Abstract
Background:
Several clinical trials have been conducted to evaluate the role of maintenance therapy in untreated diffuse large B-cell lymphoma (DLBCL) patients with complete response or partial response following standard immunochemotherapy; however, the effect of maintenance therapy remains uncertain, and a suitable maintenance strategy has not been determined because of the lack of direct/indirect comparisons.
Methods:
We performed a systematic review and Bayesian network meta-analysis (NMA) to analyze and compare the effectiveness of different maintenance regimens in newly diagnosed DLBCL patients. We searched the PubMed Medline, Embase, Web of Science, Cochrane Database of Systematic Reviews, and Cochrane Central Register of Controlled Trials for relevant papers from inception to 18 March 2021. Our study was prospectively registered in the International Prospective Register of Systematic Reviews (CRD42020168864). Data on overall survival (OS) were extracted and the treatments were ranked using the surface under the cumulative ranking (SUCRA) curve.
Results:
Eight trials and seven treatments involving 3525 patients were analyzed. OS analysis indicated that none of the drugs showed any benefit compared with non-maintenance therapy. Maintenance therapy with lenalidomide (SUCRA 69.3%) was ranked first in terms of OS.
Conclusion:
Based on the OS results observed in this NMA, we do not recommend maintenance therapy in patients with newly diagnosed DLBLC after first-line therapy.
Introduction
Diffuse large B-cell lymphoma (DLBCL) is the most common type of non-Hodgkin lymphoma (NHL), accounting for 30–40% of all NHL cases. 1 DLBCL is a potentially curable disease; during the last two decades, the addition of rituximab (R) to standard chemotherapeutic drugs including cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) has improved the outcome of patients with DLBCL.2,3 R-CHOP has therefore emerged as the standard first-line immunochemotherapy for newly diagnosed DLBCL patients. However, >30% of DLBCL patients relapse within the first 2–3 years, leading to a poor prognosis. 4
It has been shown that maintenance with rituximab is associated with improved outcomes in indolent NHL. 5 To prevent relapse and improve the survival of DLBCL patients, clinical investigators have made considerable efforts regarding subsequent treatment after front-line therapy. Recently, increasing attention has been focused on maintenance therapy following induction therapy-induced remission.
Several clinical trials have been conducted with drugs, including rituximab, lenalidomide, enzastaurin, everolimus, and thalidomide among others, to evaluate the efficacy of maintenance therapy for untreated DLBCL patients with complete response (CR) or partial response (PR) after standard immunochemotherapy.6–13 Subgroup analyses of some clinical trials indicate that maintenance therapy has differences in gender 7 and induction protocol, 8 which suggests that it is a promising strategy for future research on the choice of drugs and potentially a highly selected group of patients.
However, the effect of maintenance therapy remains uncertain, and a suitable maintenance strategy has not been determined due to the lack of direct/indirect comparisons. It has been reported that rituximab maintenance improved survival in male patients with DLBCL after standard therapy. 7 However, Habermann et al. 8 reported that no benefit was provided by maintenance therapy after R-CHOP. It has been indicated that 2 years of lenalidomide maintenance in patients responding to R-CHOP significantly improved progression free survival, without a significant impact on overall survival (OS). 11 To determine the suitable maintenance strategies and estimate the ranking and hierarchy of them in DLBCL patients, we conducted a network meta-analysis (NMA), which can compare multiple interventions under the similar condition.
We first performed a systematic literature review of randomized controlled trials (RCTs) in newly diagnosed DLBCL cases to evaluate the maintenance treatments using different agents, and then conducted an NMA to compare the OS of each agent versus the others. This paper provides evidence that can enable the design of clinical trials for the maintenance treatment of DLBCL patients in the future.
Methods
Study design
This systematic review and NMA was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.14,15 Our study was prospectively registered in the International Prospective Register of Systematic Reviews (CRD42020168864)
Search strategy and selection criteria
We reviewed studies evaluating patients newly diagnosed with DLBCL and with response to standard immunochemotherapy including rituximab that compare maintenance therapy with placebo/observation or compare different agents for maintenance with each other. We systematically searched PubMed Medline, Embase, Web of Science, Cochrane Database of Systematic Reviews, and Cochrane Central Register of Controlled Trials for relevant papers from inception to 18 March 2021. The search terms were “diffuse large B cell lymphoma” and “maintenance”. The language of the articles was restricted to English. We also conducted manual searches for the references of each study to ensure that no articles were missed.
The following eligibility criteria were considered: population, study design, interventions, comparisons, and outcomes. The inclusion criteria were as follows: (a) RCTs that evaluated the response of newly diagnosed DLBCL patients (adults) to standard immunochemotherapy; (b) parallel design RCTs fulfilling the criteria of population/problem, intervention, comparison, outcome, and study type; 15 (c) prospective phase II or phase III RCTs that included rituximab in pre-maintenance therapy and compared at least two maintenance approaches; (d) RCTs that included the comparison of agents including placebo and no maintenance; and (e) RCTs that reported survival data including OS. We excluded studies in which patients did not receive rituximab as part of the induction therapy because this is no longer considered standard therapy.
Two investigators independently reviewed and selected studies according to the PRISMA diagram. 16 All disagreements between the two reviewers (TY and XZ) were resolved by discussion with a third investigator (FZ, QY, or YL).
Data extraction
Two independent reviewers extracted the following information from all the eligible studies: name of the first author, year of publication, study phase, number of patients included in each arm, study enrollment period, months of median follow-up, pre-maintenance therapy, agents for maintenance of each arm, treatment schedule of every maintenance therapy, months of median duration of maintenance therapy, hazard ratios (HRs) and confidence intervals (CIs) for OS.
Whenever data were unavailable in the full-text studies, we estimated the HR using the ratio between the probabilities of the two arms and estimated the 95% CI using the p value. 17
Quality assessment
We used the Cochrane risk of bias tool to evaluate the risk of bias of each RCT that was included. 18 Two authors independently assessed the following domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases.
Statistical analysis
The primary outcome was the OS. We conducted the Bayesian NMA for OS in a random-effect model using the natural log transformations of HRs, and their 95% CIs, to estimate standard errors. No maintenance/placebo was selected as a common comparator to include all trials within one framework. The placebo treatment was assumed to be equivalent to no maintenance regarding the efficacy level. Our NMA does not involve direct or indirect pairwise comparisons; therefore, we conducted a Bayesian consistency framework.
The software used in our study was Stata software (version 15.1) and R-project statistical software (version 3.6.1) with the gemtc package and JAGS package. Bayesian NMA was performed in a random-effect model using Markov chain Monte Carlo methods. For our outcome measure, three Markov chains were simultaneously run in parallel for 80,000 interactions with 10,000 burn-ins per chain to obtain the posterior distribution. Treatment effects were estimated using HR and the corresponding 95% CI. We calculated the surfaces under the cumulative ranking curves (SUCRAs) to rank the probabilities of all included treatments. SUCRA represents 1 if the treatment is certain to be ranked first and 0 if it is certain to be the last.19,20
Results
Study selection and characteristics
We identified 754 articles from our original literature search. After duplicates were removed and the titles and abstracts were screened, 35 articles were considered for full-text assessment. Finally, eight articles involving 3525 patients met the eligibility criteria for NMA (Figure 1).

Literature search and selection. Preferred Reporting Items for Systematic reviews and Meta-analyses (PRISMA) flow chart of the study selection for the network meta-analysis.
The main characteristics of the eight RCTs are listed in Table 1. Most of the enrolled trials were published within the last 5 years. There were five phase III trials and two phase II trials. The median follow-up period ranged from 32 to 81 months. Most of the pre-maintenance treatments included R-CHOP or R-CHOP-like regimens. Seven maintenance options were evaluated: enzastaurin, rituximab, lenalidomide, lenalidomide plus rituximab, everolimus, thalidomide, and observation/placebo. The main results of the eight RCTs are presented in Table 2.
Selected trials – trial characteristics.

chemo, chemotherapy; NA, not available; R, rituximab; R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone.
Selected trials – trial results.
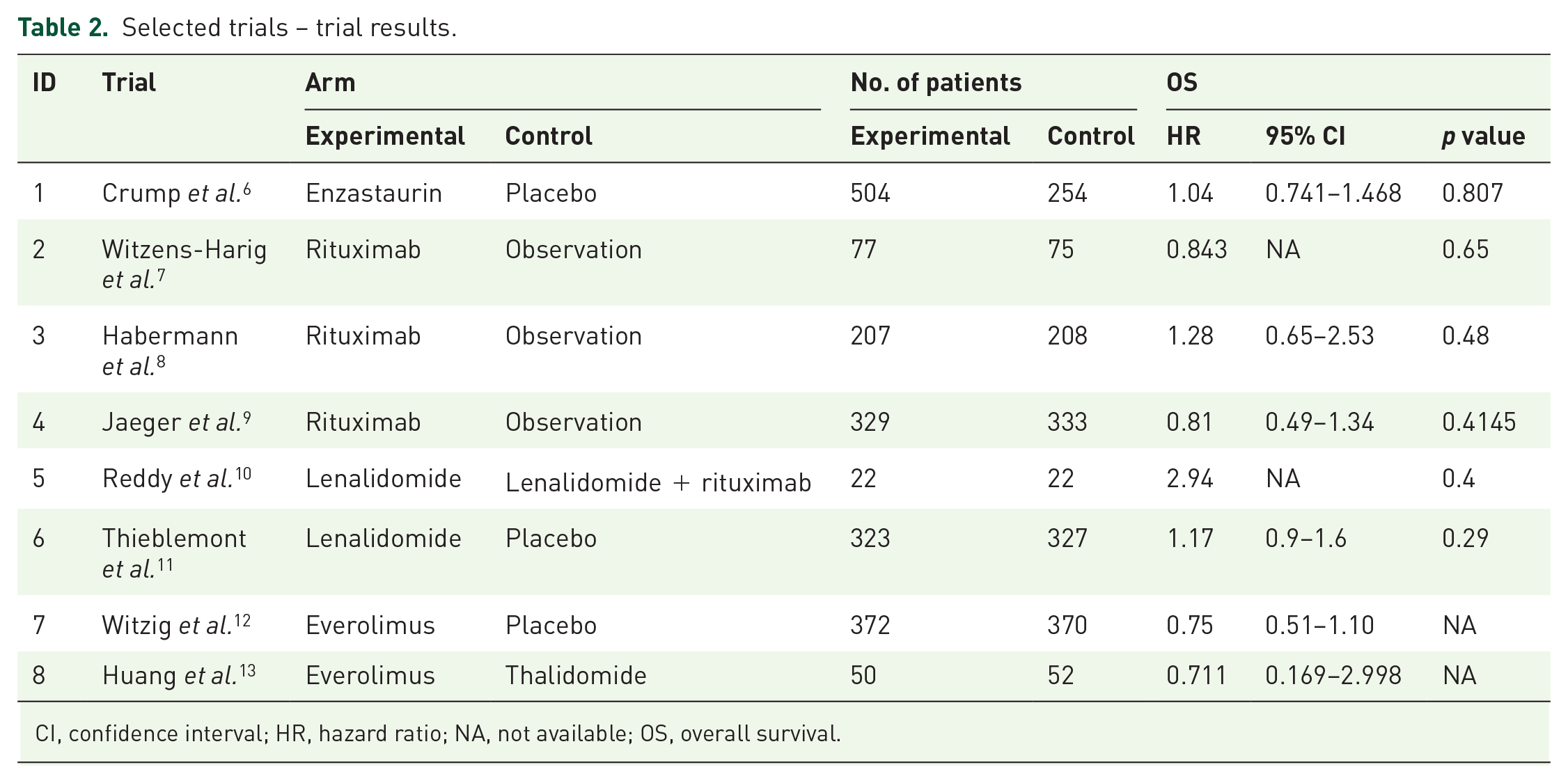
CI, confidence interval; HR, hazard ratio; NA, not available; OS, overall survival.
The network established for NMA in our outcome is shown in Figure 2, where direct comparisons between drugs are represented by solid lines.

Network plot of all eligible trials assessing maintenance treatments for untreated diffuse large B-cell lymphoma. The size of each plot represents the number of patients (in brackets) receiving the corresponding intervention. The width of each line represents the number of trials (beside the line) of corresponding comparison.
Risk of bias
The risk of bias in the included trials is shown in Figure 3. The overall quality of the included trials was adequate. Regarding random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases, all the included RCTs were rated as “low risk”.

Assessment of the risk of bias in the included studies.
NMA
Compared with non-maintenance therapy through NMA, none of the drugs showed any benefit in improving OS, and the results showed no statistical difference (Figure 4).

Forest plot of network meta-analysis results for overall survival. SUCRA represents the rank probabilities of all included treatments; it equals 1 if the treatment is certain to be ranked the first and 0 if it is certain to be the last.
Maintenance therapy with lenalidomide (SUCRA 69.3%) was ranked first in terms of OS; enzastaurin (SUCRA 58.8%) and thalidomide (SUCRA 57%) were ranked the second and third respectively (Figure 4) (Supplemental material Figure s1 online).
Discussion
Maintenance therapy is a strategy to maintain the initial response to induction therapy, prolong remission duration, delay relapse, and increase long-term survival by inhibiting cell proliferation. Maintenance therapy has been successfully used in a broad range of neoplasms, such as multiple myeloma or indolent lymphoma.21–24 In the past decades, maintenance strategies have been used in clinical trials for DLBCL.6–13 However, the role of maintenance therapy in DLBCL remains uncertain, and there is a lack of clinical trials that directly compare the efficacy of different maintenance drugs.
We conducted the present systematic review and NMA using Bayesian statistics to compare the efficacy of drugs and drug combinations for maintenance therapy in newly diagnosed DLBCL patients after standard therapy. The OS was chosen as the primary outcome and results suggest that maintenance therapy provides no benefit.
To the best of our knowledge, this study is the first NMA to estimate the comparative efficacy of different drugs used in maintenance therapy after first-line immunotherapy for newly diagnosed DLBCL patients. A systematic literature review of PubMed revealed just one meta-analysis study that assessed the role of maintenance therapy in patients with DLBCL who achieved CR or PR after first-line therapy. 25 Assessing OS as the primary outcome, our results, which showed that maintenance therapy did not affect the OS, were consistent with those of that study. There are several differences between the studies. First, in the meta-analysis by Rozental et al., not all studies included rituximab in the induction therapy; the immunochemotherapy containing rituximab is currently standard therapy for DLBCL, therefore we excluded the studies in which patients did not receive rituximab as part of the induction therapy. Second, they conducted a traditional meta-analysis to compare maintenance and non-maintenance therapy; we conducted an NMA to compare multiple maintenance drugs. Third, they performed subgroup and adverse event analysis, which we were unable to perform due to the lack of relevant data in the included literature.
Traditional meta-analysis can only collect head-to-head RCTs of two treatments, for example, a certain drug and a placebo, and then draw a conclusion about which one is preferred. NMA is a technique for comparing three or more interventions simultaneously in a single analysis by combining both direct and indirect evidence across a network of studies. There are six maintenance therapies in our study under the similar condition, therefore we use the Bayesian NMA as opposed to the classic meta-analysis. There are several advantages of NMA according to the Cochrane handbook. First, NMA can exploit all available direct and indirect evidence. Second, empirical studies have suggested it yields more precise estimates of the intervention effects in comparison with a single direct or indirect estimate. Third, NMA provides information for comparisons between pairs of interventions that have never been evaluated within individual randomized trials. In addition, the simultaneous comparison of all interventions of interest in the same analysis enables the estimation of their relative ranking for a given outcome. 26
Our study included eight RCTs, all of which reported OS as one of the outcomes. To synthesize all the available evidence of maintenance therapy, we chose OS as the primary outcome. Generally, OS is the most important end point in clinical trials. DLBCL is an aggressive lymphoma that mostly relapses within the first 2–3 years; one study indicated that patients with DLBCL treated with standard immunochemotherapy who are event-free for 2 years have the same OS as the general population. 27 Event-free survival for 2 years is an appropriate criterion to evaluate agents for maintenance therapy in newly diagnosed DLBCL patients, which provides a reliable design scheme for future clinical trials and meta-analysis.
There are several points worthy of further study according to our comprehensive understanding of maintenance therapy in patients with newly diagnosed DLBCL.
First, lenalidomide is an oral immunomodulatory agent with direct anti-tumor activity and immunologic effects. Its mechanism of action in neoplasms is yet to be fully determined. It has been used in several cancer types, such as multiple myeloma, myelodysplastic syndrome with 5q deletion, and relapsed/refractory mantle cell lymphoma. 28 Lenalidomide has significant single-agent activity in relapsed/refractory DLBCL. 29 Nowakowski et al. 30 reported that the combination of lenalidomide and R-CHOP therapy could overcome the negative prognostic impact of non-germinal center B-cell (non-GCB) phenotype on the outcome in newly diagnosed DLBCL patients. The current study evaluated the efficacy of lenalidomide as maintenance therapy for untreated DLBCL; the negative results could probably be attributed to the small sample size.
Second, rituximab has been applied as maintenance therapy in several types of lymphomas, such as follicular lymphoma and mantle cell lymphoma, with encouraging results.22,24,31,32 However, convincing evidence that maintenance with rituximab can improve the prognosis of patients with DLBCL after completion of first-line standard treatment is still lacking. Three studies that evaluated the role of rituximab maintenance therapy in newly diagnosed DLBCL patients were included in our NMA. Two of them, through subgroup analysis, concluded that rituximab maintenance therapy improves outcomes in male DLBCL patients;7,9 men with a low international prognostic index (IPI) have better outcomes. 9 However, small sample size and unplanned subgroup analysis limit the credibility of these results. This is an interesting question that needs to be addressed in future prospective trials.
Third, DLBCL is a heterogeneous disease, and the heterogeneity is derived from the clinical, pathological, and molecular biological aspects of recurrent somatic mutations in multiple genes that can affect cell regulation and proliferation. Heterogeneity leads to different therapeutic effects in patients with DLBCL. It has been reported that the prognosis is poor in DLBCL patients with the following characteristics: male, older age, more complications, higher tumor burden, higher level of lactate dehydrogenase, higher IPI score, non-GCB according to cell-of-origin (COO), higher Ki-67 proliferation index, P53 mutation, double/triple hits, and co-expression of BCL6, BCL2, and MYC.33–43 Individualized therapy according to patient characteristics is the focus of the current research. Therefore, it is necessary to conduct subgroup analysis, considering the different sources of heterogeneity in the design of clinical trials, which show exciting prospects for DLBCL maintenance therapy.
There are ongoing clinical trials that use other agents for DLBCL maintenance therapy after first-line immunochemotherapy. These drugs include bortezomib (phase III, NCT01965997), metformin (phase II, NCT03600363), and nivolumab (phase I, NCT03311958). We are awaiting exciting results from the ongoing DLBCL maintenance therapy clinical trials.
However, there are several limitations to our NMA. First, the results may be potentially affected by heterogeneity, which is caused by many factors, such as different inclusion criteria for the individual studies, inconsistent induction therapy, different protocols for a maintenance drug, and different follow-up durations. Second, very few RCTs were included in our study, and some comparisons included only one trial in our meta-analysis, such as lenalidomide versus placebo, lenalidomide versus lenalidomide plus rituximab, enzastaurin versus placebo, everolimus versus placebo, and everolimus versus thalidomide; therefore, the results may be considered as inconclusive evidence to some extent. More high-quality RCTs are essential to provide reliable proof. Third, our NMA did not conduct subgroup and adverse events analysis.
Conclusions
Our results do not support maintenance therapy in patients newly diagnosed with DLBLC, after first-line therapy. However, lenalidomide may be a potentially effective drug although the efficacy has to be confirmed by further clinical trials with more patients. The results of ongoing clinical trials which evaluate the effects of other agents such as bortezomib and metformin on maintenance therapy of newly diagnosed DLBCL patients are awaited. In addition, due to the heterogeneity of DLBCL, prospective subgroup design of clinical trials considering the sources of heterogeneity should be conducted in the future to identify the suitable drug for maintenance therapy in appropriate patients.
Supplemental Material
sj-pdf-2-tah-10.1177_20406207211018894 – Supplemental material for Maintenance therapy for untreated diffuse large B-cell lymphoma: a systematic review and network meta-analysis
Supplemental material, sj-pdf-2-tah-10.1177_20406207211018894 for Maintenance therapy for untreated diffuse large B-cell lymphoma: a systematic review and network meta-analysis by Ting Yuan, Feng Zhang, Qingmin Yao, Yanxia Liu, Xiaojuan Zhu and Peng Chen in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-3-tah-10.1177_20406207211018894 – Supplemental material for Maintenance therapy for untreated diffuse large B-cell lymphoma: a systematic review and network meta-analysis
Supplemental material, sj-pdf-3-tah-10.1177_20406207211018894 for Maintenance therapy for untreated diffuse large B-cell lymphoma: a systematic review and network meta-analysis by Ting Yuan, Feng Zhang, Qingmin Yao, Yanxia Liu, Xiaojuan Zhu and Peng Chen in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-1-tah-10.1177_20406207211018894 – Supplemental material for Maintenance therapy for untreated diffuse large B-cell lymphoma: a systematic review and network meta-analysis
Supplemental material, sj-tif-1-tah-10.1177_20406207211018894 for Maintenance therapy for untreated diffuse large B-cell lymphoma: a systematic review and network meta-analysis by Ting Yuan, Feng Zhang, Qingmin Yao, Yanxia Liu, Xiaojuan Zhu and Peng Chen in Therapeutic Advances in Hematology
Footnotes
Author contributions
Conception and design, PC and XZ; development of methodology, TY and PC; acquisition of data, TY, YM, YL and FZ; formal analysis, TY and PC; writing, TY; reviewing and editing, PC and XZ. All authors contributed to the article and approved the submitted version.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants from National Key Research and Development Program of China (2020YFC2008902), Natural Science Foundation of Shandong Province (ZR2020MH112) and China Postdoctoral Science Foundation (grant number 2020T130067ZX).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

