Abstract
Differentiation of thyroid follicular lesions from parathyroid lesions on fine needle aspiration cytology can be very challenging, both being endocrine lesions with overlap in cytomorphologic features. In some cases, it may be impossible to differentiate thyroid neoplasms from parathyroid neoplasms based on cytologic grounds alone. Clinical information, including laboratory and radiologic findings, is crucial in these difficult cases. Immunocytochemistry is a useful diagnostic tool in differentiating thyroid from parathyroid neoplasms. In this case report, we detail the challenges faced in differentiating a parathyroid lesion from a thyroid follicular lesion based on cytomorphology, and how clinical information and immunocytochemistry were applied towards making a correct diagnosis.
Introduction
Differentiation of thyroid follicular lesions from parathyroid lesions on fine needle aspiration cytology can be very challenging, both being endocrine lesions with significant overlap in cytomorphologic features.1,2 In some instances, parathyroid neoplasms may also masquerade as thyroid neoplasms on ultrasonography, due to the close proximity of these two organs and the incidence of intrathyroidal parathyroid lesions. Clinical information, including laboratory and additional radiologic tests, is extremely helpful in resolving these cases. Parathyroid hormone (PTH) assay on aspirated material and immunocytochemistry are useful adjuncts in differentiating thyroid from parathyroid neoplasms. In this case report, we detail the challenges faced in differentiating a parathyroid lesion from a thyroid follicular lesion based on cytologic grounds, and how clinical information and immunocytochemistry were applied towards making a correct diagnosis.
Case report
A 67-year-old man was investigated for a right neck lump, which was localized to the thyroid. Ultrasound-guided fine needle aspiration cytology showed a cellular aspirate with microfollicular architecture, oncocytic changes and minimal colloid, consistent with a thyroid follicular neoplasm (Figure 1). Routine pre-operative work-up showed a raised serum calcium level, resulting in a series of further laboratory investigations which showed a concurrent increase in serum parathyroid hormone level, indicating primary hyperparathyroidism. Serum free triiodothyronine (FT3), free thyroxine (FT4) and thyroid-stimulating hormone (TSH) levels were normal. In order to locate the hyperfunctioning parathyroid gland, a parathyroid scan was performed with technetium 99m-labeled sestamibi, followed by pertechnetate subtraction at 90min, which revealed no sestamibi-avid parathyroid lesion. In view of this negative finding, four-dimensional computed tomography (4D-CT) of the neck was performed, which revealed a nodule on the right side of the neck that appeared separate from the right thyroid gland itself. As a result, the patient was reassessed, the cytologic smears reviewed and adjunct immunocytochemistry performed on a destained Papanicolaou (Pap) slide. Positive PTH immunocytochemistry was in keeping with a cytological diagnosis of a parathyroid lesion (Figure 2). Resection of the lesion was performed subsequently, and histology confirmed a parathyroid adenoma.
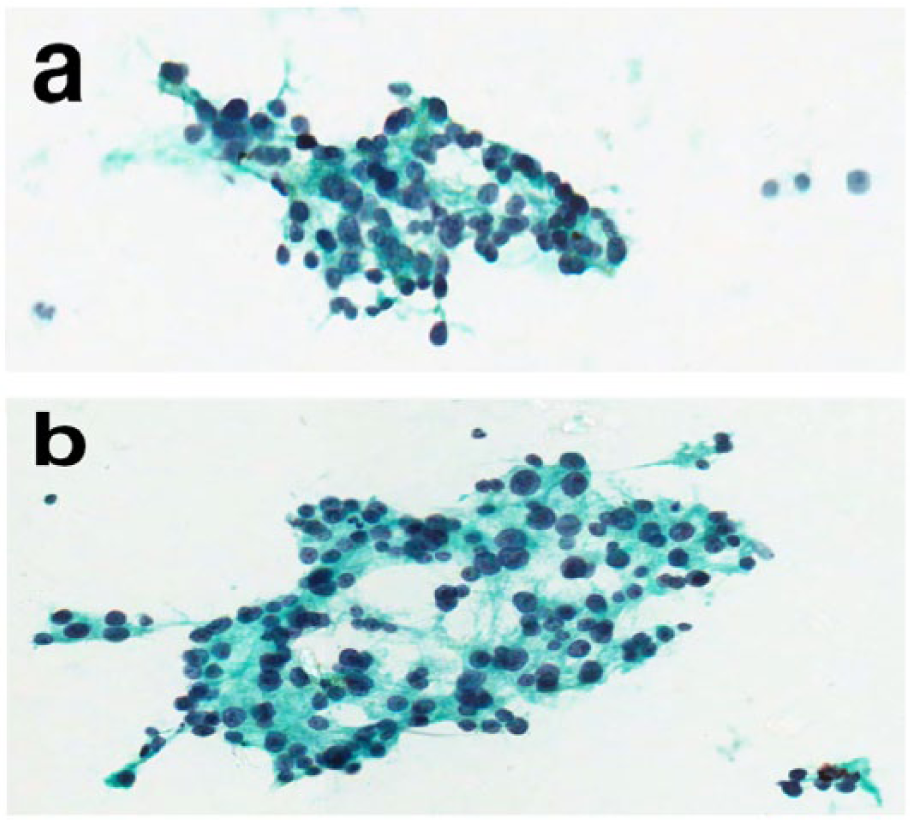
Syncytial groups (a) and microfollicular clusters (b) of cells from the parathyroid lesion, displaying variation in nuclear size and distinct nucleoli with accompanying ample lacy-to-granular cytoplasm, resulting in mimicry of and misinterpretation as a thyroid oncoytic follicular neoplasm (Papanicolaou stain).

Parathyroid hormone immunocytochemistry performed directly on the destained Papanicolaou (Pap) slide, demonstrating positive cytoplasmic granularity within the same cluster as in Figure 1(a), confirming its parathyroid origin. (Parathyroid hormone immunocytochemistry).
Discussion
Parathyroid adenomas are a major cause of primary hyperparathyroidism. 3 The clinical diagnosis of primary hyperparathyroidism in this patient initiated a series of investigations to locate the hyperfunctioning parathyroid gland. While the technetium 99m-labeled sestamibi scan gives a high yield in detecting hyperfunctioning parathyroid adenomas, 4 in this case it was non-revealing, necessitating assessment with 4D-CT. The 4D-CT finding of a nodule that appeared separate from the thyroid gland clued us into the possibility that the aspirated lesion may represent a parathyroid adenoma. PTH immunocytochemistry was performed as the cytomorphology showed no obvious discerning feature that enabled us to differentiate between a thyroid follicular lesion and a parathyroid lesion.
Cytologically, parathyroid adenomas are frequently mistaken for a follicular lesion of the thyroid. 1 Common morphologic features that have been described with regards to parathyroid cells on cytology are rather non-specific, including loosely clustered cells in syncytial or microfollicular architecture, absence of colloid, presence of single cells and naked nuclei in the background, round nuclei with coarsely granular chromatin and moderately abundant granular cytoplasm. 5 These features could also be seen in a thyroid follicular neoplasm, and hence are non-discriminatory in most cases without corroborative clinical information. Parathyroid cells are usually smaller in comparison with cells of thyroid follicular lesions. 6 Variation in nuclear size and pleomorphism, nuclear overlapping and molding have also been described in parathyroid lesions. 7
Unfortunately, there is no single cytologic feature that can distinguish parathyroid cells from thyroid follicular cells. 2 The mimicry is further heightened in lesions with oncocytic features; an oncocytic (oxyphil-cell) parathyroid adenoma may easily be mistaken for a Hürthle cell follicular neoplasm of the thyroid.
In retrospect, the cytological smears our case showed a combination of the cytomorphologic features that have been described in a parathyroid lesion. There was a predominantly syncytial and microfollicular arrangement of cells, mild to moderate variation in nuclear size and minimal colloid, as well as increased numbers of isolated cells and naked nuclei in the background. The nuclear size in general was smaller than would be expected for a thyroid follicular neoplasm, although the presence of variation in nuclear size was a confounding factor. On examination under high magnification, the chromatin had a more stippled appearance and denser texture as compared with oncocytic thyroid follicular cells. However, the diagnosis of a parathyroid lesion could be made with confidence only in the presence of a positive staining result on PTH immunocytochemistry and corroborative clinico-radiological findings.
In situations where a lesion is suspected to be of parathyroid origin, aspirated material can be sent for PTH immunoassay; a high concentration strongly supports the diagnosis of a parathyroid tumor. However, if no material is available for PTH assay, as in our case, PTH immunocytochemistry may be performed on a destained Pap slide. The presence of cytoplasmic granular staining is confirmatory of parathyroid origin.8,9
Conclusion
Thyroid follicular neoplasms are much more commonly encountered than parathyroid adenomas. However, it is important to be aware that parathyroid lesions can occasionally masquerade as thyroid follicular lesions, clinically as well as cytologically. Correlation with clinical and laboratory findings, in conjunction with the use of immunocytochemistry, is essential to avoid misdiagnosis.10,11
Footnotes
Declaration of conflicting interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
