Abstract
Objective
Traditional autologous chondrocyte implantation (ACI) involves arthroscopically harvesting a cartilage biopsy (stage 1), followed by arthrotomy 3 to 4 weeks later to apply a periosteal patch and implant culture-expanded chondrocytes underneath (stage 2). This study aimed to determine if patch application during stage 1 rather than stage 2 improved clinical outcome.
Design
A randomized controlled trial was conducted from 1998 to 2001. Patients were randomized to receive either traditional ACI (control/late) or ACI with “early” patch during stage 1 (intervention/early). Clinical outcome (Lysholm score) was assessed pre-operatively and annually post-operatively.
Results
Seventy-seven patients were recruited, with 40 patients randomized to the early and 37 to the late patch group. The overall mean pre-operative Lysholm score was 51.8 (range 11-89) and significantly improved by 11.1 points (95% confidence interval [CI] = 4.8 to 17.4) at mean 12.7 years (range 1.5-23.7) follow-up. Latest mean Lysholm scores for the early and late groups were 68.4 (95% CI = 19 to 100) versus 56.7 (95% CI = 18 to 98). Adjusted for covariate imbalances, no evidence was found for a difference between the groups (mean difference = 8.5, 95% CI = −5.2 to 22.2, P = 0.22). Twenty-year survival until any re-operation or arthroplasty was 59.6%/82.1% for the early and 56.8%/69.5% for the late group, with no evidence for a difference.
Conclusion
ACI is an effective durable treatment for cartilage defects, with high levels of patient satisfaction and low failure rates. No evidence was found that applying the periosteal patch at the time of chondrocyte harvest improved long-term Lysholm scores or survival until any re-operation or arthroplasty.
Keywords
Introduction
Articular cartilage (AC) is a smooth tissue that covers the articular surfaces of bones and is produced by chondrocytes. 1 Chondral and osteochondral defects can arise as part of musculoskeletal trauma.2,3 Unfortunately, AC has a limited ability for repair, and these defects can lead to permanent damage to the joint surface, pain, and functional disability. The treatment of these defects is challenging, especially in younger individuals where the use of a partial or total knee replacement is not advisable.1-3 A multitude of options has been developed to treat focal chondral or osteochondral defects. These include options of arthroscopic shaping such as debridement 4 and chondroplasty, 5 repair options such as drilling 6 and microfracture,7-9 and restoration options such as osteochondral allografts,10,11 mosaicplasty,7,12-15 and autologous chondrocyte implantation (ACI). 3 The latest available evidence favors ACI over other forms of treatment for appropriate sized lesions due to its superior long-term outcomes and cost-effectiveness.1-3,16 The current guidance in the United Kingdom is particularly specific, advising ACI for use in patients with no previous AC defect surgery, minimal osteoarthritic damage to the knee, a defect over 2 cm2, and the procedure to be performed at a tertiary referral center. 16 Our Good Manufacturing Practice (GMP) cell laboratory was established in 1997, offering ACI treatment for chondral and osteochondral defects.17,18 As part of our routine follow-up, a self-administered modified Lysholm score was gathered yearly. 19 This allowed us to report our mid- to long-term outcomes for ACI of the knee, with 81% showing improvement and a prediction of a sustainable and successful outcome at 15 months post-operation. 3
ACI was first reported in 1994 as a form of cartilage repair treatment for chondral defects 20 and involves a 2-stage approach. The first stage requires a small full-thickness cartilage biopsy to be harvested arthroscopically, usually from a low-weightbearing area of the trochlea. In a GMP licensed laboratory, chondrocytes are isolated from the biopsy by mechanical and enzymatic treatment.21,22 The cells are then culture-expanded over 3 to 4 weeks to produce (generally) several million chondrocytes. The second stage, approximately 3 to 4 weeks later, involves a small knee arthrotomy through a medial or lateral parapatellar approach. A periosteal patch is sutured to the surrounding cartilage, and the culture-expanded autologous chondrocytes are implanted underneath.1,3 Regardless of its name, the traditional procedure has gradually evolved: for instance, the chondrocytes are now almost always applied onto a porcine collagen scaffold in vitro or intra-operatively and anchored to cover the defect using only fibrin glue.1,2
One aspect of traditional ACI that has not changed is the timing of the patch application, which always happens at the second stage. However, applying the patch at the first stage could have advantages. The patch would have 3 weeks to integrate with the surrounding cartilage, which could provide a better seal. If it had partially loosened at the second stage, the attachment could be restored and the whole construct would likely be stronger than when initially applied. A randomized controlled trial was therefore undertaken by the late Professor James Richardson to determine the clinical outcomes of ACI in the knee following either early (first stage) or late (second stage) periosteal patch application. This study aims to report the long-term outcomes of that trial.
Patients and Methods
This was a pragmatic single-center randomized controlled trial with approval from the Shropshire Health Authority District Medical Ethics Committee in January 1998 (98/5/RJ). Using a closed sealed envelope system, consenting patients were randomized to having either a traditional ACI procedure (with periosteal patch attachment at stage 2, i.e., control or late patch) or an early attachment (i.e., intervention). Consenting and randomization were done by a member of staff who did not know the patient and was not involved in the treatment. The traditional ACI (late group) had a first stage arthroscopic biopsy and a second stage mini arthrotomy with implantation of the periosteal patch and cells. The early group had a first stage mini arthrotomy and biopsy with implantation of the periosteal patch and a second stage wet arthroscopy during which the chondrocytes were injected under the patch. In both groups, the periosteal patch was obtained from the proximal medial tibia, cut to the size of the chondral defect. The patch was then sutured around the edge with 6-0 Vicryl (Ethicon, Leeds, UK) and reinforced with fibrin glue (Tisseel, Immuno AG, Vienna, Austria). In the early group, fibrin glue was also applied at the injection site during stage 2, immediately after injecting the cells. Patients were consented for the trial and followed up yearly. Due to the nature of the intervention, blinding was not possible for either the patient or treating surgeon.
All patients were offered a knee arthroscopy and biopsy at 1 year post-operatively. Biopsies of the repair tissue formed at the site of the treated defect were obtained using a juvenile Jamshedi needle and snap-frozen in liquid nitrogen-cooled hexane. Cryosections with a thickness of 7 µm were mounted onto polysine-coated microscope slides and stained with hematoxylin and eosin (H&E). General tissue morphology and the surface architecture were assessed via bright light microscopy and collagen fiber orientation (indicative of cartilage morphology such as hyaline or fibrocartilage) via polarized light (
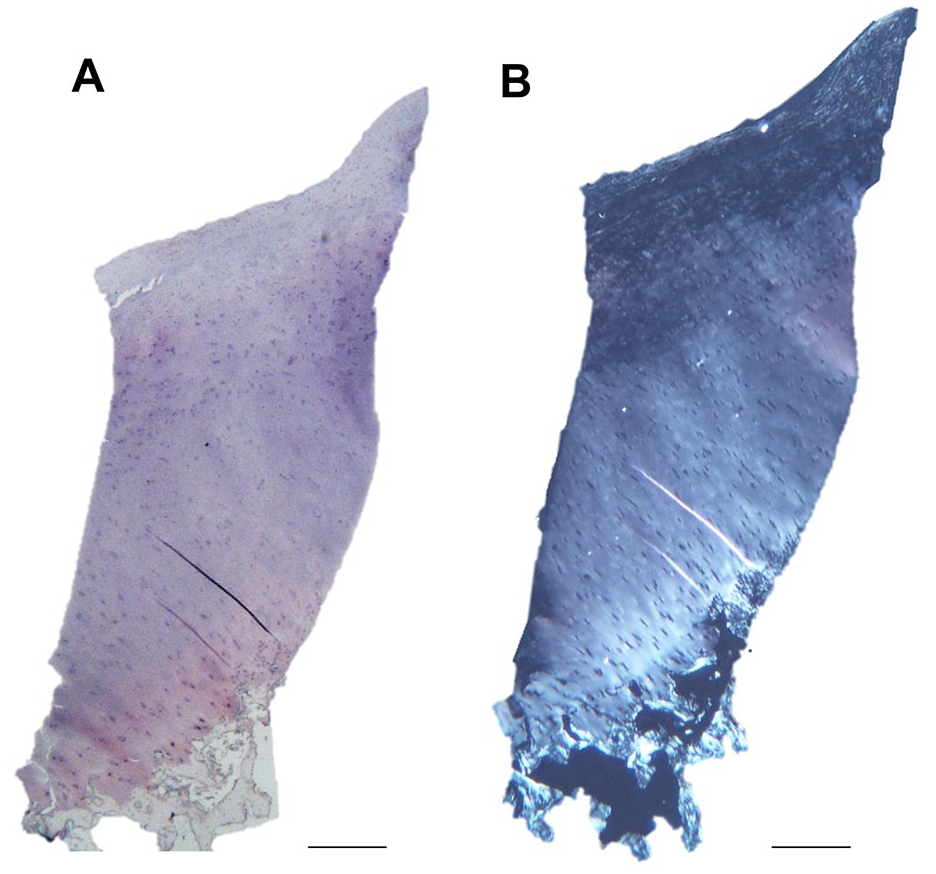
Histological analysis of the repair tissue biopsies. Representative images of the repair tissue formed displaying a hyaline morphology from both the early (
Inclusion criteria were as follows: (1) age 16-55 years, (2) femoral or patellar AC defects identified on MRI or at arthroscopic examination, (3) ligamentous stable knee (patients with anterior cruciate ligament [ACL] reconstruction were included if performed more than 1 year ago and the knee was stable on examination), (4) intact subchondral bone plate on arthroscopy and MRI, (5) written and informed consent prior to entry into the trial, and (6) willing and able to return for scheduled follow-up evaluations. Exclusion criteria were as follows: (1) disabling osteoarthritis or other joint disease, (2) untreated malalignment of the patella, (3) rheumatoid arthritis, diabetes, infection or family history of mesenchymal tumors, (4) drugs or treatments within the last 3 months that affect cartilage healing, (5) being >150% of the ideal body weight, (6) known history of anaphylaxis to any of the products used in preparation of the chondrocytes, and (7) any contraindications or medical disorders that would interfere with the conduct of the trial. The Consolidated Standards of Reporting Trials (CONSORT) were used to report on the trial.
Basic demographics such as age at operation, gender, laterality, and body mass index (BMI kg/m2) were collected. The defect location was documented as medial femoral condyle (MFC), lateral femoral condyle (LFC), patella, or trochlea. The size of the defect treated was measured in square centimeters (cm2) 23 and recorded on a specially designed knee map. The randomization was stratified by anatomical site of the defect (femur vs. patella) and implemented using 2 computer-generated random sequences that were put into sequentially numbered opaque envelopes.
Outcome Measures
The primary outcome was the patient’s clinical function, assessed using the modified Lysholm scale. 19 The Lysholm scale has been validated for assessing cartilage defect patients and assesses knee function based on symptoms of pain, instability, swelling, locking, support, and limping.19,24 The Lysholm scale was administered pre-operatively, post-operatively, and annually thereafter.
The secondary outcomes were time to failure and quality of repair tissue at the defect. Time to failure was analyzed for 2 definitions of failure. The first one represented failure of the treatment and was based on re-operation for any clinical reason indicating treatment failure, such as further ACI or mosaicplasty, osteotomy, a washout for infection, or conversion to knee replacement. Second-look arthroscopies were excluded, as they formed part of the research protocol and routine practice at the time. The second definition represented failure of the treatment to stop progression of the articular damage to end-stage osteoarthritis and was based on conversion to knee replacement. The quality of the repair tissue was defined by the morphology of the tissue and the surface architecture. Finally, serious adverse events (deep infection and deep-venous thrombosis [DVT]) were collected, as well as patch-related adverse events.
Statistical Analysis
Continuous variables were presented as means and ranges, and categorical variables as numbers and percentages. Standardized mean differences (SMDs), calculated as Cohen’s effect sizes, were used to assess balance between the 2 groups.25,26 A Kaplan-Meier survival analysis was performed to analyze time to failure, and the 2 groups were compared using the log-rank test. The use of location as stratifying variable was accounted for by performing a Cox regression analysis with location as covariate. Functional outcome was analyzed using analysis of covariance (ANCOVA), with location as covariate. In cases of severe baseline imbalance (SMD >0.25) where the unbalanced variable could have a prognostic influence (e.g., age, sex, baseline Lysholm score), we did additional regression analyses using the unbalanced variables as covariates. Differences in the tissue morphology and surface architecture between the groups were assessed using a Fisher’s exact test. All analyses were performed using R version 4.0.5 (R Foundation for Statistical Computing, Vienna, Austria) using the packages “survival” and “stdReg.” A 2-sided P-value below 0.05 was considered significant. A pre-trial calculation was performed assuming biserial correlation, and with a 20% standard deviation (SD) of endpoints explained by the intervention (r2 = 0.2), a minimum of 21 patients would be required in each group to obtain 80% power at the 2-sided P = 0.05 level.
Results
The trial ran between February 1998 and June 2001, when it was halted with the advent of alternatives to periosteum, such as a porcine collagen I/III membrane (Chondrogide®). All eligible patients who were approached were also happy to enter the trial, and at that point, 77 patients (56 males, 21 females) had been recruited into the study (
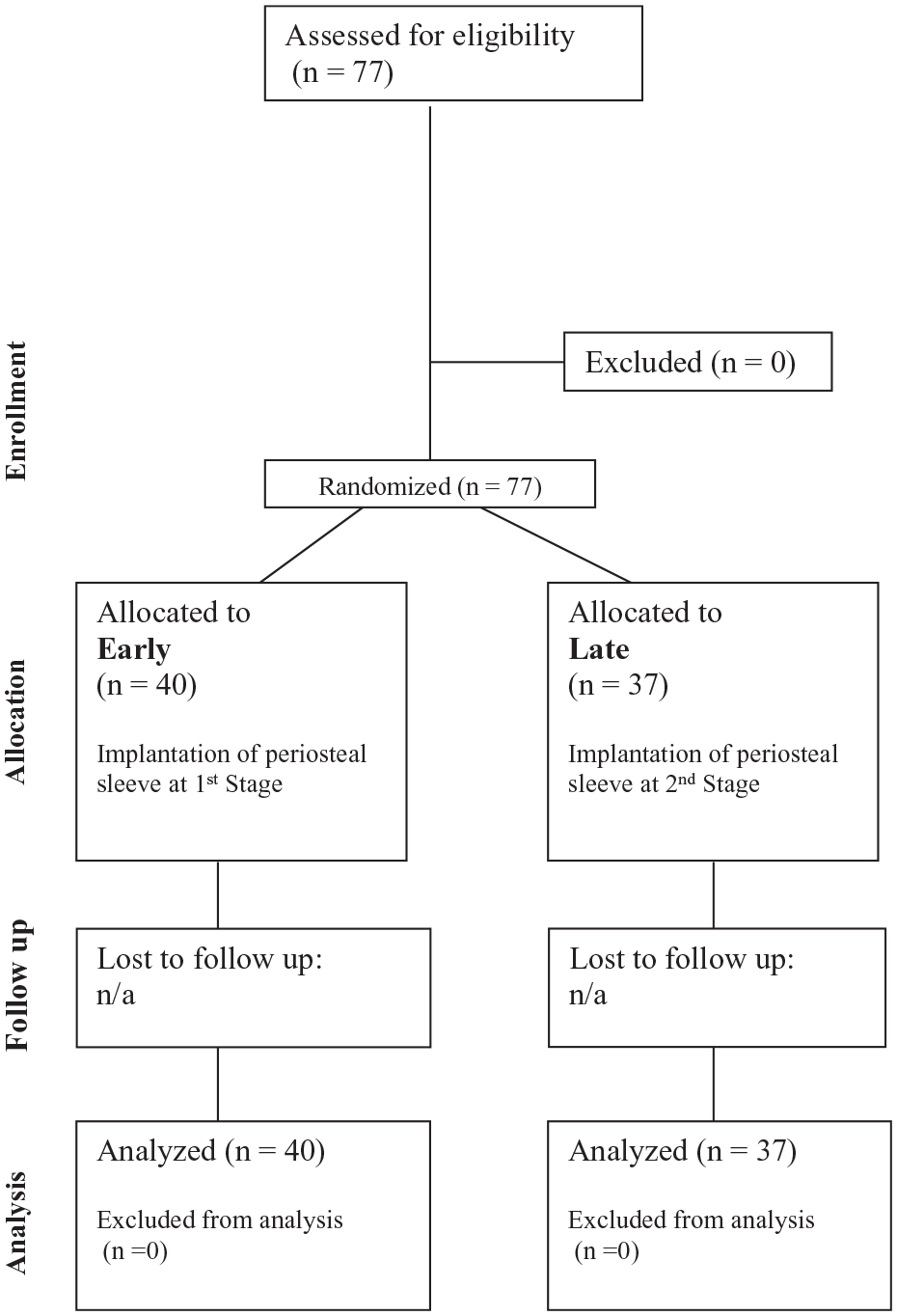
CONSORT diagram.
Baseline Demographics and Clinical Variables.

IQR = interquartile range; SMD = standardized mean difference, which was calculated as Cohen’s effect size. 25
Values in bold indicate SMDs > 0.25 (severe covariate imbalance).
Overall, the mean latest Lysholm score was 62.9 (SD = 24.5, range = 18-100) at the mean latest follow-up of 12.7 years and had improved significantly compared to baseline (mean improvement = 11.1, 95% CI = 4.8 to 17.4, P < 0.001, paired t test).
In terms of the primary outcome, at 12 months, no evidence was found for a difference between the 2 groups ( Table 2 ). However, at latest follow-up, the early group had a significantly higher mean Lysholm score than the late group (mean difference = 12.2, 95% CI = 1.0 to 23.3, P = 0.033, Table 2 ).
Clinical Outcomes.

SAE = serious adverse event; RR = relative risk.
Difference in mean Lysholm scores and survival rates, and their P-values, based on analysis of covariance and Cox regression using stratifying variable as covariate. Values in bold indicate statistical significance.
As for the secondary trial outcome, 26/77 patients (34%) required a re-operation, mainly a knee arthroplasty (12/26;
Table 2
). Most repair treatments such as a second ACI or mosaicplasty were performed because the repair tissue had failed, but in the 3 cases of early failure, the re-operation occurred because symptoms had not improved, and arthroscopy showed that repair tissue had failed to form. In all, 12/40 (30%) early group participants and 14/37 (38%) late group participants had a re-operation, resulting in comparable 20-year survival rates of 60% and 57% for the 2 groups (
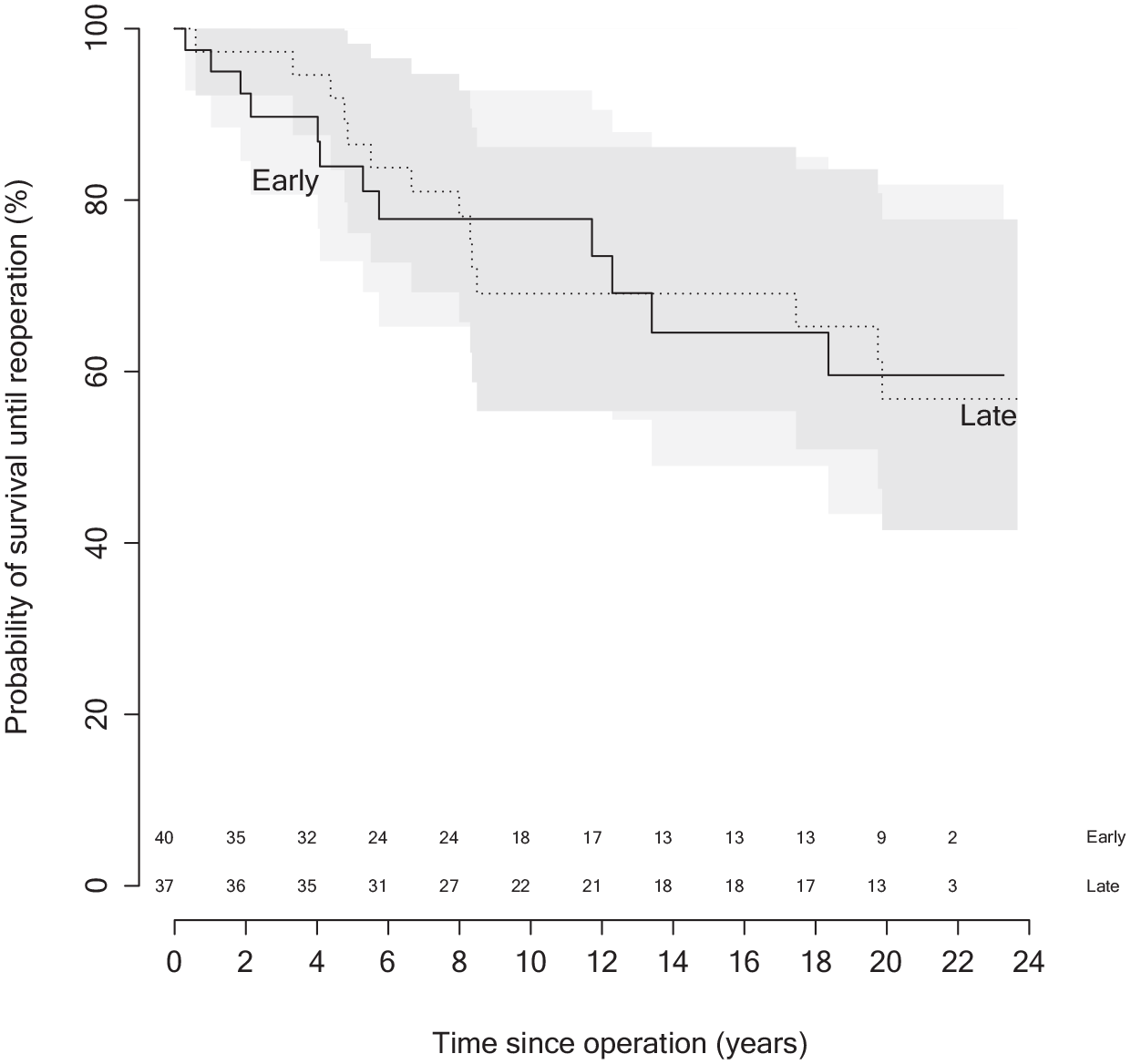
Kaplan-Meier survival analysis comparing between early and late group (P = 0.98, log-rank test). The numbers above the horizontal axis represent the number at risk for each time point.
A multivariable analysis was performed to determine if the results would change if adjusted for the unbalance in age, gender, defect area, and baseline Lysholm score. In regard to the latest Lysholm score, the adjusted mean difference reduced from 12.2 to 8.5 and was not significant (95% CI = −5 to 22, P = 0.22, Table 3 ). The adjusted differences in 20-year survival rates between early and late were larger (7% and 13% for any re-operations and arthroplasty, respectively) but still not significant in either case ( Table 3 ).
Adjusted Differences Using Analysis of Covariance and Cox Regression.
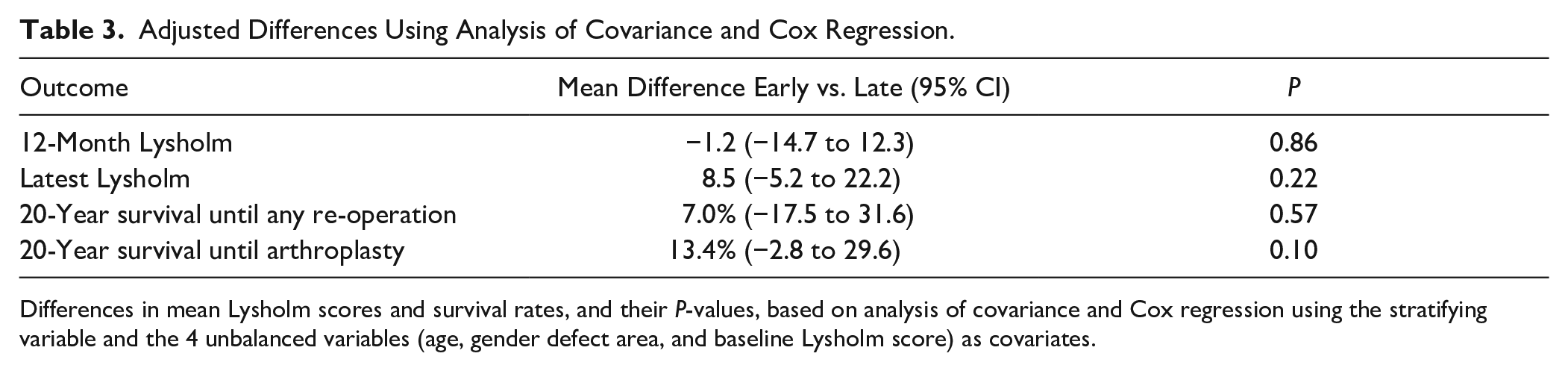
Differences in mean Lysholm scores and survival rates, and their P-values, based on analysis of covariance and Cox regression using the stratifying variable and the 4 unbalanced variables (age, gender defect area, and baseline Lysholm score) as covariates.
With regard to the repair tissue type, biopsies of repair tissue (n = 57) were obtained at a mean of 11.4 months (SD = 1.5, range = 9-15) and 11.8 months (SD = 1.2, range = 9-14) for the early and late groups, respectively. Two biopsies from each group could not be assessed because no tissue remained on the slide after processing. A further 7 biopsies in each group could not be assessed for the surface architecture as they were incomplete (e.g., had been artifactually damaged during processing or were not full depth and had no orientation). The tissue morphology was variable within each group and showed similar variability between the groups, with no significant difference (P = 0.37; Table 4 ). The majority of biopsies in both groups exhibited a fibrocartilage morphology, and there was a higher tendency to form hyaline cartilage in the early group compared to the late group, but this was not significant (P = 0.34). The surface architecture of the biopsies was also variable both within and between each of the groups, with no significant differences (P = 0.71; Table 4 ).
Tissue Morphology and Surface Architecture of the Repair Tissue Formed.

Two patients, both in the early group, had a deep infection, and 2 further patients, both in the late group, had DVT ( Table 2 ). Regarding patch complications, there were 2 cases in the early group where the patch was found to be detached at the time of cell implantation and needed re-attachment, which was achieved during arthroscopy by suturing with 6-0 Vicryl, using a grabber to manipulate the needle. There was a single case in the late group where revision surgery 7 months post-ACI was possibly related to patch detachment.
Discussion
To our knowledge, this is the first published study comparing placement of the periosteal patch during the first stage of ACI (chondrocyte harvest) against the conventional technique of placing the patch during the second stage (chondrocyte implantation). The intended benefit was to give the patch time to adhere to the surrounding cartilage and get an opportunity to inspect the patch integrity during the second stage and rectify any deficiencies as necessary.
At 12 months, no evidence was found for a difference in functional outcome as measured by the Lysholm score. However, at latest follow-up patients with an early patch reported 12 points higher mean Lysholm scores than those in the late group, a significant difference. After adjusting for the lack of balance between the groups in age, sex, defect size, and baseline Lysholm score, this difference was smaller (8.5 points) and no longer significant. It was also below the minimum clinically important difference of 13.6 points. 27 We conclude therefore that also no evidence was found that the long-term functional outcome differed between the groups.
It is difficult to explain why any potential difference in Lysholm score only became apparent with long-term data. This seems counter to the common observation that Lysholm scores typically improve in the first year following ACI and subsequently plateau.3,28 It also seems logical that a technique conceived with the aim of detecting and reducing early patch-related complications would primarily see benefits in the short term. However, the number of patch-related complications was small: only 2 cases in the early group had a detached patch at the time of cell implantation and a single case in the late group showed signs of failure related to patch detachment. This small rate is in contrast to the high detachment rate of two third found in goat and rabbit studies,29,30 suggesting patch detachment risk is much smaller in humans. Moreover, almost 90% of implanted human chondrocytes attach to cartilage or subchondral bone within 24 hours, 31 suggesting the patch only has a short-term role keeping the chondrocytes within the defect. On the contrary, an analysis of the repair tissue of failed ACI treatment, 1.5 to 6 months after the procedure, found evidence of displaced periosteal patch in all cases. 32 Our trial found no evidence for a difference in survival rates between the 2 groups. Three patients had an early failure, with only 1 (from the late group) showing signs of patch detachment. Given the low rates of patch detachment and the lack of evidence for a difference in 12-month functional outcome, the short-term benefit of inserting the patch at the harvest procedure does indeed seem negligible.
The randomization was stratified by defect location (femur vs. patella/trochlea) and almost perfectly balanced in this aspect. This stratification was based on information at the trial inception, suggesting that patellar and trochlear defects had worse outcome than femoral defects. Current knowledge still is that lesions in the patella have inferior outcomes compared with other locations within the knee following ACI,33-35 unless any malalignment is corrected simultaneously as tends to occur in current practice. However, it is now clear ACI of trochlear lesions tends to have a better outcome than patellar or even condylar lesions.34,36-38 Seen in this light, the 2 groups were less balanced: the early group had 1 patellar and 3 trochlear defects, whereas the late group had 4 patellar and no trochlear defect. Nevertheless, even when judged by detailed defect location, the imbalance between the groups was small (0.20) and below the level of 0.25 indicating serious imbalance. The imbalance in patellar and trochlea locations is therefore unlikely to have influenced our conclusions.
There were 2 (5%) deep infections in the early patch group. Deep infection is a concern for any operation. Although both cases occurred long after ACI was performed (2 and 4 years post-operatively), this observation begs the question whether implanting periosteum early can lead to increased deep infection rates. This figure is much higher than that found by Harris et al., 39 who found only 2 cases of deep infection in 3,321 knees after periosteum ACI. In relation to periprosthetic joint infection, the risk is higher in males, smokers, patients with a higher BMI and patients with a history of diabetes, rheumatoid arthritis, depression, steroid use, and previous joint surgery. 40 In this trial, the gender ratio and BMI were reasonably balanced, but we did not assess the smoking status. We excluded patients with rheumatoid arthritis or diabetes, but at the time ACI was recommended when other treatments had failed, and therefore, all patients would have had previous joint surgery.16,41 In arthroplasty, Quinlan et al. 42 found a significant increased risk of periprosthetic joint infection following early aseptic revision surgery for both hip and knee arthroplasty, most likely due to the increased operation time and open arthrotomy needed for the revision. In our case, all patients end up having an arthrotomy and an arthroscopy, with the late patch (traditional ACI) arguably having a longer arthrotomy because it involves suturing the patch and implanting the cells. It is therefore not clear why this complication should occur more often in the early group.
The study failed to show any significant difference in the quality of the repair tissue formed following either an early or late application of the periosteal patch during the ACI procedure. Although this finding appears consistent with the Lysholm score results observed, earlier studies found little to no correlation between histological and clinical outcomes.43-46 Whether this quality of repair is maintained throughout the life course of the repair tissue is unknown as repeat biopsies were not taken. However, previous studies have shown that cell therapy-repaired cartilage remodels with time.47,48 More recently, however, studies have demonstrated that the use of a collagen membrane produces a superior quality repair tissue compared to periosteum, including a significantly better surface architecture. 43
Besides being a trial of patch insertion timing, our data have additional value in demonstrating that ACI is an effective and durable technique for cartilage regeneration. Moreover, the vast majority of patients in the trial (84%) had at least 1 prior procedure to the defect, suggesting a prior procedure is not necessarily detrimental. This study suffers from the same limitations as other studies through a lack of consensus as to what constitutes failure. Most commonly, this is defined as the need for further surgery. There were 26 patients (33%) who required a further procedure in our series. Our figures exclude post-operative arthroscopies which were at this time routinely offered to all patients to visualize the treated defect and to provide prognostic advice and histological evidence of structural repair. This rate of further surgery compares favorably with the rate found in other case series in the literature39,49 even allowing for the longer follow-up. Likewise, the Lysholm scores increased by a mean of 10.9 points indicating patients also improved subjectively.
Using arthroplasty as an end point, we found 12 (15.6%) patients who required knee arthroplasty. This corresponds with previously reported outcomes at our institution, 33 but was higher than the 5% reported in one of the largest published longer-term durability studies of ACI. 49 The caveat here is that patellar lesions were not included in that large study, and their follow-up period was shorter. Furthermore, Knutsen et al. 50 reported a rate of 2.5% and 15% 51 at 5 and 15 years, respectively. Overall, we were surprised by the scant literature recording the rates of arthroplasty following failed ACI. In a meta-analysis from Harris et al., 39 the rate of conversion to arthroplasty was found to be 0.3%, but they admit many studies did not record this data point. It is likely therefore that the prevalence of knee arthroplasty following ACI is underestimated in the literature. Recently, knee arthroplasty following ACI was compared to that in the normal population, utilizing the Norwegian arthroplasty registry. 52 In 20 years follow-up, the risk of arthroplasty following ACI was 19.1%, which was 416 times the risk to the normal population. Older age, high BMI, deep lesions, and multiple lesions were significant risk factors for a joint arthroplasty.
As with many studies on this topic, this study has many limitations. One important limitation was the lack of balance between the groups. Although the randomization used stratification by location and did indeed achieve balance in this characteristic, other characteristics such as age and baseline Lysholm scores showed severe imbalance. Severe imbalance is almost inevitable in small trials, 53 which we tried to prevent by using stratified randomization. However, the number of patients with patellar/trochlear defects was small, and therefore, the effect of stratification on reducing imbalance was also small, and this may explain the large imbalance in other predictors. A better strategy would have been to include the baseline Lysholm score, with 2 strata split by the expected median baseline Lysholm score, a strategy we used in the later trial.54,55 However, by doing additional regression analyses using the unbalanced variables (such as Lysholm score) as covariates, we have essentially addressed this design issue at the analysis stage. In addition, this trial was also a single-center study with the majority of patients being treated by a single surgeon. This renders it susceptible to selection bias. Furthermore, we were not able to ascertain the type of surgical procedure to the defect prior to ACI. However, because the number of prior procedures was reasonably balanced between the 2 groups, we do not expect this to have introduced a bias. Finally, with the development of new generations of patch surfaces being used, the results utilizing those should be tested in a similar fashion to confirm our observations.
Conclusion
In conclusion, we found no evidence that placement of the periosteal patch at the time of chondrocyte harvest instead of chondrocyte implantation improved long-term Lysholm scores or survival until re-operation or knee replacement in patients undergoing ACI. Our results also show that ACI is an effective and durable technique for cartilage regeneration, with good patient outcomes and low failure rates.
Footnotes
Authors’ Note
The reported work was done at the Robert Jones and Agnes Hunt Orthopaedic Hospital NHS Foundation Trust, Oswestry, SY10 7AG, UK.
Acknowledgments and Funding
The authors dedicate this article to the late Professor James Richardson, the orthopedic surgeon responsible for this trial, who initiated and implemented it. The authors acknowledge Mr. Paul Harrison and the John Charnley (OsCell) GMP-laboratory at the RJAH Orthopaedic Hospital. The authors are grateful to Versus Arthritis (grants 18480, 19429, 21156, and 20815) and the Medical Research Council (MR/L010453/1 and MR/N02706X/1) for supporting this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for the reported trial was obtained from the Shropshire Health Authority District Medical Ethics Committee (98/5/RJ).
