Abstract
Background
A systematic review of the outcomes of combination therapy using Yttrium 90 radioembolization (Y90) and sorafenib/nivolumab for patients with hepatocellular carcinoma (HCC), with hepatic vein (HV) or inferior vena cava (IVC) invasion. The aim of this study is to summarise the results of different studies that used the combination therapy for HCC patients with tumor thrombosis involving the HV or IVC.
Method
A literature search was performed using keywords in Medline and Google Scholar limited to publications from 2010 to 2021. There were 173 articles identified during the initial literature search. During abstract screening, 81 articles were excluded. Another 83 did not contain information on hepatic vein or IVC invasion. Therefore, 9 articles met the eligibility criteria and were included in the synthesis.
Results
In total, 37 patients with hepatic vein or IVC invasion were identified. There were 31 patients who were given sorafenib, 7 were given nivolumab and 1 was given both sorafenib and nivolumab. Among the 37 patients, 21 had hepatic vein invasion, 22 had IVC invasion and 6 had both HV and IVC invasion. The median OS was 20.55 months and median PFS was 8.18 months. For the results, 23 patients were evaluated via modified RECIST (mRECIST) criteria and 14 were evaluated via RECIST 1.1.
Conclusion
The combination of local and systemic therapies demonstrated potential results for increased response rates, OS and PFS benefits. Further studies are required to determine the long-term outcomes of the combination therapy for this group of patients.
Introduction
Hepatocellular carcinoma (HCC) is one of the leading cancers in the world, with increased incidence in progression to advanced liver disease. 1 Common risk factors for HCC include chronic hepatitis B and hepatitis C, increased alcohol intake, non-alcoholic fatty liver disease (NAFLD) and toxin exposure. 2 Macrovascular invasion is typically observed in advanced HCC, with the most common vessel being the portal vein. In fewer cases, HCC patients experience invasion of the inferior vena cava and hepatic veins (HCC-HV/IVC). 3
The current treatment of early-stage HCC utilizes curative strategies involving surgical resection, liver transplantation and ablation. 4 For advanced-stage HCC with invasion of the portal veins, hepatic veins and IVC, alternative treatment modalities include chemoembolization, radioembolization and systemic therapy. 4 Yttrium 90 radioembolization (Y90) is a mode of intra-arterial therapy for unresectable HCC, to reduce the size of the tumor and bridge patients to surgical resection or liver transplantation.5,6 For patients with unresectable HCC, systemic therapy can also be considered. These include sorafenib, nivolumab, lenvatinib, pembrolizumab and cabozantinib. Sorafenib is a tyrosine kinase inhibitor and was the first Food and Drug Administration (FDA) approved first-line systemic therapy for HCC in 2007. 7 Nivolumab is a PD-1 inhibitor that has recently received approval by FDA for treatment of HCC in patients previously treated with sorafenib. 8 In this systematic review, we will be focusing on the outcomes of combination therapy using Y90 and these FDA-approved systemic agents (sorafenib and nivolumab) for HCC-HV/IVC. It has been proposed that the combination could enhance the immunotherapy effect. 9
Material and methods
A literature search was performed using keywords in Medline and Google Scholar. Keyword searches of ‘HCC’, ‘Y90’, ‘systemic therapy’, ‘sorafenib’, ‘nivolumab’, ‘hepatic vein’, ‘IVC’ were used. The publication language was limited to English only and studies without the full text were excluded. There were 173 articles identified during the initial literature search. During abstract screening, 81 articles were excluded. Of the remaining 92 articles, 83 did not contain information on hepatic vein or IVC invasion. Therefore, only 9 articles met the eligibility criteria and were included in the synthesis. Figure 1 shows the flowchart of the screening process for the systematic review. Among the 9 articles, 7 were from USA, 1 was from Switzerland and 1 was from Italy. The evaluation was done based on the median overall survival (OS), median progression-free survival (PFS) and the local response rate. Flowchart of literature screening for the systematic review.
Results
Characteristics of studies
Characteristics of included studies.
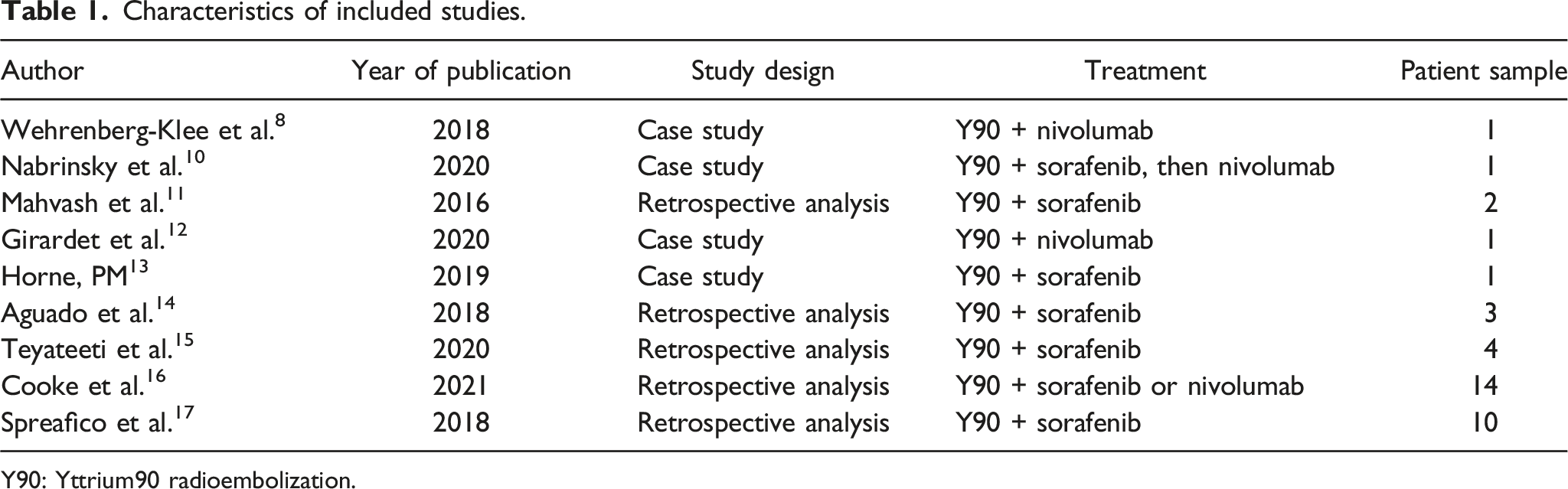
Y90: Yttrium90 radioembolization.
Treatment response
Local response rate and survival outcome data of included studies.

Y90: Yttrium90 radioembolization; OS: overall survival; PFS: progression-free survival; IVC: inferior vena cava.
Discussion
Treatment modalities for patients with advanced HCC 18 include chemoembolization, radioembolization and systemic therapy. 19 Newer studies have assessed the combinations of local and systemic therapies for advanced HCC, evaluating response rates, OS and PFS. 19 SORAMIC was a randomised controlled trial that compared the combination of Y90 radioembolization plus sorafenib with sorafenib alone in patients with unresectable HCC. 20 The median OS for SORAMIC was 12.1 months for the combination therapy and 11.4 months for sorafenib alone. 20 Another study evaluated the outcomes of combining Y90 radioembolization and sorafenib. 21 For BCLC stage B patients, the OS was 20.3 months and the PFS was 15.2 months. 21 A recent study also compared the OS between Y90 radioembolization and systemic therapy in HCC with major vascular invasion. The median OS for radioembolization was 7.1 months and for systemic therapy, it was 4.9 months. 22 An ongoing trial (STOP-HCC) will further evaluate the safety and effectiveness of combining sorafenib and Y90 radioembolization compared to sorafenib alone in unresectable HCC. 23
In our study, we focused on the outcomes of combination therapy using Y90 and sorafenib/nivolumab for HCC-HV/IVC patients. Patients with unresectable HCC with tumor thrombosis in the HV or IVC usually have poorer survival outcomes. 24 Systemic therapy such as sorafenib is commonly used in unresectable HCC as non-invasive options are preferred. 16 These articles demonstrate the potential effectiveness of combining Y90 radioembolization with sorafenib or nivolumab. Benefits of Y90 radioembolization include high tumor response rates and the ability to bridge patients to liver transplantation. 25 Nivolumab, a PD-1 inhibitor, has been demonstrated to produce robust anti-tumoral immune response which complements the immunogenic effect of Y90 radioembolization. 15 Sorafenib, an antiangiogenic agent, also normalizes tumor vasculature and improves the delivery of oxygen to the tumor which complements the immunogenic effect of Y90 radioembolization. 10
The main limitation of our study is the small reported number of HCC patients with HV or IVC invasion that have undergone Y90-systemic combination therapy. Moreover, the optimal interval and dosage of sorafenib or nivolumab when used with Y90 radioembolization have not been fully explored. 10 Therefore, future studies including multi-centre trials could evaluate the role of Y90-systemic combination therapy in HCC-HV/IVC invasion. Currently, there is considerable interest in the combination of Y90 and atezolizumab with bevacizumab. We are presently conducting a multi-centre trial to study the value of this combination.
Conclusion
The combination of local and systemic therapies demonstrated better results with increased response rates, OS and PFS benefits. This review of literature suggests that the combination of systemic (sorafenib or nivolumab) and local therapy (Y90 radioembolization) is effective and safe in patients with HCC-HV/IVC. However, the majority of literature on this topic comprises small studies and case reports which limit generalizable conclusions. Comparative longitudinal studies are warranted to elucidate the effect of systemic versus local therapy in HCC-HV/IVC.
Footnotes
Author contributions
SC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data
The datasets generated and analysed during the study are available from SC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
