Abstract
Background:
Fever without source in infants is a common clinical problem that accounts for many ambulatory care visits and hospitalisations. Currently, there is no reliable method of identifying those at risk of serious infection (SI).
Objective:
The goal of this study was to determine the incidence and identify the predictors of SI in febrile infants who presented to the emergency department (ED).
Methods:
This was a single-centre retrospective cohort study of children presenting to a Singapore tertiary hospital paediatric unit between 1 July 2018 and 31 December 2018. Children were included if they were aged 0–90 days and presented to the ED with a fever. SI was defined as urinary tract infection (UTI), sepsis, bacteraemia, meningitis (viral and bacterial), enterocolitis, osteomyelitis, abscess or pneumonia.
Results:
Of the 659 infants, 161 (24.4%) were diagnosed with SI. Meningitis (49.7%) was the most common SI, followed by UTI (45.3%), enterocolitis (5.6%), sepsis (3.1%) and bacteraemia (2.5%). Factors significantly associated with SI were aged 29–60 days, male sex, Severity Index Score (SIS) <10, absolute neutrophil counts >10×109/L, C-reactive protein (CRP) >20 mg/L and procalcitonin >0.5 ng/mL. Multivariate analysis entering all these items retained only male sex, SIS <10 and CRP >20.
Conclusion:
Among hospitalised infants aged 0–90 days, the incidence of SI was 24.4%, and invasive bacterial infection was 0.6%. Meningitis was the most common SI followed by UTI. SIS and CRP can be used to predict SI in infants <90 days old.
Introduction
Fever without source in infants is a common clinical problem that accounts for many ambulatory care visits and hospitalisations. Young febrile infants (aged 0–90 days) often present with non-specific symptoms, and often it is difficult to distinguish between infants with a viral infection and those with early serious bacterial infection. Therefore, the management of infants <90 days old with fever without source has been challenging. In the literature, the rates of serious bacterial infections were 11–34% in those aged <30 days and 13–19% in those aged 30–90 days.1,2 To predict serious bacterial infection risk better, a number of clinical and laboratory criteria are used in paediatric institutions to stratify the risk, with the aim of identifying low-risk infants who can forego empirical antibiotics.3,4 More recently, the ‘Step by Step’ approach by Kupperman et al. incorporated new biomarkers, such as procalcitonin (PCT), which have shown good sensitivity and negative predictive value. 5 However, more studies are needed to evaluate the clinical prediction tools in improving the outcome of this infant group.
In this study, we sought to establish the incidence of serious infection (SI) and to identify predictors of SI in febrile infants aged 0–90 days presenting to the emergency department (ED). This will help us to understand the infection risk of these febrile infants better.
Methods
Study design and setting
We performed a single-centre retrospective study of infants aged 0–90 days presenting with fever without source at the ED and who underwent an evaluation for SI. Institutional Review Board approval was obtained with a waiver of informed consent (2020/2795).
Patient population
Using the institutional data repository of electronic medical records, we identified all children presenting to the ED with fever between 1 July 2018 and 31 December 2018. This study period was selected, and the patient sample was drawn as part of a quality improvement initiative done in the department. Infants were included if they were <90 days old and had a fever, which was defined as a temperature of ⩾38°C. Temperature was measured at triage by a nurse using an axillary or rectal thermometer. In our hospital, all febrile infants younger than three months are hospitalised, and therefore this formed a natural pool of febrile infants with complete outcomes that can be tracked. The management and investigations are decided by the ward physicians. It is usual practice for neonates <28 days old with fever to undergo blood, urine and cerebrospinal fluid (CSF) cultures. Older infants may undergo limited investigations, depending on clinical assessment. All infants are monitored in the hospital until the fever has resolved for at least 24 hours before being discharged.
Patient exclusion
The exclusion criteria were as follows: (a) pre-term infants with a gestational age of <37 weeks; (b) infants with significant neonatal complications requiring a prolonged stay (>7 days) in the neonatal intensive care unit; (c) any pretreatment of oral or intravenous antibiotics within the past week; (d) patients with co-morbid conditions predisposing them to severe or recurrent bacterial illness, including genetic, congenital, chromosomal, malignancy, neuromuscular or neurodevelopment abnormalities; (e) patients with central venous catheters or ventriculoperitoneal shunts; and (f) patients transferred from another hospital inpatient setting.
Study definition
SI was defined as urinary tract infection (UTI), sepsis, bacteraemia, meningitis (viral and bacterial), enterocolitis, osteomyelitis, abscess and pneumonia.6–8 We included viral meningitis as part of the definition of SI because it is known to be associated with long-term health sequelae such as cognitive impairment and poor developmental outcomes.9,10 Invasive bacterial infection (IBI) was defined as isolation of a bacterial pathogen in a blood or CSF culture.11,12 The rest of study definitions are shown in Table 1.
Study definition.
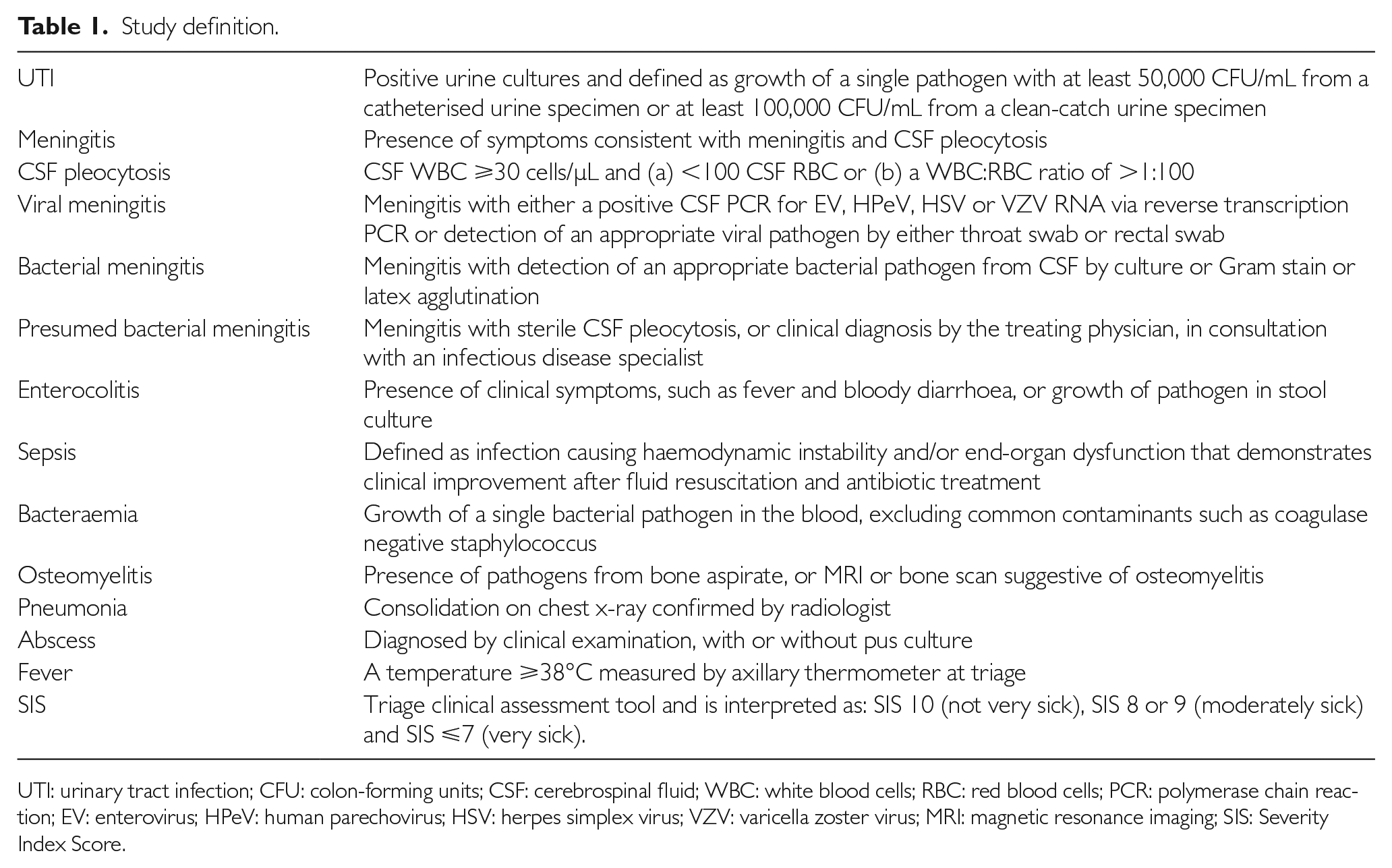
UTI: urinary tract infection; CFU: colon-forming units; CSF: cerebrospinal fluid; WBC: white blood cells; RBC: red blood cells; PCR: polymerase chain reaction; EV: enterovirus; HPeV: human parechovirus; HSV: herpes simplex virus; VZV: varicella zoster virus; MRI: magnetic resonance imaging; SIS: Severity Index Score.
Data extraction
The following data were recorded: demographics (age in days, sex and race), final discharge diagnosis, length of stay and follow-up. We also recorded Severity Index Score (SIS), which is a validated composite index score used to triage patients in the ED based on respiratory effort, colour, activity or level of consciousness, temperature and functional status of playing. A score of 10 points is designated as ‘not sick’, 8 or 9 points as ‘moderately sick’ and ⩽7 points as ‘very sick’. 6 Therefore, any score of <10 denotes a sick child who require further assessment and treatment. Biochemical results, including total white blood cell count, absolute neutrophil count (ANC), platelets, C-reactive protein (CRP), PCT, urinalysis and CSF analysis, were collected. In addition, microbiology culture results from urine, blood, CSF, stool and other sites (abscess, wounds) as well as nasopharyngeal aspirate for common viruses were recorded.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows v23.0 (IBM Corp., Armonk, NY). Normally distributed data are expressed as the mean±standard deviation, non-normally distributed data as the median and interquartile range (IQR) and categorical data as frequencies and percentages. Statistical comparisons were performed using Student’s two-tailed t-test and chi-square testing for all numerical and categorical values, respectively. Significance was set at p<0.05.
To find out the risk factors associated with SI in infants ⩽90 days old, we dichotomised age, sex, SIS score, ANC, CRP and PCT, and these were first evaluated using univariate analysis. Based on the earlier definition, an SIS score of <10 indicated an abnormal assessment, which in turn could be an early indicator of evolving sepsis. Therefore, we chose the SIS cut-off point of 10 and dichotomised it into 10 and <10. The cut-off chosen for ANC, CRP and PCT were those commonly used in evaluating serious bacterial infection.13,14 The factors that were significant in the univariate analysis were then subjected to multivariate logistic regression analysis. Adjusted odds ratios (aOR) are presented with their corresponding 95% confidence intervals (CI), and statistical significance was set at p<0.05.
Results
Patient demographics and clinical characteristics
The patient demographics and characteristics are shown in Table 2. A total of 659 infants were analysed. Most of the infants included were ⩽28 days old (55.8%), followed by infants 29–60 days old (24.2%) and those 61–90 days old (20%). The median age of the SI and non-SI study population was 20 days (IQR 5–50 days) and 32 days (IQR 15–58 days), respectively. There were 396 (60.1%) males. The ethnic composition was Chinese (56.3%), followed by Malay (26.9%), Indian (7.9%) and other ethnic groups (8.9%).
Clinical characteristics of infants with or without SI.
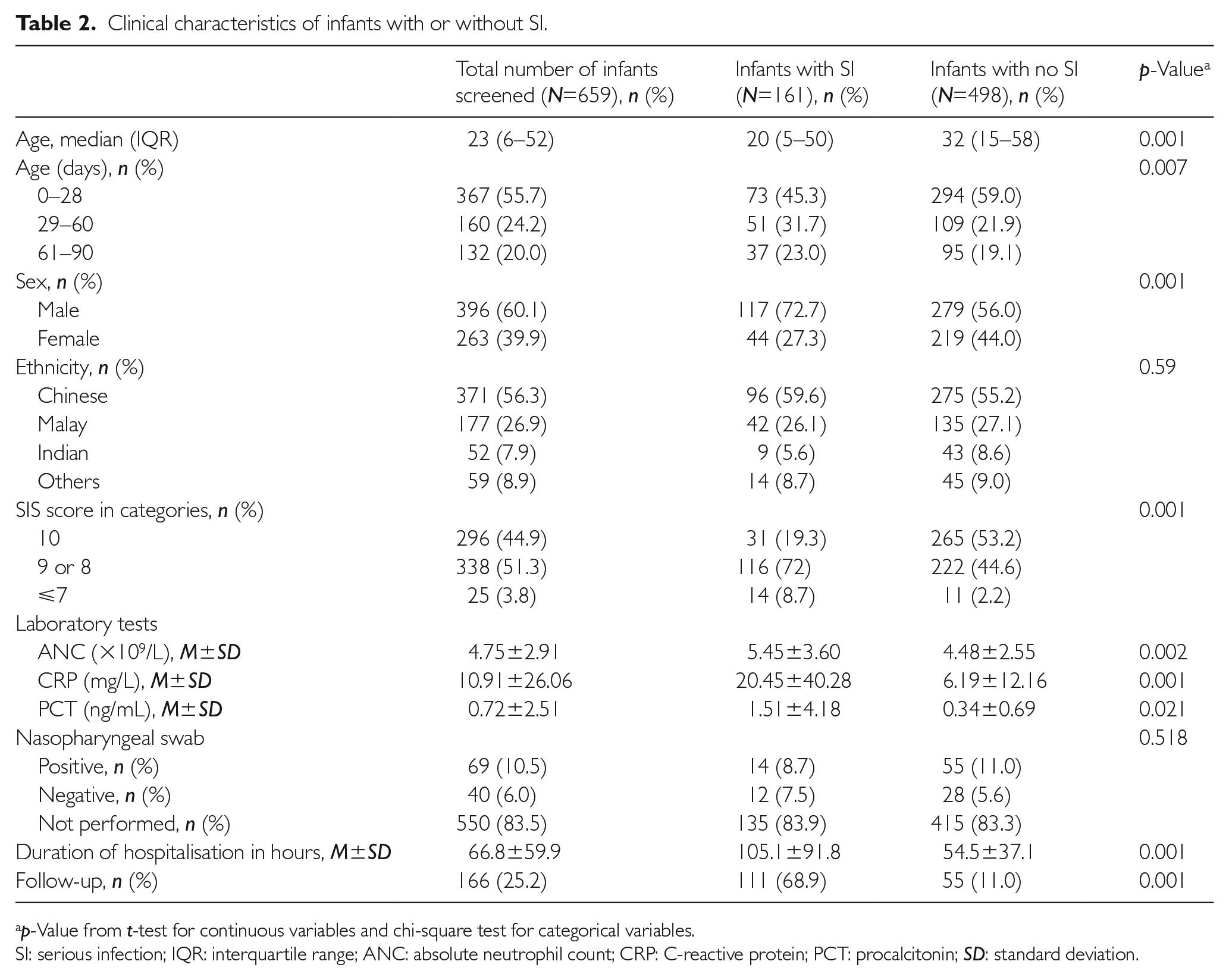
p-Value from t-test for continuous variables and chi-square test for categorical variables.
SI: serious infection; IQR: interquartile range; ANC: absolute neutrophil count; CRP: C-reactive protein; PCT: procalcitonin; SD: standard deviation.
SI result
Of the 659 infants, 161 (24.4%; 95% CI 21.3–27.9%) infants were diagnosed with SI. The male-to-female ratio in infants with SI was 2.7. Comparing age groups, the incidence of SI was highest in infants ⩽28 days old (45.3%; 95% CI 37.9–53.1%), followed by infants 29–60 days (31.7%; 95% CI 25–39.2%) and infants 61–90 days old (23.0%; 95% CI 17.2–30.1%). SIS score, mean value of ANC, CRP and PCT were significantly higher in infants with SI compared to those without SI (Table 2). Only 16.5% of the infants had nasopharyngeal aspirate for common respiratory viruses performed. Of these, 14 (8.7%) with SI had a positive nasopharyngeal aspirate for common respiratory viruses compared to 55 (11.0%) infants with no SI (p=0.518). A significant proportion of infants with SI had longer hospitalisations and required follow-up compared to infants without SI (p<0.001).
Table 3 shows the bacterial cultures by age group in infants with SI. Infants aged 0–28 days were more likely to receive a complete evaluation for SI, including blood, urine and CSF cultures, compared to infants 29–60 days and 61–90 days old (94.5% vs. 90.2% vs. 75.7%, respectively). Infants 61–90 days were more likely to have blood and urine cultures done without CSF cultures (16.2% vs. 4.1% in infants 0–28 days old and 9.8% in infants 29–60 days old).
Bacterial cultures by age group in infants with SI.

Table 4 shows the SI comparison between age group. Meningitis (49.7%) was the most common SI, followed by UTI (45.3%), enterocolitis (5.6%), sepsis (3.1%) and bacteraemia (2.5%). Among the 80 infants with meningitis, 72 (90%) were viral, and eight (10%) were bacterial. Both incidences of viral and bacterial meningitis declined with increasing age. Fifty-one (72.2%) viral meningitis cases were secondary to human enterovirus (EV), 13 (18.1%) to human parechovirus (HPeV) and one (1.4%) to herpes simplex virus. Of the eight infants with bacterial meningitis, one had a CSF culture positive for group B streptococcus (GBS), and the remaining seven infants had sterile pleocytosis (CSF culture negative) bacterial meningitis. UTI was the second most common SI, with an overall rate of 45.3%, of which Escherichia coli and Enterococcus sp. were the commonest Gram-negative and Gram-positive pathogens, respectively. Nine infants were diagnosed with enterocolitis. Five infants with sepsis were identified, of whom one required admission to the high dependency unit. There were 11 infants with multiple infections, of whom seven had concomitant UTI and EV meningitis; one had E. coli bacteraemia and enterocolitis; one had disseminated GBS with UTI, meningitis and bacteraemia; and two had sepsis with enterocolitis.
Serious infection: comparison between age groups.
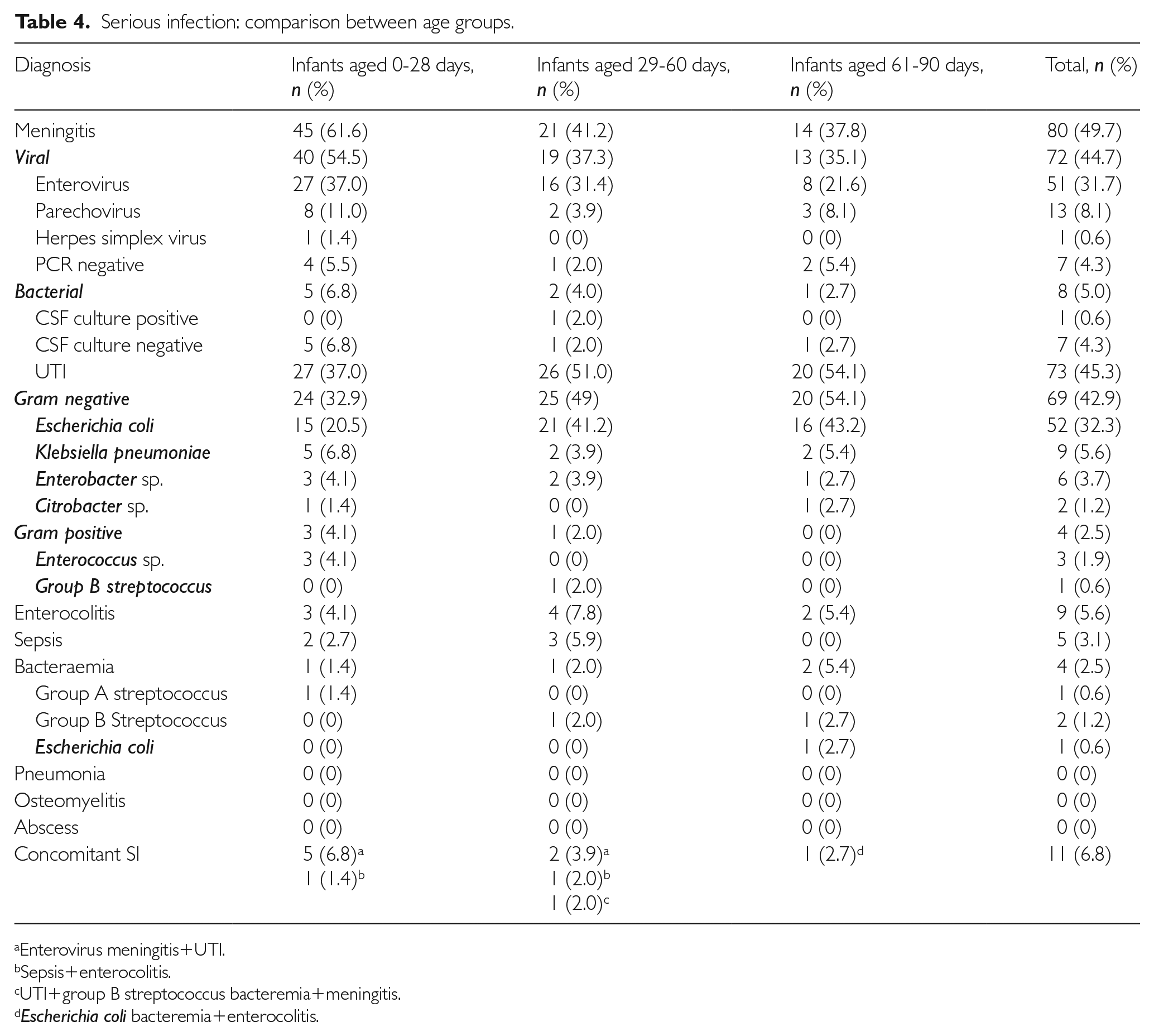
Enterovirus meningitis+UTI.
Sepsis+enterocolitis.
UTI+group B streptococcus bacteremia+meningitis.
Escherichia coli bacteremia+enterocolitis.
IBI result
Our study cohort had four (0.61%) IBI cases. GBS was isolated in two out of the four infants with bacteraemia. One of the two GBS bacteraemic infants had concomitant GBS meningitis. The other two infants with bacteraemia had E. coli and group A streptococcus isolated. When comparing infants with IBI to those without IBI, only CRP was significantly higher (94.77±160.33 vs. 9.30±21.95; p<0.001).
Univariate and multivariate analysis
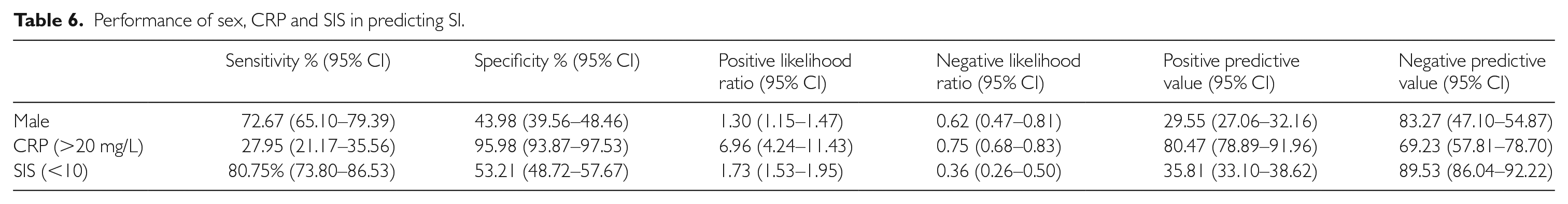
Univariate analysis (Table 5) showed that age 29–60 days (OR-1.88; 95% CI 1.24–2.87; p=0.003), male sex (OR=2.09; 95% CI 1.41–3.08; p<0.001) and SIS score <10 (OR=4.77; 95% CI 3.10–7.33; p<0.001) had a statistically significant relationship with outcome of SI. Investigations showed that ANC >10×109/L (OR=2.94; 95% CI 1.46–5.92; p=0.003) and CRP >20 mg/L (OR=9.27; 95% CI 5.27–16.30; p<0.001) were statistically significant in predicting SI in our population. In the multiple variable analysis, male sex (aOR=1.93; 95% CI 1.26–2.96; p=0.002), SIS <10 (aOR 4.69; 95% CI 2.95–7.47; p<0.001) and CRP (aOR=8.16; 95% CI 4.15–16.07; p<0.001) remained significantly associated with SI. The sensitivity, specificity, positive and negative likelihood ratio, positive and negative predictive values of male sex, CRP and SIS in predicting SI are summarised in Table 6.
Univariate and multivariate analyses of predictors for serious infection.

aOR: adjusted odds ratio; CI: confidence interval; OR: odds ratio.
Performance of sex, CRP and SIS in predicting SI.
Discussion
Overall, the incidence of SI was 24.4% in our study sample, which was higher than the 7–12.8% previously reported in other studies.1,2,4 This is likely due to our broad definition of SI, which included viral meningitis. Conventionally, most studies include only serious bacterial infection. However, a definition of SI that includes viral aetiologies is more pragmatic because viral meningitis is known to be associated with significant long-term cognitive and neurological sequelae.10,15
Our study demonstrated an increased incidence of meningitis, predominantly viral meningitis, as a cause of SI, especially in younger febrile infants aged 0–28 days. EV and HPeV were the two commonest causes of viral meningitis in infants. These two viruses usually cause mild self-limiting respiratory and gastrointestinal infections in children. Occasionally, however, they may be responsible for more serious illness, including encephalitis, hepatitis, myocarditis or fulminant sepsis. 9 None of the reported sepsis in our study was related to EV or HPeV meningitis. Long-term neurological complications have also been reported in Australia, Northern Europe and the USA.9,16 Our study did not evaluate the long-term sequelae, but 86.1% of the infants with viral meningitis were followed up to monitor their neurodevelopment.
The overall incidence of bacterial meningitis in our young febrile infant cohort was 1.2%, which is consistent with the results of other studies, ranging from 0.4% to 1.2%. 17 Most of the bacterial meningitis in our cohort had negative CSF cultures and was diagnosed based on clinical history and risk factors in the presence of significant CSF pleocytosis, in consultation with an infectious disease specialist. Afifi et al. reported low rates of positive CSF culture samples (8%) in suspected cases of bacterial meningitis, and it is therefore not uncommon to obtain negative CSF cultures in patients with suspected bacterial meningitis. 18 In view of this, molecular methods, such as polymerase chain reaction (PCR), are gaining traction to provide an additional adjunct in identifying aetiological pathogens of bacterial or viral meningitis. 19 It has been shown that the PCR method is more sensitive and rapid compared to culture in detecting the infectious agents. 20 BioFire@ FilmArray Meningitis/Encephalitis Panel, which is one of the commercial multiplex PCR assays, only became available in our centre towards the end of this study period. This technique can potentially help us to identify pathogens in those infants with suspected bacterial meningitis with negative CSF cultures.
UTI remains an important cause of SI in neonates and infants. 21 It may be the first indicator of underlying genitourinary structural abnormalities. Our study found UTI to be the second commonest cause of SI, of which the most common pathogens were E. coli and Klebsiella pneumoniae. Approximately 5% of infants with UTI have bacteraemia identified.22–24 In our cohort, only one patient with UTI (1.3%) had associated bacteraemia and meningitis secondary to GBS. This underlines the importance of completing the work-up, including urinalysis and blood culture, as it will affect the duration of antibiotics prescribed for the treatment of UTI. Infants with bacteraemic UTIs tend to be given longer courses of parenteral antibiotics than non-bacteraemic children with UTI. In our institution, we treat young infants with bacteraemic UTI with parenteral antibiotics for at least seven days followed by oral antibiotics.
Sepsis is an important cause of mortality and morbidity in infants. As the symptoms are non-specific, clinical suspicion often leads to early initiation of empirical antibiotic treatment. We identified five cases of culture-negative sepsis. All five infants were resuscitated and received early antibiotic treatment. Culture-negative sepsis has been well described in the neonatal population, which could be due to the small volume of bloods obtained for culture, low level of bacteraemia or fastidious bacteria, fungi or viruses.25,26
Our IBI rate was 0.6%, which was low compared to the 2–5% reported in other studies.13,27 GBS accounted for half of all cases of IBI in our cohort, and all cases were late-onset GBS. The significant reduction in early-onset GBS was likely due to good antenatal follow-up in Singapore and increased use of intrapartum antibiotic prophylaxis for maternal GBS. However, late-onset GBS remains an important pathogen in IBI in young febrile infants.
There was substantial variation in the evaluation of these young febrile infants in terms of inflammatory biomarkers and cultures obtained. Most of the variations were in infants 29–60 and 61–90 days old, with lumbar puncture performance decreasing with age (Table 3). There were two possible reasons why the evaluation of young febrile infants demonstrated such variability: (a) the low rates of bacteraemia and meningitis with changing SI epidemiology worldwide 28 and (b) availability of a discriminative biomarker to differentiate SI and non-SI infants. In our study, we evaluated three biomarkers: ANC, CRP and PCT. All three biomarkers had significant higher mean values for infants with SBI compared to those without. CRP was the only significant biomarker in predicting SI in multivariate analysis. This finding concurs with previous studies which describe CRP as the most discriminatory biomarker for young infants with serious bacterial infection compared to other biomarkers.29–31 ANC and PCT are the other two well -studied biomarkers. However, their reported sensitivity and specificity in the literature are variable.32–37 Our result showed that both ANC and PCT were not significant in predicting SI, and this is likely due to our small sample size and differing cut-off values used.
Our multivariable analysis demonstrated that male sex, SIS <10 and CRP >20 mg/L were significant predictors for SI. A SIS of score <10 has good sensitivity of 80.75% (95% CI 73.80–86.53) and negative predictive value of 89.53% (95% CI 86.04–92.22) in differentiating ‘well’ and ‘sick’ infants and therefore predictive of SI. On the other hand, CRP >20 mg/L has a good specificity of 95.98% (95% CI 93.87–97.53) and positive predictive value of 80.47% (95% CI 78.89–91.96) for SI. Results from previous studies also suggest that CRP may have some value in a low-resource setting, but it was not recommended to be used as a standalone tool to predict SI. 38 To our knowledge, no other study has found sex to be a significant risk factor for SI. This could be due to the relatively small sample size or the fact that UTIs are more common in uncircumcised male infants.
Limitations
Our study has certain limitations. First, it was a single-centre study, and therefore our results must be cautiously extrapolated to other settings. Another limitation of this study includes those inherent to a retrospective design, such as selection bias. It is possible that many children who presented to the ED with fever were missed during the study period due to incorrect diagnosis by the ED physicians. However, we believe that this number was small because patients were likely to return to the ED if symptoms progressed or they were persistently febrile. In view of the variation among clinicians in the management of febrile infants, not all diagnostic tests were performed in all infants. It is possible that some SI cases were missed. Again, we believe the number is small. All febrile infants were being monitored closely in the hospital and examined twice daily. Any changes in the clinical status would be picked up by the medical team, and further evaluation would be conducted. If they remained well and afebrile for this long period of observation (at least 24 hours before discharge), a serious infection can be excluded without the need to perform additional tests.
Conclusion
Among hospitalised infants 0–90 days old, the incidence of SI was 24.4% and IBI was 0.6%. Our study showed meningitis was the most common SI (50.3%) followed by UTI (45.3%). Late-onset GBS infection accounted for 50% of the IBI cases in our cohort. SIS score and CRP can be used to predict SI in febrile infants <90 days old.
Footnotes
Acknowledgements
None.
Availability of data and materials
The data sets generated and analysed during the current study are available from corresponding author.
Authors’ contributions
Z.X.K. conceptualised the study and drafted the initial manuscript. C.C., Z.M.Y., J.C.K., S.C., N.W. and K.T.P. were participated in the study design and data collection. C.Y.C. provided critical input. All authors approved the final manuscript as submitted.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
This study was exempted from review by the Singhealth Centralised Institutional Review Board (CIRB 2020/2795), as it was part of a quality improvement project.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Paediatric Academic Clinical Program J.M. Gomez Faculty Development Fund in Patient Safety and Clinical Quality.
Informed consent
This study was exempted from review by the Singhealth Centralised Institutional Review Board (CIRB 2020/2795), as it was part of a quality improvement project.
