Abstract
Introduction:
Pleural fluid adenosine deaminase (pfADA) is a simple, rapid and inexpensive surrogate marker for tuberculous pleural effusion (TPE). A nationwide cut-off of 40 U/L is currently used based on overseas data. There is a need to optimise the diagnostic utility of pfADA by establishing a local cut-off value. In this study, we aimed to describe the demographics and clinical characteristics of patients with TPE and non-TPE; determine the sensitivity and specificity of current pfADA of 40 U/L; and establish a new local pfADA cut-off for TPE.
Methods:
We conducted a single-centre, observational, prospective study of patients with exudative pleural effusion and pfADA measured from 1 October 2019 to 30 April 2020 at Queen Elizabeth Hospital, Malaysia.
Results:
The diagnosis of analysed patients (n = 93) included TPE (n = 41), malignancy (n = 28), parapneumonic effusion (n = 12) and other causes (n = 12). The mean pfADA was 51.15 U/L (standard deviation (SD) = 13.77) among TPE group and 18.86 U/L (SD = 12.33) among non-TPE. When analysis was restricted to TPE patients, the local pfADA cut-off is 29.6 U/L, with a sensitivity of 97.6% and specificity of 90.4%. The current pfADA of 40 U/L has a sensitivity of 87.8% and specificity of 92.3%.
Conclusion:
We established a local pfADA cut-off of 29.6 U/L for TPE. Optimising the utility of pfADA helps to enhance clinicians’ treatment confidence of TPE when initial work-up is inconclusive.
Introduction
Tuberculosis is a major public health issue in Malaysia. Incidence of tuberculosis in Malaysia was reported at 92 per 100,000 population in 2019, according to the latest World Health Organisation global tuberculosis report. 1 Tuberculous pleural effusion (TPE) is the most common extra-pulmonary manifestation of tuberculosis.1,2 In Malaysia, TPE remains the leading cause of exudative pleural effusion, followed by malignant pleural effusion (MPE) and parapneumonic effusion (PPE). 3 A prompt delineation of TPEs from other common causes of exudative pleural effusions are very much needed for therapeutic and public health reasons.
Despite being a common clinical entity, diagnosing TPE can be challenging. Conventional smear microscopy of pleural fluid has low yield rates of below 10%. 4 Pleural fluid cultures using solid media are positive in less than 30% of cases. 4 Even rapid molecular tests such as Xpert Mycobacterium tuberculosis/Rifampicin resistance (Xpert MTB/RIF) report a wide range of sensitivity, from 28% to 81% in pleural fluid and 90% in pleural tissue, albeit with a high specificity of 90% to 100%. 5 While medical thoracoscopy has a sensitivity of up to 100% for TPE, and is able to improve culture and Xpert MTB/RIF yield rates, it is not always available in resource-limited settings.6–8
Adenosine deaminase plays an important role in the development and maintenance of the immune system in humans.9–11 Pleural fluid adenosine deaminase (pfADA) is useful to aid in the diagnosis of TPE as it is inexpensive, simple to perform and has a high sensitivity and specificity.12–16 A pfADA cut-off level of 40 U/L has been used in local settings to diagnose TPE. This value was derived based on data obtained from earlier studies conducted overseas. 17 Subsequent studies, however, revealed that a low pfADA level of <40 U/L in TPE is occasionally observed in countries with high tuberculosis burden. 18 pfADA levels lower than 40 U/L have been reported in 7%, 12% and up to 18.8% in three separate studies among patients with confirmed TPE.18–20 Patients with TPE can be erroneously denied treatment by failing to consider the possibility of low pfADA in certain cases. Realising that the incidence rates of TPE differ between countries and regions, local studies from Thailand and Hong Kong demonstrated that the diagnostic accuracy of pfADA can be further enhanced by establishing a local pfADA cut-off value.10,21
In this study, we aimed to: (a) describe the demographics and clinical characteristics of patients with TPE and non-TPE; (b) determine the sensitivity and specificity of the current pfADA value of 40 U/L in diagnosing patients with TPE; and (c) establish a new local diagnostic pfADA cut-off value for patients with TPE.
Methods
We conducted a single-centre, observational, prospective study on pleural fluid analysis of patients with pleural effusions who underwent thoracocentesis, chest drain insertion or medical thoracoscopy from 1 October 2019 to 30 April 2020 at Queen Elizabeth Hospital, Malaysia. Patients were recruited as outpatients from the respiratory clinic or as inpatients from medical and respiratory wards. Patients with exudative pleural effusions that had pfADA levels measured were included in the study’s analysis. Patients with uncertain diagnosis or with presumptive diagnosis (e.g. empirical treatment of TPE based on clinical and radiological characteristics without confirmatory bacteriological evidence) were followed up for two months after initial recruitment to ascertain the final diagnosis. Patients who do not fulfil case definitions, patients with transudative pleural effusions, patients who declined pleural investigations, patients with pfADA levels measured by different labs, patients with missing or rejected pfADA levels and patients lost to follow-up were excluded. Duplicated specimens – that is, a second sample from one patient – were also excluded.
The following data were collected from the study subjects: gender, age (at time of subject recruitment), duration of clinical symptoms (more or less than two weeks), presenting symptoms (cough, dyspnoea, chest pain, fever, haemoptysis, night sweats, weight loss and reduced appetite), smoking history, co-morbidity (diabetes mellitus, hypertension, chronic kidney disease, liver disease, dyslipidaemia, heart disease, cerebrovascular accident), nature of pleural fluid (exudative or transudative based on Light’s criteria), pleural fluid cytology results, pleural fluid or pleural biopsy histopathological and bacteriological (acid fast stain or Xpert MTB/RIF) findings, pfADA levels and the definitive diagnosis.
We aimed to determine the sensitivity and specificity of the pfADA value at its current cut-off level of 40 U/L, as well as to identify a new optimal local pfADA diagnostic cut-off level among TPE patients. We utilised the receiver operating characteristic (ROC) curve and Youden Index (the point of maximal summation of sensitivity and specificity) to test the reliability of the current pfADA value and to establish a new local pfADA value. This study was conducted in accordance with the latest amended Declaration of Helsinki. The study protocol was approved by the Medical Research and Ethics Committee, Ministry of Health, Malaysia (approval number: NMRR-19-2339-49855 Investigator initiated research (IIR)).
Case definitions
Pleural fluid was considered exudative if one of the following Light’s criteria was present: (a) pleural fluid protein to serum protein ratio greater than 0.5; or (b) pleural fluid lactate dehydrogenase (LDH) to serum LDH ratio greater than 0.6; or (c) pleural fluid LDH level greater than two-thirds the upper limit of the laboratory’s reference range of serum LDH. The diagnosis of TPE was established by (a) histological demonstration of caseating granulomas on pleural biopsy specimens; and/or (b) bacteriological confirmation of Mycobacterium tuberculosis on acid-fast staining or rapid molecular test (Xpert MTB/RIF) of pleural fluid or biopsy materials; and/or (c) positive pleural fluid or pleural biopsy cultures for M. tuberculosis; and/or (d) definite radiological and clinical improvement after two months of anti-tuberculosis medications as per clinician’s judgement. Patients with caseating granulomas on pleural biopsy specimens were followed up for two months after initiation of anti-tuberculous medications to ascertain radiological and clinical improvements. This was to exclude other granuloma-associated conditions such as sarcoidosis and tumour-related sarcoid reactions.
Pleural effusions were classified as malignant when malignant cells were identified on pleural fluid cytology and/or pleural biopsy results. PPEs referred to any effusions secondary to infections such as bacterial pneumonia, lung abscess or bronchiectasis. A definitive diagnosis of PPE was made in the presence of clinical and radiological improvements with antibiotics or when pleural fluid or pleural biopsy materials had positive microbiological results.
Laboratory methods
Throughout our study period, pfADA levels were measured using a Dirui adenosine deaminase reagent kit (Dirui Industrial Co. Ltd.) on an AU400® chemistry analyser (Beckman Coulter, Brea, CA, USA). The detection range was 0.28 U/L to 200 U/L. All tests were conducted at the Public Health Laboratory Kota Kinabalu, Malaysia. The reagent kits and analyser were validated and calibrated at regular intervals according to the manufacturer’s recommendations.
Statistical analysis
Numerical data are expressed as mean and standard deviation (SD) if they follow normal distribution; for non-normally distributed data, median and interquartile range (IQR) are reported instead. Categorical data are expressed as frequencies and percentages, and comparisons were analysed using the Chi-square test or Fisher’s exact test, as appropriate. Continuous variables were analysed using Student’s t-test. Demographic and clinical characteristic factors associated with TPE status were evaluated using the aforementioned inferential tests. Areas under curve of ROC curve of pfADA values used to diagnose TPE were computed. A Youden Index was then calculated based on the sensitivity and specificity of the corresponding pfADA values. The pfADA value that provides the maximum value of the Youden Index was chosen as the optimal cut-off value. All tests of significance were two-sided, as we took a p-value of <0.05 as statistically significant. Statistical analyses were done using IBM SPSS Statistics for Windows (IBM Corp., Armonk, NY, USA).
Results
Subject demographics and clinical characteristics
During the study period, a total of 219 independent pleural fluid specimens were collected for analysis. We evaluated 97 patients with exudative pleural effusion and with pfADA taken after excluding cases with transudative pleural effusions and duplicated specimens. Four cases were further removed due to incomplete follow-up and data collection (Figure 1). Of the 93 cases that were analysed, 61 patients were male (65.6%). The median age of patients was 55 years old (IQR 33.5 years old). Patient demographics and their clinical characteristics are shown in Table 1. Fever (p = 0.005), diabetes mellitus (p = 0.025) and presence of kidney disease (p = 0.040) are found to be associated with TPE status. Patients from TPE group were significantly younger (mean age 43.54 ± 20.20 years old versus 56.77 ± 17.97 years old in non-TPE group; p = 0.001) (Table 2).
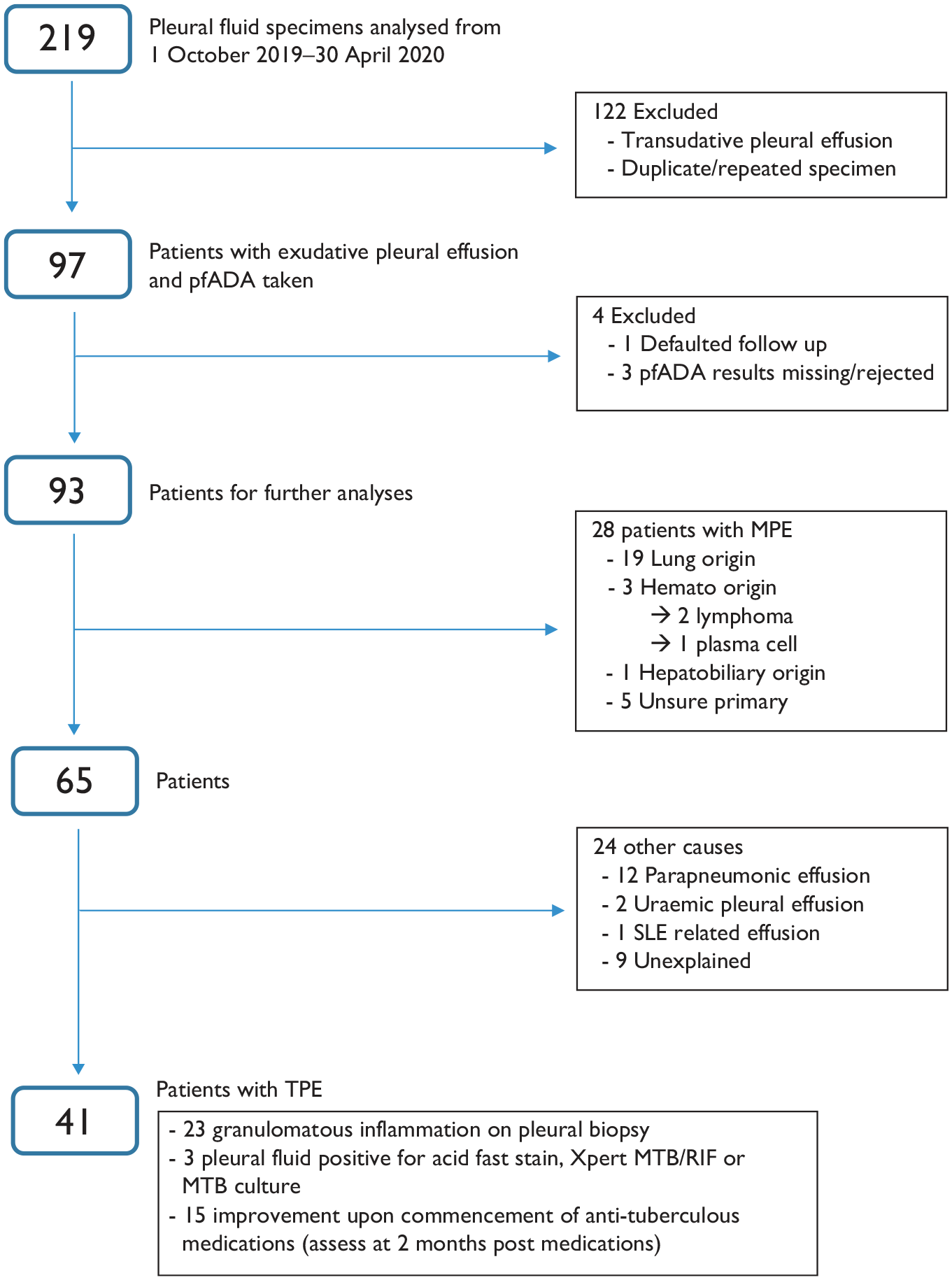
Study flow and aetiologies of pleural effusions.
Patient demographics and clinical characteristics.

IQR: interquartile range.
Factors associated with TPE and non-TPE status.

TPE: tuberculous pleural effusion; SD: standard deviation.
Fisher’s exact test was used because assumption for Chi-square test not satisfied.
Independent t-test.
pfADA levels in various exudative pleural effusions
The most common cause of exudative pleural effusion was TPE, with 41 cases (44.1%). Other conditions in decreasing frequencies were: MPE 28 (30.1%), PPE 12 (12.9%), unexplained 9 (9.7%), uraemic pleural effusion 2 (2.2%) and connective tissue disease related 1 (1.1%). Table 3 illustrates the diagnoses of patients and their corresponding pfADA values.
Diagnosis of pleural effusion among patients with pfADA measured.

TPE: tuberculous pleural effusion; MPE: malignant pleural effusion; PPE: parapneumonic effusion; SLE: systemic lupus erythematous; pfADA: pleural fluid adenosine deaminase; SD: standard deviation.
Among 41 patients with TPE, the diagnosis was (1) histologically suggested by pleural biopsy results in 23 (56.1%); (2) bacteriologically confirmed by positive acid-fast stain or Xpert MTB/RIF results or positive culture yield for M. tuberculosis in three (7.3%); and (3) diagnosed clinically and radiologically as evidenced by improvement after two months of anti-tuberculosis medications as per clinician judgement in 15 (36.6%). Calculated mean (SD) and range (min–max) of pfADA values in TPE patients were 51.15 U/L (SD = 13.77) and 79.88 U/L (16.85–96.73), respectively. Only three patients with TPE had positive sputum smears for acid-fast bacilli.
Among 28 patients with MPE, 24 (85.7%) were diagnosed by demonstrating malignant cells on pleural fluid or pleural biopsy samples. The remaining four (14.3%) cases were diagnosed based on clinical and radiological grounds. MPEs were caused by lung carcinoma in 19 (67.9%) patients, haematological malignancy in three (10.7%), hepatobiliary cancer in one (3.6%) and unknown primary in five (17.9%). The mean (SD) and range (min–max) pfADA among patients with MPE were 17.85 U/L (SD = 10.19) and 42.65 U/L (5.35–48.00), respectively.
Among 12 patients with PPE, the diagnosis was bacteriologically confirmed by positive pleural fluid culture results in two (16.7%), with the remaining cases diagnosed based on clinical and radiological improvements upon commencement of antibiotics. The mean (SD) and range (min–max) pfADA values were 23.84 U/L (SD = 18.15) and 64.62 U/L (3.99–68.61), respectively. The proportion of patients with different aetiologies according to the optimal pfADA cut-off point of 29.6 U/L can be found in Table 4. We demonstrated that TPE was unlikely when the pfADA level was more than 100 U/L. All patients with TPE had pfADA levels of lesser than 96.73 U/L in our study.
Proportion of patient according to optimal pfADA cutoff value of 29.6.

TPE: tuberculous pleural effusion; MPE: malignant pleural effusion; PPE: parapneumonic effusion; SLE: systemic lupus erythematous; pfADA: pleural fluid adenosine deaminase.
Optimising pfADA level for the diagnosis of TPE
To determine the sensitivity and specificity of the current pfADA value of 40 U/L in diagnosing TPE among patients with exudative pleural effusions, we performed a step-by-step analysis by first excluding transudative effusions and repeat samples followed by stepwise exclusion of non-TPE cases. Based on our analysis, the pfADA value of 40 U/L had a sensitivity of 87.8%, specificity of 92.3%, positive predictive value (PPV) of 90.0% and negative predictive value (NPV) of 90.6%. We further analysed the ROC curve and utilised the Youden Index (Figure 2) in order to identify the best cut-off pfADA value for the diagnosis of TPE. Again, by analysing patients with negative findings for other non-TPE causes, a local cut-off pfADA value of 29.6 U/L was established for the first time. This pfADA value corresponded to a Youden Index of 0.879, and had a sensitivity of 97.6%, specificity of 90.4%, PPV of 88.9% and NPV of 97.9%.

ROC curve of pfADA level to identify a new local cut-off level for TPE.
Discussion
We analysed the demographics and clinical characteristics of patients with TPE versus non-TPE. TPE patients were more likely to present with fever and were younger compared to non-TPE patients (p = 0.005). We postulate that the age differences could be explained by a higher incidence of non-TPE cases, especially MPEs among older patients. Besides, the incidence of TPE in endemic areas is higher in younger patients, as primary infection predominates over reactivation as the mechanism of development of disease. 9 In addition, our data revealed a significantly higher proportion of patients with diabetes mellitus (p = 0.025) and kidney disease (p = 0.040) among patients with TPE. Renal impairment is associated with an acquired immunodeficiency state due to functional abnormalities of neutrophils, compromised lymphocyte function and reduced natural killer and monocyte activity. 22 Patients with diabetes mellitus have up to three times higher risk of developing active tuberculosis when compared to people without diabetes mellitus.23,24
We further performed a subgroup analysis of pfADA levels between different age groups (younger age group <55 years old versus older age group >55 years old). The mean pfADA level among older patients (age >55) was 26.92 U/L, while the mean pfADA level among younger patients (age <55) was 39.14 U/L (p = 0.004). When analysis is restricted to patients with TPE, the mean pfADA was 48.14 U/L among older patients (age >55) and was slightly higher at 52.71 U/L among younger patients (age <55) (p = 0.32). Previous studies have demonstrated a negative correlation between pfADA levels and age. 25 A similar trend is observed in our study samples, although the results were not statistically significant. We postulate that the relatively small sample size of our study might be responsible for such findings.
As a surrogate marker for TPE, pfADA values need to be correlated with other clinical and radiological findings. Using local data, we demonstrated that TPE is unlikely when pfADA values were more than 100 U/L, echoing previous studies. 10 There is a need to consider non-TPE causes such as empyema- or lymphoma-related pleural effusion when pfADA exceeds 100 U/L. 26 Although a diagnostic cut-off value of 40 U/L has been used nationwide for TPE based on earlier overseas data, there is a need to establish a local cut-off value to further optimise the diagnostic accuracy of pfADA.10,21 pfADA assay is inexpensive, rapid and simple to perform, requiring only a small amount of pleural fluid for analysis. Its characteristics enable rapid decision-making at the time of initial diagnostic thoracentesis, which can be performed safely at day-care or outpatient settings. Among patients with a pfADA level of more than 29.6 U/L, with early negative findings for MPE or other infective causes from initial analysis of pleural fluid, it is probably appropriate to consider empirical treatment for TPE. This option is attractive in resource-limited settings, among patients who refused further more invasive tests or among patients without a good ‘window’ to perform medical thoracoscopy due to a small amount of pleural effusion or dense septations. Nonetheless, it is vital to exercise caution due to a 11.1% (1 minus PPV) chance of falsely diagnosing TPE among non-TPE patients. On the other end of the spectrum, there is a 2.1% (1 minus NPV) chance of misclassifying non-TPE among patients with true TPE. Thus, the availability of pfADA should not deter healthcare providers from offering other standard-of-care tests such as medical thoracoscopy or Abrams needle pleural biopsy whenever possible. Unnecessary treatment of TPE exposes the patient to potential toxic effects of anti-tuberculous medications, while misclassifying true TPE patients as non-TPE will inadvertently lead to delay in attaining competing differential diagnoses, especially MPE. Hence, we recommend serial follow-ups to look for clinical and radiological improvements upon commencement of anti-tuberculosis medications amongst all patients diagnosed with TPE. A proposed algorithm is presented in Figure 3.

Proposed algorithm to distinguish TPE from non-TPE in patients with exudative pleural effusion.
pfADA assays can be adapted to various analytical systems, including the AU400® chemistry analyser (Beckman Coulter, Brea, CA, USA) used in our study. One previous study using the Cobas® 6000 analyser (Roche Diagnostics, Meylan, France) recommended a cut-off value of between 25 and 30 U/L for TPE diagnosis, consistent with our proposed local cut-off value. 27 This raises the question of whether cut-off values will differ according to different analytical systems. We performed a literature review in an attempt to answer this question. A comparison study conducted using different analytical systems – namely, Advia 1800 (Siemens Healthcare Diagnostics, Saint Denis, France) and the Cobas® 6000 analyser – showed a good correlation between the two analysers (r2 = 0.88; t-test for correlation coefficient, p < 0.001). 28 We believe that further large-scale comparison studies will be useful to address the level of consistency between various pfADA sample analysers. Published data on sustained good correlation between analysers can further boost a more widespread usage of pfADA for TPE diagnosis.
We accept that a limitation is the size of the study and, therefore, increasing generalisability and applicability of findings may have been found with a larger sample size. Besides, misclassification bias might occur, partly owing to the natural history of TPE. TPE can be a self-limited condition, and the natural history of untreated TPE is characterised by spontaneous resolution in some patients with potential of subsequent reactivation. 8 This phenomenon might lead to misclassification of TPE as non-TPE. Another possible source of information bias was uncertain aetiology of pleural effusion, which were grouped as non-TPE in our analysis. Nonetheless, efforts were made by researchers to follow-up cases with non-confirmatory initial diagnosis for at least two months after first encounter and data collection to minimise the impact of such bias. To the authors’ best knowledge, this is the first study to establish a local cut-off value of pfADA for TPE in Malaysia. We hope that our study will lead to further research to refine the diagnostic (or even prognostic) utility of pfADA among TPE and non-TPE patients in different clinical settings.
Conclusion
For the first time, we have established a local pfADA diagnostic cut-off value of 29.6 U/L for TPE. pfADA appears to be an accurate and rapid yet minimally invasive test to aid in diagnosing TPE. It enhances clinicians’ treatment confidence of TPE, thereby improving patient care, especially when other initial work-up for TPE is inconclusive. In the future, we hope that the generalisability and applicability of our findings can be further enhanced by performing multi-centre studies with larger sample sizes.
Footnotes
Authors’ contributions
NCH and ISK contributed to the design and implementation of the research. KCN, AS, QZL and WJT carried out the data collection. MYL performed data analysis and calculations. NCH and HYR wrote the manuscript. KKSK supervised the project. All authors discussed the results and contributed to the final manuscript.
Availability of data and materials
The datasets generated and/or analysed during the current study are available from NCH (corresponding author,
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethical approval
Ethical approval for this study was obtained from the Medical Research and Ethics Committee, Ministry of Health, Malaysia (approval number NMRR-19-2339-49855 (IIR)).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patients for their anonymised information to be published in this article.
