Abstract
An uncommon cause of primary hyperparathyroidism is a cystic parathyroid adenoma. This paper describes two patients with hypercalcemia and right knee disease. Their serum calcium concentration was high, phosphorus concentration was low, and parathyroid hormone (PTH) concentration was high. Ultrasound and computed tomography scans of the neck indicated a cystic mass near the thyroid. Parathyroid scintigraphy showed no focal uptake in one patient and low tracer concentration in the cystic mass in the other patient. Following resection of the cystic masses, both were pathologically confirmed to be a cystic parathyroid adenoma with predominantly cystic degeneration. The calcium and PTH concentrations gradually decreased to the reference range. Both patients were stable at their last follow-up. The diagnosis of a functional cystic parathyroid adenoma is highly challenging because of the different clinical manifestations and negative result on parathyroid tracer scintigraphy. For patients with high serum calcium and PTH concentrations and a cystic mass in the neck, resection of the mass and subsequent postoperative pathological diagnosis is necessary even if the clinical diagnosis of a parathyroid adenoma cannot be confirmed preoperatively. Decreases in the PTH and serum calcium concentrations indicate successful resection of a functional parathyroid adenoma.
Keywords
Introduction
Primary hyperparathyroidism is an uncommon disease with a reported incidence of approximately 21 cases per 100,000 persons per year. 1 A solitary parathyroid adenoma accounts for approximately 85% of these cases. 2 Rarely (1%–2% of cases), primary hyperparathyroidism may be caused by cystic degeneration of a parathyroid adenoma. 3 Such cases present a diagnostic challenge for clinicians. For imaging doctors who are not familiar with cystic degeneration of a parathyroid adenoma (especially a complete cystic adenoma), the cystic characteristics are a confounding factor for accurate diagnosis. In fact, a parathyroid cyst is not a specific histopathological diagnosis but only a rare feature of a parathyroid adenoma.
We herein present two cases of primary hyperparathyroidism caused by cystic parathyroid adenomas that were atypical because of their large size, cystic character, and negligible or low tracer concentration when compared with the typical solid parathyroid adenomas on sestamibi scintigraphy. These findings illustrate the diagnostic difficulties related to the unusual clinical presentations in such cases. In addition, we review the available literature on this clinical condition.
Case Report
The present study was carried out in accordance with the Helsinki Declaration and was approved by the Institutional Review Board of the First Affiliated Hospital, Zhejiang University School of Medicine (reference no. 2020-670). Because of the retrospective observational nature of the study, the requirement for informed consent was waived. All patient details have been de-identified. Written informed consent for treatment was obtained from each patient. The reporting of this study conforms to the CARE guidelines. 4
Case 1
A man in his early 40s was admitted to our hospital with a 7-day history of hypercalcemia in November 2012. Seven days before admission, the patient had fallen off a motorcycle and sustained a right knee injury and patellar fracture. He was then admitted to a local hospital and found to have hypercalcemia prior to the knee operation. His serum calcium concentration was not recorded in detail. The patient’s clinical condition progressed rapidly, and he developed symptoms of obvious thirst, polyuria, decreased appetite, severe nausea, vomiting, and constipation. He had no history of diabetes or hypertension. There were no unusual aspects of his medical history, family health history, or history of marriage and childbirth. He was awake, alert, and oriented. Physical examination findings were normal. Blood tests revealed hypercalcemia (4.1 mmol/L; reference range [N], 2.03–2.54 mmol/L), an elevated parathyroid hormone (PTH) concentration (593.5 pg/mL; N, 15–65 pg/mL), a low phosphorus concentration (0.82 mmol/L; N, 0.85–1.51 mmol/L), and a normal 25-hydroxyvitamin D concentration. His albumin concentration was 45 g/L (N, 40–55 g/L), and his corrected serum calcium concentration was 4.0 mmol/L (N, 2.1–2.6 mmol/L). His 24-hour urinary calcium was 10.45 mmol (N, 2.5–7.5). He had a high blood urea nitrogen concentration of 9.8 mmol/L (N, 2.9–8.2 mmol/L) and a high serum creatinine concentration of 120 µmol/L (N, 59–104 µmol/L). His urine calcium-to-creatinine ratio was 0.023, calculated as follows: [urine calcium (mmol/L) × serum creatinine (µmol/L)/[adjusted serum calcium (mmol/L) × urine creatinine (µmol/L)].
Neck ultrasound examination revealed a cystic mass closely related to the right superior pole of the thyroid gland. The mass was about 3.4 × 2.7 × 3.7 cm in size and showed a clear boundary with separations inside of the cyst (Figure 1(a)). Neck computed tomography (CT) showed a cystic-solid mass of about 4.1 × 3.6 cm above the right thyroid gland. The solid part of the mass had a plain CT value of about 45 HU, which was significantly higher after enhancement. The boundary between the mass and the superior pole of the thyroid was not clear, while that between the mass and the posterior blood vessels and sternocleidomastoid muscle was evident (Figure 1(b)). Double-phase parathyroid scintigraphy with technetium (Tc)-99m-sestamibi single-photon emission CT (SPECT)-CT was performed after intravenous injection of 25 mCi Tc-99m-sestamibi. Static imaging of the anterior cervical region at 15 minutes, 1.5 hours, and 2.5 hours revealed the following: in the early phase, the imaging of the bilobed thyroid was clear, the tracer distribution was basically uniform, and no obvious concentration or sparse defect areas were observed; in the delayed phase, the thyroid imaging subsided, and no abnormal tracer concentration was found in the anterior cervical region. SPECT imaging of the parathyroid gland indicated no typical parathyroid adenoma (Figure 1(c)).
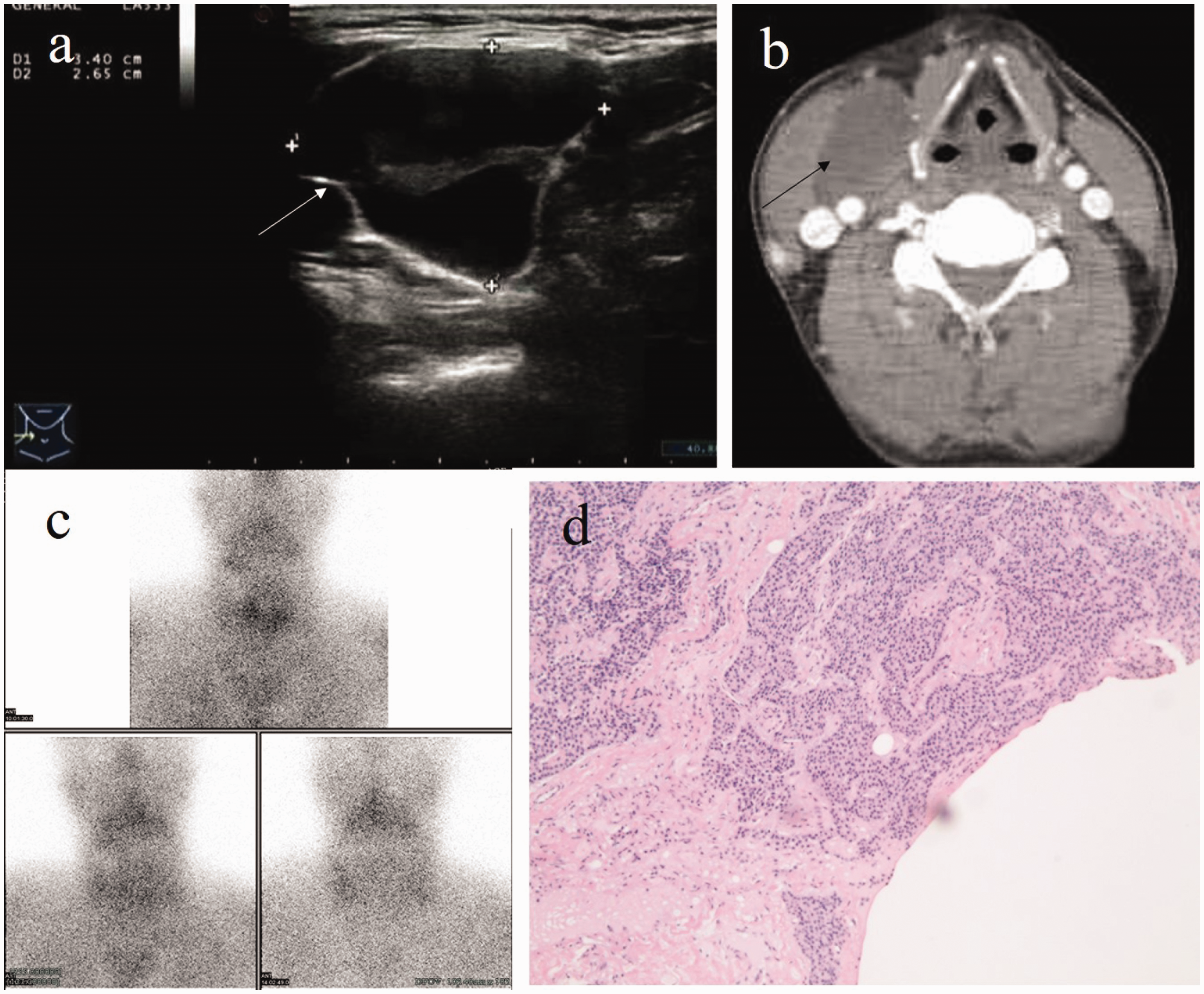
Imaging findings in Case 1. (a) Neck ultrasound indicates a cystic mass next to the right superior pole of thyroid gland. (b) Neck computed tomography shows a cystic-solid mass above the right thyroid gland. (c) SPECT imaging of the parathyroid gland indicates no typical parathyroid adenoma. The arrows denote the cystic parathyroid adenoma and (d) The histopathologic diagnosis is a right parathyroid adenoma with central cystic change (×10).
The patient’s clinical symptoms along with his elevated serum calcium, creatinine, and urea nitrogen concentrations suggested that he was in parathyroid crisis. He was initially given intravenous infusion of saline fluids and intramuscular calcitonin. Ultrasound-guided fine needle aspiration was then performed, and 25 mL of bloody fluid was drained. Two pieces of parenchymal tissue were cytologically examined, revealing a small piece of parathyroid tissue with adenoma cells containing round hyperchromatic nuclei. No atypical cells were seen, and the PTH concentration in the cystic fluid was not measured. The patient then underwent resection of the right parathyroid adenoma and right partial thyroidectomy. The intraoperative findings showed a soft yellow mass measuring 5.0 × 3.0 × 3.0 cm in the upper pole of the right thyroid gland. Its surface was smooth, and it contained dark red cystic fluid. The base was closely adhered to the carotid artery, and the top was near the submandibular gland. A thin capsule was seen on gross observation. The histopathologic diagnosis was a right parathyroid adenoma with hemorrhagic cystic change (Figure 1(d)) and right nodular goiter with a small amount of parathyroid tissue scattered inside. The serum PTH concentration was 440 pg/mL before the mass resection and 119 pg/mL after the mass resection. Multiple serum calcium measurements during the operation revealed that the free calcium fluctuated by approximately 2.0 mmol/L. After surgery, the calcium concentration gradually decreased; at 4 days after the mass resection, the calcium concentration was 2.23 mmol/L, the phosphorus concentration was 1.1 mmol/L, and the PTH concentration was 28.3 pg/mL.
Case 2
A woman in her early 60s was admitted to our hospital with a 1-month history of hypercalcemia in March 2017. One month earlier, she had been found to have hypercalcemia with a significantly increased PTH concentration during right knee arthroplasty. She presented no other symptoms. Her medical history and family health history were not unusual. At the time of admission, she was in good mental condition and had normal physical examination findings. Her serum calcium concentration was 3.12 mmol/L (N, 2.03–2.54 mmol/L), phosphorus concentration was 0.65 mmol/L (N, 0.85–1.51 mmol/L), albumin concentration was 39.8 g/L (N, 40–55 g/L), and corrected calcium concentration was 3.13 mmol/L (N, 2.1–2.6 mmol/L). All other electrolytes and 25-hydroxyvitamin D were within the reference range. Her PTH concentration was 413.0 pg/mL (N, 15–65 pg/mL), and her 24-hour urinary calcium was 6.7 mmol (N, 2.5–7.5 mmol). Her urine calcium-to-creatinine ratio was 0.013.
Neck ultrasound revealed several nodules in the bilateral thyroid. The larger nodule on the left side was cystic and solid, about 4.3 × 3.2 × 6.3 cm, and mainly cystic with a clear boundary. Color Doppler flow imaging showed no abnormal blood supply (Figure 2(a)). Nodular goiter was considered, and no obvious abnormality was found in the bilateral parathyroid area by ultrasound. Neck CT showed a huge cystic-solid mass of about 4.3 × 3.2 cm in the posterolateral left lobe of the thyroid gland; the plain CT value was about 36 HU, and the enhancement was obvious with separation. It was necessary to differentiate between a parathyroid adenoma and a thyroid nodule (Figure 2(b)). SPECT-CT imaging of the parathyroid gland in the delayed phase (2 hours) showed mild tracer concentration in the middle and upper polar regions of the left lobe of the thyroid gland, and the possibility of uptake by a parathyroid adenoma or thyroid nodule was considered (Figure 2(c)).

Imaging findings in Case 2. (a) Neck ultrasound indicates a huge cystic-solid mass in the posterolateral left lobe of the thyroid gland. (b) Neck computed tomography shows a giant cystic solid mass in the left thyroid gland. The arrows denote the cystic parathyroid adenoma. (c) SPECT imaging indicates mild tracer concentration in the middle and upper polar regions of the left lobe of the thyroid gland and (d) The pathologic examination suggests a left parathyroid adenoma (×10).
The patient was initially clinically managed with intravenous fluids and intramuscular calcitonin. She then underwent resection of the left parathyroid adenoma and left thyroidectomy. The pathologic examination indicated left thyroid nodular goiter and left cystic parathyroid adenoma (Figure 2(d)). The bean-shaped mass was mixed cystic and solid, but mainly cystic. The tumor cells were of the same size and arranged in a solid sheet and trabecular shape, and they contained round and deeply stained nuclei, occasional small nucleoli, and vacuolar or weakly eosinophilic cytoplasm. Two days after the operation, the patient’s calcium concentration was 2.28 mmol/L, phosphorus concentration was 0.67 mmol/L, and PTH concentration was 15.7 pg/mL.
Discussion
Primary hyperparathyroidism is a rare disease with an annual incidence rate of about 21 cases per 100,000 persons. 1 A solid parathyroid adenoma is the etiology of primary hyperparathyroidism in 85% of cases, whereas rare etiologies include multiple adenomas and parathyroid hyperplasia. 2 A less common cause of this condition is a parathyroid carcinoma or cystic parathyroid adenoma. During physical examination, a smooth and flexible mass can be palpated in the anterior cervical area or lateral cervical area; the mass often moves with swallowing because it is usually tightly adhered to the thyroid gland. 5 Therefore, a cystic parathyroid adenoma can be misdiagnosed as a parathyroid cyst, 5 thyroid nodule, 6 or other neck mass.
Preoperative diagnosis of a cystic parathyroid adenoma is essential to avoid intraoperative cyst rupture and prevent subsequent parathyromatosis. 7 Cervical ultrasound is the first choice for screening and usually reveals several common thin-walled cystic ultrasound features, clear separation from the thyroid, and the presence of a polar feeding vessel. The accuracy of SPECT in the diagnosis of parathyroid cysts has been rarely reported. Some patients show tracer concentration. 8 Even in patients with a high PTH concentration in the cystic fluid, negative results are obtained in as many as 32% of patients. 3 This mainly depends on the number of parathyroid cells in the lining cystic wall, which is associated with expansion of the cyst and the diffusion of tracer uptake activity. Case 1 is an example of false-negative sestamibi scintigraphy, where the negative result was due to the decrease of tracer uptake caused by compression and dilution of the parathyroid tissue in the capsule wall. 9 When differentiating between a cystic parathyroid adenoma and a thyroid cyst, it is helpful to determine the PTH and thyroid hormone concentrations in the cyst fluid by fine needle aspiration of the cyst. The cystic fluid of thyroid cysts contains high thyroid hormone and thyroglobulin concentrations, 5 whereas the cystic fluid of cystic parathyroid adenomas contains a high PTH concentration. 10 Because the PTH content of parathyroid cyst fluid is high, the PTH concentration in the cyst fluid cannot be used to distinguish between a functional cystic parathyroid adenoma and a nonfunctional parathyroid cyst. In such cases, only measurement of the plasma PTH and calcium concentrations can reveal the difference.
The possible origins of cystic parathyroid adenomas include degeneration, hemorrhage, and infarction of a parathyroid gland, a parathyroid adenoma, or rarely a parathyroid carcinoma. The possible origins of parathyroid cysts include accumulation of parathyroid colloid vesicle secretion, coalescence of vesicular-canalicular or glandular-like rudiments (Kürsteiner canals) during development of the embryonic parathyroid gland, enlargement or fusion of normal parathyroid microcysts, 11 and remnants of the third and fourth branchial clefts. 12 Cysts originating from accumulation of secretions, fusion of microcysts, or remnants of glandular structures or branchial clefts have thin walls, clear cystic fluid, and flattened cuboid to low columnar epithelium. 13 However, cysts originating from degeneration, hemorrhage, and infarction are not lined with epithelium, and the cystic fluid is bloody or chocolate-colored 5 and mixed with varying degrees of inflammatory and xanthomatous tissue responses.
Inactivation of the hyperparathyroidism–jaw tumor syndrome gene, CDC73 (formerly known as HRPT2), was recently established as a genetic mechanism in the development of parathyroid tumors. Its encoded protein parafibromin has tumor-suppressor properties that play an important role in tumor development in the parathyroids, jaws, and kidneys. Inactivating HRPT2 mutations and therefore loss of parafibromin expression have been described in a few cases of parathyroid adenomas with cystic features. 14 A limitation of the present clinical study is that parafibromin immunohistochemistry was not performed to confirm this hypothesis.
Parathyroid crisis, also known in the literature as acute hyperparathyroidism, parathyroid storm, parathyrotoxicosis, hypercalcemic crisis, and calcium intoxication, is a life-threatening emergency characterized by severe hypercalcemia (>3.5 mmol/L) accompanied by signs and symptoms of multiorgan failure. 15 Although severe hypercalcemia is common in patients with malignant tumors, it also develops in patients who have primary hyperparathyroidism with an elevated PTH concentration. 15 The clinical manifestations of parathyroid crisis include gastrointestinal and neurological symptoms, renal failure, and cardiac rhythm abnormalities. Our patient in Case 1 had a high corrected serum calcium concentration of 4.0 mmol/L. He exhibited gastrointestinal features such as nausea, vomiting, and constipation. Given today’s advanced medical technology, primary hyperparathyroidism is usually diagnosed early in patients with asymptomatic and mild hypercalcemia; however, giant cystic adenomas such as those reported herein are rare.
Treatments for parathyroid cysts or cystic adenomas include aspiration, injection of sclerosing agents, and surgical resection. For a functioning cystic adenoma, surgery may be the best choice. 5 Perioperative plasma PTH monitoring is helpful to judge whether the operation was successful. A decreased plasma PTH concentration indicates that resection of the functional adenoma was successful; however, if the PTH concentration does not decrease, it is necessary to determine whether the remaining parathyroid glands are complicated by hyperplasia. 16 The calcium concentration should also be monitored after the operation because hypocalcemia may occur after resection of hyperfunctioning cysts. 3
The main limitation of this retrospective observational study is that the number of cases was small; therefore, the findings are not suitable for general recommendations in clinical practice. Additionally, the PTH, thyroid hormone, and thyroglobulin concentrations were not detected in the fluid aspirated from the cyst and therefore could not be used to assist with determination of the cyst type.
For patients with high serum calcium and PTH concentrations and a cystic mass in the neck, resection of the mass with subsequent postoperative pathological diagnosis is necessary even if the clinical diagnosis of a parathyroid adenoma cannot be confirmed preoperatively. Decreased PTH and serum calcium concentrations indicate successful resection of a functional parathyroid adenoma.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221106419 - Supplemental material for Giant cystic parathyroid adenoma: two case reports and a literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605221106419 for Giant cystic parathyroid adenoma: two case reports and a literature review by Junwei Weng, Weibin Zhou and Yunfei Feng in Journal of International Medical Research
Footnotes
Author contributions
J.W., W.Z., and Y.F. were involved in the acquisition of data and drafting of the manuscript. J.W. is the corresponding author and organized the study. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Data availability statement
All data generated or analyzed during this study are included in this published article. The original research data obtained during the current study are available from the corresponding author on reasonable request.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
