Abstract
The association between emphysematous pyelonephritis (EPN) and diabetes mellitus is well known. EPN as the cause of sepsis in hepatocellular carcinoma (HCC) is rare and unreported. We report a case of EPN in a non-diabetic HCC patient, in which the clinical features of decompensated chronic liver disease masked the more sinister urological emergency to a certain degree. A computed tomography scan of the abdomen revealed a mixture of gas and fluid in the left retrorenal space, in keeping with left EPN. Nevertheless, the course of clinical deterioration was rather rapid that any surgical intervention was not feasible.
Introduction
Emphysematous pyelonephritis (EPN) is a rare acute severe necrotising infection of the renal parenchyma and its surrounding tissue that can lead to gas formation. Diabetes mellitus (DM) is the most common associated factor, as nearly as 95% of patients with EPN have uncontrolled DM.1,2 Whilst EPN may be associated with other factors such as an anatomic anomaly, neurogenic bladder, alcoholism and drug abuse, the association between EPN and hepatocellular carcinoma (HCC) is almost unheard of and is unreported. 2 We hereby report such a rare case of EPN in a non-diabetic HCC patient.
Case report
A 60-year-old non-diabetic male was diagnosed as having HCC in a private centre following evaluation of abdominal distention. The diagnosis was confirmed with a four-phase computed tomography (CT) scan of the liver which revealed an ill-defined heterogeneous mass on the right lobe of the liver involving segments VI and VII, measuring approximately 7.3 cm×5.3 cm×6.1 cm (AP×W×CC), as well as additional features suggestive of liver cirrhosis and elevated alpha-fetoprotein level. The liver cirrhosis was cryptogenic, as hepatitis B and C screening was negative, there was no history of excessive alcohol consumption and no other evidence of autoimmune disorder or inherited disease. The initial calculated Child–Pugh score from the referring hospital was 14 (Child–Pugh class C), and the patient was already in stage D HCC based on the Barcelona Clinic Liver Cancer staging system. His first admission for decompensated liver cirrhosis was medically treated, and hence did not necessitate peritoneal paracentesis. He subsequently underwent selective internal radiation therapy at another centre two months later, which was successful, with neither procedure-related nor infectious complications after the procedure. It is noteworthy that there was no pre-existing urological abnormality necessitating any intervention or long-term placement of a catheter. He did not receive any immunosuppressive medication, and his regular medication comprised only spironolactone and propranolol.
Four months after his initial diagnosis, he presented to our centre with worsening abdominal distension, leg swelling, reduced effort tolerance and progressive malaise. He reported having an intermittent fever, as well as a reduced amount of urination. On examination, he was alert and conscious but was noted to be pale and jaundiced. His initial vital signs were stable, although he did have an occasional spike of temperature throughout admission. There were stigmata of chronic liver disease, that is, spider naevi, gynecomastia, gross ascites, splenomegaly and peripheral oedema.
Blood investigations on admission revealed a white blood cell count of 26.3×109/L, haemoglobin of 92 g/L and platelet count of 231×109/L. There was coagulopathy, with an activated partial thromboplastin time of 54.40 seconds, prothrombin time of 24.40 seconds and international normalised ratio of 2.29. Renal function was acutely deranged, with urea of 18.8 mmol/L and creatinine of 312 µmol/L. Liver enzymes were markedly elevated: aspartate aminotransferase 143 IU/L, alkaline phosphatase 293 IU/L and alanine transaminase 37 IU/L, with a total bilirubin of 110 mmol/L (direct 77 mmol/L, indirect 33 mmol/L) and serum albumin of 20 g/L. The calculated Child–Pugh score was 19 (Child–Pugh class C). Urinalysis revealed proteinuria and microscopic haematuria.
The initial assessments were decompensated chronic liver disease precipitated by possible spontaneous bacterial peritonitis (SBP), and acute kidney injury secondary to hepato-renal syndrome. He was medically treated with empirical antibiotics, but a diagnostic peritoneal paracentesis was not promptly performed due to coagulopathy.
However, during admission, the patient complained of worsening generalised abdominal pain. His general condition further deteriorated as he became hypotensive, requiring inotropic support. A plain abdominal radiograph showed an enlarged left kidney shadow (Figure 1), whilst an urgent CT scan of the abdomen confirmed the finding of gas and fluid mixture in the left retrorenal space, measuring 9.6 cm×10.5 cm×22.3 cm (AP×W×CC; Figure 2). This finding was in keeping with a diagnosis of left EPN.

Plain abdominal radiograph shows an enlarged left kidney shadow (single arrow) with extensive mottled appearance within the left renal fossa and extends until the lower pelvic region (double arrows).
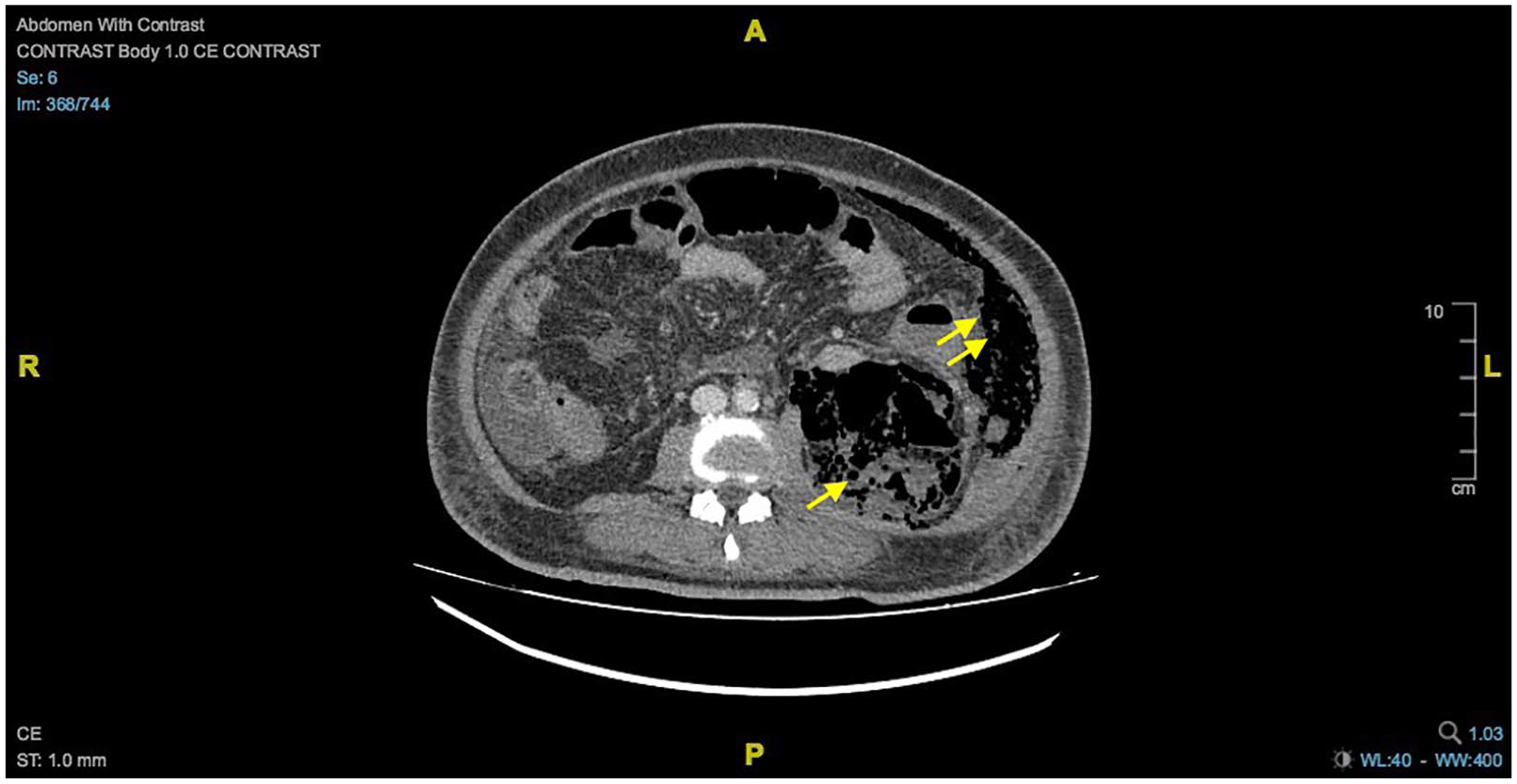
Computed tomography scan of the abdomen shows heterogenous gas collection mixed with fluid in the left retrorenal space (single arrow). There is the presence of an air pocket within the left lateral abdominal wall muscle (double arrows).
He had rapid clinical deterioration. Thus, surgical intervention was not feasible, and the patient passed away soon after due to overwhelming sepsis. Posthumously, the final septic work-up revealed evidence of
Discussion
EPN is a rare necrotising gas-forming infection of the renal parenchyma which is potentially life-threatening if not promptly detected and managed. Clinical features of EPN are variable, ranging from fever, upper urinary tract infection, acute renal impairment and disturbances of consciousness or to a more severe feature of septic shock. 2
In our case, the predominant features of decompensated chronic liver disease disguised the more sinister urological emergency that he presented with to a certain degree. The diagnosis of SBP should be made based on ascitic fluid analysis mainly to distinguish between this and other intraabdominal surgically treatable sources of infection. The decision of deferring paracentesis due to moderate coagulopathy might be disputable in our case, given the relatively low risk of haemorrhagic complications before the procedure. 3 However, individual physicians may have different thresholds of pre-procedure coagulation profile in real-world clinical practice.
Prompt radiological diagnosis is vitally important, as early recognition may expedite medical treatment, potentially avoiding more invasive intervention, including nephrectomy. 4 Radiological confirmation of gas within the kidney with or without the involvement of the collecting system is the hallmark EPN diagnosis.1,2,4 Our case highlights the importance of prompt imaging study in the quest for another surgical source of sepsis, particularly when the presumed SBP is not responding to the empirical broad-spectrum antibiotic.
DM is the most common associated factor in patients with EPN.1,2,4,6 Other associated factors that have been reported include drug abuse, neurogenic bladder, alcoholism and anatomic anomaly. 2 The postulated pathogenesis behind the generation of EPN is the presence of pathogenic bacteria which are capable of mixed acid fermentation, high glucose tissue levels in diabetic patients as well as impairment of tissue perfusion. 6
Nevertheless, the association of EPN with a liver-related problem such as chronic liver disease or HCC is almost unheard of or have been rarely reported. Kim et al. reported a case of bilateral EPN in a patient with uncontrolled DM and HCC with hepatitis B virus infection, whilst Nana et al. encountered a case of right EPN in a diabetic patient with autoimmune liver cirrhosis.7,8 However, in these two case reports, the presence of DM confounds the potential association between EPN and liver disease. Our patient, on the other hand, was not diabetic. Thus, the association between EPN and his liver disease is unclear. A highly possible explanation is that HCC and DM are both immunocompromised states, making the patient more susceptible to severe infections such as EPN. The pre-existing liver cirrhosis with HCC in our patient, however, might not be the only potential risk factor for developing severe EPN, since other factors which have yet to be proven might have been present.
Multivariate analysis by Lu et al. identified several predictors of mortality in patients with EPN: hypoalbuminemia, shock at initial presentation, bacteraemia, the need for haemodialysis and polymicrobial infection. 9 The first three prognostic factors were present in our patient, hence the higher risk of mortality imposed.
Factors that may influence the occurrence of infections in HCC include a decreased number and function of Kupffer cells which lead to reduced reticuloendothelial function, reduced immunoglobulin M antibodies and complement levels, decreased number and function of neutrophils and reduced synthesis of opsonisation factors. Portosystemic shunting and impaired lymph flow could further augment those factors, leading to increased risk of infection, especially of urinary origin. 10
Conclusion
In summary, EPN is a life-threatening infection in immunocompromised patients that should be promptly detected and managed. In decompensated liver cirrhosis, other surgically treatable causes of intraabdominal sepsis should be suspected and explored with radiological modalities whenever the patient does not respond well to the empirical treatment for SBP. Lastly, mild to moderate coagulopathy is not an absolute contraindication for abdominal paracentesis, and correction of coagulopathy should not delay diagnostic paracentesis.
Footnotes
Acknowledgements
None.
Authors’ contributions
H.H.W.H. managed the patient. H.H.W.H. and S.N.M.N. prepared the manuscript. N.M. and L.Y.Y. edited and approved the final draft.
Availability of data and materials
Data sharing is not applicable to this article, as no data sets were generated or analysed during the current study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Ethical approval was not sought for the present case report because it is not required as per university guideline. This study was completed in accordance with the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from legally authorised representatives before the study.
