Abstract
Background:
Chronic Back Pain (CBP) may lead to a reorganization of brain function, which can be observed through the indicator of degree centrality (DC). Traditional pain research has predominantly focused on static measurements of brain function within classical frequency bands, which may not fully capture the complexities of chronic pain. This study not only employed static frequency division but also incorporated dynamic analyses to capture the evolving nature of brain activity in chronic pain conditions.
Methods:
This study included a total of 31 patients with CBP and 33 age- and gender-matched healthy controls. Spontaneous brain activity was investigated by traditional DC, DC in subfrequency bands (slow-5, slow-4) and dynamic DC (dDC). Differences in brain regions between the two groups were obtained using two-sample t-tests. The association of abnormal brain regions with pain intensity and psychological tests were analyzed in parallel.
Results:
Compared to classical frequency band, the number of brain regions with changes in DC values in the slow-5 frequency band is greater. The right angular gyrus was found in both the slow-5 frequency band and the classical frequency band, while the left putamen was only found in the slow-5 frequency band. The dDC values were changed in left hippocampus, and right putamen, which were all different from the brain regions that the static DC (sDC) value altered.
Conclusion:
This study demonstrates that incorporating frequency and dynamic analysis in addition to traditional DC metrics can better understand the functional characteristics of the brain in CBP.
Keywords
Introduction
Chronic back pain (CBP) is one of the leading causes of disability worldwide, affecting 60%–70% of the population at some point in their lives, with its prevalence increasing each year. 1 It significantly reduces the quality of life for patients and imposes a substantial burden on both individuals and society. CBP is more complex in nature compared to acute back pain. A growing body of evidence suggests that chronic pain is not only associated with molecular or structural changes in peripheral inputs and spinal circuits but also accompanies a reorganization of the brain’s morphology and function. 2 Thus, gaining insights into the brain’s functional characteristics in CBP could provide more information for the treatment, diagnosis, and prevention of this condition.
Resting-state functional magnetic resonance imaging (rs-fMRI) has been widely used in pain research by evaluating spontaneous fluctuations of blood oxygen level-dependent (BOLD) signals, which indirectly respond to neuronal activity and observe alterations in brain function. This method has the advantages of being noninvasive and highly reproducible. Several methods have been proposed and proven to be effective in characterizing the features of the brain function, such as the amplitude of low-frequency fluctuations (ALFF), regional homogeneity (ReHo), independent component analysis (ICA), and seed-based functional connectivity (FC). However, these methods often focus on localized or specific network-based connectivity. Degree centrality (DC) can compensate for this limitation, as it quantifies the overall connectivity strength of a node within the entire brain network on a global connectome scale. 3 This approach allows DC to provide a comprehensive measure of how connected each brain region is within the whole-brain network, complementing the more targeted connectivity insights provided by the other methods. DC has been used for pathophysiological exploration of CBP. One study found a significant increase in DC values in the sensorimotor network of patients with CBP compared to normal controls. 4 Another study on DC similarly found that patients with CBP tend to have unstable and inefficient brain networks. 5 These findings indicate that DC can examine the connectivity changes at the voxel level within the whole-brain functional network of CBP patients, providing a new window into understanding the brain physiology associated with chronic pain.
Most studies on brain function in patients with CBP have focused on the classical frequency range of rs-fMRI. However, studies have indicated that different frequency bands are associated with distinct neural oscillation functions, and rs-fMRI signals exhibit varying characteristics and sensitivities to specific brain activity patterns across different frequency ranges.6–8 A regional coherence study on tension headache found that abnormalities in spontaneous neural activity in patients with tension headache were frequency-specific and contained more information in the slow-5 band compared to the slow-4 band. 9 The results of another ALFF study on trigeminal neuralgia (TN) similarly reflect this phenomenon. 10 The slow-5 frequency band may be more sensitive to the detection of abnormalities in brain function related to pain. However, there are no studies exploring in depth whether CBP patients also exhibit frequency dependence in global brain function.
Additionally, although rs-fMRI scans are conducted while subjects are at rest, the spontaneous neuronal activity of the brain is dynamic, exhibiting time-varying activities. 11 This dynamic nature reflects the continuous fluctuations in neural interactions and connectivity. For this reason, we used dynamic DC (dDC), a method that combines static DC (sDC) with a sliding window to reflect the temporal dynamics of remote functional connectivity. 12 dDC has been applied to the study of major depressive disorder (MDD), 13 schizophrenia, 14 and depressive mania, 15 among other mental illnesses. In the field of pain, dDC has been utilized in the research of brain functions in TN. 16 This study has revealed that following the activation of pain, there are alterations in both the sDC and dDC values across various brain regions in patients with this condition. These changes are found to be mutually reinforcing and complementary. However, the application of dDC in CBP has not been documented in the literature.
Therefore, here we studied the frequency-dependent and time-dependent changes of global brain functional properties in patients with CBP. In this study, we measured traditional DC, DC in subfrequency bands (slow-5, slow-4) and dDC to observe the differences between the CBP and HC groups, and explored the relationship between patients’ DC variants and their pain intensity and psychological assessments. This is the first study to apply the combination of static multi-frequency and dynamic DC to assess changes in functional connectivity in CBP patients. Our hypotheses were: (1) DC values in CBP patients are significantly abnormal relative to HC, and these changes may be related to disease severity; (2) DC in the slow-5 band is more sensitive to changes in functional connectivity in CBP patients compared to the slow-4 band and classical frequency ranges; (3) The brain region results from dDC differ from those of sDC, with dDC offering a beneficial supplement to sDC.
Material and methods
Participants
Data for this retrospective study were obtained from the Open Pain Project (OPP) database (https://www.openpain.org). The original data collection was conducted by investigators at Northwestern University. As documented in the database, the original study protocol was approved by the Northwestern University Institutional Review Board (IRB) and carried out in accordance with the Declaration of Helsinki. All participants provided written informed consent prior to inclusion. The final sample included 34 patients with CBP and 34 age- and sex-matched healthy controls (HC), all of whom were right-handed. According to the original OPP protocol, participants underwent a comprehensive clinical assessment and MRI scanning; questionnaires including the Visual Analogue Scale (VAS) for pain and the Beck Depression Inventory (BDI) were administered approximately 1 h before scanning. Inclusion criteria for CBP patients included a diagnosis by a board-certified physician, average pain intensity ≥ 40/100, and symptom duration ≥ 16 weeks. Exclusion criteria included other chronic pain conditions, systemic diseases, or severe depression (BDI > 19). HC were required to be pain-free for at least 52 weeks. General exclusion criteria for all subjects included age outside the 18–75 range, history of neurological or psychiatric disorders, substance abuse, and MRI contraindications.
MRI data acquisition and image preprocessing
MRI data were acquired on a 3-Tesla Siemens Trio wholebody scanner. Rs-fMRI data preprocessing was conducted using the Data Processing and Analysis of Brain Imaging (DPABI6.1) and Statistical Parametric Mapping 12 (SPM12) toolbox based on the MATLAB platform (MathWorks, Natick, MA, USA). The preprocessing procedure is similar to our previous study. 16 For details on the imaging data acquisition and preprocessing pipeline, please refer to Supplemental Methods S1. Three patients and one HC were excluded due to large head motion (>2.5 mm maximum displacement, 2.5° rotation or framewise displacement (FD) exceeded 0.2 throughout the scanning), and the remaining 31 patients with CBP and 33 HCs were subjected to further analysis.
Traditional DC calculation
After preprocessing, band-pass filtering was applied in classical frequency band (0.01–0.08 Hz). For the weighted graph, DC is defined as the sum of the weights from the edges connected to the node. Compared to the binary version of DC, weighted DC provides a more accurate representation of the centrality of functional brain networks.
17
Pearson’s correlation of time series between every voxel and other voxels in the whole brain gray matter calculated the correlation matrix
DC in subfrequency bands (slow-5, slow-4) calculation
Distinct from the traditional DC approach, the computation of DC within subfrequency bands (slow-5 and slow-4) necessitates the application of band-pass filtering post-preprocessing, targeting the specific frequency ranges of slow-5 (0.01–0.027 Hz) and slow-4 (0.027–0.073 Hz). The method of calculating the metric is the same as the traditional DC calculation. In order to maintain consistency with traditional DC, we used a Gaussian kernel with half height and the full width of 6 mm for spatial smoothing.
DDC calculation
The dynamic metrics was calculated using a sliding window method. The most important parameter in resting-state dynamic computation is window length. A short window length may increase the risk of introducing spurious fluctuations in the observed dDC, and a long window length may hinder the characterization of the temporal variability dynamics of the dDC. 18 In line with our previous study, 19 we used a 50 TR (100 s) sliding window length and a 2 TR (4 s) step size. Based on the method similar to sDC, after calculating the DC of all voxels in the time window, each participant will obtain multiple window-based DC graphs. Then, we calculated each participant’s standard deviation per voxel in all window-based DC plots to measure the dynamic changes in DC. In order to maintain consistency with sDC, we used a Gaussian kernel with half height and the full width of 6 mm for spatial smoothing. To ensure the reliability of the results, dDC results were further validated using dynamic methods with sliding window lengths of 50 TRs (100s) and moving steps of 5 TRs (10s), as well as sliding window lengths of 100 TRs (200s) with a moving step of 2 TRs (4s) (Supplemental Figures S1, S2 and Tables S1, S2).
Statistical analysis
In DPABI software, an independent two-sample t-test was used to compare traditional DC, DC in subfrequency bands and dDC values between CBP patients and HCs within the gray matter mask, to observe inter-group differences. We regressed out three covariates: age, sex, and head motion. The resulting f-maps were corrected for multiple comparisons using Gaussian random field theory (GRF), with a voxel-level threshold of p < 0.001 and a cluster-level threshold of p < 0.05. Spearman correlation analysis was performed between clinical characteristics (VAS, BDI) and the values of traditional DC, DC in subfrequency bands and dDC.
Results
Demographic information and clinical characteristics
A total of 31 CBP patients and 33 HCs were ultimately included in the data analysis. It is noteworthy that there were no significant differences between the CBP group and the HC group in terms of age (p > 0.05) and gender (p > 0.05; Table 1), however, there was a difference in the BDI scores between the two groups (p < 0.05; Table 1), indicating CBP patients suffered from more negative mood and depression.
Demographics and clinical characteristics of the participants.

BDI: beck depression inventory; CBP: chronic back pain; HC: healthy controls; VAS: visual analogue scale.
p-values for sex distribution obtained by the chi-square test.
p-value obtained by analysis of variance.
Group differences in traditional DC
Prior to examining group differences, we first validated the spatial distribution of the weighted DC maps. As illustrated in Supplemental Figure S3, the group-level mean maps for both the CBP and HC groups exhibited typical spatial patterns, with primary hubs predominantly located within the canonical default mode network (DMN). This spatial consistency confirms the reliability of our DC calculation. Subsequently, a significant inter-group difference was noted in the right angular gyrus (Angular_R), characterized by decreased DC values in the CBP group compared to the HC group (voxel p < 0.001, cluster p < 0.05, GRF corrected, cluster size > 53 voxels; Table 2, Figure 1).
Brain areas changed by traditional DC across the two groups.

Angular_R: right Angular gyrus; MNI: Montreal Neurological Institute.

Brain regions with significant alterations of traditional DC between CBP and HC.
Group differences in DC in subfrequency bands (slow-5, slow-4)
Within the slow-5 frequency band, significant differences were noted between groups in the left putamen (Putamen_L) and the right angular gyrus (Angular_R), with decreased DC values in the CBP group compared to the HC group (Table 3, Figure 2).
Brain areas changed by DC across the two groups in subfrequency bands.

Angular_R: right Angular gyrus; MNI: Montreal Neurological Institute; Putamen_L: left Putamen.
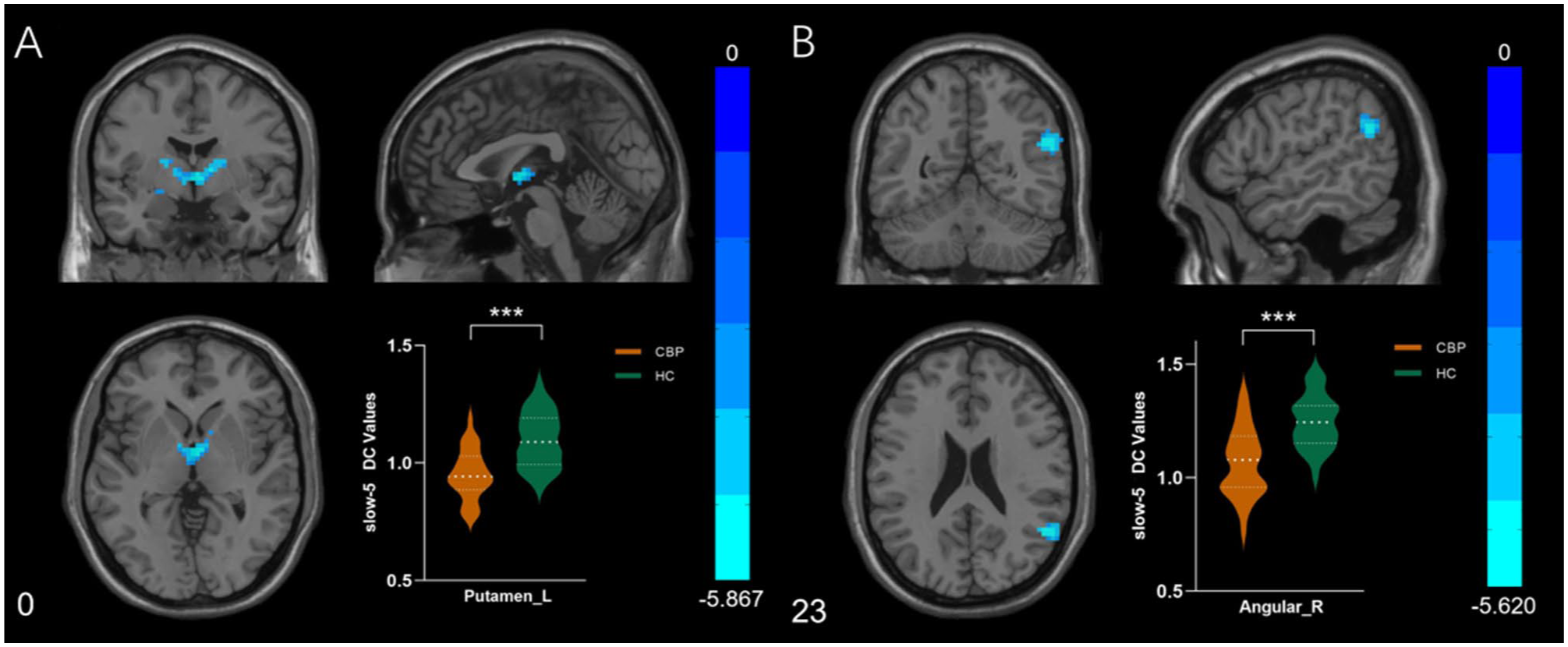
Brain regions with significant alterations of DC between CBP and HC in slow-5 frequency band. (a) Significant decrease localized in the left Putamen. (b) Significant decrease localized in the right Angular gyrus.
In the slow-4 frequency band, no significant inter-group differences in brain regions were observed.
Group differences in dDC
Notable inter-group differences were identified in the left hippocampus (Hippocampus_L) and the right pallidum (Pallidum_R), with increased dDC values in the CBP group compared to the HC group (Table 4, Figure 3).
Brain areas changed by dDC across the two groups.

Hippocampus_L: left Hippocampus; MNI: Montreal Neurological Institute; Pallidum_R: right Pallidum.
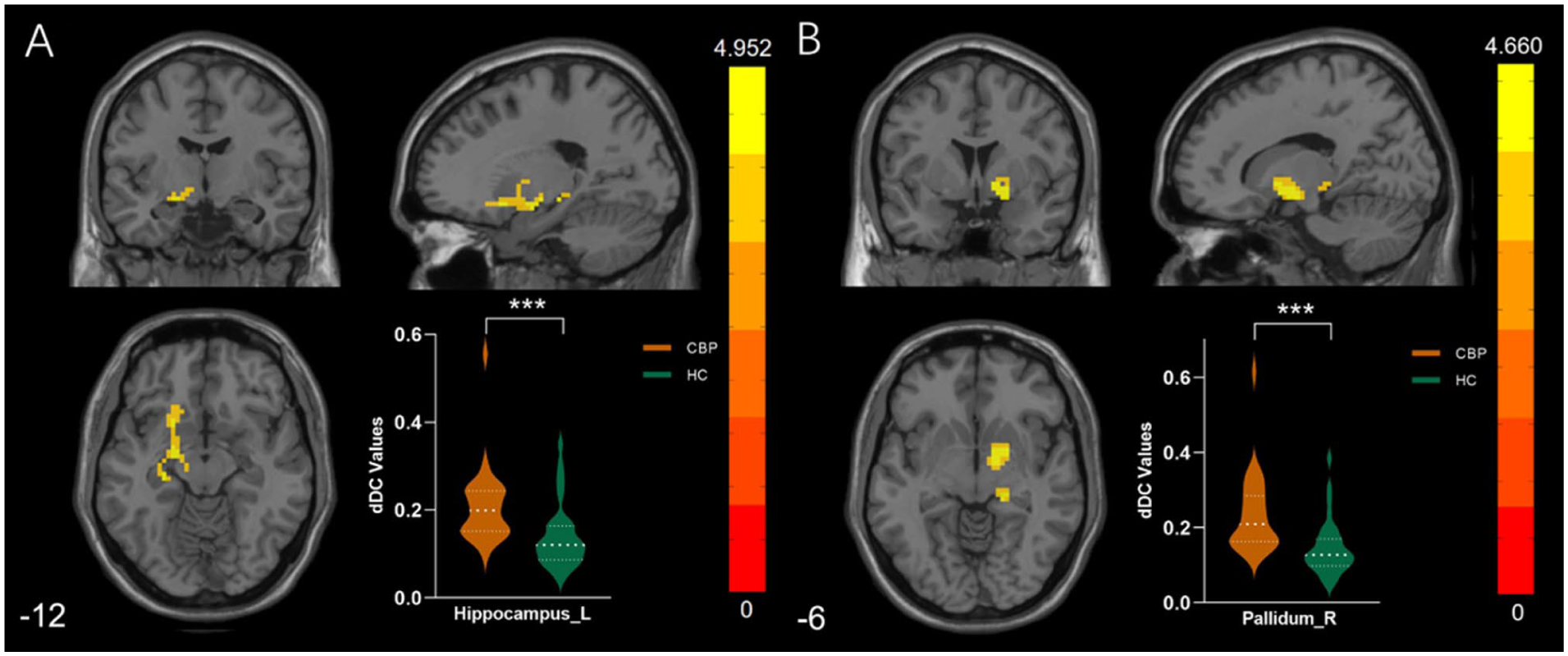
Brain regions with significant alterations of dDC between CBP and HC. (a) Significant increase of temporal instability localized in the left Hippocampus. (b) Significant increase of temporal instability localized in the right Pallidum.
Correlation between the demographics and indicators of altered brain region
The dDC values in the right pallidum demonstrated a positive correlation with both VAS and BDI scores (Table 5, Figure 4).
Correlation between dDC value and neuropsychological test in patients.

BDI: beck depression inventory; Pallidum_R: right Pallidum; VAS: visual analogue scale.

Correlation between dDC value and neuropsychological test in patients.
Discussion
CBP represents a prevalent and critical issue across clinical, social, economic, and public health domains, being one of the most common chronic pain conditions globally. 20 However, there is a lack of knowledge about how the brain adapts to this disease and how this brain adaptation is related to the intensity of pain and negative emotions. To elucidate this relationship, we constructed brain connectome networks, measured their DC, and investigated their interactions with questionnaire results related to pain and depression. DC characterizes voxel centrality by assessing various connections between a given voxel and other voxels at the level of the whole brain, avoiding the subjectivity of predefining regions of interest, thus ensuring high sensitivity, specificity, and retesting reliability. 21 Unlike seed-based FC and ICA, the two most common indicators of functional connectivity, DC considers a given region’s relationship to the entire functional connectome, not just its relationship to individual regions or isolated larger components, and is able to capture the complexity of the functional connectome as a whole. Our aim is to determine how the brain’s functional connectivity network reorganizes under conditions of CBP.
To the best of our knowledge, this study is the first to assess DC alterations in CBP patients under a combination of static conditions across multiple frequency bands and dynamic. This approach addresses the limitations of most studies in this field, where previous research typically focused only on classical frequency band and static. When comparing CBP patients with controls, we identified significant differences in traditional DC, DC in subfrequency bands and dDC in various brain regions, including the right angular gyrus, left putamen, left hippocampus, and right pallidum. In addition, dDC changes in the right pallidum were found to be strongly correlated with pain intensity and psychological assessments in the CBP group. Most notably, the inter-group differences in the slow-5 frequency band are more pronounced than those in the classical and slow-4 frequency bands. Additionally, we observed that dDC can provide more differential brain region results compared to sDC. These findings suggest that patients with CBP exhibit abnormal changes in whole-brain functional connectivity during intrinsic brain activity, which are influenced by specific frequency bands and exhibit time-dependence.
In this study, diminished DC values were noted in the right angular gyrus of patients with CBP relative to the HC group, suggesting a decreased level of direct brain connectivity for this region. The angular gyrus, located at the junction of the temporal, parietal, and occipital lobes, receives visual input from the occipital lobe, auditory input from the temporal lobe, and somatosensory input from the parietal lobe, and is involved in higher brain functions such as language and reading comprehension, mathematical cognition, spatial cognition, social cognition, and memory retrieval. 22 More importantly, it plays a crucial role in the DMN. Prior research has indicated that CBP can lead to the disruption of the DMN. There is a prevailing view that chronic pain may induce neuroplastic changes in the brain, which in turn could affect the stability of the DMN. 23 Such alterations in DMN activity are linked to a range of symptoms commonly observed in CBP patients, including depression, anxiety, sleep disorders, and decision-making anomalies. Our findings reveal a decrease in direct connectivity in the right angular gyrus among CBP patients when compared to the HC group, which lends further support to this perspective. Additionally, multiple studies on chronic pain have discovered changes in the connectivity of the angular gyrus, with Lo Buono et al. 24 observing increased functional connectivity in the left angular gyrus of patients with migraine with aura compared to patients with migraine without aura. Gupta et al. 25 found that females with urogenital chronic pelvic pain syndrome (UCPPS) showed greater centrality in the left angular gyrus compared to HC females, and that females with UCPPS, compared to men with UCPPS, showed less centrality in the left angular gyrus. These results are not entirely consistent with ours, and we speculate that this may be related to different stages of chronic pain, or it could be that chronic pain from different diseases has its own specific brain activity patterns. It can be affirmed that under conditions of chronic pain, the connectivity of the angular gyrus is highly susceptible to change, and further exploration into how this region deals with pain should be pursued in the future.
Within the slow-5 frequency band, a reduction in DC values was observed in both the right angular gyrus and the left putamen of CBP patients in contrast to the HC group. The putamen is a major component of the basal ganglia and an important site of brain plasticity. The putamen is primarily involved in motor control but also plays an important role in the regulation of pain sensation, analgesic responses, and pain signaling. 26 This is because the putamen is a key node in the thalamocortical-basal ganglia circuit, which is the main circuit affected by pain, and that the basal ganglia not only receive pain signals from direct and indirect pain pathways, but also receive input signals through the cortical and subcortical areas that make up the basal ganglia-thalamo-cortical circuits. 27 A previous study has expanded on this premise and found that in a chronic pain condition known as fibromyalgia, the connectivity of the right nucleus accumbens (NAcc) with mesolimbic circuit regions (including the putamen, thalamus, and ventral pallidum) is reduced. 28 All of these brain areas, including the putamen, play a vital role in motivation and reward processing. Additionally, a study of complex regional pain syndrome (CRPS) observed a significant reduction in GM volume in the bilateral putamen in patients with CRPS compared to the normal population. 29 Another study on tension headache (TTH) found significantly lower ReHo values in the bilateral putamen in the TTH group compared to healthy controls. 9 These studies concluded that functional abnormalities in the putamen are associated with pain processing. Our results similarly support this notion, showing decreased functional connectivity of the putamen in patients with CBP. However, patients with CBP present with both pain and some motor deficits. We believe that functional abnormalities of the putamen may be related to both, and a more in-depth comparison between motor and sensory tasks in subsequent studies may help clarify whether altered functional connectivity of the putamen is caused by motor-related abnormalities or pain-related processing.
In the aforementioned results, a decrease in the DC of the right angular gyrus can be observed in both the classical frequency band and the slow-5 frequency band. In contrast, changes in the DC of the left putamen are only detected in the slow-5 frequency band. The brain’s oscillations encompass a broad range of frequency bands, with low-frequency oscillations (LFO) divided into slow-5 (0.01–0.027 Hz), slow-4 (0.027–0.073 Hz), slow-3 (0.073–0.198 Hz), and slow-2 (0.198 Hz–0.25 Hz). 7 These bands are generated by distinct oscillators with specific properties and physiological functions. The slow-2 band is associated with white matter signal, while the slow-3 band is related to physiological noise. Both the slow-4 band, which reflects activity in the basal ganglia, and the slow-5 band, which reflects cortical neuronal activity, fall within the classical frequency range (0.01–0.08 Hz) that is used to represent spontaneous brain activity. Consequently, the majority of brain function studies are conducted within this classical frequency range. However, an increasing number of studies have found that intrinsic activity varies across different frequency bands, and analysis that removes interfering signals from other bands can more specifically reveal the spontaneous activity of particular sub-bands. Subfrequency band analysis may hold significant physiological importance in understanding the pathogenesis of chronic pain. This is well demonstrated in our study, where the slow-5 frequency band showed the most results in brain regions and included the classical frequency band. This suggests that different brain regions show overlapping and significant variations in DC in different frequency bands and complement each other to some extent. The right angular gyrus, which overlaps in the classical and slow-5 frequency bands, may play a more stable and important role in the brain’s adaptation to CBP and should be analyzed with emphasis in future studies. In our study, the slow-5 band contains more information. This is consistent with other previous pain studies9,10 where brain function exhibits specific frequencies and patterns of involvement, and the slow-5 band may be instrumental in better exploring how the functional connectivity network of the brain in patients with CBP is reorganized.
The brain is a highly dynamic system characterized by fluctuating temporal and spatial functional patterns. 30 dDC reflects the strength of functional connectivity of a voxel with all other voxels throughout the brain over time, providing additional complementary information for research. 31 In the present study, two brain regions with significant changes in dDC values were identified: the left hippocampus and the right pallidum, which were different from the brain regions with changes in sDC values. The dDC of the two brain regions increased in the CBP patients compared to the HC group, suggesting that the left hippocampus and the right pallidum have an elevated status and role in the whole-brain network. The hippocampus is a crucial region for memory formation and learning, particularly associated with the formation of long-term explicit memory, and it also plays a role in processing pain stimuli and nociception. 32 A Study has indicated that chronic pain can inhibit noradrenergic neurotransmission in the hippocampus by locally producing the inflammatory cytokine TNFα, thereby affecting hippocampal function and leading to an increase in anxiety, depression, and learning and memory deficits in patients with chronic pain. 33 Another study has found that mice with spared nerve injury (SNI) exhibit a decrease in the expression and phosphorylation of extracellular signal-regulated kinase in the hippocampus, reduced neurogenesis, and alterations in short-term synaptic plasticity. 34 They also further measured the volume of the hippocampus in humans with CBP, complex regional pain syndrome (CRPS), and osteoarthritis (OA), discovering a significant reduction in the volume of the bilateral hippocampus in CBP and CRPS patients, successfully linking the hippocampal abnormalities in mice to chronic pain in humans. However, our research reported an increase in the dDC value in the left hippocampus of CBP patients, showing a contrary result. There are also numerous studies that yield results similar to ours. Cobos et al. 35 found that the right hippocampus of patients with chronic headache had higher overall efficiency, shorter path lengths, and higher clustering coefficients compared to the healthy population, all of which were positively correlated with headache frequency, while Liu et al. 36 observed elevated ReHo values in hippocampal regions in primary dysmenorrheal patients with chronic menstrual pain. A study has provided an explanation for the contradictory results observed among studies: they found that patients with chronic pain initially had more extensive hippocampal connectivity compared to healthy controls, but that functional connectivity between the patient’s hippocampus and the medial prefrontal cortex declined dramatically as pain persisted. 37 Thus, the increase in hippocampal functional connectivity we observed may be temporary, eventually leading to decreased functional connectivity and volume atrophy of the hippocampus as the disease progresses.
Like the putamen, the pallidum is a major component of the basal ganglia and a major node of the cortex-basal ganglia-cortex circuit. Interestingly, in our study, unlike the left putamen where sDC was diminished, the right pallidum had increased dDC values and was positively correlated with pain intensity. It has been suggested that the larger DC in the pallidum may be an adaptive response to prolonged pain episodes and that prolonged painful stimuli may lead to an increased need for pain modulation manifested by relatively high DC values in the pallidum. 38 Another mouse study 39 suggests that this may be related to the cholinergic circuitry originating from the ventral pallidum. In a mouse model of persistent pain, the ventral pallidum is activated by pain-like stimuli and becomes abnormally active during the period of ongoing pain. This activation leads to the comorbid pathophysiology of hyperalgesia, anxiety, and depression-like behaviors through the cholinergic circuitry. Our findings can support this theory. In our study, the BDI scores of CBP patients were significantly different from those of the healthy population, and the dDC values of the right pallidum of CBP patients were positively correlated with the BDI scores, which implies that functional alterations in this brain region are associated with depressive symptoms in addition to pain intensity. Depression and chronic pain are highly co-morbid, and the pallidum is thought to be associated with pain and depression. It is very likely that chronic pain leads to depressive-like behaviors in patients with chronic pain by affecting the connectivity efficiency of the pallidum, and this hypothesis needs to be further confirmed in subsequent research. Targeting the pallidum and its derived ventral pallidum cholinergic circuitry may be a promising therapeutic strategy for treating the comorbidity of chronic pain and depression.
The absence of significant dDC alterations in canonical pain-sensory (e.g. insula, ACC) or emotion-related regions (e.g. amygdala) warrants consideration. First and foremost, these findings align remarkably well with the pain chronification model described by Hashmi et al. 40 This model postulates a functional shift of pain representation from nociceptive to affective-reward circuitry as pain persists. Our results support this framework, suggesting that in the chronic stage of CBP, pathological dynamism is anchored primarily within these emotional and mnemonic networks rather than persisting in primary sensory-processing regions. Secondly, from a methodological perspective, the absence of significant findings in sensory cortices and the amygdala may be attributed to the strict statistical thresholds we employed (GRF correction: voxel p < 0.001, cluster p < 0.05). As noted by Hashmi et al., 40 the ability to capture the full extent of pain-related brain activity can depend heavily on statistical power and detection thresholds. We adopted this conservative approach to rigorously control for false positives, adhering to current standards for cluster-level inference. 41 However, as demonstrated by recent large-scale methodological studies, stringent multiple comparison corrections can inevitably mask meaningful brain regions with subtler effect sizes. 42 Therefore, while ensuring high specificity, our strict criteria might have increased the risk of Type II errors, potentially masking subtler dynamic variability within the sensory and emotional matrix.
A core finding of this study is that different analytical indices identify abnormal brain regions with marked spatial specificity. Rather than reflecting inconsistency, these differences robustly demonstrate the complementary value of multi-level fMRI analyses, as each index captures distinct dimensions of brain functional reorganization. Specifically, traditional DC focuses on quantifying cumulative connectivity strength at the whole-brain level, 3 thereby revealing macro-scale functional impairment of the right angular gyrus as a critical information-integration hub. In contrast, frequency-specific analyses further dissociate different frequency-dependent physiological processes 7 : abnormalities in the putamen were detected only within the slow-5 frequency band and were masked in traditional frequency analyses, highlighting the heightened sensitivity of this approach in uncovering pathology-specific alterations in subcortical nuclei. Along the temporal dimension, dDC sensitively captures transient fluctuations and instability of functional connections 11 ; anomalies in the left hippocampus and right pallidum emerged exclusively through dDC, suggesting that the pathophysiology of these limbic-reward nodes is driven not by changes in average connectivity strength, but by dynamic maladaptation. In summary, by integrating traditional DC, DC in subfrequency bands, and dDC, we reconstructed a comprehensive spatiotemporal signature of functional reorganization in CBP. Relying on any single metric alone would have failed to fully resolve the complex pathophysiological mechanisms underlying this disorder.
Although the clinical diagnosis of CBP mainly relies on the patient’s self-report and the process is relatively straightforward, it is still difficult to fully understand the heterogeneity of its clinical manifestations. The three indicators integrated in this study construct a complementary evaluation framework, which provides a basis for clinical classification. First, traditional DC can evaluate the integrity of the DMN network. This study observed that the connectivity strength of the DMN core area (the right angular gyrus) was significantly reduced. This is consistent with the view in the field of chronic pain: DMN dysfunction is a key mechanism in pain chronification, reflecting the reorganization of the brain’s intrinsic functional network by pain. 23 Therefore, traditional DC can be used as an objective indicator to assess the degree of brain network functional damage caused by pain. Second, sub-frequency analysis reveals subcortical abnormalities that are difficult to detect in traditional full-band analysis. This study found that the functional abnormality of the putamen was specifically manifested in the slow-5 frequency band, while it was masked in traditional broadband analysis. This frequency-specific phenomenon is highly consistent with the research results in the field of Parkinson’s disease (PD): Wang et al. 43 clearly pointed out in a sub-frequency study of PD patients that the functional abnormalities of the striatum (including the caudate nucleus and putamen) were particularly significant in the slow-5 frequency band and more specific than in the slow-4 frequency band. This indicates that the slow-5 frequency band has unique sensitivity in capturing hidden subcortical pathology involving the striatal circuit and can effectively reveal the potential neural mechanisms related to motor control disorders in CBP patients. Finally, dDC, as a dynamic indicator, reveals the link between brain functional instability and patients’ depressive mood. This finding is supported by the latest research on MDD: Sun et al. 44 pointed out that the dDC variability in areas such as the hippocampus of MDD patients is increased, and this network instability is related to clinical symptoms. Consistent with this, the abnormal fluctuations in dDC in the right pallidum in this study were positively correlated with BDI scores, suggesting that the dynamic instability of the pallidum as a node in the reward circuit may be the neural basis for depressive symptoms associated with CBP. Clinically, monitoring dDC fluctuations in this area may help identify patient subtypes with a high risk of depression. In summary, this multi-dimensional analysis strategy not only overcomes the limitations of single indicators but also provides an actionable imaging pathway for achieving individualized symptom assessment and precision diagnosis and treatment of CBP.
This study has several limitations, including the potential influence of participants’ varying educational levels on brain function outcomes, which warrants consideration as a covariate in regression analyses. Additionally, patients with CBP may experience symptoms such as sleep disturbances and anxiety, in addition to suffering from pain and depressive mood. However, the open pain database lacks general information on the level of education and does not include scales such as the Pittsburgh Sleep Quality Index and the Self-Rating Anxiety Scale. We will collect more comprehensive general data and clinical information in our subsequent research and include more scales. Second, our study was cross-sectional, and alterations in functional connectivity may vary at different stages of CBP, leading to incomplete matching of results from numerous previous studies. Therefore, the next step should include longitudinal investigations to examine changes in functional connectivity during disease progression. Thirdly, CBP can be divided into various types based on the specific causes, such as lumbar disk herniation and nonspecific low back pain. In the future, we plan to expand the sample size and conduct subgroup studies targeting the different etiologies of CBP. Finally, this study did not combine functional and structural indicators to further analyze and explore the correlation between them. Future studies should explore the relationship between functional and structural changes in the brain.
Conclusion
This study demonstrated that the use of DC metrics provides a better understanding of the functional abnormalities of underlying brain networks in CBP patients. The observed frequency dependency and temporal variability complement traditional metrics, enriching our insights into the brain’s functional dynamics in CBP. These findings offer valuable insights into the neural mechanisms underlying CBP.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251412603 – Supplemental material for Exploring frequency-dependent and dynamic changes in brain connectivity of chronic back pain patients using degree centrality analysis
Supplemental material, sj-docx-1-mpx-10.1177_17448069251412603 for Exploring frequency-dependent and dynamic changes in brain connectivity of chronic back pain patients using degree centrality analysis by Hanjun Hu, Luoyu Wang, Jiayi Deng, Yi Lin, Xue Tang, Xiuhong Ge and Zhongxiang Ding in Molecular Pain
Footnotes
Acknowledgements
We express our sincere gratitude to the OPP, along with its principal investigators and study participants, for generously sharing the clinical and neuroimaging data. Their contribution to this high-quality open-source dataset made this retrospective study possible.
Author contributions
HJH: Formal analysis, Investigation, Methodology, Writing – original draff, Writing – review & editing. LYW: Methodology, Validation, Writing – original draff, Writing – review & editing. JYD: Methodology, Software, Writing – original draff. YL: Methodology, Validation, Writing – original draff. XT: Software, Visualization, Writing – original draff. XHG: Methodology, Writing – original draff. ZXD: Conceptualization, Validation, Writing – review & editing. All authors reviewed the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Ningbo Clinical Research Center for Medical Imaging (No. 2021L003), the Ningbo Key Laboratory of Digital Imaging and Medical-Engineering Interdisciplinarity (No. 2024031), and the Project of National Key Clinical Specialty (Department of Medical Imaging, No. 2024017).
Ethics approval and consent to participate
The studies involving human participants were reviewed and approved by the Institutional Review Board of Northwestern University and conducted in accordance with the principles set forth in the Declaration of Helsinki. All patients/participants provided written informed consent to take part in this study. Written informed consent was also obtained from the individuals concerned for the publication of any potentially identifiable images or data included in this article.
Declaration of AI and AI-assisted technologies in the writing process
I hereby declare that this work was completed independently by myself, and no artificial intelligence (AI) or AI-assisted technologies were used in the writing process.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
