Abstract
Background:
Robust clinical documentation of side-effects communication is an integral component of good patient care, endorsed by the Singapore Medical Council Ethical Code and Ethical Guidelines and further highlighted by the Modified Montgomery Test. In addition to ensuring quality of care, good clinical documentation can help mitigate potential medico-legal risks against doctors and health-care providers.
Objectives:
This audit-cum-quality-improvement project aimed to enhance clinical documentation of side-effects discussion of newly prescribed medication in psychiatry outpatient clinics to 100% in a 12-month period.
Methods:
A baseline measurement revealed that 40% of new cases seen in general and geriatric psychiatry outpatient clinics from March to June 2017 had evidenced clinical documentation of at least one or more side effects. PDSA methodology was employed to bring about improvements and test change interventions.
Results:
Through three audit cycles between January and December 2018 and a fourth round of data collection in April 2019, documentation rates showed marked improvement from a baseline of 40% to 91%. The IT document tool was the most effective intervention which was successfully adopted and implemented into the standard documentation template for new case assessments.
Conclusions:
The project was a success overall, with improvements in documentation rates rising to 91%. Through change interventions, systemic factors rooted in patient safety have been embodied in everyday clinical practice.
Introduction
Robust clinical documentation of the communication of side effects or material risks of treatment with patients is an integral component of good patient care, as endorsed by the Singapore Medical Council Ethical Code and Ethical Guidelines 2016 1 and further highlighted by the Modified Montgomery Test.2,3 The Singapore Medical Council Handbook on Medical Ethics explicitly states that doctors are required to inform patients of the more common drug interactions or side effects or those likely to be significant to specific patients and to maintain clear, legible, accurate and contemporaneous medical records of sufficient detail. In addition to ensuring the quality of care, good clinical documentation can help mitigate potential medico-legal risks against doctors and health-care providers.
Changi General Hospital is a public hospital with more than 1000 beds, caring for a community of over 1 million located in the eastern part of Singapore. The hospital has a Department of Psychological Medicine which currently has service provision for general adult psychiatry, geriatric psychiatry, liaison, substance misuse and psychotherapy services, with outpatient services being the predominant mode of service delivery. Outpatient clinic sessions are conducted by doctors in general adult psychiatry and geriatric psychiatry service, with both service categories being involved in other sub-speciality clinics.
In October 2015, a preliminary baseline audit on side-effects communications of newly prescribed psychotropic medications was undertaken on a sample of 18 new patients, assessed in outpatient clinics run by general adult consultant psychiatrists and trainees in Changi General Hospital, Singapore. The results revealed that only 22% (4/18) of patients had recorded evidence in electronic clinical documents of side-effects communication. This study excluded patients seen by psychiatrists from the geriatric psychiatry service and was therefore not representative of the entire medical workforce from the Department of Psychological Medicine.
As part of the departmental strive to achieve excellence in clinical care, a consultation exercise with departmental medical and interprofessional colleagues was conducted in May 2017 regarding the conduct of a departmental quality-improvement project (QIP), and a consensus was achieved to revive the earlier work undertaken in October 2015. In October 2017, a QIP multidisciplinary task force embarked on the revival of the previous study through a more robust baseline measurement in a representative sample which included new patients assessed by all departmental doctors. The task force set out to address the potential problem area of poor documentation. It aimed to improve documentation rates of communication of one or more side effects to 100% over a 12-month period in new cases assessed by all departmental doctors.
Background
At the very essence of good medical care is relevant and appropriate communication which is essential to enhance care delivery through a range of interventions which may involve medications. Communication with patients regarding new medications prescribed, rooted in maximising patient participation and involvement, forms the cornerstone of an enhanced patient–doctor relationship. An observational study in the USA which combined patient and physician surveys and audiotaped office visits from 185 outpatient encounters involving family physicians, internists and cardiologists assessed the quality of physician communication with patients on newly prescribed medications. The study revealed that side effects were addressed in only 35% of medications, 4 and physicians often failed to communicate the key elements involved in usage of newly prescribed medications. 4 In this study, education on psychiatric and analgesic medications was better than others, but the quality of communication was poor even for these medications. 4 Other published literature has shown that physicians discuss adverse effects less than one third of the time.5,6 These studies have highlighted that the quality and content of patient–doctor communication is crucial. Indisputably, adherence can be enhanced by involving patients in medication preference and attitude towards potential side effects. 7
In psychiatry, prescribed medications are not devoid of harm, with antipsychotic medications being associated with adverse effects such as extrapyramidal side effects, increased cardiovascular risks and metabolic side effects.
8
Sodium valproate, commonly used for its mood-stabilising properties, is associated with major congenital malformations (in particularly neural tube defects) and a serious form of rash manifesting as Stevens–Johnson syndrome.
8
Lithium, another mood stabiliser used in psychiatry, is associated with renal impairment.
8
The landmark case law in the UK, the Montgomery Test, is a stark reminder of doctors’ duties and obligations to discuss material risks.9–11 Another landmark case in Singapore,
In light of the above, we undertook an audit using quality-improvement methodology to examine and improve the documentation rates by departmental doctors of their communications of side effects of newly prescribed psychotropic medications.
Methods
Baseline measurement
The data collection for retrospective baseline measurement took place in October 2017. The time frame was chosen to coincide with the new batch of trainees joining the department, all of whom were invited to participate in this project, with two out of three trainees accepting the invitation. The key standard for our baseline measurement was whether the communication of at least one side effect to a newly prescribed medication was recorded by the respective doctors in electronic clinical records. If electronic entries stated that side effects were ‘discussed’, these entries were not considered as objective evidence of discussions. Our criterion for appropriate documentation was clear descriptions of specific side effect(s) discussed, and the minimum number of side effects that a doctor should discuss was set at one. The supporting clinical audit definitions were set as follows:
Audit criteria
Psychiatric doctor completed documentation of at least one or more side effects to newly prescribed psychotropic medications for new cases assessed in psychiatric outpatient clinics.
Audit indicator
Numerator: Number of completed documentation of at least one side effect that was documented for newly prescribed psychotropic medications for new cases assessed in psychiatric outpatient clinics. Denominator: Number of newly prescribed psychotropic medications for new cases assessed in psychiatric outpatient clinics.
Audit standard
The standard was set at 100% documentation of at least one or more side effects to newly prescribed psychotropic medications of new cases assessed in psychiatric outpatient clinics.
Data integrity was ensured at the outset with two medical officers and the first author (QIP lead consultant) undertaking a pre-data collection trial of data recording on a sample of five patients. Following this trial, data collection proceeded. Baseline data were retrospectively obtained from outpatient electronic records over three months from March 2017 to June 2017 on newly prescribed psychotropic medication at either initial assessment or subsequent reviews of new cases. Out of 728 new cases who were started on psychotropic medications during this period, 303 were extracted, which amounted to a representative sampling of 42% of all new prescriptions. A random-sampling method for data extraction was used, using the random-number function in Microsoft Excel, and the study QIP specialist stratified the extracted data based on various classes of psychotropic medications which included antidepressants, antipsychotics, benzodiazepines, cognitive-enhancing medications, mood stabilisers and hypnotics. Baseline data extracted by three data collectors were rechecked by the first author to ensure data accuracy and integrity.
Baseline measurements revealed that among general psychiatrists, overall documentation rates were 40% (86/217), with 42% (86/205) for specialists and 0% (0/12) for non-specialists. Among geriatric psychiatrists, overall documentations rates were 40% (34/86), with 40% (28/70) among specialists and 38% (6/16) among non-specialists.
For subsequent audit cycles, the quality-improvement task force agreed on prospectively examining new psychotropic medications prescribed during initial outpatient assessments of new cases by psychiatrists over two weeks following the implementation of a change intervention. The time frame and inclusion of new prescriptions generated at initial assessment were identified as appropriate aim measures to make the data collection manageable and achievable.
Design
Following baseline measurement results, the quality-improvement task force brainstormed on the possible factors leading to low documentation rates, which included physician-, patient-, system- and process-related factors.
The key standard and outcome measurement for post-intervention audit cycles were the same as for the baseline measurement. The data were collected by the first author and two psychiatry trainees for the baseline measurement and the first two audit cycles. The third audit cycle and fourth data collection were done by the first author and study pharmacist. One of the two psychiatry trainees who was re-posted into the department assisted again with the fourth data collection. Any clarifications on data collection or study design were addressed through discussions with the remainder of the task force which involved two other consultant psychiatrists, one of whom was the departmental chief, and a quality-improvement specialist.
All new patients assessed and prescribed a new psychotropic medication by doctors from the department were eligible for study inclusion. Electronic records were accessed by the data collectors and Microsoft Excel spreadsheets were used to facilitate data extraction. The results of documentation rates were categorised into general adult and geriatric psychiatry subgroups of doctors.
Ethical approval was not applicable, as the study was a quality-improvement initiative and falls within the exemption category.
The quality-improvement task force deliberated that active engagement of departmental colleagues was crucial to achieve the study aim. Engagement from the outset and at every stage was deemed necessary to refine change interventions through early identification of its strengths and limitations.
Strategy
Audit cycle 1 using PDSA methodology
We presented the results of the baseline measurement to departmental colleagues in our weekly continuing professional development sessions in early November 2017, in which possible factors leading to poor documentation, the driver diagram and possible change interventions were explored. Following discussions, the key drivers of our project were condensed into a driver diagram. Figure 1 illustrates the driver diagram, which summarises the primary and secondary drivers contributing to the achievement of the project aim. Raising awareness on medico-legal risks posed to doctors by the hospital solicitor was agreed as the first intervention following departmental discussions. A medico-legal educational awareness session on implications of poor medical documentation was implemented towards the end of November 2017, with the target audience being departmental doctors. Data on documentation rates were prospectively collected over a two-week period (8–21 January 2018). The overall time period for the first audit improvement cycle was about two months. We predicted that raising awareness on medico-legal risks would be a driver for improved documentation rates, and the positive verbal feedback generated after this session supported our prediction that the intervention was likely to be impactful. For general psychiatrists, documentation rates improved from 42% to 52% (
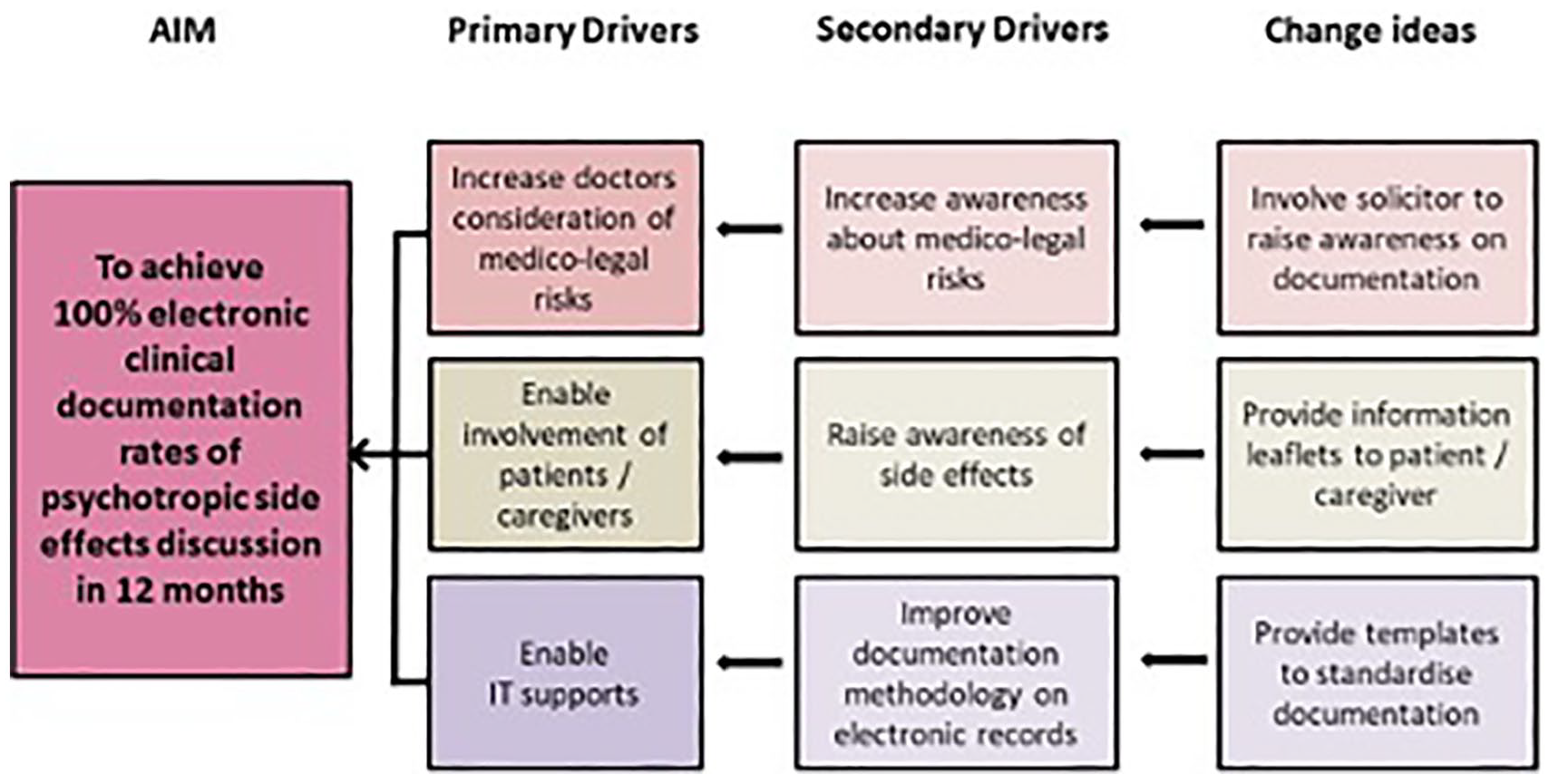
Driver diagram.
Audit cycle 2 using PDSA methodology
Results from the first change intervention were discussed at the departmental continuing medical education session in March 2018. The driver diagram and further change interventions were discussed, and the second intervention – namely the availability of patient information leaflets – was agreed on and implemented towards the end of the month. These patient information leaflets on psychotropic medications were developed to facilitate informed discussions and documentation of these discussions. The leaflets included information on indications, benefits and side effects. Data on side-effects documentation rates were prospectively collected over two weeks (2–15 April 2018). The period for the second audit improvement cycle was about two months. The quality-improvement task force predicted that brochures would serve as reminder aids, thereby enhancing documentation rates. In comparison to the first intervention, documentation rates improved from 52% to 62% (
Audit cycle 3 using PDSA methodology
Following results from the second intervention, we discussed and brainstormed at a department meeting in June 2018 on the third change intervention. It was agreed to incorporate an electronic recording aid, designated especially for side-effects communications into the clinical documentation template. This recording aid served as a reminder for doctors and augmented the process of documentation. There was a four-month time frame for the electronic aid to be implemented, and it was made available in November 2018. Data on documentation rates were prospectively collected over two weeks (3–16 December 2018). The time frame for this audit cycle was longer than the previous two interventions at five months. In comparison to the second intervention, documentation rates improved from 62% to 92% (

Results of three audit cycles using PDSA methodology.
Fourth measurement
Following the results from the third intervention, the team conducted another round of data collection over two weeks in April 2019 (1–14 April 2019). Results of the fourth round of data collection revealed that among general psychiatrists, documentations rates were 93% (
Results
Overall, there has been a substantial departmental improvement from a baseline of 40% to 91%.
The first intervention was aimed at addressing poor awareness among doctors on the significance of robust medical documentations through a medico-legal seminar. Documentation rates improved to 53% (
The second intervention made patient information leaflets available to facilitate informed discussions and promote documentation of such information discussed. Documentation rates improved to 63% (
The third intervention involved incorporating an electronic recording aid into the clinical document template. These served as a reminder for doctors to document during their clinic sessions and aimed to augment the process of documentation. Documentation rates improved to 87% (
The fourth measurement results showed improved documentation rates for general psychiatrists at 90% (
Lessons and limitations
The study was an inter-professional initiative with doctors, pharmacists and a QI specialist, thus adding to the strength of the study design. Inclusion of departmental colleagues as key stakeholders at every stage ensured that the project aim, methodology and change interventions were fit for purpose. There were thus opportunities for collaborative learning and continual professional development which gave further impetus for this project.
We minimised errors in data extraction with pre-data trial testing with trainees at the outset to ensure data integrity. We were cognisant of the trainee turnover during the study and built in measures with choice of data collectors who would continue the project following trainee placement departure. Re-involvement of one of the two trainees (previously involved in baseline measurement and two study cycles) in the fourth data collection with a refresher session with the first author and study pharmacist on data collection and study design further ensured data integrity. This is another strength of our study. The baseline data from a representative sample and data extracted using random-sampling methodology added to the study strengths.
The team chose to audit new cases but not follow-up cases. Auditing new cases was a change driver, enabling departmental colleagues to instigate changes in practice from their initial assessment in outpatient clinics and follow it through patients’ journeys. This was also for pragmatic reasons, as it was more feasible to capture the new cases where psychotropic medications were initiated over that period.
Co-ownership and engagement enhanced buy-in of departmental colleagues and may have minimised resistance to change. Continuous and consistent feedback may have impacted on the change process.
The third intervention was the documentation tool set up on an IT platform which automatically provides an entry field on new case consultation notes for patients assessed in psychiatric outpatient clinics. This high-impact intervention prompted all doctors to document side effects, and it enhanced the workflow by invoking the entry field for new consultation notes. In terms of our shared learning, the IT document tool was the most effective intervention, which was successfully adopted and implemented into the standard documentation template for new case assessments. Also, through the recursive process of the documentation of side effects, that is, using the entry field as a prompt to document side effects, practice changes were likely to be enshrined in everyday patient care. The use of automation 12 through the IT platform also eliminates the potential cost, which is usually associated with a paper documentation approach. Also, it increases ease of accessibility and consistency 12 for the psychiatric doctor to perform the necessary documentation.
We wish to acknowledge the following limitations. The key standard for documentation included the recording of at least one side effect of newly initiated psychotropic medications. This may be regarded as too lenient or lax, and there should be an agreement on what is crucial when documenting common versus serious side effects. We had gathered the department’s consensus in establishing at least one side effect for the purpose of this QIP. Having departmental doctors discuss and record at least one side effect ensures that communication of side effects has occurred during the clinic consultation. In this project, we measured the salience of communicating and documenting side effects. It is noteworthy that merely noting that side effects were explained in electronic medical records during the psychiatric consultation was not regarded as good enough, and through this QIP endeavour, we ensured explicit recording of side effects. It was, however, beyond the scope of this project to examine the specific side effects that were documented.
Despite our enthusiasm and efforts, progress made was relatively gradual and slow, with significant improvements observed only after the third intervention. While we are unable to explain the tardy progress made entirely, it is noteworthy that improvements after the third intervention were sustained three months later in the general psychiatry group, demonstrating early signs of sustainability of tested interventions.
With regards to the geriatric psychiatry group, progress was also gradual and slow. Suboptimal recording of side effects communications by one psychiatrist may have skewed the results of the third intervention. Therefore, we wish to acknowledge that the small sample size of the two-week data collection may have introduced some bias into the results post intervention.
We were unable to achieve improvement to 100% but are close to achieving our target. The approximate time frame for this project was across 18 months, which was six months longer than the original target of 12 months. The medico-legal seminar and the development of the educational brochures took time to plan, execute and implement. This may have contributed to some of the delays to the project time frame and gaps between the audit cycles. There were delays in implementing the third intervention, which involved incorporating electronic aids, contributing to the overall time-lag factor. The lag has been offset by the success of this intervention.
The working group planned for the sustainability of the interventions. The IT intervention was a critical factor which has aided sustainability. Department consensus to use documentation of side effects as a clinical quality indicator in May 2019 also helped to ensure ongoing sustainability of the interventions. Further time points for data collection to assess long-term sustainability of improvements would undoubtedly be useful. The working group has reflected on the methodology, experience and learning. Embedding documentation of side effects as an entry field in new case consultation notes was unwittingly the most effective intervention supporting the process of change.
There were gaps and delays in the audit cycles through the time needed for planning and implementation of the interventions. However, these delays may have been helpful for departmental doctors, ensuring adaptability and self-reflection, which are necessary to implement practice changes.
We have also reflected on other factors which may have contributed to the success of this project. The department functioning as ‘a whole unit’ to enact changes in a practice centred around improvement in patient care. The leadership exercised by the working group and the head of the department are additional insights that have transcended into practice changes. We aspire to share these reflections and learning through this publication. Despite some of the limitations highlighted above, the project was an overall success, with improvements in documentation rates rising to 91%. Through change interventions, systemic factors rooted in patient safety have been embodied in everyday clinical practice.
Conclusion
This project is especially relevant, given the current health-care landscape in Singapore. Medical negligence case law serves as a timely reminder to doctors and health-care providers of the salience of robust documentation practice. This audit using quality-improvement methodology has shown that documentation of the communication of side effects to prescribed medications in the Department of Psychological Medicine has improved to 91% from a baseline measurement of 40% through change interventions. This project serves as a crucial reminder that robust documentation of patient communications is at the very core of good clinical practice. Shared decision making involving patients in treatment decisions and embedding robust documentation into practice, in addition to enhancing care, confers added benefits to both individual health-care providers and their employers through the potential diminution of medico-legal risks. Systemic and organisational approaches to patient safety have been addressed through this audit and quality-improvement endeavour.
Although our target of 100% documentation rates was not achieved by the end of the project, the quality-improvement task force managed to increase documentation rates substantially from 40% to 91%. Enabling IT support to enhance documentation was the most effective and high-impact intervention, which has been incorporated into our standard documentation template for new case assessments in psychiatric outpatient clinics.
Footnotes
Acknowledgements
None.
Authors’ contributions
B.B. was the lead author involved in the study design, data collection, writing the first draft and subsequent revisions. P.J., S.A. and B.Y.Z. were involved in the study design, data collection and contributed to the write-up and revisions. T.L.L. and P.L.H. were involved in the study design and contributed to the write-up and revisions. A.W.B. was involved in the study design and data analysis, and contributed to the write-up. All authors reviewed and edited the manuscript and approved the final version.
Availability of data and materials
The data set is available from the corresponding author.
Ethical approval
A formal application for ethical approval is not applicable as this study was a quality-improvement project. SingHealth Centralised Institutional Review Board, Singapore advised exemption from a formal application for ethical approval as this study was an audit with a focus on quality improvement.
Informed consent
Informed consent is not applicable in this audit and quality-improvement study. Anonymised patient data were used in line with the quality-improvement initiative of this project and therefore informed consent was not applicable.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
