICUS® system using allogeneic donor erythrocytes for machine priming. 148 × 106/kg CD3+ cells were harvested, resulting in a final product of 3.26 × 106/kg CAR T-cells. As bridging therapy prior to tisagenlecleucel, the patient received F1 and F2 courses according to the ALL-relapse study of the Berlin-Frankfurt-Münster Group (ALL-Relapse BFM 2002) (Clinicaltrials.gov registration number: NCT00114348) 15 with adjacent lymphodepletion (fludarabine, cyclophosphamide).
During bridging therapy, the patient had another soft tissue infection. Nevertheless, she remained MRD positive by PCR at a level of 7 × 10–1. Tisagenlecleucel was tolerated well; the patient did not develop cytokine release syndrome (CRS) or immune effector cell-associated neurotoxicity syndrome (ICANS). Molecular remission in the BM was achieved on Day + 21. The patient proceeded to a second allogeneic HSCT using PB stem cells from her mother (who was HLA matched at 9 out of 10 loci) after fractionated total body irradiation (TBI) to a total dose of 8 Grey (Gy) and etoposide (60 mg/kg). GvHD prophylaxis consisted of post-transplant cyclophosphamide, tacrolimus, and mycophenolate mofetil. Toxicities of the second allogeneic HSCT included another soft tissue infection and mild veno-occlusive disease.
Engraftment in the PB was achieved on Day + 23. BM on Day + 32 showed complete donor chimerism with negative MRD by PCR. Immunosuppression was tapered and withdrawn on Day + 39 at complete absence of acute GvHD. Molecular remission and complete donor chimerism were confirmed on Day + 60, and Day + 100, and day +180.
Description of case 2
The second case we present is a 16-year-old girl with a late, isolated BM ALL relapse after allogeneic HSCT (Figure 2 and Table 1). The patient was initially treated according to the Associazione Italiana di Ematologia ed Oncologia Pediatrica (AIEOP)-BFM ALL 2009 protocol (Clinicaltrials.gov registration number: NCT01117441) and was transplanted from an unrelated donor matched at 9 out of 10 loci in CR1 after administration of blinatumomab. The latter was used to lower her pre-transplant MRD and led to an MRD load of 2 × 10–4 prior to allogeneic HSCT. Conditioning consisted of 12 Gy TBI in combination with etoposide (60 mg/kg). BM was used for the graft. Pre- and post-transplant organ toxicities included severe necrotic pancreatitis with concomitant stomach perforation and polyomavirus BK-associated haemorrhagic cystitis requiring prolonged antiviral treatment. After allogeneic HSCT, the patient developed acute GvHD of the skin (grade IV), which responded well to multimodal immunosuppressive therapy. Nephrotoxic side effects of both immunosuppressive and antiviral treatment resulted in stage IV renal insufficiency (glomerular filtration rate 20 mL/min/1.73 m2; serum creatinine 2.0–2.5 mg/dL).
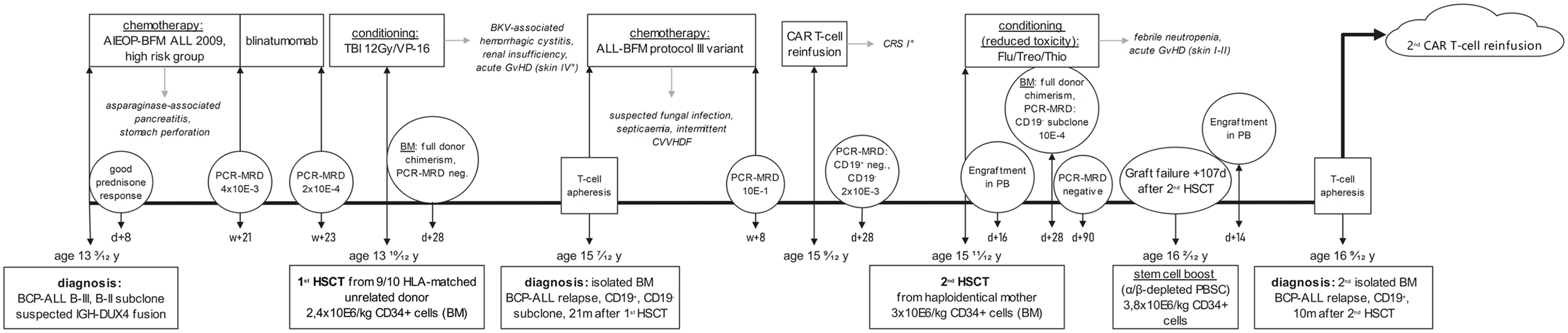
Timeline of case 2.
After successful haematopoietic engraftment with full donor chimerism at Day + 28, the BM at 21 months after allogeneic HSCT revealed a late, isolated BM relapse (partially CD19 +, CD22 +). Despite the CD19- subclone (49% of the cells), the multidisciplinary leukaemia and cellular therapy board opted for CAR T-cell therapy with tisagenlecleucel as an attempt for remission induction therapy followed by allogeneic HSCT.
While awaiting CAR T-cell production, the patient received bridging chemotherapy derived from the ALL-BFM protocol III variant (Clinicaltrials.gov registration number: NCT00430118) 16 under continuous veno-venous haemodiafiltration (CVVHDF). MRD as measured by PCR remained at a level of 1 × 10–1. During chemotherapy, the patient developed multiple pulmonary lesions with pleural effusion and both hepatic and intracerebral lesions, which were considered of fungal origin and finally treated with isavuconazole. Due to rapid deterioration of the patient’s general condition, tisagenlecleucel was given without prior lymphodepleting chemotherapy. It was well-tolerated with only grade I CRS and no neurotoxicity. Follow-up imaging showed clearance of the hepatic and pulmonary lesions; the intracerebral lesions remained unchanged. BM puncture on Day + 28 showed molecular remission of the CD19+ clone; however, as expected, the CD19- clone persisted with an MRD level of 2 × 10–3 by PCR.
The presence of the CD19- clone led us perform the second allogeneic HSCT very soon after tisagenlecleucel infusion (Day + 59), using the patient’s haploidentical mother. Due to stage IV renal insufficiency, the patient received a reduced-intensity dose-adjusted chemoconditioning with fludarabine, treosulfan and thiotepa under concomitant CVVHDF. Chemoconditioning was a reduced-toxicity regimen based on the For Omitting Radiation Under Majority age (FORUM) study in paediatric ALL. 17 Post-transplant cyclophosphamide, mycophenolate mofetil and tacrolimus were used as GvHD prophylaxis. Overall, the second allogeneic HSCT was well-tolerated. BM on Day + 28 showed full donor chimerism; however, the CD19- leukaemic clone persisted with an MRD level of 1 × 10–4 by PCR. Thus, immunosuppression was reduced to the lowest possible level. Three months after allogeneic HSCT, MRD by PCR in the BM was negative for the first time.
Secondary graft failure prompted us to administer a stem cell boost (CD3/19 and α/β depleted PB stem cells) on Day + 107, leading to stable engraftment. Unfortunately, a second isolated BM relapse of the patient’s CD19+ BCP-ALL occurred on Day + 180. After a profound discussion with the patient and her family regarding her dismal prognosis, a mutual decision for another CAR T-cell infusion, using allogeneic T-cells transplanted at the second HSCT, was reached and induced a third CR at the time of writing.
Discussion
Treatment options for systemic ALL relapse after allogeneic HSCT include polychemotherapy,18,19 immunotherapeutic approaches,20–23 donor lymphocyte infusions24,25 and second allogeneic HSCT.6,26–28 As there is currently no standard therapy for this situation and, thus, posing a therapeutic dilemma to the medical treating team, individualized treatment is often pursued.21,28 To the best of our knowledge, this report is the first which directly compares the efficacy of tisagenlecleucel for remission induction in two children with relapsed ALL after their first HSCT, who represent both ends of the paediatric age spectrum and their specific challenges. Especially data on the use of tisagenlecleucel in infant ALL are scarce for various reasons. Meeting the specified cell dose targets for successful production of CAR T-cells can be challenging in low-weight infants. 29 Thus far, only few cases have been reported.12,13
General considerations
Kuhlen et al. 6 showed that paediatric patients with ALL relapse after first allogeneic HSCT seem to fare better if salvage therapy includes a second allogeneic HSCT (3-year event-free survival of 33.9%). Notably, in that trial there was some selection bias, given that the children who were eligible for second allogeneic HSCT presented with therapy-sensitive relapse. As a MRD level >10–4 is associated with a significantly higher risk for relapse after allogeneic HSCT15,17,30–32 pre-transplant reduction of disease burden to the lowest possible MRD is pivotal.
For CD19+ ALL relapse, immunotherapeutic approaches include monoclonal antibodies such as blinatumomab (a first-in-class bispecific T-cell engager) and CD19-directed CAR T-cell therapy. 6 For CD22+ ALL relapse, apart from epratuzumab which is not available anymore, inotuzumab ozogamicin (an antibody–drug conjugate) constitutes an option, taking into consideration the fact that antibody-based therapies are used either for remission induction therapy or reducing MRD during a patient’s disease course, followed by allogeneic HSCT to achieve long-term survival. CAR T-cell therapy represents a possible standalone curative approach without the need for previous CR, which strikingly contrasts to allogeneic HSCT.
Inotuzumab ozogamicin is a monoclonal anti-CD22 antibody linked to the cytotoxic ozogamicin and is, thus, specifically directed to malignant cells. 33 Rytting et al. 34 first described the promising activity of inotuzumab ozogamicin with an acceptable toxicity profile in children with relapsed/refractory ALL, leading to a phase I/II clinical trial of this treatment in this patient group (EudraCT 2016-000227-71). While the risk of veno-occlusive disease should be taken into account, remission induction rates are very impressive, approaching 80% and usually being accompanied by MRD negativity. This suggests using inotuzumab ozogamicin as for remission induction therapy while blinatumomab might have its place as for consolidation therapy elements.
The bispecific T-cell engaging monoclonal antibody blinatumomab is directed to both CD19 on B-cell lymphoblasts and host CD3 cytotoxic T-cells. 9 In adult patients with relapsed/refractory BCP-ALL, single-agent blinatumomab has been shown to significantly improve median overall survival (7.7 months) compared with conventional treatment with chemotherapy (4.0 months). 22 Meanwhile, in paediatric patients with relapsed/refractory BCP-ALL, single-agent blinatumomab was shown to significantly improve CR rates (39% CR after two cycles) 35 compared with conventional therapy (17%), with 52% of the responders achieving MRD negativity. 36 In the paediatric cohort, observed toxicities were mostly cytopenia; grade ⩾ 3 CRS was rare (5%) and associated with a higher disease burden. Neurological toxicities, which are frequently seen in adults, occurred in only 3% of the paediatric cohort. In two recent randomized trials for first ALL relapse, outcome parameters, such as overall and event-free survival rates as well as molecular remission rates, were significantly better in the blinatumomab group compared with standard chemotherapy, thus, making the combination of blinatumomab and allogeneic HSCT the ‘new’ standard of care in first high-risk relapse of CD19+ BCP-ALL.37,38
CAR T-cell therapy with tisagenlecleucel reprograms T-cells to specifically eliminate the host’s CD19+ B cells. Tisagenlecleucel is approved by the European Medicines Agency for the treatment of paediatric and young adult patients with relapsed/refractory CD19+ BCP-ALL and adults with relapsed/refractory mature B-cell non-Hodgkin lymphoma. The phase II ELIANA trial (NCT02435849) showed that a single autologous CAR T-cell infusion after lymphodepletion with fludarabine/cyclophosphamide achieved an overall CR rate of 81% with MRD negativity in all responders.10,11 Successive real-world data indicate comparable initial CR rates of 85.5%, as well as 12-month event-free survival and overall survival rates of 52.4% and 77.2%, respectively. 39 Grade ⩾ 3 adverse events included CRS (16.1% grade ⩾ 3), which is associated with high leukaemic burden, and ICANS (9% were grade ⩾ 3). 39
Case 1
In infant leukaemia, the outcome of relapsed ALL with KMT2A gene rearrangement is very poor – pursuing an allogeneic HSCT has been reported to be an important component of relapse therapy.40,41 Furthermore, a risk of lineage switch to acute myeloid leukaemia has been reported too.42–44 Even though this is a rare event (approximately 1%), affected patients have a very poor prognosis. 43 Consequently, we pursued a second allogeneic HSCT in patient 1, although we considered standalone CAR T-cell therapy as a valuable alternative.
During her primary disease, the patient suffered from severe acute toxicities and presented with resistance to chemotherapy. Further severe infections occurred subsequent to the first allogeneic HSCT, which are a known risk after HSCT from matched related donors in paediatric patients with BCP-ALL due to intensified GvHD prophylaxis. 45 For both reasons, we did not consider prolonged conventional chemotherapy to be a viable option for pre-transplant reduction of leukaemia burden in this patient in order to achieve CR before allogeneic HSCT.
As the patient presented with a CD19+ relapse, we decided to apply CD19-directed immunotherapy. While tisagenlecleucel is probably associated with a higher risk of the patient developing therapy-related complications compared with blinatumomab,11,23,36 its superiority in remission induction with MRD negativity prompted us to administer tisagenlecleucel in our patient.
As the relapse was partially CD22 +, the patient would have been eligible for the clinical trial of inotuzumab ozogamicin mentioned above. However, there was no slot available. Hence, conventional moderate bridging chemotherapy was applied. Similarly, to initial treatment, the patient suffered from severe infections and presented with chemotherapy-resistant disease. Subsequent application of tisagenlecleucel was well-tolerated and resulted in MRD negativity, allowing progression to second allogeneic HSCT. This experience fits to the real-world data on the use of tisagenlecleucel in infant ALL describing an MRD-negative remission induction rate of 69% with only 21% development of CRS of grade >3 and no neurotoxicity. 12
Due to the patient’s lack of response to chemotherapy and the preceding application of age-appropriate chemoconditioning prior to first allogeneic HSCT, an individualized conditioning regimen was discussed. Considering the patient’s high risk of relapse, TBI and etoposide were used with a reduced irradiation dose of 8 Gy. TBI is usually restricted to patients >4 years, as it is associated with severe long-term side effects such as multiorgan dysfunction and neurocognitive abnormalities in younger children46–48 and is also associated with other well-known side effects in all age groups.49–51 In 2005, an analysis described the efficacy of TBI-based conditioning (12–15.75 Gy) in 39 infants with ALL (mostly in first CR), with an estimated 3-year overall- and disease-free survival of 50% and 42.2%, respectively, without excessive long-term toxicities. 52 Results of the phase III FORUM study described significantly higher overall survival and lower relapse rates after TBI-based conditioning compared with chemoconditioning, 17 which seems to apply especially to patients receiving allogeneic HSCT in second CR. 53 Although there are no data supporting a favourable toxicity profile of 8 Gy compared with the standard dose of 12 Gy in TBI-based conditioning for ALL in infants, we decided to apply the lower dose with an aim to reduce toxicity while still achieving an optimal anti-leukaemic effect. This approach resulted in tolerable acute toxicity, persistence of MRD negativity and good graft function.
Encouragingly, patient 1 is still in CR and in a good general condition >140 days after CAR T-cell bridging therapy and second allogeneic HSCT.
Case 2
The optimal post-remission therapy for adolescents or young adults with ALL in CR1 remains a matter of debate. Some recent publications tend to favour a paediatric-style post-remission chemotherapy over allogeneic HSCT in terms of OS or DFS for patients >16 years. 54 Patient 2, however, underwent a myeloablative allogeneic HSCT in CR1 at the age of 12 years after persistent high MRD levels according to the stratification in the AIEOP-BFM ALL 2009 front-line protocol. Twenty-one months post-HSCT, our patient relapsed. Considering the dismal prognosis of ALL relapse after allogeneic HSCT, a second HSCT appeared pertinent. Several options for pre-transplant MRD reduction were discussed.
Due to combined acute and long-term toxicity (pancreatitis with stomach perforation and stage IV renal failure) and the chemotherapy-resistant nature of the patient’s primary disease, conventional chemotherapy was deemed unsuitable. As she presented with CD22- ALL relapse, inotuzumab ozogamicin was not a treatment option either. Despite the CD19- subclone, we considered CD19-targeted treatment to be the best option for this patient. Due to its limited toxicity profile compared to polychemotherapy and high impact on disease burden, tisagenlecleucel was considered the best option. However, as a second HSCT seemed inevitable, we opted against additional anti-leukaemic treatment to target the low-level CD19- subclone in an attempt to avoid any unnecessary toxicity.
Moderate dose-adjusted bridging chemotherapy with concomitant CVVHDF was applied. The patient’s overall condition deteriorated and chemotherapy hardly reduced disease burden. Conversely, administration of tisagenlecleucel was well-tolerated and resulted in MRD negativity of the patient’s CD19+ subclone. As anticipated, the CD19- subclone persisted, which reinforced our decision to pursue a second allogeneic HSCT.
Reduced-toxicity conditioning is an option in heavily pre-treated patients at risk of early transplant-related mortality and long-term morbidities. 55 Due to her stage IV renal insufficiency and prior TBI, the patient received a reduced-toxicity regimen with concomitant CVVHDF. Subsequently, she achieved complete donor chimerism and MRD negativity.
As our patient developed graft dysfunction, she received a stem cell boost around Day + 100, eventually leading to stable engraftment.
Unfortunately, the girl developed a second isolated BM relapse around Day + 180 after her second allogeneic HSCT. As she presented with a purely CD19+ relapse and showed stable renal function, a second CAR T-cell infusion was successfully performed at the time of writing, considering the patient’s wish, and using her allogeneic T-cells transplanted by the second HSCT.
As the two described patients have biologically distinct leukaemias, direct comparison of their individual outcome remains speculative. Noticeably, some associations between their clinical course and disease outcome can be backed by literature. Higher pre-transplant MRD burden is associated with worse outcome.15,17,30–32 While patient 1 was MRD negative at the time of second HSCT, patient 2 had a persistent CD19- clone at a molecular level of 2 × 10–3. Furthermore, patient 2 had already received TBI for her first HSCT, which has been shown to provide a better leukaemic control post-HSCT compared to myeloablative chemoconditioning. 17 Long-term toxicities and morbidities prompted us to apply a reduced-intensity chemoconditioning for the second allogeneic HSCT, which might have additionally increased the relapse risk of patient 2 in combination with an inferior anti-leukaemic NK-cell and T-cell alloreactivity due to secondary graft failure.17,53
In summary, many therapies are currently being studied for the treatment of relapsed ALL, from immunomodulatory agents to adoptive cell therapy, bispecific T-cell engagers, unconjugated and conjugated antibodies, oncolytic virotherapy, and checkpoint inhibitors. Ongoing translational research is increasing our understanding of immune-based therapies and will most likely shape the next generation of clinical trials of primary and relapsed childhood ALL.
Both of the cases described here demonstrate the challenges associated with, in particular, systemic ALL relapse after first allogeneic HSCT: treating physicians are often confronted with chemotherapy-resistant disease and persisting organ damage inflicted by previous therapy. Novel immunotherapeutic agents such as CD19-directed CAR T-cells can induce MRD-negative remission, even with resistant disease, and long-term survival. However, CAR T-cells might also play a role as bridging therapy to a second allogeneic HSCT. Due to an acceptable toxicity profile, CAR T-cells are suitable for heavily pre-treated patients.
We are fully aware that our decision to opt for a second allogeneic HSCT in patient 1 is debatable, not only because we did not rely on a standalone therapy with tisagenlecleucel, but also because we used a TBI-based conditioning regimen in a child <4 years of age. However, the only unbiased study-population-based data for relapse of KMT2A-rearranged infant ALL that are available underline the importance of HSCT in this setting. 56 Furthermore, the risk of a lineage switch might be increased under the selective anti-CD19 pressure caused by the CAR T-cells. A prospective clinical trial of the efficacy and safety of CAR T-cell therapy for relapsed or refractory infant KMT2A-rearranged ALL would be of benefit for informed treatment decisions in this patient group. In lieu of these data, clinicians must rely on real-world data or case series to form their decisions. Prospective studies should collect data on those patients successfully infused with CAR T-cells, as well as those who do not achieve successful cell apheresis, cell production or infusion due to early events. As CAR T-cell therapy will change the landscape of the next generation of clinical trials, we believe that it will also address the management of the very high-risk group of infant KMT2A-rearranged ALL, thereby avoiding the short- and long-term toxicities of allogeneic HSCT in this vulnerable age group. Apart from CAR T-cells and blinatumomab or inotuzumab ozogamicin, future treatment of KMT2A-rearranged infant leukaemias might also include direct menin/KMT2A targeting, 57 with a clinical phase I/II trial currently ongoing to determine the safety and efficacy of the menin-KMT2A inhibitor SNDX-5613 (NCT04065399).
In patient 2, a further therapy component was definitely needed to eradicate the CD19- clone, which could only be achieved by a second allogeneic HSCT. If this could have also been possible by targeted therapies, for example BCL2 inhibitors (venetoclax), either alone or in combination with chemotherapy remains open. Preliminary results from phase I/II studies in children with different malignancies including r/r ALL were promising, reporting response rates of more than 60% and a favourable side effect profile.58–60 In theory, a combination of novel compounds with low-dose chemotherapy could additionally help bridging heavily pre-treated patients until a potentially curative treatment is available.
Conclusion
In general, systemic ALL relapse after first HSCT represents a challenge, and optimal therapy remains to be defined. Nevertheless, immunotherapy such as CAR T-cells has the potential to move to this particular setting, preferably as a standalone therapy but alternatively as bridging therapy to a second HSCT.
Footnotes
Acknowledgements
All colleagues involved in the development of this article and the patients’ treatment are listed as authors, as they contributed in the above-named ways. The authors thank the patients and their families for supporting this article.
Ethics approval statement and Informed consent statement
Ethics approval was not required for this case series, primarily, as it did not constitute research, and second, because a written informed consent from the patients and their guardians for publication were available.
Consent for publication
Patients and their guardians provided written informed consent for the publication of patient information in the present article.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by St. Anna Children’s Hospital and funded by St. Anna Children’s Cancer Research Institute (CCRI).
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: AA has received honoraria for lectures, consultancy, or advisory board participation from the following companies: Jazz Pharmaceuticals, Amgen, Novartis, MSD, Servier, and Gilead. He has received compensation for travel expenses from Jazz Pharmaceuticals. MD has received honoraria for lectures, consultancy, or advisory board participation from the following for-profit companies: Beckman-Coulter, Daiichi Sankyo, and Servier. The following for-profit companies have supported research conducted by MD with benefits or honoraria made to his institution: Beckman-Coulter, Becton-Dickinson, Exbio, and Jannsen. BH has received compensation of travel expenses from AOP Orphan Pharmaceuticals, AMGEN, and Amomed Pharma.
The remaining authors declare no conflict of interest.
Availability of data and materials
Not applicable.
