Abstract
Keywords
Introduction
Stroke is a leading cause of long-term adult disability, requiring extensive rehabilitation. 1 Providing appropriate therapy is challenging because stroke is a heterogeneous condition, meaning therapeutic interventions are not one size fits all. Therefore, physiological biomarkers have an important role in guiding therapeutic strategies. Briefly, stroke recovery biomarkers can be defined as indicators of disease state that can be used as a measure of underlying physiological processes to understand outcome or predict recovery or treatment response.2,3 Recently, an international expert consensus identified that the presence of an upper-limb motor-evoked potential (MEP) in response to transcranial magnetic stimulation (TMS) to the ipsilesional motor cortex (M1) was a predictor of good motor outcomes. 2 MEPs provide indication that the descending motor pathways are intact, an important determinant of recovery potential. 4
Resting state functional connectivity (RSFC) is an additional measure that may provide insight to understand mechanisms of stroke recovery and outcomes. Therefore, it has been identified as a developmental priority in stroke recovery and rehabilitation by consensus from international experts. 2 Importantly, RSFC can be performed in patients with a wide range of impairments, including those with severe stroke, and is able to interrogate multiple brain networks simultaneously. 2 A relatively novel, yet safe, technique is to quantify RSFC using electroencephalography (EEG). Previous investigations have reported that EEG connectivity correlated with upper-limb impairment following stroke, suggesting that EEG measures of connectivity can provide a meaningful marker of behavior. 5 Furthermore, EEG studies have demonstrated that interhemispheric RSFC is associated with the excitability of MEP generating circuits in chronic stroke, 6 which might suggest an interaction between interhemispheric RSFC and descending motor pathway integrity. However, it is not clear how MEP status might influence the relationship between EEG-based measures of interhemispheric RSFC and upper-limb behavior in stroke. Investigation of RSFC could provide mechanistic insight to understand functional network activity after stroke and how this may differ based on MEP status. This information may be a necessary first step toward development of combined brain structure and function markers of behavior. The purpose of this study was to determine (1) whether interhemispheric RSFC, quantified with EEG, is different between stroke survivors based on integrity of descending motor pathways; (2) whether interhemispheric RSFC is associated with upper-limb behavior in chronic stroke; and (3) if the relationship between interhemispheric RSFC and upper-limb behavior differs based on MEP status. We hypothesized that MEP positive (MEP+) stroke survivors would have greater interhemispheric RSFC of the sensorimotor region compared with MEP negative (MEP−) stroke survivors and that greater interhemispheric connectivity would be associated with less impairment and greater activity of the upper limb.
Materials and Methods
Participants
A total of 36 people with stroke (26 male, aged 64.4 [SD = 11.1; range = 43-93] years) and 25 healthy adults (17 male, aged 67.3 [SD = 6.7; range = 52-77] years) were recruited. Potential stroke participants were eligible for inclusion if they had experienced a first-ever ischemic or hemorrhagic stroke with motor impairment (Fugl-Meyer Upper Extremity [FM-UE] motor scale of <66) more than 6 months prior to inclusion, were ≥18 years of age, and had no contraindications for magnetic resonance imaging (MRI) or TMS. 7 Exclusion criteria were language or cognitive impairments and a history of craniotomy or craniectomy because skull defects are known to affect the EEG signal. Healthy adults were eligible for inclusion if they were ≥18 years old and had no contraindications for TMS. 7 All study participants provided written informed consent, and ethical approval was provided by the University of South Australia Human Research Ethics Committee and University of Adelaide Human Research Ethics Committee.
Experimental Protocol
Stroke survivors attended 2 experimental sessions. At the first session, an MRI scan was performed to determine lesion location, lesion volume, and integrity of the corticospinal and interhemispheric white matter pathways. The second session was performed within 5 days of the first and involved an EEG recording to determine RSFC and TMS to determine MEP status as a functional measure of integrity of the descending motor pathways. Following this, 3 upper-limb behavioral outcome measures were performed. Healthy adult participants attended a single experimental session that comprised an EEG recording to determine RSFC and TMS to determine MEP status.
Behavioral Measures
Upper-limb assessments included the Action Research Arm Test (ARAT), FM-UE, and grip strength. The ARAT is a validated and reliable measure of paretic upper-limb activity.8,9 It consists of 19 items, and scores range from 0 to 57, with higher scores indicating greater arm activity. The FM-UE is a validated and reliable measure of sensorimotor impairment.10,11 The FM-UE consists of 33 items, with scores ranging from 0 to 66 and higher scores indicating less upper-limb impairment. Finally, grip strength of the paretic hand was measured using a hand dynamometer (SH5001 Saehan Hydraulic Hand Dynamometer, Saehan Co, Masan, Korea) with the best (maximal) response of 3 attempts recorded. Previous studies have reported that grip strength assessed with a hand dynamometer has excellent reliability and is a valid assessment of upper-limb impairment. 12
EEG Acquisition
EEG data were obtained in 2 separate laboratories for stroke participants and healthy adults, respectively. Acquisition and analyses were identical, except for minor differences that were corrected during data preprocessing to ensure that all data were identical prior to analysis. For all participants, 3 minutes of resting EEG was acquired using an ASA-lab EEG system with 64 channel Waveguard cap (ANT Neuro, Enschede, Netherlands). Signals were sampled at 2048 Hz, amplified 20×, online filtered (stroke participants: 1-45 Hz; healthy adults: DC-553 Hz), and online referenced to CPz (stroke participants) or common average (healthy adults). Signal impedance was kept to <5 kΩ using EEG conductive gel. Participants were seated comfortably in a quiet room during data recording. Prior to recording data, participants were asked to relax, keep their eyes open and look straight ahead at a fixation point positioned at eye level, and refrain from speaking or moving and not to actively engage in any cognitive or mental tasks.
EEG Preprocessing and Analysis
EEG data were exported to MATLAB 9.3.0 (MathWorks, Inc, Natick, MA) for preprocessing and analysis with the EEGLAB and FieldTrip toolboxes.13,14 Preprocessing steps included removal of bad channels, re-referencing to the average, filtering with a second-order Butterworth filter with a bandpass 1 to 45 Hz using the EEGLAB filter function for healthy adult data, and segmenting data into 180 epochs of 1 s each. Data were then submitted to an independent component analysis for denoising. 13 Nonphysiological components were identified using a semiautomated component classification algorithm (tesa_compselect function) within the TESA toolbox. 15 Functional connectivity was determined using the debiased weighted phase lag index (dwPLI) between all electrodes. 16 The dwPLI is a conservative approach to estimate connectivity based on phase consistency and weighting against zero phase lag relationships to limit spurious detection of connectivity resulting from effects of volume conduction and common reference problems.16,17 Values for dwPLI range from 0 (no phase coupling; negative values can incidentally occur) to 1 (maximal phase coupling). The primary interhemispheric connectivity measure of interest was calculated between 2 regions of interest approximating the left (C1, C3, CP1, CP3) and right (C2, C4, CP2, CP4) sensorimotor cortex. The average connectivity value for all possible combinations of interhemispheric pairs of electrodes were determined for δ (1-3 Hz), θ (4-7 Hz), α (8-13 Hz), and β frequencies (14-30 Hz). Several secondary control analyses were performed. First, whole scalp interhemispheric connectivity was determined by calculating dwPLI between all electrodes overlying the ipsilesional and contralesional hemispheres. Second, dwPLI between individual electrode pairs within the identified regions of interest (ie, C1 and C2; C3 and C4; CP1 and CP2; CP3 and CP4) were determined. Finally, EEG power was determined within the ipsilesional and contralesional regions of interest and for the whole ipsilesional and contralesional hemisphere. Briefly, spectral power was extracted from preprocessed EEG data by submitting the time series in each channel to a discrete fast Fourier transform and then normalizing by epoch length. Absolute power for each electrode was calculated in 1-Hz bins and expressed relative to total power between 1 and 30 Hz.
Electromyography
Surface electromyography (EMG) was used to determine the presence of a MEP evoked by TMS. Ag-AgCl surface electrodes (Ambu, Ballerup, Denmark) were positioned in a belly-tendon montage over the first dorsal interosseous (FDI) muscle of the paretic hand (right hand for healthy adults). Skin was prepared by cleaning with alcohol and lightly abrading (NuPrep), and a ground strap was positioned around the wrist. Signals were sampled at 5 kHz (CED 1401), amplified 1000× (CED 1902), filtered (20-1000 Hz), and stored for offline analysis (Signal v4.09, Cambridge Electronic Design, Cambridge, UK).
Transcranial Magnetic Stimulation
Single, monophasic, TMS pulses were applied to the ipsilesional M1 (left M1 in healthy adults) at 0.2 Hz ± 10% to determine MEP status (70 mm figure-of-eight magnetic coil; Magstim 200 stimulator, Magstim, Whitland, Dyfed, UK). The coil was positioned tangentially over the scalp to induce a posterior-anterior current across the hand M1. Stimulation intensity was increased incrementally, and the coil position was systematically moved anterior-posterior and medial-lateral in small increments to attempt to evoke a consistent MEP. To be categorized as MEP+, at least 5 out of 10 MEPs with peak-to-peak amplitude ≥50 µV within a 15- to 30-ms window after the TMS pulse were required. Where an MEP could not be found, even at 100% of maximal stimulator output, participants were asked (where possible) to perform an active contraction of the FDI to confirm that a consistent MEP could not be determined. If this did not result in a consistent MEP, the participant was considered MEP−. For MEP+ stroke participants, corticospinal excitability was determined by evoking 30 MEPs at 120% resting motor threshold (RMT; defined as the minimum stimulus intensity required to evoke a MEP with peak-to-peak amplitude ≥50 µV in at least 5 out of 10 consecutive trials in the relaxed FDI) and measuring peak-to-peak amplitudes. 18
Magnetic Resonance Imaging
MRI was performed using a Siemens 3T MAGNETOM Skyra (Siemens, Germany) with a 64-channel head coil. The scan protocol was as follows: T1-weighted image, voxel 1 mm3, repetition time (TR) = 2300 ms, echo time (TE) = 2.98 ms, flip angle = 9°; T2-weighted fluid-attenuated inversion recovery (FLAIR) images, voxel 1 mm × 0.5 mm × 0.5 mm, TR = 5000 ms, TE = 393 ms; diffusion MRI was performed with a diffusion-weighted spin echo pulse sequence with diffusion gradients along 64 directions (voxel 2 mm3, TR = 4200 ms,
MRI Preprocessing and Analysis
Image processing was carried out using FSL (FMRIB Software Library, Oxford, UK).19,20 T1-weighted images were registered to T2-weighted FLAIR using FLIRT.21,22 Lesion masks were manually drawn by an experienced investigator blind to MEP status on the T1-weighted image, as informed by the T2-weighted FLAIR image. The overlap between the lesion and a reference corticospinal tract (CST) was determined as a marker of injury to descending motor pathways. Lesions masks for each participant were registered to standard MNI (Montreal Neurological Institute) space using FNIRT.19,20,23 The reference CST was derived from the Johns Hopkins University (JHU) white matter tractography atlas and overlap volume between the lesion mask and reference CST determined.24 -26
Diffusion data were analyzed using FMRIB’s Diffusion Toolbox. Briefly, B0-inhomogeneirty distortion was corrected using FSL’s Topup function. 27 Head motion and eddy current induced field inhomogeneities were corrected using FSL’s Eddy function. 28 DTIfit then fitted a diffusion tensor at each voxel to estimate fractional anisotropy (FA). A region of interest for the left and right posterior limb of the internal capsules (PLICs) were generated from the JHU white matter tractography atlas.24,25 The PLIC regions of interest were then registered to subject-specific FA maps. Mean FA values were calculated for each PLIC (range 0-1: 0, isotropic diffusion; 1, anisotropic diffusion). An FA asymmetry index was calculated as FAAI = (FAC − FAI)/(FAC + FAI), where FAC is FA of the contralesional PLIC and FAI is FA of the ipsilesional PLIC. Negative FAAI values indicate reduced FA in the contralesional PLIC, and positive FAAI values indicate reduced FA in the ipsilesional PLIC. Structural integrity of interhemispheric white matter pathways was determined at a region of interest corresponding to sections III (motor) and IV (sensory) as described by Hofer and Frahm. 29 The region of interest was manually drawn in the participants’ native space and mean FA values extracted. Similar to previous studies, 30 the mask included the center slice and 4 adjacent slices.
Statistical Analysis
Statistical analyses were performed using SPSS software (IBM SPSS Statistics for Windows, Version 24.0, Armonk, NY) with significance level set at
Results
Demographics and Clinical Characteristics
Participant demographics are reported in Table 1, and individual participant structural imaging is provided in Figure 1. Of the 36 stroke participants, 26 were found to be MEP+ and 10 MEP−. All healthy adult participants were MEP+. There were no differences in age, gender, time since stroke, lesion location, and lesion volume between participants (Table 1). The first component of the principle components analysis accounted for 87% of the variance in the ARAT, FM-UE, and grip strength and was used as the combined behavioral measure (see Table 2 for individual and combined behavioral measures). Those in the MEP+ group demonstrated greater upper-limb function compared with the MEP− group (Table 2). As expected, FAAI correlated with the principle behavioral component (ρ = −0.569,
Participant Demographics and Clinical Characteristics. a
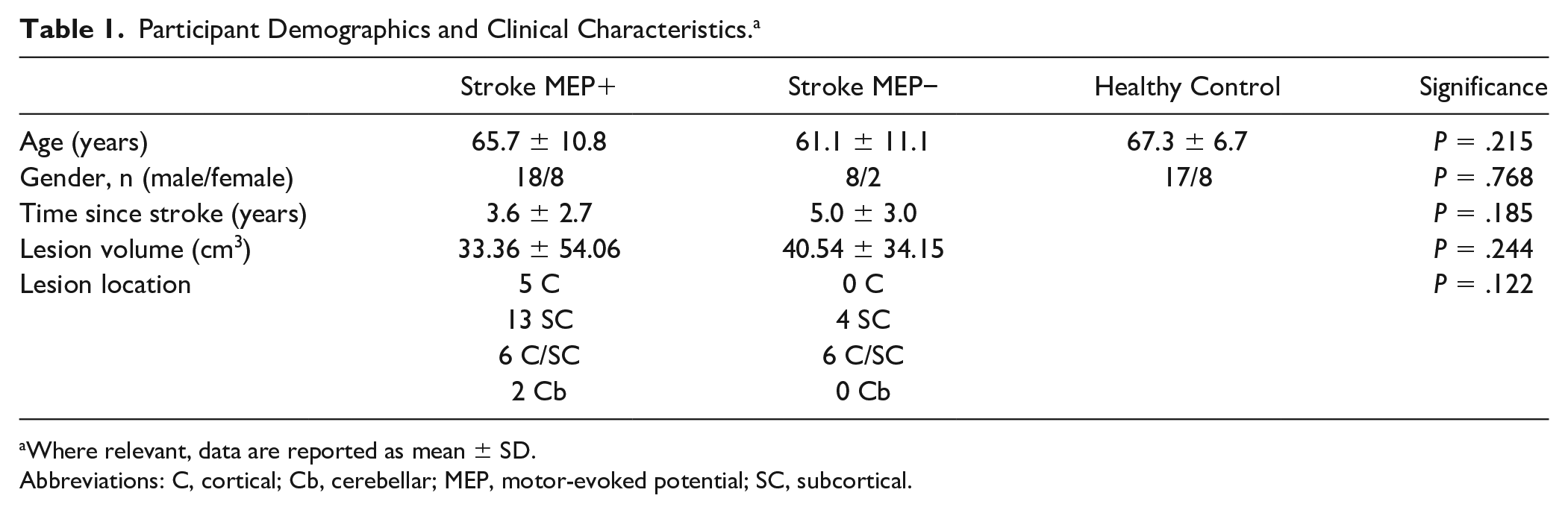
Where relevant, data are reported as mean ± SD.
Abbreviations: C, cortical; Cb, cerebellar; MEP, motor-evoked potential; SC, subcortical.
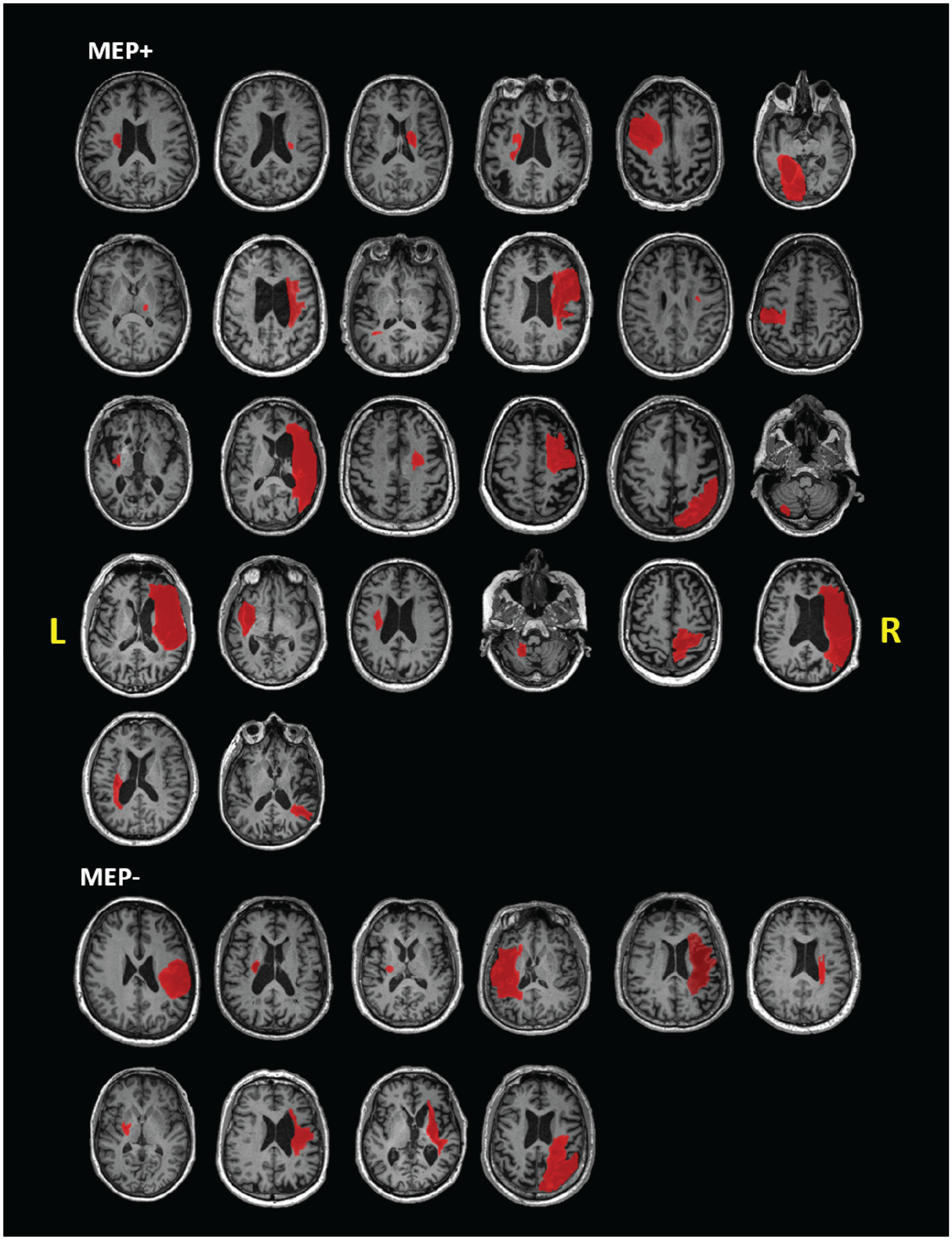
Individual structural T1-weighted images for each stroke participant: images are shown in the axial plane at the level of the largest cross-sectional area of the lesion. Lesion tracings for each participant are shown in red.
Behavioral and Neurophysiological Measures.
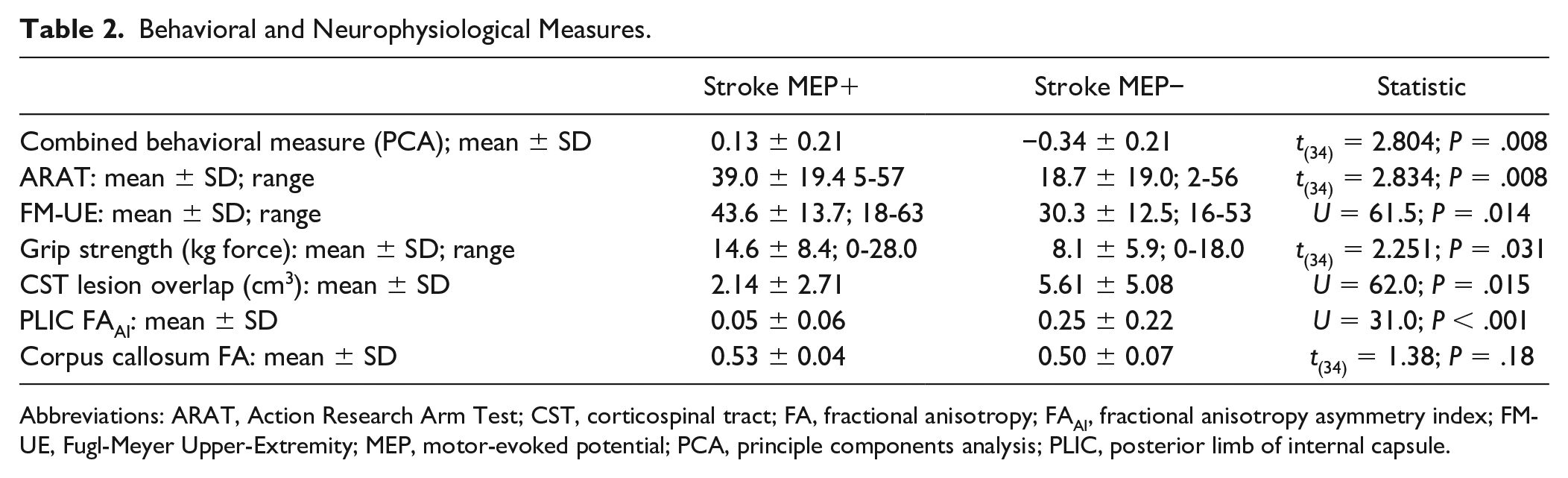
Abbreviations: ARAT, Action Research Arm Test; CST, corticospinal tract; FA, fractional anisotropy; FAAI, fractional anisotropy asymmetry index; FM-UE, Fugl-Meyer Upper-Extremity; MEP, motor-evoked potential; PCA, principle components analysis; PLIC, posterior limb of internal capsule.
Assessment of CST Integrity
MEP− stroke participants had greater CST lesion overlap and more positive FAAI values (Table 2). For MEP+ stroke survivors, MEP amplitude was negatively correlated with FAAI of the PLIC (ρ = −0.532;
RSFC and MEP Status
MEP+ stroke participants had stronger β-frequency connectivity between the ipsilesional and contralesional sensorimotor regions of interest compared with the MEP− participants, and this relationship survived correction for multiple comparisons (
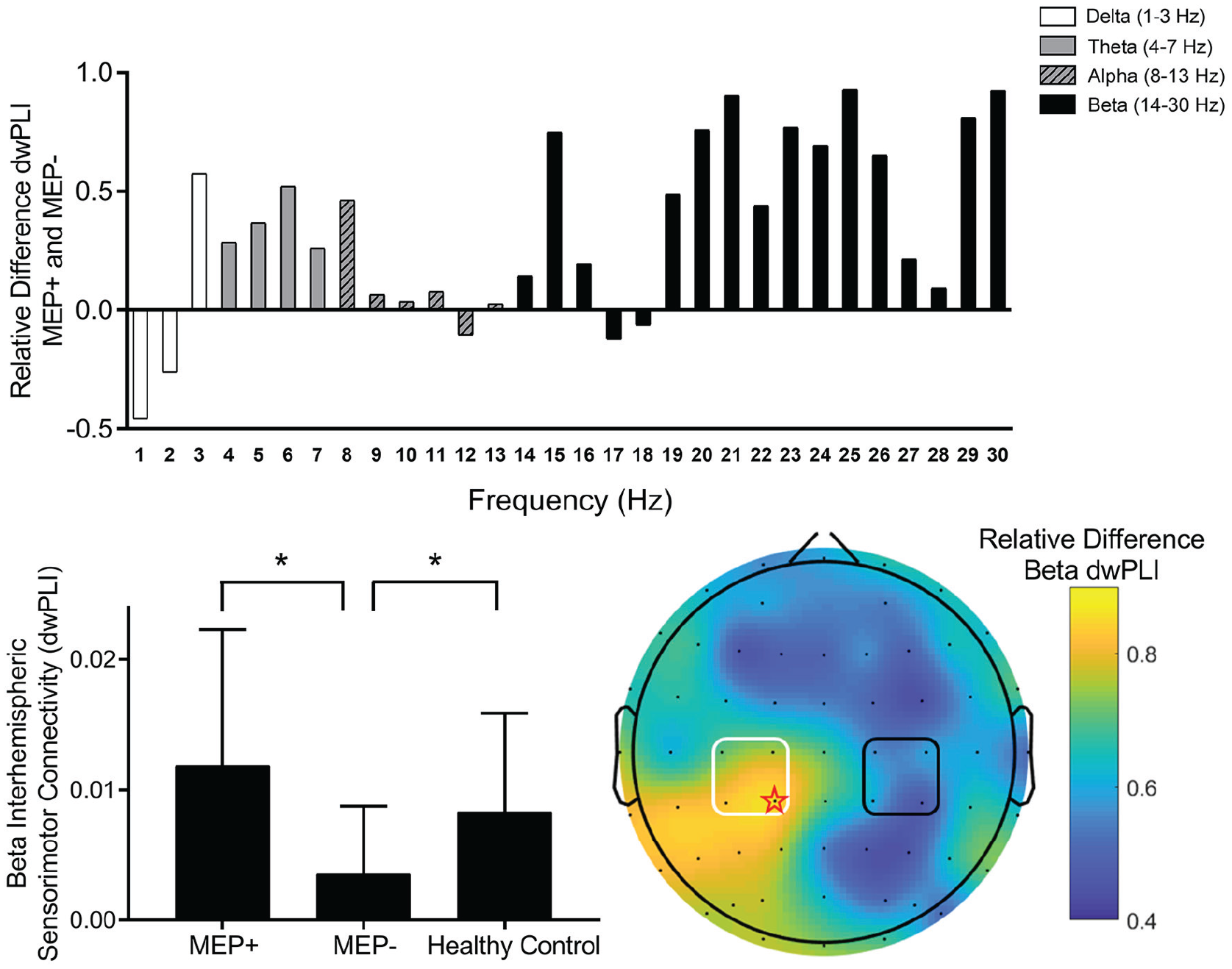
Resting state functional connectivity between regions approximating the ipsilesional sensorimotor and contralesional sensorimotor areas. Top: Relative difference in debiased weighted phase lag index (dwPLI) shown in 1-Hz bins between electrode clusters approximating the ipsilesional and contralesional sensorimotor regions. Calculated as the difference in connectivity between motor-evoked potential positive (MEP+) and MEP− stroke survivors divided by sum. Positive values indicate greater connectivity in MEP+ stroke survivors. Note that the frequency range of β (14-30 Hz) appears to show the largest change between groups. Bottom left: There was a significant difference between MEP+ and MEP− stroke survivors in dwPLI between electrodes approximating the ipsilesional and contralesional sensorimotor cortex for β frequency (14-30 Hz). This difference appeared to reflect abnormal dwPLI values for MEP− stroke survivors because they differed compared with healthy adults (data are shown as mean and SD). Bottom right: A topographical plot of relative difference in dwPLI between MEP+ and MEP− stroke survivors for connectivity between electrodes approximating the ipsilesional sensorimotor cortex and all other electrodes across the scalp (calculated as difference in connectivity between MEP+ and MEP− stroke survivors divided by sum for dwPLI between the ipsilesional sensorimotor cortex region of interest and all other electrodes). The ipsilesional sensorimotor seed region is shown as a black square. It is noteworthy that maximal change between MEP+ and MEP− stroke survivors was observed at CP1 (red star), which falls within the region of interest for electrodes approximating the contralesional M1 (white square). Note that, for illustrative purposes, the right hemisphere has been modeled as the stroke affected hemisphere.
Whole scalp interhemispheric connectivity was not statistically different between MEP+ and MEP− stroke survivors for any frequency (all
There was a significant difference in β dwPLI between MEP+ and MEP− stroke participants and healthy adults (H(2) = 7.788,
Interhemispheric RSFC and Structural Integrity of White Matter Pathways
There was a significant correlation between β dwPLI and lesion overlap (ρ = −0.304;
RSFC Is Associated With Upper-Limb Behavior
There was a moderate correlation between the combined behavioral measure and β dwPLI (
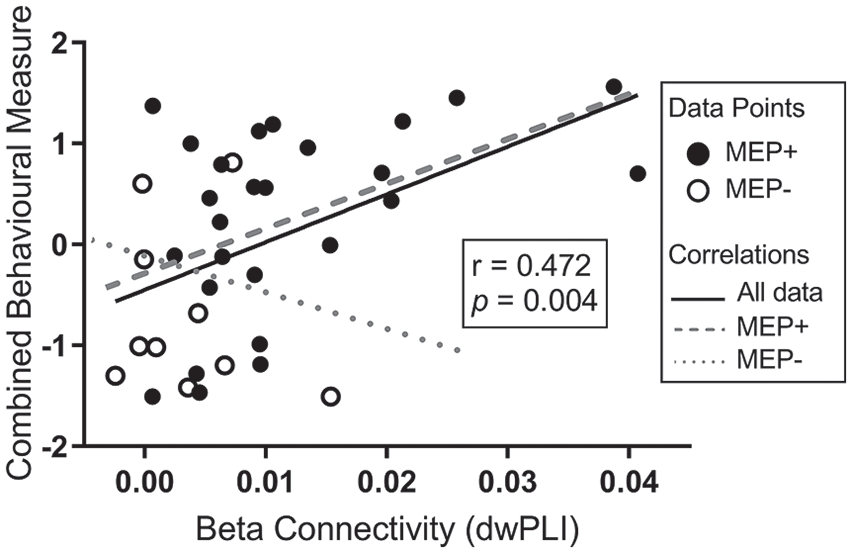
Association between upper-limb behavior and β frequency resting state functional connectivity between regions approximating the ipsilesional sensorimotor and contralesional sensorimotor areas. Closed circles represent motor-evoked potential positive (MEP+) stroke survivors and open circles, MEP− stroke survivors. The solid black regression line represents the linear relationship between β debiased weighted phase lag index (dwPLI) and all stroke participants. The gray dashed line represents the linear relationship between β dwPLI and MEP+ stroke participants. The gray dotted line represents the linear relationship between β dwPLI and MEP− stroke participants. It is noteworthy that the correlation between upper-limb behavior and resting state functional connectivity appears to be driven by the MEP+ group.
Regression Models to Account for Variance in Upper-Limb Behavior. a
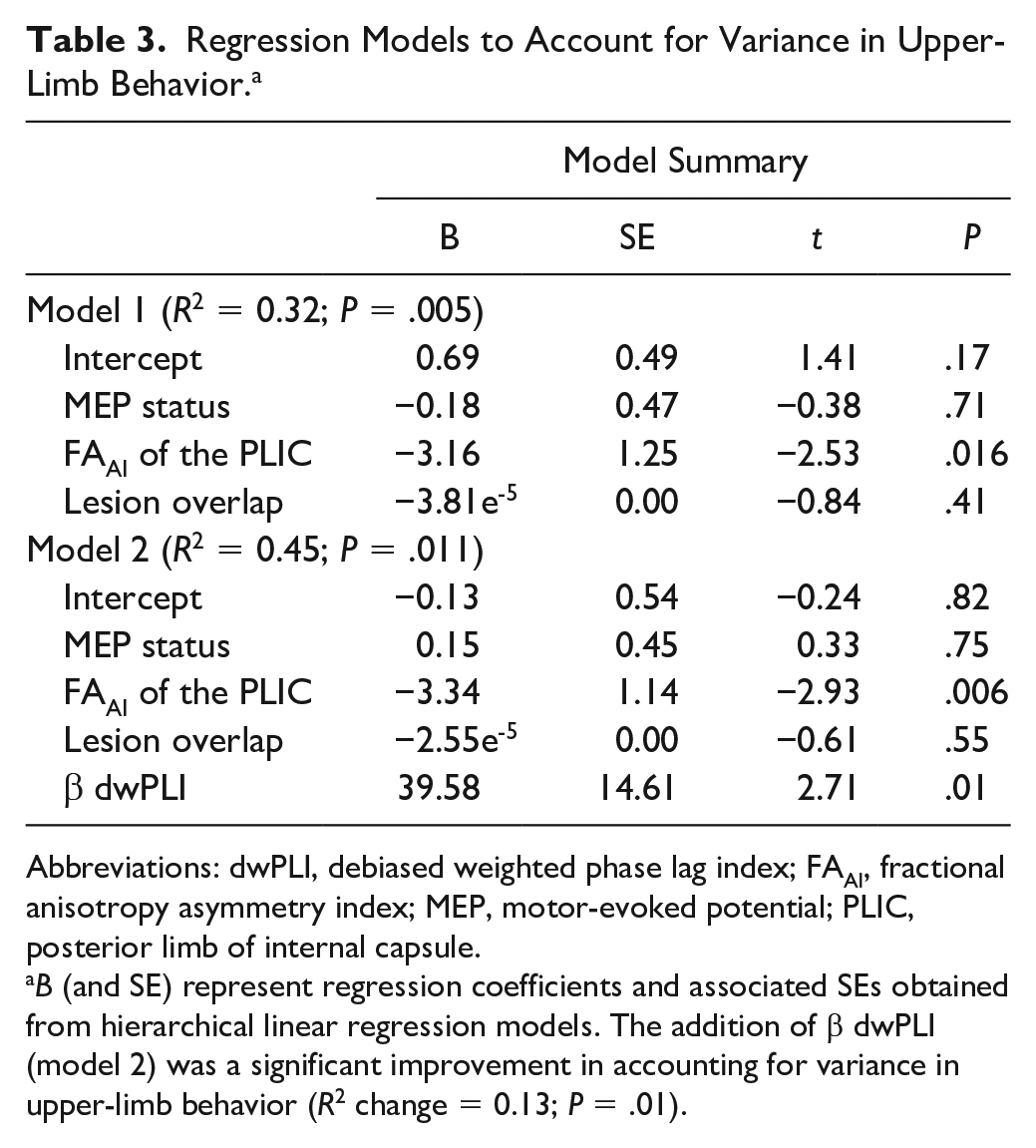
Abbreviations: dwPLI, debiased weighted phase lag index; FAAI, fractional anisotropy asymmetry index; MEP, motor-evoked potential; PLIC, posterior limb of internal capsule.
Discussion
This study explored the association between RSFC, MEP status, and upper-limb behavior in chronic stroke. The key findings were the following: (1) A functional measure of CST integrity (MEP status) was associated with structural measures of CST integrity, indicating that MEP status is a good marker of descending white matter integrity; (2) β frequency RSFC was significantly greater in the MEP+ group, most likely reflecting a normalization of brain function because it was similar to a control group of healthy adults; and (3) β frequency RSFC moderately correlated with upper-limb behavior in chronic stroke survivors, with interhemispheric connectivity greater in stroke survivors with better upper-limb ability. The novelty of this study was that we demonstrated that a regression model combining measures of descending motor pathway integrity to account for variance in upper-limb behavior was improved with the addition of interhemispheric RSFC. Furthermore, the relationship between RSFC and upper-limb behavior was influenced by descending motor pathway integrity. Specifically, stronger interhemispheric RSFC for MEP+, but not MEP−, stroke survivors was associated with greater upper-limb behavior. These findings provide insight to understand the role of interhemispheric networks in chronic stroke upper-limb behavior and how this association may be modulated by descending motor pathway integrity.
RSFC is a relatively new approach to quantify brain activity. It may be that RSFC has importance and value in stroke because it does not require participants to perform voluntary movement or cognitive tasks, such as those required for task-based functional MRI or movement-related EEG. These tasks can be particularly challenging as a result of stroke impairments. As evidence of the potential utility of resting state measures, small studies using functional MRI have reported that interhemispheric connectivity of the sensory or motor cortex is associated with motor impairment in chronic stroke31,32 and may strengthen within several months following stroke. 33 In comparison to MRI, EEG has reduced spatial resolution and suffers from volume conduction, which can lead to spurious detection of nonexistent relationships between brain regions.17,34 In an attempt to overcome this, several different analytical approaches have been developed to quantify functional connectivity based on oscillatory phase with nonzero time lag, such as dwPLI used in this study.16,17 An advantage of EEG is greater temporal resolution, allowing investigation of several oscillatory frequencies that have been shown to reflect activity of different brain regions or behaviors.35 -38 Furthermore, EEG data can be rapidly obtained in a range of locations, including research laboratories or clinical environments, with little cost in comparison to MRI, enhancing potential utility as an assessment tool.
This study demonstrated that β dwPLI of the sensorimotor cortex is moderately correlated with upper-limb behavior. Several control analyses provide confidence that this result was specific to β frequency RSFC within the a priori selected regions of interest (Figure 2) and did not appear to be driven by integrity of corpus callosum pathways or other measures of neural activity such as EEG power. In support, previous studies demonstrating both α and β RSFC are associated with upper-limb recovery and behavior.39,40 However, we observed that these correlations are primarily driven by the MEP+ stroke survivors. Although caution should be applied when interpreting this result given the relatively small sample of MEP− stroke survivors, our results advance understanding of RSFC in stroke by identifying that (1) the addition of β RSFC may be complementary to measures of CST integrity and (2) the association between upper-limb behavior and interhemispheric RSFC appears to be most relevant for stroke survivors with an intact descending motor pathway. Although outcomes of this study only provide an association between neurophysiological measures (MEP status and RSFC) and upper-limb behavior in chronic stroke, these promising results do warrant further investigation.
The current study provides some insight into understanding motor network activity in chronic stroke. Although it is unclear from the current results whether stronger RSFC is a consequence or cause of better upper-limb behavior, our results appear to suggest that both sensorimotor cortices contribute in some capacity to shape motor control of the paretic upper limb when descending motor pathway integrity is preserved. Because interhemispheric RSFC was similar between MEP+ stroke survivors and healthy adults, this pattern of connectivity may reflect a relatively normal motor control state. In support, brain stimulation and imaging studies in healthy adults have reported a cooperative interaction between hemispheres, with both the ipsilateral and contralateral sensorimotor cortex playing an active role in motor learning and motor control.41,42 Conversely, our results indicate that interhemispheric RSFC is lower in MEP− stroke survivors and does not correlate with upper-limb behavior in this subset of participants. This finding is not underpinned by microstructural properties of interhemispheric white matter pathways. Therefore, it appears that functional interhemispheric sensorimotor networks are unlikely to contribute to paretic upper-limb motor control, or alternatively, RSFC could be lower as a consequence of impaired upper-limb behavior. In either instance, it appears that a different pattern of network communication may be important for upper-limb behavior in MEP− stroke survivors. For example, earlier MRI studies have reported increased contralesional hemisphere activity in response to paretic hand function for severely affected stroke survivors,43,44 suggesting that networks within this hemisphere might shape motor output when there is greater ipsilesional white matter disruption. Furthermore, a recent model proposed that structural reserve may help stratify stroke survivors to 1 of 2 models of motor control. Where structural reserve is low, such as compromised integrity of ipsilesional descending motor pathways, there is limited ability to restore a physiologically normal pattern of motor control that includes, in-part, interhemispheric communication. 45 Our results are consistent with this model and provide further insight by demonstrating that RSFC of interhemispheric sensorimotor networks is not associated with upper-limb behavior where there is low structural reserve of the ipsilesional descending motor pathways. A novel interpretation of the current findings would be to explore whether interventions that increase β interhemispheric RSFC would improve upper-limb behavior. This approach would help decipher if a causal relationship exists between RSFC and upper-limb behavior in stroke. Innovative techniques that may be capable of achieving this include transcranial alternating current stimulation, which has been reported to entrain neural oscillations in a frequency-specific manner and change behavior,46,47 or EEG biofeedback training, which has been shown to modulate frequency-specific neural oscillations and improve motor performance in stroke. 48 Although speculative at this stage, but informed by the current findings, we would hypothesize that increasing β interhemispheric RSFC will improve upper-limb behavior in MEP+ stroke survivors but fail in MEP− stroke survivors. This remains to be tested in further experimental studies.
There are limitations to this study that should be acknowledged. First, this was a sample of convenience that recruited chronic stroke survivors from the community. This sampling strategy resulted in an uneven sample size between MEP+ and MEP− groups, with comparatively fewer MEP− stroke participants. This uneven sample may have affected our results when comparing MEP subgroups, and our findings should be viewed cautiously. However, we note that (1) our data replicate known literature that MEP+ stroke survivors exhibit greater upper-limb behavior compared with MEP− stroke survivors 4 and (2) the correlation between upper-limb behavior and RSFC clearly diverged when separated by MEP status, with MEP− stroke survivors having a much smaller effect size in the opposite direction. These observations provide some confidence that the reported results are likely to be genuine and not primarily driven by an unequal sample between groups. Although these early results provide indication that RSFC is associated with upper-limb behavior in stroke, further studies are needed to (1) investigate the role of RSFC across different phases of stroke recovery with larger samples of those categorized as MEP− and 2) evaluate whether RSFC assessed soon after stroke has any additional predictive power for recovery and response to treatment, over and above existing neurophysiological and neuroimaging predictors. Second, our inclusion criteria limited us to select those participants who were safe for TMS because we categorized participants based on MEP status. At this stage, we do not know whether RSFC is associated with behavior in participants who are not safe for TMS. Third, our selected measure of RSFC is based on phase synchronization of EEG oscillations. It is unclear whether similar relationships would be observed for measures that estimate connectivity from amplitude. Finally, we did not obtain upper-limb behavioral measures in the healthy control participants. Although these participants were free of any impairments that may affect upper-limb performance and would be expected to score well, the addition of behavioral assessments may have provided greater context to interpret the relative upper-limb performance of stroke survivors in this study.
In conclusion, the results of this study provide better understanding of the functional significance of RSFC in chronic stroke. Our findings suggest that interhemispheric network activity in stroke survivors differs based on integrity of descending motor pathways. Specifically, stronger interhemispheric RSFC was observed for MEP+ compared with MEP− stroke survivors, and correlations between upper-limb behavior and RSFC were driven by MEP+ stroke survivors. Furthermore, a regression model comprising measures of descending motor pathway integrity to account for variance in upper-limb behavior was improved with the addition of interhemispheric RSFC. The ease of obtaining RSFC measures with EEG suggest that this measure is worthy of further investigation to understand network activity in stroke and uncover potential biomarkers of stroke recovery or response to therapy.
Footnotes
Acknowledgements
The authors acknowledge Alberto Lazari for assistance provided for the MRI analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Sylvia and Charles Viertel Charitable Foundation Clinical Investigator Award (VTL2016CI009). BH is supported by a research fellowship from the National Health and Medical Research Council of Australia (1125054). MRG is supported by an NHMRC-ARC Dementia Research Development Fellowship (1102272).
