Abstract
Ewing sarcoma family tumors (ESFT) pose diagnostic challenges, which largely depend on the primary site of involvement and tumor stage. Despite advancements in treatment, metastatic ESFTs remain associated with poor outcomes. This case describes a 21-year-old woman who, in July 2022, presented with a left breast mass identified through ultrasound and CT scan, along with abdominal distention. A biopsy of the breast mass confirmed metastatic extraskeletal Ewing sarcoma. Further imaging revealed an ovarian mass, with subsequent biopsy confirming ovarian origin as extraskeletal Ewing sarcoma. The breast mass was identified as metastatic based on imaging features, including irregular margins and CT scan confirmation of widespread metastasis. Histopathology and immunohistochemistry confirmed Ewing sarcoma, consistent with the ovarian mass pathology that was the primary site. She underwent 15 cycles of VDC/IE chemotherapy ((vincristine, doxorubicin, and cyclophosphamide) for 2 days and 5 days IE (ifosfamide etoposide)), resulting in tumor cytoreduction. However, in less than 2 years, she developed metastases to the dura, spine, and bone, with optic nerve involvement. Despite treatment with radiotherapy and two cycles of high-dose Ifosfamide chemotherapy, her condition deteriorated, and she passed away in April 2024. This case underscores the complexity of managing metastatic ESFTs. Further research is needed to improve outcomes and establish treatment protocols for this malignancy.
Introduction
The Ewing sarcoma family of tumors (ESFT) are a type of tumors that is highly aggressive, characterized by the presence of shared pathognomonic chromosomal translocations, these tumors include Ewing sarcoma of bone, extra-skeletal Ewing sarcoma (EES), atypical Ewing and Askin tumor of the chest wall. They appear as a small round blue cell that arises from some mesenchymal or neural crest-derived stem or progenitor cell from bones, soft tissues, and viscera.1,2
Methods for diagnosis and detection of metastasis, include imaging such as magnetic resonance imaging (MRI) and Positron emission tomography (PET) scan, and the definitive diagnosis is made by histological, immunochemistry, and molecular analysis. 1 In histopathology, these appear as round blue tumor cells. It is domineering to go for molecular and or cytogenetic techniques, in addition to histopathology, to confirm the diagnosis, especially in challenging cases such as those that arise in uncommon sites.1,3 In immunohistochemical analysis, most cases have cytoplasmic membranous positivity for MIC2/CD99 and FLI1.2,3 While molecular genetic analysis through reverse transcriptase (RT) PCR or fluorescence in situ hybridization (FISH) plays an additional role in diagnosis when it ends in uncertainty, and involves common chromosomal translocations such as t (11;22) (q24;q12) and t (21;22) (q22;q12). Fusion between the FLI gene on 11q24 and the EWSR1 gene on 22q12, creating an EWS/FLI-1 happened in t (11;22) (q24;q12) translocation. However, other rarer fusion partners of the EWSR1 gene, such as ERG, ETV1, and TAF15, have also been identified in various subtypes of Ewing sarcoma. 1
Treatment for the Ewing sarcoma typically involves chemotherapy and surgery, while for extraskeletal Ewing sarcoma, treatment includes chemotherapy with surgery or radiation therapy, depending on the tumour’s location and resectability. In cases of metastatic extraskeletal Ewing sarcoma, a more aggressive approach combining chemotherapy, radiation therapy, and, if feasible, surgical resection is necessary to manage the spread. 1 Each of these types had a different treatment and prognosis, therefore a precise diagnosis is elemental. Unfortunately, it is difficult to differentiate them from other malignancies, especially when they arise in extra-skeletal or visceral locations, such as in the female genital tract, for the molecular and cytologic data play a great role. As in previous reports, the majority were found to be positive for CD994–6 and demonstrated the pathological EWSR1 fusion with other partner genes, The most common fusion partner is the FLI1 gene, resulting in the EWSR1-FLI1 fusion, which is present in approximately 85% of cases. Other less common partners include ERG, ETV1, E1AF, and FEV, among others.4,5
There are few cases reported about ESW in the female genital tract, Cases involving ovaries have mostly been associated with an aggressive clinical course. Many of these cases have failed more than one line of chemotherapy and were of high mortality. In this report, we are presenting a rare case of ESW arising from the ovary, and metastasis to breast, managed with tumor cytoreduction and chemotherapy.
Case description
A 21-year-old woman, previously diagnosed with bipolar disorder, was referred to Najah National University Hospital (NNUH) In July 2022, due to the incidental discovery of a mass in her left breast 2 weeks earlier. She also reported a month-long increase in abdominal size and a weight gain of 10 kg, despite experiencing a loss of appetite and persistent nausea. Before referral, a series of investigations had been conducted. A breast ultrasound revealed a large, well-defined soft tissue mass in the inner quadrant of the left breast, measuring 4.2 × 2.8 cm with irregular edges. Additionally, an abdominal ultrasound indicated a sizable soft tissue mass measuring 12 × 10 cm in the left ovary, accompanied by cystic lesions and a significant amount of ascites. These findings raised concerns regarding possible ovarian or breast malignancy.
Upon physical examination, the patient’s cardiac evaluation yielded normal findings. However, thoracic examination revealed reduced air entry in the middle and lower right lung, accompanied by bronchial breathing, which suggested possible consolidation or pleural involvement. Abdominal examination showed a distended abdomen with shifting dullness and a prominent fluid thrill suggestive of ascites, these findings raised concern for peritoneal metastasis or fluid accumulation due to other malignancies. Examination of the left breast identified a 5 × 5 cm mass located at nine o’clock, which was hard and warm without noticeable skin or nipple changes, which suggested a solid, non-inflammatory lesion, warranting further evaluation. A gynaecological examination showed no genital ulcers or bleeding. Upon admission, to investigate further, subsequent chest and pelvis CT scans demonstrated a large lobulated mass in the left breast, measuring approximately 6.5 × 4.8 cm Figure 1). A separate lesion with lobulated margins was observed in the right lung, extending from the right hilum to the pleural surface, occupying most of the lower lobe and encasing the right mediastinal structures. However, no collapse or invasion of the right main bronchus resulted in a shift of the mediastinum and heart to the left side. Another pleural-based lesion, measuring approximately 1.4 × 1 cm, was found in the right middle lobe. Additionally, a 2 × 3.5 cm pleural-based lesion was observed in the left lower lobe, accompanied by multiple mildly enlarged lymph nodes, These findings raised further suspicion for metastatic spread from an unknown primary site. For further characterization of the pelvic mass, a Pelvis MRI showed a large heterogeneously enhancing pelvic mass lesion with solid and cysti components, measuring about 10 × 10 × 13 cm (APXRLXCC) The imaging suggested this mass was likely a metastatic lesion originating from the ovaries, contributing to the ascites observed on examination (Figure 2). Tumor markers showed elevated CA-125 and LDH levels, while other markers were within normal limits. To be noted that the tumor markers CA125 and LDH were measured using serum-based assays, and were measured using serum-based chemiluminescent immunoassays on an automated platform. Chest CT scan with contrast. (A) a large right lung lesion with lobulated margin. (B) showing a large lobulated left breast mass, axial View. Pelvis MRI of a large heterogeneously enhancing pelvic mass lesion with solid and cystic components, measuring about 10 × 10 × 13 cm (APXRLXCC).

A biopsy of the breast mass was performed, and while awaiting the results, the patient underwent peritoneal tapping to manage the ascites. Five days later, the breast histopathology revealed extensive infiltration by a tumor composed of undifferentiated small round blue cells with clear cytoplasm, arranged in sheets with widespread areas of necrosis, stromal sclerosis, and increased apoptosis. Immunophenotyping indicated that the tumor cells were positive for CD99 in a diffuse membranous pattern and positive for NKX2.2 Ewing in a diffuse molecular pattern, and were diffusely positive for vimentin, FLi-1, and CDI17, with patchy positivity for BCL2. The tumor cells were negative for several other markers, confirming a diagnosis of extraskeletal Ewing sarcoma. In addition, FISH testing confirmed the EWSR1-FLI1 fusion.
Subsequently, the patient underwent the VDC/IE chemotherapy protocol, consisting of alternating cycles of VDC (vincristine, doxorubicin, and cyclophosphamide) for 2 days and IE (ifosfamide etoposide) for 5 days, with complications including peritonitis and neutropenic fever, both managed with appropriate medications. After completing four cycles of chemotherapy, a follow-up chest CT scan showed significant changes (Figure 3), including a reduction in the size of lung and pleural-based lesions, with the breast mass no longer visible. A laparoscopic right unilateral salpingo-oophorectomy revealed no residual viable tumor in the specimen, indicating an effective treatment outcome. Follow-up chest CT scan with oral and IV contrast showed Non-visualization of the previously described left breast mass and a significant decrease in the size of the right lung mass.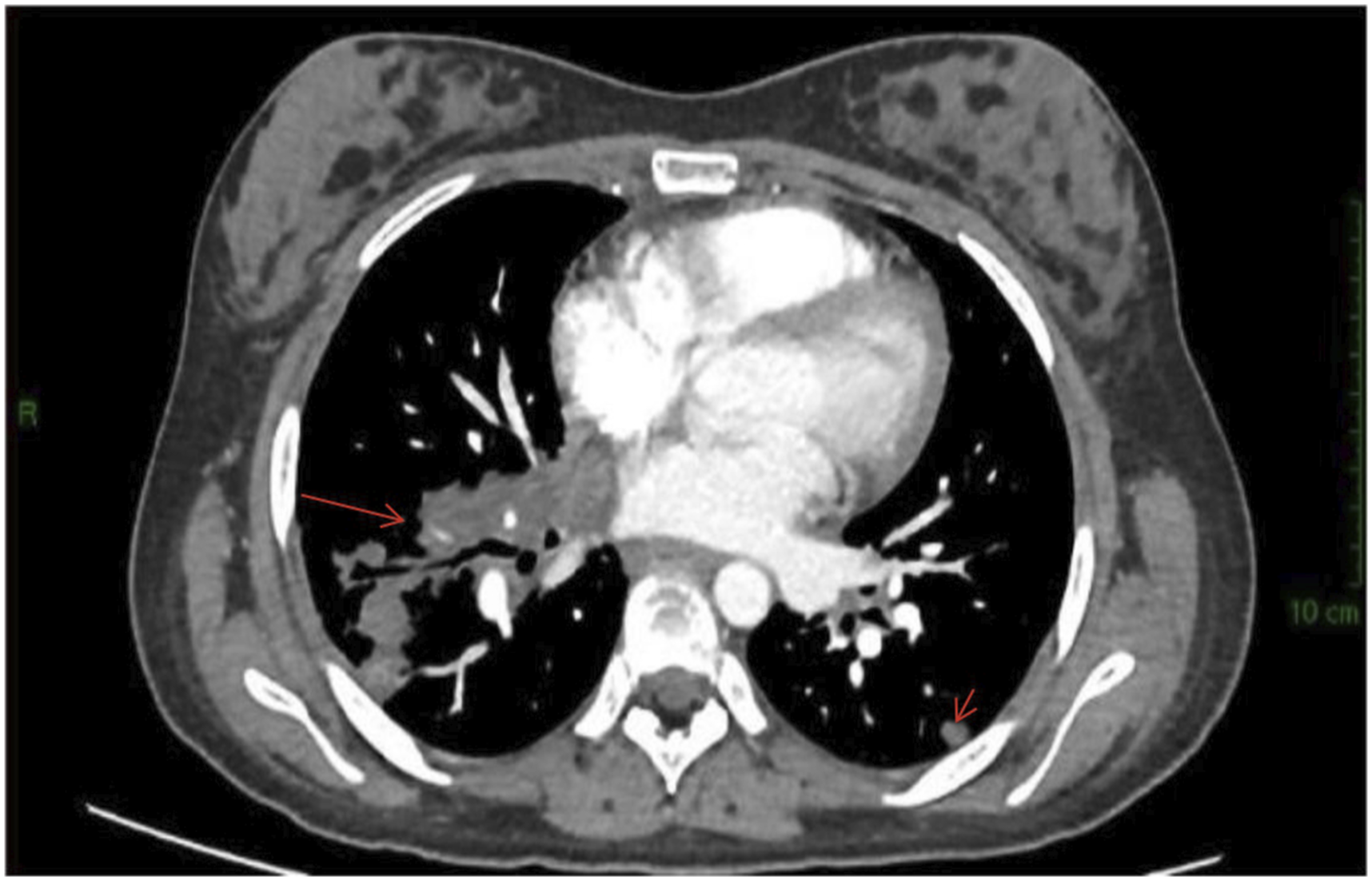
The patient underwent an additional 11 cycles of alternating VDC/IE chemotherapy without complications. A subsequent follow-up CT scan showed a further decrease in the size of the lung mass and resolution of the pleural-based lesions, with tumor markers gradually decreasing throughout treatment.
After completing more than a year and a half of follow-up post-diagnosis, the patient concluded chemotherapy and maintained good overall health. However, in August 2023, she reported severe back pain and diminished vision acuity in her left eye, which eventually led to complete vision loss. Brain and spine MRI revealed dural, spine, and bone metastases (Figure 4), including involvement of the optic nerve, for which she underwent radiotherapy followed by two cycles of high-dose Ifosfamide chemotherapy. Unfortunately, her condition progressively deteriorated, showing no response to treatment. Tragically, in April 2024, she passed away. MRI with IV contrast. (A) Brain MRI showed an extra axial dural based enhancing lesion seen in the parafalcine region of the left frontoparietal lobe, measuring about 2.2 × 2 × 0.7 cm, suggestive of metastasis. (B) Spine MRI showed Multiple metastatic bone deposits involving the visualized vertebral bodies.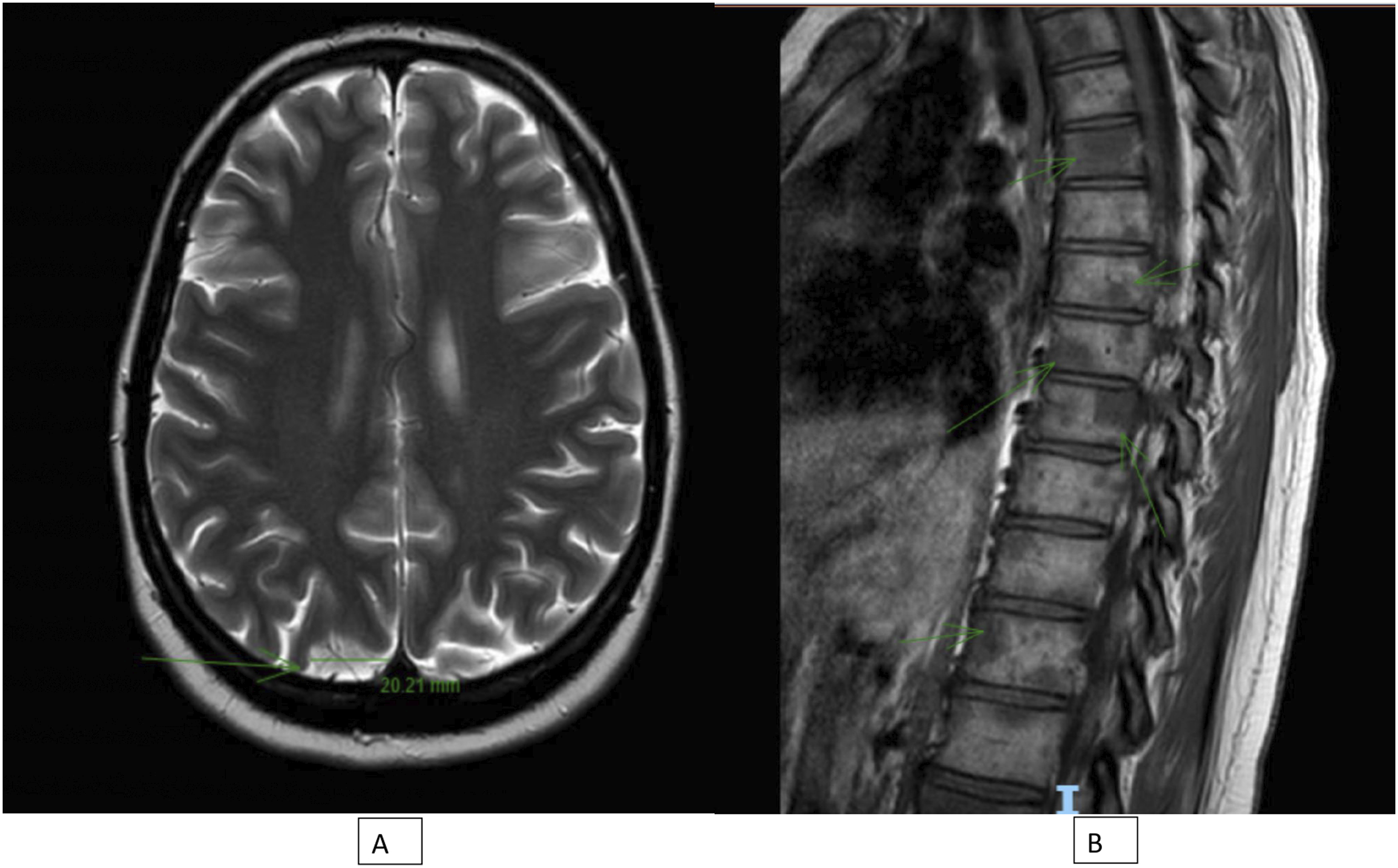
Discussion
This case presents an unusual manifestation of Ewing sarcoma originating from the ovary and presenting with a breast mass. Common presentations documented in previous reports of primary ovarian Ewing sarcoma include complaints of acute abdominal pain, palpable masses in the flanks, weight loss, vaginal bleeding, or hirsutism.7,8 However, unlike these typical symptoms, our case presented with a breast lump. Additionally, similar to other types of ovarian cancer, abdominal distention due to ascites is not uncommon,6,9 as observed in our case.
Nevertheless, exceedingly rare cases of Ewing sarcoma with breast metastasis have been reported, often presenting with metastasis as the primary observation. For instance, Kwak et al. described a case of a breast mass that was later identified as a metastatic extraskeletal Ewing sarcoma originating from the back. 10 Generally, in metastasis to the breast from any malignancy, the metastasis itself is the initial presentation in 16%–30% of cases.10–12 In such instances, accurate differentiation between metastasis involving the breast and primary breast pathology is crucial, as both may exhibit similar radiographic features. Surgery such as radical mastectomy is unnecessary for the treatment of such metastases.
This case represents the first reported instance of breast metastasis from ovarian Ewing sarcoma. Analysis of the limited literature on ovarianEwing sarcoma suggests a tendency for an occurrence at young ages, consistent with other tumors within the Ewing sarcoma family, typically between the second and third decades of life, as seen in our case.4,13
The diagnosis of extraskeletal Ewing sarcoma arising from the ovary is primarily confirmed through morphological features, immunohistochemical analysis, and molecular studies. Morphologically, these tumors exhibit small hyperchromatic round blue cells with scanty cytoplasm arranged in lobules, often accompanied by variable amounts of fibrillar cell processes and areas of necrosis. Immunohistochemically, these tumor cells typically demonstrate strong immunoexpression of CD99 (MIC2 protein) and often express FLI-1. Additionally, fluorescence in situ hybridization (FISH) analysis commonly reveals a break-apart signal pattern of EWS gene rearrangement, while RT–PCR detects the classical t (11;22) (q24;q12) translocation, characteristic of the ESWR–FLI1 fusion gene, consistent with findings in our case.4,7,14 While some reports indicate nucleus staining for CD56, synaptophysin, and NK2 homeobox 2 (NKX2.2), 7 these markers were not positive in our case. Differentiation between central Ewing sarcoma and extraskeletal Ewing sarcoma (peripheral) relies mainly on morphological and FISH analyses. Immunohistochemical analysis may show the expression of CD99 and FLI-1 in central Ewing sarcoma as well. Thus, the diagnosis of Ewing sarcoma is based on the presence of EWSR1 rearrangement by FISH analysis and the absence of morphological features typical of the central nervous system. 4
The conclusion that the ovarian mass is the primary site of the tumor, while the breast mass is metastatic, is based on a thorough assessment of clinical, imaging, and histopathological data. Imaging studies, including CT scans and MRI, as a sizeable mass in the ovary; solitary involvement rather than bilateral ovarian involvement; and the characteristics of ovarian mass, showing a heterogeneous solid-cystic composition. On the other hand, the breast mass exhibited features typical of metastatic involvement. Histological examination of the breast mass confirmed metastatic extraskeletal Ewing sarcoma, with key markers such as CD99 and FLI-1 present. The absence of similar metastatic markers in the ovarian mass, alongside the patient’s elevated CA-125 levels, further supported the diagnosis of a primary ovarian lesion. Additionally, the metastatic spread of Ewing sarcoma often involves the lungs and pleura, making breast involvement less common.
Management of ovarian Ewing sarcoma varies due to its rarity, and there are no standardized treatment protocols. Typically, a surgical approach is employed, which may involve cytoreduction by cystectomy with unilateral salpingo-oophorectomy for fertility preservation, as in our case with a young female patient. Alternatively, some cases may require total abdominal hysterectomy with bilateral salpingo-oophorectomy. Adjuvant chemotherapy and/or radiotherapy are often administered following surgery. Various chemotherapy regimens have been described, with the VDC (vincristine, doxorubicin, cyclophosphamide) alternating with IE (ifosfamide, etoposide) regimen being commonly used and recommended in the literature. Other reported regimens include VCD (vincristine, cyclophosphamide, doxorubicin) alone, PT (carboplatin, paclitaxel) protocol, and AD (ifosfamide, epirubicin, dacarbazine). This approach applies to both primary and metastatic disease.4,6,9,13–15
Generally, this neoplasm carries a poor prognosis, with high mortality, particularly worsening when it progresses to metastatic disease. Survival rates range from 10.8 months to 3 years. 6 This represents the first reported case of ovarianEwing sarcoma with breast metastasis, managed with unilateral salpingo-oophorectomy and chemotherapy. Though the patient has shown remarkable improvement, having completed 15 cycles of chemotherapy, unfortunately, she had brain and bone metastasis, which led to deterioration less than 2 years after diagnosis.
This case report has several limitations. Notably, the absence of pathology images reduces the ability to visually substantiate the pathology findings; this limitation arose from the loss of pathology blocks by the patient’s family prior to manuscript preparation. Furthermore, the retrospective nature of this report meant some clinical details were unavailable. Despite these challenges, the case highlights a rare presentation and provides valuable insights into diagnostic and clinical considerations.
In conclusion, this report discusses a case of extraskeletal Ewing sarcoma presenting with multifocal involvement, highlighting the initial favorable response to intensive chemotherapy and surgical intervention. The diagnosis of ESW relies on clinical, morphological, immunohistochemical, and molecular analyses to differentiate primary breast cancer from EWS, which is an infrequent entity associated with a poor prognosis. While the Given the rarity and aggressive nature of this disease, prognosis remains poor. Although a standardized treatment regimen is yet to be established, an individualized approach, including surgical resection followed by chemotherapy (commonly VDC alternating with IE), remains the cornerstone of management in achieving optimal outcomes.
Footnotes
Acknowledgements
We extend our sincere appreciation to the pathology and radiology teams for their invaluable contributions to this research. Their expertise and insights have been integral to the success of this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Contributorship
RA conceptualized the study, curated data, and drafted the initial manuscript. MH led the methodology design. Additionally, RA managed project administration, allocated resources, and provided supervision. Both RA and YD contributed to writing, reviewing, and editing the manuscript. YD also contributed to refining the methodology.
