Abstract
Purpose
Secondary language areas, including the pre-supplementary motor area (pre-SMA), dorsolateral prefrontal cortex (DLPFC), and the visual word form area (VWFA) play important roles in speech, but have been under-evaluated in the realm of resting-state (rs)-fMRI. The purpose of this study is to determine the incidence that secondary language areas and contralateral language areas can be localized using seed-based correlation (SBC) rs-fMRI.
Methods
We retrospectively reviewed 40 rs-fMRIs for functional connectivity (FC) to secondary language areas in cases where FC to Broca’s or Wernicke’s area near tumor in the left hemisphere were successfully generated using SBC analysis. Logistical regression was used for statistical analysis.
Results
SBC rs-fMRI with a seed in the left Broca’s or Wernicke’s area ipsilateral to the tumor was performed in the 40 patients. 72.5% of cases showed FC to the left DLPFC, 67.5% to left pre-SMA, and 52.5% of cases had FC to right Broca’s area. In addition to other correlations, we found older patients have a lower incidence of FC to the right Wernicke’s area when seeded from both left Broca’s and left Wernicke’s area (p-value = .016, odds ratio = 0.94).
Conclusion
SBC rs-fMRI can detect left hemispheric secondary language areas as well as right hemispheric primary and secondary language areas. The left DLPFC showed the highest incidence of FC, followed by the left pre-SMA when seeded from both left Broca’s and Wernicke’s area. Logistics regression also showed in some instances, differences in the incidence of FC to language areas was dependent on age, seed location, and gender.
Keywords
Introduction
It is well documented that task-based fMRI (tb-fMRI) speech paradigms result in BOLD activation in primary language centers: Broca’s and Wernicke’s areas.1,2 However, there are also secondary language areas such as pre-supplementary motor area (pre-SMA), dorsolateral prefrontal cortex (DLPFC), and visual word form area (VWFA) that can be detected on tb-fMRI and resting-state fMRI (rs-fMRI).3–6 Localizing these eloquent secondary language areas is important for presurgical planning in efforts to preserve language functions of brain tumor patients.
The pre-SMA as defined by Penfield and Welch is a region located in the dorsomedial cortex of the superior frontal gyrus (Brodmann’s cortical area 6c) 3 and has been recognized to be a contributor to many language functions including word reading, letter-based fluency, and word-stem completion. 4 The DLPFC, another well-established secondary language area, is located in the middle frontal gyrus with a role in verbal working memory and expressive language functions such as semantics, grammar, and word generation. 5 The VWFA is located in the fusiform gyrus and is known for its role in reading and elaborating visual information. 6
While these three secondary language centers have all been individually documented in their importance to language using tb-fMRI and some rs-fMRI studies, to our knowledge, they have never been analyzed all together in a single, cohesive rs-fMRI study. In addition, the incidence of FC to the right primary and secondary language areas when seeding from left Broca’s and Wernicke’s area has not been studied.
While often considered a gold standard, tb-fMRI language mapping may be limited in patients who are young, neurologically impaired, or poor at performing the task paradigms due to tumor effects. Resting-state fMRI does not rely on patient performance. If a patient is unable to perform tb-fMRI or if there are limited tb-fMRI results, seed-based correlation (SBC) rs-fMRI has been shown to identify Broca’s and Wernicke’s area ipsilateral to tumor. 7
SBC is becoming more practical due to its algorithmic simplicity and straightforward interpretation. 8 SBC is becoming a more attractive approach for preoperative mapping due to its ease to interpret and reliable nature. 9 The purpose of this study is to determine the incidence of FC to secondary and contralateral language areas in patients with brain tumors when seeding left Broca’s and Wernicke’s areas using SBC rs-fMRI and to determine if there were any significant differences in incidence of FC based on seed location, age, gender, lobe in which tumor is located, pathology, and IDH status (if applicable).
Methods
We retrospectively reviewed 40 rs-fMRIs for detecting secondary and contralateral language areas in cases in which FC to left Broca’s or left Wernicke’s area ipsilateral to tumor was successfully generated using a seed-based correlation (SBC) analysis.
Approximately 95% of right-handed individuals and 70% of left-handed individuals are left hemispheric dominant for language. 10 Therefore, we decided to choose cases in which tumor, rs-fMRI seed placement, and successful FC were all in the left hemisphere. In our study, handedness was assessed by a neuropsychologist administering an Edinburgh Handedness Inventory for presurgical evaluation of each patient. Of the 40 patients in our study that met our inclusion criteria, 38 were right-handed, one was mixed-handed, and one was left-handed.
Subjects
After obtaining IRB approval from our institution with waiver of informed consent for this retrospective study on 8/20/2019, a retrospective review of 134 patients with brain tumors who underwent both task-based fMRI and resting-state fMRI for presurgical brain mapping between 9/1/2017 and 3/31/2019 was performed. Eighty-five patients had adequate tb-fMRI language mapping and had no need for further processing of rs-fMRI imaging. In the remaining 49 patients, rs-fMRI was processed due to no or limited tb-fMRI data. The predominant reason for no or limited tb-fMRI results was poor patient performance as determined by a neuropsychologist who was present during the fMRI study.
Among the 49 patients in which rs-fMRI was processed, in 5 cases we were unable to obtain FC to left Broca’s or Wernicke’s area. In two of these cases, the unsuccessful rs-fMRI was attributed to patient head motion during the rs-fMRI sequence acquisition. This was determined by reviewing the rs-fMRI motion graphs. In one patient, the FC obtained was in a nonspecific anatomic location outside left Wernicke’s area. Finally, in 2 cases, rs-fMRI failure was due to an unknown system instability. In the remaining 4 cases, FC to left primary language area of interest was successful, but only after the seed was placed in a contralateral right primary language area after initial seed placement in a left primary language area apart from tumor failed. Based on our inclusion criteria, all 9 cases were excluded from the study. The clinical and rs-fMRI post-processed data for the remaining 40 patients were then collected for this retrospective study. The age, sex, tumor location, pathology, and IDH status were documented for each patient.
Technique
MRI acquisition
fMRI and structural MR imaging were performed using a 3T MRI scanner (GE Healthcare, Waukesha, Wisconsin) with an eight-channel head coil. Functional images were acquired using a T2*-weighted gradient-echo echo-planar imaging sequence (repetition time/echo time = 2000/25 ms, matrix size = 64 × 64, field of view = 24 × 24 cm, slice thickness = 4 mm with no intersection gap). Thirty-two slices were acquired per dynamic to cover the entire brain. High-resolution FLAIR and 3D spoiled gradient-echo T1-weighted sequences were acquired for anatomic reference. The rs-fMRI acquisition was obtained prior to the tb-fMRI speech paradigms in all cases. During these 6 min, the patient was asked to do the following: close their eyes, not fall asleep, not think of anything, and keep their head still.
Workflow and processing pipeline
The IClinfMRI software was used to process rs-fMRI data using the MATLAB 2014a platform. (The MathWorks, Inc., Natick, MA, USA). The software was built upon in-house scripts and calls functions in free for non-commercial use software such as dcm2nii (https://www.nitrc.org/projects/dcm2nii/), AFNI (version 16.2.09), 11 SPM12 (v6685) (Welcome Department of Cognitive Neurology, Institute of Neurology, London, UK). The resting-state datasets were pre-processed through slice timing, motion correction, de-spiking, detrending, regressing out covariates (including six motion parameters and two averaged fluctuations over masks of white matter and cerebrospinal fluid), band-pass filtering of 0.01–0.08 Hz, and 4-mm FWHM smoothing. The dcm2nii is used to convert images from Digital Imaging and Communications in Medicine (DICOM) to Neuroimaging Informatics Technology Initiative (NIfTI) formats. Functions in both AFNI and SPM12 are adopted for data analysis. For further details of the pre-processing used in this study please refer to Hsu et al. 12 After the pre-processing, seed-based analysis was applied to detect the language network.
rs-fMRI seed placement
As part of our SBC rs-fMRI post-processing workflow, seed placement guided by left Broca’s or Wernicke’s area tb-fMRI activation apart from tumor was attempted first. If tb-fMRI activation was not available or if this method of seeding was unsuccessful, then the ReHo method with the use of 3dReHo 13 embedded in the resting-state fMRI module was utilized. The ReHo analysis summarizes the local FC by measuring the temporal similarity between a given voxel and its neighboring voxels with the use of Kendall coefficient of concordance. 14 The ReHo map has been proposed as an alternative to guide seed selection. 15 In our resting-state fMRI module, the preprocessed rs-fMRI data before spatial smoothing was fed into the ReHo analysis for obtaining ReHo map. 16 In some of the datasets, we placed the seed on the ReHo map confined within a mask obtained from meta-analysis (ReHo + MA). For the analysis of language network, the meta-analysis result was downloaded from the Neurosynth (http://neurosynth.org/) by using the term “language” that resulted from 1101 studies 17 and then inversely normalized the meta-analysis maps from standard space to the native space using SPM12. Since it was corrected for a false discovery rate (FDR) of 0.01, we did not apply an additional threshold but constrained the result within the brain regions covering the Broca’s area (posterior inferior frontal gyrus including pars triangularis, pars opercularis) and Wernicke’s area (posterior superior and middle temporal gyrus), implemented by using the LONI Probabilistic Brain Atlas. 18 The final mask was then dilated for 4-mm to consider the altered functional anatomy in patients.
For each seed location, a sphere of 6-mm radius was defined as the seed region and a reference time course was generated by averaging the time courses over the voxels within the region. The rs-fMRI connectivity map was computed using Pearson correlation between the reference time course and that of each voxel in the brain (voxel size = 3.75 × 3.75 × 4 mm3). The correlation coefficient map was then converted to a Fisher’s z map. A Z-value threshold ranged from 0.6 to 1.0 was applied to optimize the visualization of the language network and to standardize the display conditions. For each patient, multiple seeds were selected, and each generated a functional connectivity map. 12 The results were determined by one of two clinical imaging physicists (P.H. and H-L.L.; both with more than 10 years of clinical fMRI experience) who processed the rs-fMRI data. Of note, >1 mm translation or >1° rotation in any direction for all post processed rs-fMRI cases were regarded as motion degraded. Functional connectivity to ipsilateral left hemisphere secondary language areas and contralateral right hemisphere language areas was then determined by a neuroradiologist (V.A.K) with expertise in rs-fMRI.
Statistical analysis
Univariate logistic regression was conducted separately for each region to evaluate significant differences in connectivity between different language areas with respect to several factors of interest. We evaluated whether the FC to different regions listed in the dataset (left DLPFC, left pre-SMA, left VWFA, right Broca’s area, right Wernicke’s area, right DLPFC, right pre-SMA, and right VWFA) were significantly different with respect to seeding from left Broca’s or left Wernicke’s area, age, gender, lobe in which tumor is located, pathology, and IDH status (if applicable). p-values and odds ratios were reported. In addition, another round of logistic regression analysis was performed to test the association of seed location with presence or absence of FC, after controlling for age and gender.
Results
For the 40 brain tumor patients in our study, the age range was from 16 to 77 years old (average of 53.75) with 20 patients being female and 20 patients being male. Pathology revealed that there were 11 diffuse astrocytoma (IDH mutant), 6 oligodendroglioma, 3 pleomorphic xanthoastrocytoma, 19 glioblastomas (IDH wildtype), and 1 metastatic brain tumor. Regarding tumor location, there were 13 located in the left frontal lobe, 14 in the left temporal lobe, 10 located in the left parietal lobe, 2 in the left occipital lobe, and 1 in the insular cortex.
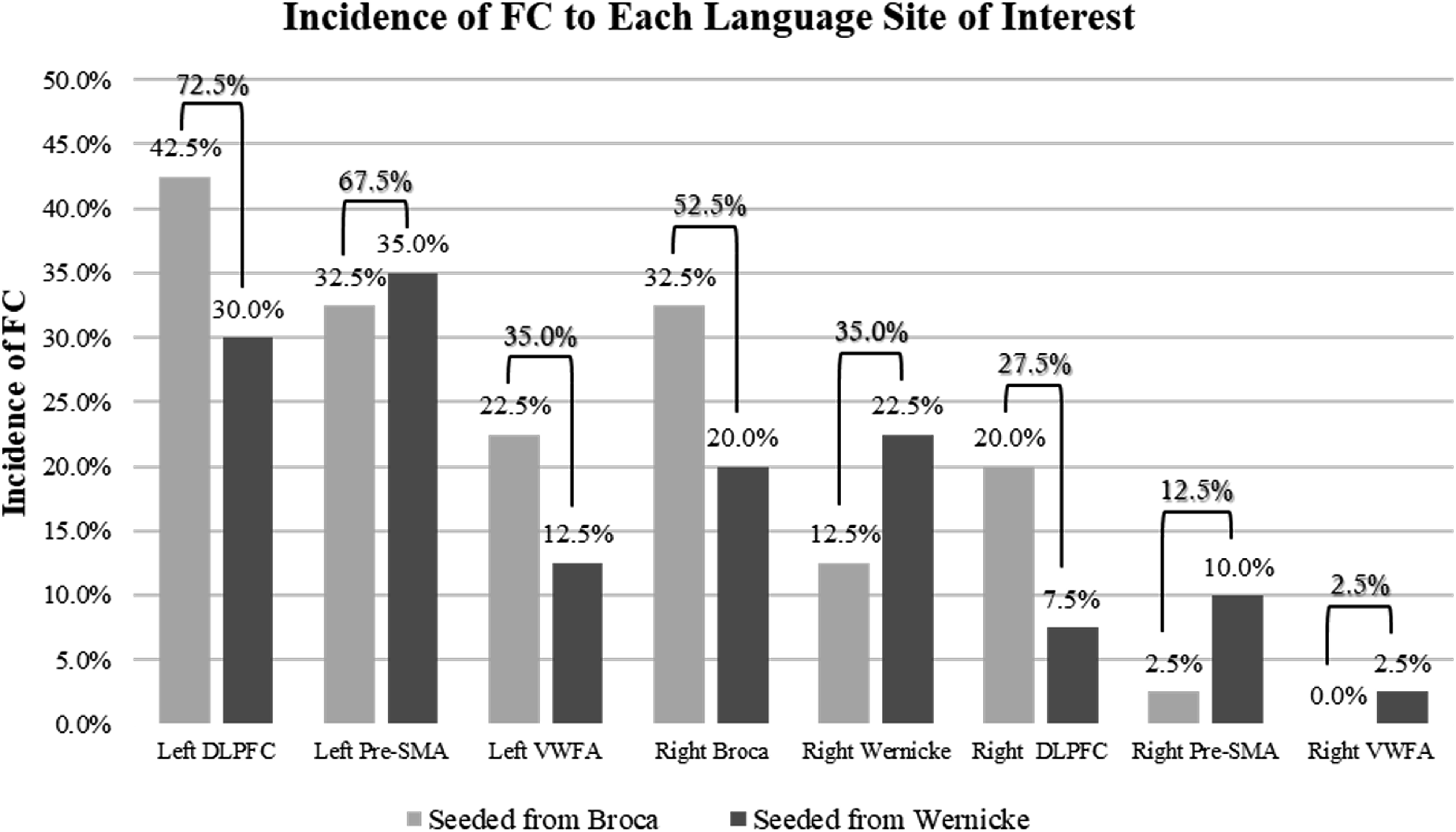
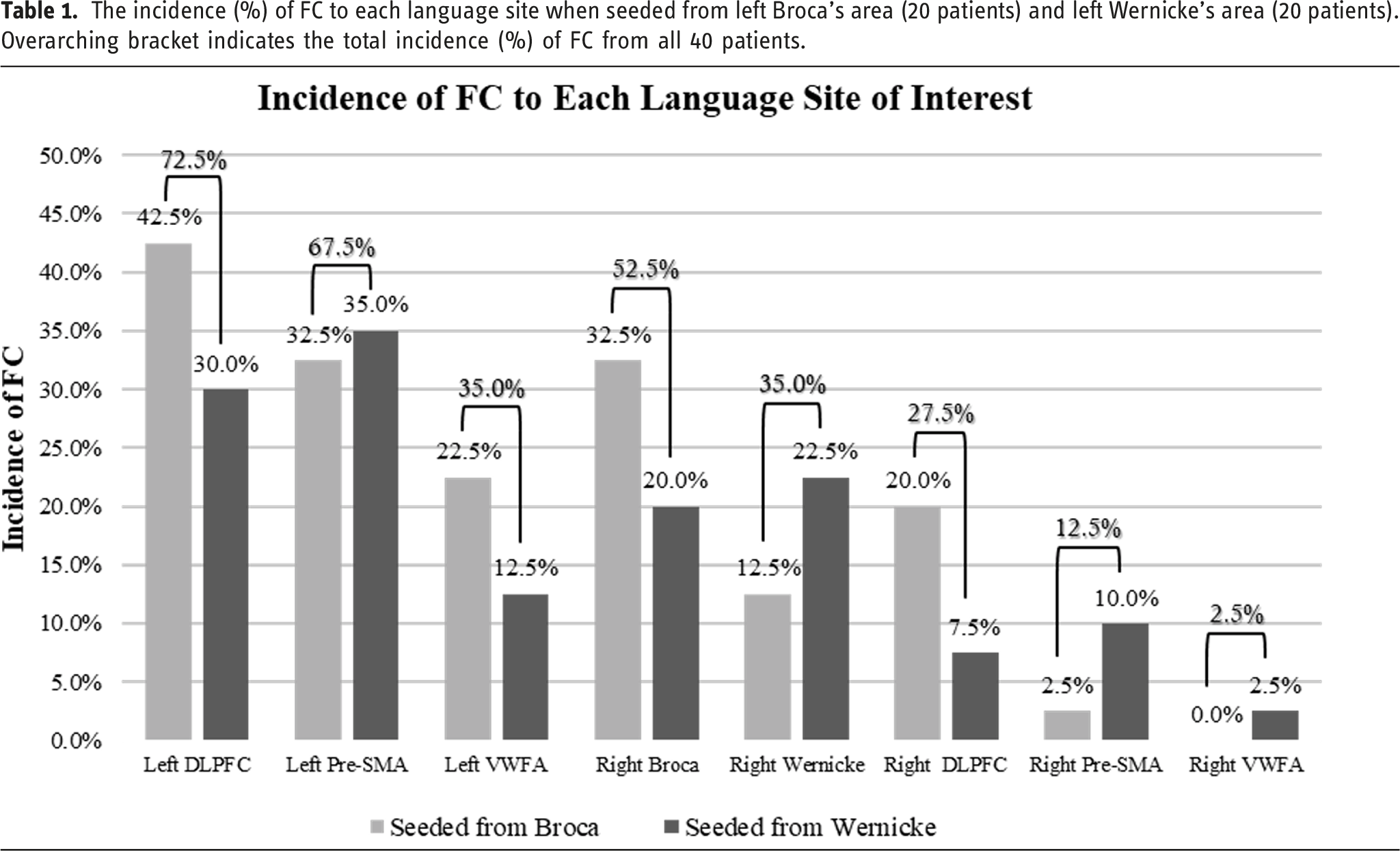
The incidence (%) of FC to each language site when seeded from left Broca’s area (20 patients) and left Wernicke’s area (20 patients). Overarching bracket indicates the total incidence (%) of FC from all 40 patients.
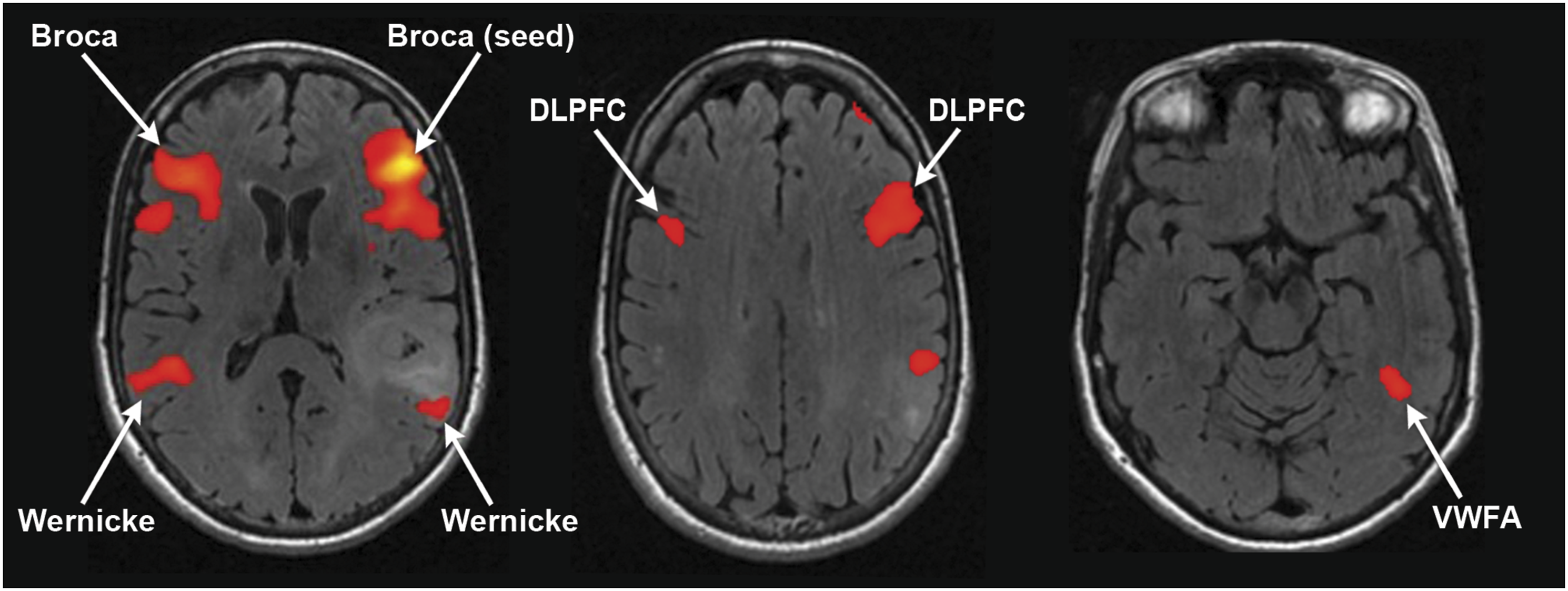
61-year-old right-handed female with a left temporal glioblastoma. Regional homogeneity was used to guide seed placement in left Broca’s area. This demonstrated FC to the contralateral right Broca’s area, bilateral Wernicke’s area, bilateral DLPFC, and the left hemisphere VWFA.
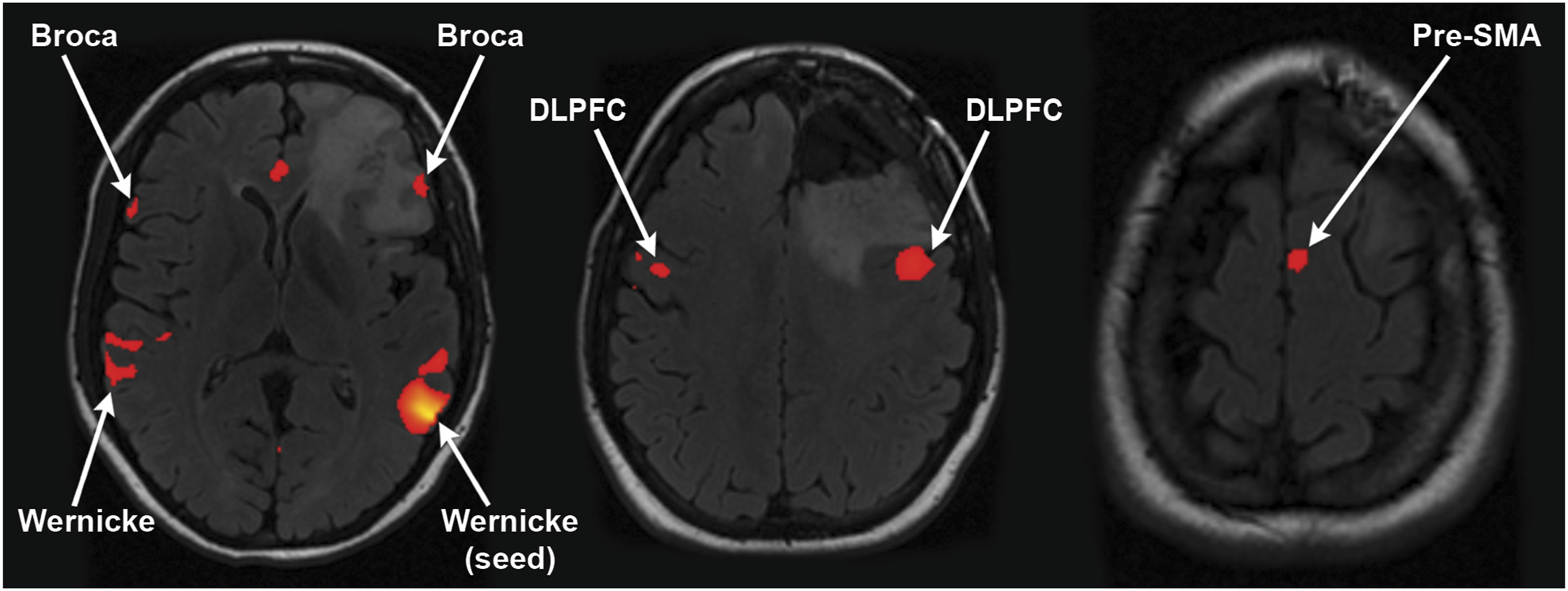
24-year-old right-handed female with a left frontal lobe IDH mutant astrocytoma, grade 4. A seed was placed in the left Wernicke’s area guided by tb-fMRI activation (not shown). This resulted in FC to bilateral Broca’s area, right Wernicke’s area, bilateral DLPFC, and the left pre-SMA.
Within our set of 40 patients, 20 were seeded from left Broca’s area and 20 were seeded from left Wernicke’s area ipsilateral to tumor (Table 1). Among the cases seeded from left Broca’s area, the greatest incidence of FC was found to the left DLPFC at 42.5%, followed by the left pre-SMA and right Broca’s area both at 32.5%. Among the cases seeded from left Wernicke’s area, the greatest incidence of FC was found to the left pre-SMA at 35.0%, which was closely followed by the left DLPFC at 30.0%.
The Fisher test did not reveal any significant differences in FC in any of the language areas when considering seed location, age, gender, lobe in which tumor is located, pathology, and IDH status (if applicable).
However, there was significant FC between several areas using a logistic regression method. There was a decreased incidence of FC to the right Wernicke’s area with increasing age (p-value = .016, odds ratio = 0.94) when seeded from both left Broca’s and left Wernicke’s area. When age and gender are controlled for, FC to right Broca’s area (p-value = .01, odds ratio = 4.75) was significantly higher when seeded from left Wernicke’s area than when seeded from left Broca’s area. Significantly greater FC to the left DLPFC area was observed with respect to male gender (p-value = .03, OR = 0.08) when seeded from left Broca’s or Wernicke’s area. When seeding from left Broca’s area, a significantly higher incidence of FC was found to the left VWFA (p-value = .04, odds ratio = 0.079) compared to seeding from left Wernicke’s area.
Discussion
The DLPFC, pre-SMA, and VWFA are eloquent language areas that are also valuable to localize for presurgical planning in efforts to preserve language functions. This study explores the use of SBC rs-fMRI to identify the incidence of FC to secondary language areas as well as contralateral primary and secondary language areas when seeding from Broca’s area or Wernicke’s area ipsilateral to tumor in the left hemisphere.
We found that the left DLPFC showed the highest incidence of FC (72.5%), followed by 67.5% FC to left pre-SMA, and 35.0% to left VWFA with seed placement in left Broca’s area or left Wernicke’s area in all 40 patients. Overall, 70.0% of cases showed FC to at least one right hemisphere primary or secondary language area. For incidence (%) of FC to each respective contralateral right hemisphere language area, see Table 1.
We found a significantly decreased incidence of FC to right Wernicke’s area with respect to increasing age when seeded from both left Broca’s or Wernicke’s area using logistic regression. The difference in language connectivity between a young patient versus an old patient may vary; neuroanatomy has shown that there are undeniable changes with age due to atrophy of the gray and white matter and neurochemical alterations which may affect FC. 19 In a study consisting of 10 healthy young subjects with a mean age of 23.5 years/o (range of 21–36 years/o) and 10 healthy older subjects with a mean age of 59.4 years/o (range of 54–76 years/o), Broca’s and Wernicke’s area were both determined to have significantly lower FC in the older group. 19 Additional studies suggest that as individuals age, there is more symmetrical activation of the bilateral hemispheres, hence hemispheric asymmetry reduction in older adults (HAROLD). 20 The posterior–anterior shift in aging (PASA) model 21 is another theory in which older populations show lower brain activity in the occipital lobe and increased activity in the frontal lobe. The exact impact that age has on incidence of FC to language areas require more studies to elucidate.
Our results also showed differences in the incidence of FC to language areas depending on the location of the SBC seed. Seeding from the left Broca’s area showed significantly higher FC to the left VWFA. The VWFA is a language area dedicated to visual word recognition.22,23 Structural neuroanatomy literature has established that there are three major white matter tracts that pass in close proximity to the VWFA: the inferior fronto-occipital fasciculus (IFOF), the inferior longitudinal fasciculus (ILF), and the vertical occipital fasciculus (VOF). 24 Literature findings suggest that the VWFA has preferential connectivity to left Wernicke’s area 25 through the VOF or temporal-parietal/superior parietal lobule (TP-SPL) tract that extends dorsally from the VWFA to Wernicke’s area.26,27 A study done by Welcome et al. demonstrated that lesions impacting the VWFA or deafferentation of the fiber tracts between Wernicke’s area and the VWFA would cause development of alexia without agraphia. 28 However, our study demonstrated that seeding from left Broca’s area produced significantly greater incidence of FC to left VWFA compared to when seeding from left Wernicke’s area. Imaging studies have strongly suggested that the IFOF terminates in the inferior frontal gyrus 29 and potentially involves the Broca’s area. Bouhali et al. have also demonstrated that there is FC to the left Broca’s area from the left VWFA 24 Further studies are need to investigate FC between left Broca’s area and the left VWFA.
Another finding from our study was that seeding from left Wernicke’s area correlated with a significantly higher incidence of FC to right Broca’s area compared to when seeding from left Broca’s area. The crossed frontal aslant tract connects left premotor areas to the contralateral right premotor and supplementary motor areas 30 demonstrating one way in which the hemispheres communicate. A separate study suggested that damage to the cortico-subcortical circuits facilitating transhemispheric communication after resecting a tumor in the basal ganglia could be compensated for by increased left FC. 31 Xu et al. proposed that in cases where transhemispheric FC between language centers was decreased, an increase in left FC could compensate and help patients regain speech fluency over time. 31 It appears from these studies that the FC within the same hemisphere and trans-hemispherically work in tandem; when left-sided FC is compromised, right-sided FC compensates and vice versa.30,31 Further studies will be necessary to shed light on the exact connection between the left Wernicke’s area to right Broca’s area.
Finally, we found a significantly higher incidence of FC to the left DLPFC observed with respect to males. The difference in gender performance of language paradigms in a presurgical evaluation is limited. Gender differences in language and subsequent mapping of these areas may reflect variance in language processing, location of tumor, resilience to disease, neuroplasticity, or even language organization and function. 32 Others offer discussion that gonadal hormones may influence transhemispheric communications. For instance, one study suggests that there is reduced cortico-cortical transmissions during the luteal cycle phase of a female’s menstrual cycle because the increased level of progesterone decouples the hemispheres and reduces the extent of language lateralization. 33
Overall, we noted that there was global FC in language areas in both hemispheres. Independent component analysis (ICA) is another well-established means of evaluating FC in contrast to seed-based correlation (SBC). In fact, ICA is typically regarded as the better methodology for evaluating global networks, while SBC effectively targets a specific language area based on prior knowledge. 34 However, because we found FC in the bilateral hemispheres, it is useful to know that SBC can also demonstrate global language FC. No study to our knowledge has explicitly described whether SBC versus ICA is better for detection secondary language areas (DLPFC, pre-SMA, and VWFA) and this would be useful for future research.
Functional plasticity can be defined as the shifts in neuron connections without altercation to the anatomical connections. 35 Briganti et al. demonstrated how gliomas, regardless of grade, have been able to change the FC of language-related areas with additional long-distance effects to the right hemisphere. 36 There are several factors that influence the possibility of reorganization. For example, slower growing and low-grade gliomas may provide greater time for plastic changes to occur. 37 For example, isocitrate dehydrogenase (IDH) mutant low-grade gliomas show reduced aggressiveness and increased longevity. 38 Another component that may influence the possibility of reorganization includes eloquent language areas and their proximity to tumor. In our study, brain tumors located in the left frontal lobe and left temporal lobe showed near equivalent incidence of FC to the right hemisphere. Wang et al. reported that left frontal lesions reduced FC in the Broca’s and Wernicke’s area whereas left temporal lobe lesions only affected Wernicke’s area. 39 Buklina et al., found that contralateral FC appeared more consistent in tumors affecting the left frontal lobe suggesting that Broca’s area plasticity is more dependent on tumor location. 40 More studies are needed to understand the correlations between tumor location, plasticity, and incidence of FC to language areas.
FC to right-sided language areas in our study reinforces the concept of known right homolog language areas which may take on a greater role because of possible tumor-induced plasticity in the presence of left hemisphere brain tumors. Some neurosurgeons have attempted to conduct multistep removals of gliomas to allow for plasticity-driven recovery to counter potential clinical deficits 41 while others have implemented cortical electrical stimulation and behavioral training prior to surgical resection of tumors to accelerate plastic changes. 42 The presence of right hemispheric FC to language areas could be predictive of cognitive improvement in a post-surgical setting and in turn—better preservation of life quality amongst brain tumor patients. 43
Limitations
In this study, thresholding was used to optimize Broca’s and/or Wernicke’s area FC since they are the primary language centers. If thresholding was tailored specifically to secondary language areas, there may have been an increased incidence of FC to these sites. In addition, the strength of FC (z-value) at the secondary language areas was not considered in the statistical analysis. In addition, our study did not directly compare tb-fMRI activations and rs-fMRI FC to secondary language areas, and this should be performed in future studies.
Another limitation is that we included the review of only one senior neuroradiologist (V.A.K.) with expertise in rs-fMRI in confirming ipsilateral secondary and contralateral language area FC. Gujar et al. found in a retrospective analysis, that there was variability amongst the reviewer rs-fMRI language localization based on years of experience. In future studies, it would be beneficial to include at least two or more experienced rs-fMRI neuroradiologists in the review panel. 44
Our study classified tumor location by lobes (i.e., frontal, temporal, parietal, occipital, insular, and temporal), but only had two tumors in the left occipital lobe and one in the left insula. Pasquini et al. suggested that lesions in the frontal lobe may cause a larger detriment in connectivity to other locations and catalyze more plastic changes. 45 They further describe that fast growing and high-grade glioma may have a higher chance of diaschisis and work as a “whole brain disease” that causes far too extensive damage even in the initial stages limiting possibilities for language reorganization. 45 Another study has shown location of tumor may have effect on FC to language areas. 40 A larger population of cases that intentionally studies tumor location, tumor size, and extent of glioma infiltrative disease in evaluating the incidence of FC to secondary language areas would be valuable in delving deeper into understanding how these factors effect FC. It would also be useful to evaluate the incidence of secondary language area FC in a healthy cohort of subjects without brain tumors or other neurological disorders.
Conclusion
We found SBC rs-fMRI can detect left hemisphere secondary language areas (DLPFC, pre-SMA, VWFA) as well as right hemispheric primary and secondary language areas which may be of value for presurgical planning. This is also the first study using SBC rs-fMRI to correlate FC from left-sided primary language areas to all three left hemisphere secondary language areas as well as right hemisphere language areas. Overall, we found the left DLPFC and left pre-SMA showed the highest incidence of FC when seeding from left Broca’s or left Wernicke’s area in all 40 patients. We found increasing age resulted in a lower incidence of FC to right Wernicke’s area for both left Broca’s and left Wernicke’s areas. In addition, seed location and gender played a factor in the incidence of FC to some of the language areas. Our findings provide preliminary insights into the incidence of FC to secondary and contralateral language areas using SBC analysis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding from the National Cancer Institute of the National Institutes of Health under award number P30 CA016672, and the National Cancer Institute of the National Institutes of Health under award number R01 CA258788 (HL).