Abstract
Purpose
The primary aim is to evaluate the relationship between MOCART scores and patient satisfaction, as evaluated by achievement of (1) the Patient Acceptable Symptomatic State (PASS) and (2) the minimal clinically important difference (MCID) for Knee Injury and Osteoarthritis Score Quality of Life (KOOS QoL), for patients undergoing autologous chondrocyte implantation (ACI) for focal Grade IV patellofemoral chondral defects. The secondary aim is to determine the threshold MOCART score which predicts the ability to meet the PASS and the MCID for KOOS QoL.
Methods
Patients undergoing ACI for grade IV patellofemoral chondral defects by a single surgeon from 2017 to 2020 were identified by search of the EMR. To determine PASS status, patients were asked, “Do you consider your current level of symptoms to be acceptable?” KOOS QoL scores were also collected. Patients with 6-month postoperative knee MRI, PASS scores, and minimum 2-year follow-up data were included. Paired t tests and Wilcoxon Rank-Sum tests were used to evaluate the relationship between MOCART scores and (1) PASS achievement and (2) achievement of the MCID for KOOS QoL (12.8).
Results
Thirty-four patients were included, with a median age of 35.1 years [IQR: 24.6, 37.1], and BMI of 24.0 kg/m2 [IQR: 21.5, 28.1]. The median time to postoperative MRI was 6.7 months [IQR: 5.8, 7.9], and average follow-up time was 3.7 ± 1.2 years. Twenty-five patients (74%) achieved PASS, and 18 patients (out of 27 who had postop KOOS QoL Scores, 67%) achieved the MCID for KOOS QoL. Patients who achieved PASS had higher average MOCART scores (61.8 ± 16.0) than those who did not achieve PASS (45.0 ± 12.8, p=0.011), whereas patients who achieved the MCID for KOOS QoL did not have higher MOCART scores than those who did not achieve the MCID (61.9 ± 18.3 versus 53.3 ± 17.1, P = 0.25). There was no relationship between age, sex, lesion size, and lesion location and ability to achieve PASS or MCID for KOOS QoL (p>0.05). A threshold MOCART value of 55 was associated with the highest AUC on ROC analysis for likelihood of achieving PASS (0.778) and MCID for KOOS QoL (0.667).
Conclusion
Higher MOCART scores are associated with an increased likelihood of achieving PASS following patellofemoral ACI. Moreover, MOCART scores > 55 predict the ability to achieve PASS and the MCID for KOOS QoL following patellofemoral ACI.
Level of Evidence:
IV
Introduction
Autologous chondrocyte implantation (ACI) is a commonly used technique for the treatment of focal chondral defects about the knee. The third-generation matrix-associated chondrocyte implantation (MACI) has been found to be successful in the treatment of Grade IV chondral defects of the patella and trochlea.1-3 The patellofemoral joint can be particularly challenging to treat given the geometry of the joint and high contact pressures experienced relative to body weight.4-6
Magnetic resonance imaging (MRI) can be used to evaluate the appearance of an ACI graft as it matures. In particular, MRI allows for evaluation of graft integration and defect filling, neither of which can be assessed accurately arthroscopically.7,8 In order to quantify ACI graft maturity on MRI, Marvolits et al. 9 described the Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) Score. The MOCART score, which ranges from 0 to 100, with 100 indicating graft structure similar to that of native hyaline cartilage, is comprised of multiple categories, including graft fill, graft signal intensity, and graft integration (Table 1).
MOCART Scoring System.
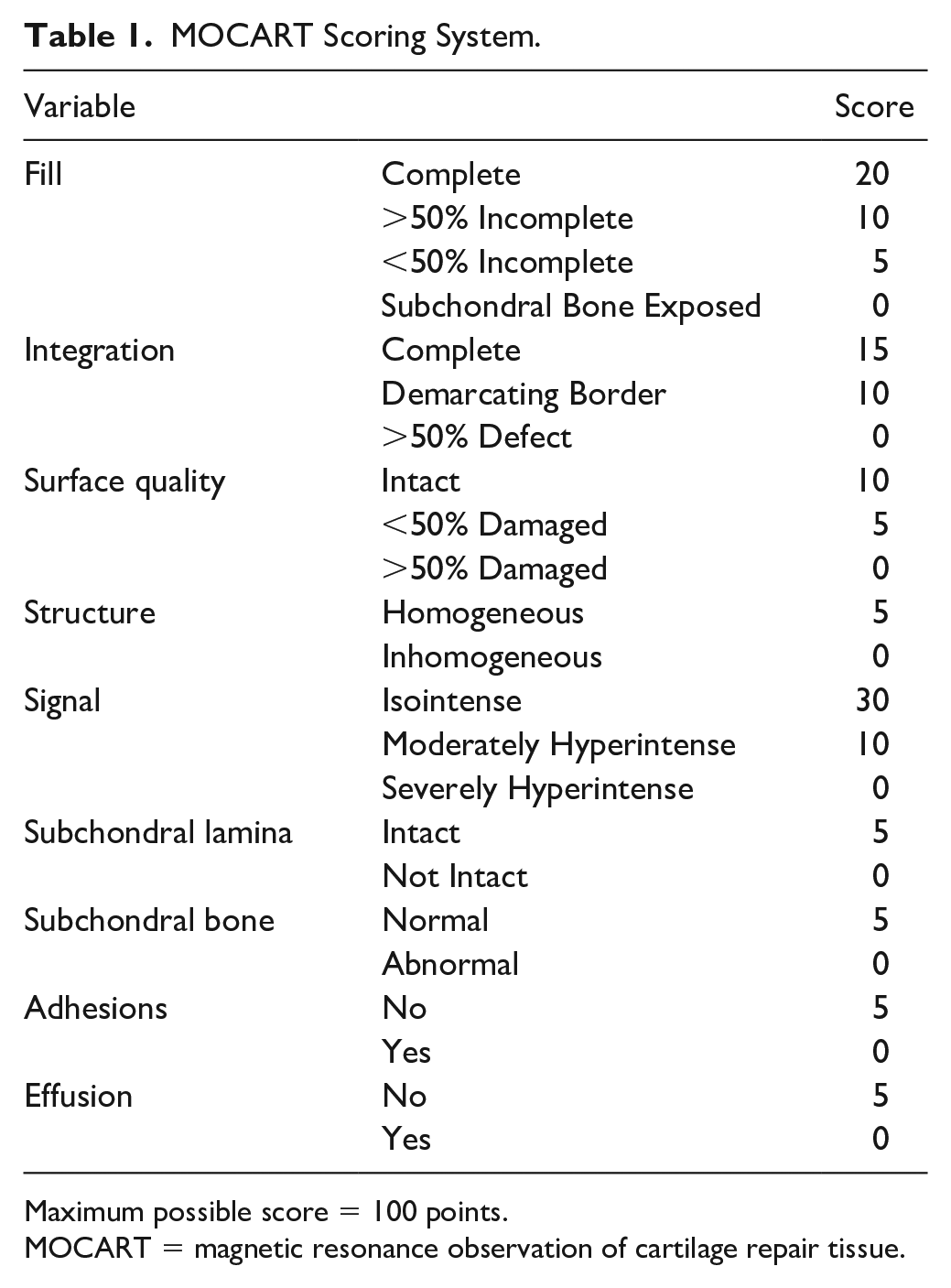
Maximum possible score = 100 points.
MOCART = magnetic resonance observation of cartilage repair tissue.
Previous investigations of the relationship between MRI appearance of patellofemoral ACI grafts and clinical outcome measures have demonstrated positive correlations between either individual components or the overall MOCART score and PROMs, including the Tegner activity, 10 Lysholm-Gillquist, 10 International Knee Documentation Committee Subjective Knee Evaluation Form (IKDC), 10 Cincinnati knee rating scores, 10 Knee Injury and Osteoarthritis Scores (KOOS), 9 and Lysholm scores. 11 However, the relationship between MRI appearance of patellofemoral ACI grafts and the patient acceptable symptomatic state (PASS) has yet to be elucidated. It is critical to consider the PASS in lieu of correlations between PROMs and MRI appearance given that correlations between scores do not correspond with a clinically significant improvement in patient symptoms.
The primary aim of the present study is to evaluate the relationship between MOCART scores at 6 months postoperative and patient satisfaction, as determined by ability to meet (1) PASS and (2) the minimal clinically important difference (MCID) for KOOS Quality of Life (QoL), at minimum 1 year postoperative for patients undergoing autologous chondrocyte implantation (ACI) for focal Grade IV chondral defects about the patellofemoral joint. The secondary aim of the present study is to determine the threshold MOCART score which predicts the ability to meet the PASS and MCID for KOOS QoL at minimum 1 year postoperatively in these patients. The authors hypothesized that higher MOCART scores at 6 months postoperative would be associated with an increased likelihood of achieving the PASS and MCID for KOOS QoL at minimum 1 year postoperatively.
Materials and Methods
Patient Selection/Patient Reported Outcome Measures
Following Institutional Review Board approval (IRB study #: 2020-2123), 44 patients undergoing ACI for large (>1.5 cm2) grade IV chondral defects of either the patella or trochlea between 2017 and 2020 by a single sports medicine fellowship-trained surgeon (S.M.S.) were identified. Informed consent was obtained for each patient prior to enrollment in the study. Patients were contacted via telephone call and asked a single “Yes/No” question to assess PASS status, “Do you consider your current level of symptoms to be acceptable?” KOOS QoL scores were collected via electronic survey.
Patients were included in the study if they had a knee MRI obtained at 6 months postoperatively, PASS data at minimum 1 year postoperatively, and minimum 2 year follow-up data available for survival analysis. Of note, routine postoperative MRI are obtained in our ACI patients to evaluate ACI healing. Patients were excluded from the study if they did not have PASS data available (n = 9) or if they did not have minimum 2-year follow-up data available for survival analysis (n = 1).
Failure of the ACI graft was defined as conversion to patellofemoral joint arthroplasty.
Surgical Technique
Stage 1—MACI biopsy
For the first stage of the procedure, a standard diagnostic arthroscopy is performed, and the chondral lesion size and depth is assessed with an arthroscopic probe. A gauge is used to detach a small piece of cartilage from the intercondylar notch (nonweightbearing area), and a grasper is used to remove the piece of cartilage from the joint. The biopsy tissue is then placed in Vericel (Vericel Corporation; Cambridge, MA) culture medium and sent to Vericel Corporation for culture and graft preparation.
Stage 2—MACI implantation
Approximately 6 weeks following the first stage, the MACI graft is available for implantation. Either a medial or lateral parapatellar approach to the knee is performed, the patella is everted, and the chondral defect is exposed and sized. The chondral defect is debrided to a stable rim with a curette. The MACI graft is placed in the defect with fibrin glue placed under and around MACI graft. If the graft is determined to be unstable after the knee is cycled through flexion and extension, interrupted 6-0 Vicryl sutures are placed at the periphery of the MACI graft to secure the graft to the adjacent healthy cartilage.
Postoperative rehabilitation
Patients are placed in a hinged knee brace immediately postoperatively. The hinged knee brace remains locked in extension for ambulation, and the patients may weight bear as tolerated for the first 6 weeks postoperatively. Patients begin to work on range of motion immediately postoperatively, starting from 0-30 degrees using a continuous passive motion machine and advancing up to 0° to 90°. Starting 6 weeks postoperatively, the patients may begin to wean themselves from the hinged knee brace, and at that time, they may begin full range of motion as tolerated.
Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) Scores
MOCART scores were determined by a single musculoskeletal fellowship-trained radiologist on postoperative MRI obtained at an average of 7.0 ± 2.3 months postoperative (range: 3.2-16.2 months). Variables included in the MOCART scores included fill, integration, surface quality, structure, signal, subchondral lamina integrity, subchondral bone appearance, presence or absence of adhesions, and presence or absence of an effusion (Table 1). 9
Statistical Analysis
Means and standard deviations or medians and interquartile ranges (IQR) were used for continuous data, and categorical data was reported as frequencies and percentages. Data for MOCART score and follow-up time were normally distributed per the Shapiro-Wilk test; therefore, paired t tests were used for analysis. Data for time between surgery and MRI, time to PASS/KOOS QoL scores, time between patient-reported outcome measures (PROMs) and postoperative MRI, age, BMI, and lesion size were not normally distributed, and Wilcoxon Rank-Sum tests were used for analysis. The minimal clinically important difference (MCID) used for KOOS QoL scores in the analysis was 12.8 per the results of prior studies. 12 Area under curve (AUC) analysis was performed on receiver operating curves (ROC) created to determine the threshold value for MOCART scores which predicts ability to meet PASS and MCID for KOOS QoL. Log-rank test was used to evaluate differences in survival probability between patients that did meet PASS versus those that did not meet PASS. Statistical analysis was performed with STATA (Version 17.0, StataCorp). Statistical significance was determined at P < 0.05.
Results
Of the 44 patients identified, 34 were included and 10 were excluded for lack of follow-up data (Figure 1). The median age was 35.1 years [IQR: 24.6, 37.1], and BMI was 24.0 kg/m2 [IQR: 21.5, 28.1]. There were 23 women and 19 right knees included. The median time to postoperative MRI was 6.7 months [IQR: 5.8, 7.9], median time to PASS scores was 2.3 years [IQR: 1.5, 3.5], median time to KOOS QoL scores was 2.1 years [IQR: 1.7, 2.6], and average follow-up time was 3.7 ± 1.2 years (range: 2.1-6.5 years, Table 2).
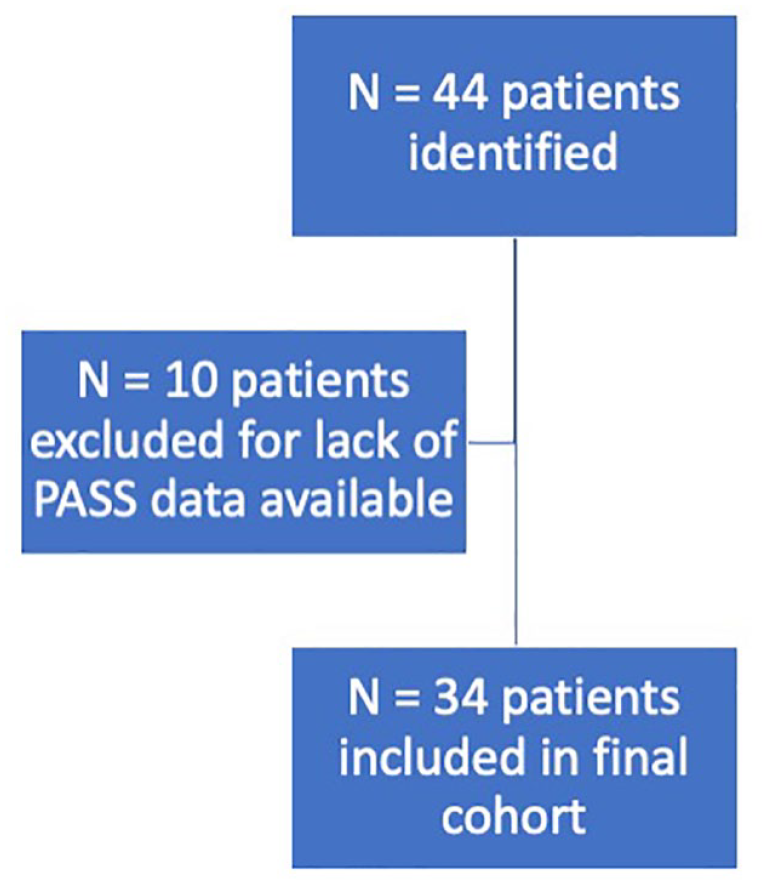
Patient cohort.
Demographic Information.
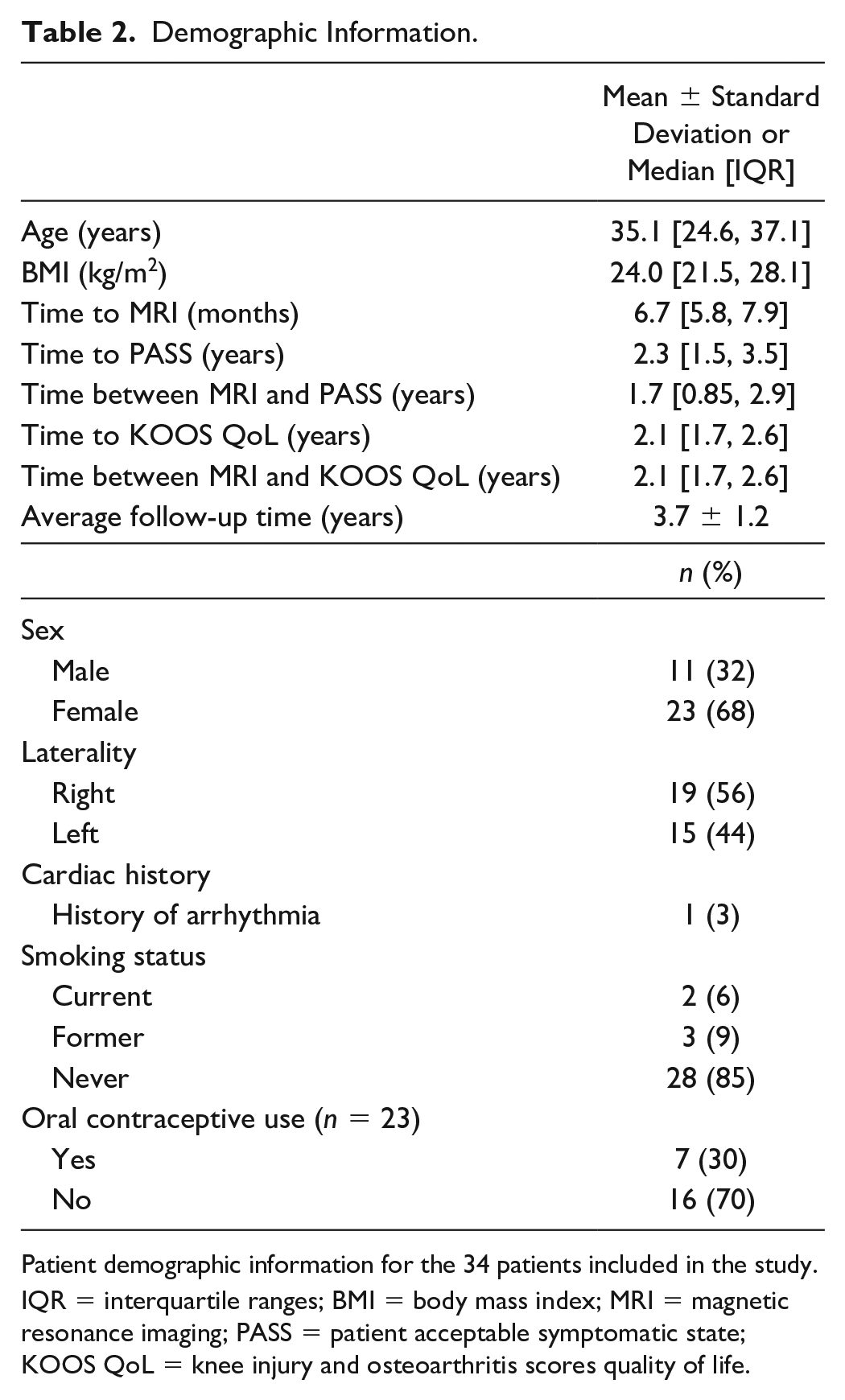
Patient demographic information for the 34 patients included in the study.
IQR = interquartile ranges; BMI = body mass index; MRI = magnetic resonance imaging; PASS = patient acceptable symptomatic state; KOOS QoL = knee injury and osteoarthritis scores quality of life.
With regard to demographic data, one patient (3%) had a history of cardiac arrhythmia. Two patients (6%) were current smokers, and 3 (9%) were former smokers. Seven patients (30%) had active oral contraceptive use at the time of surgery (Table 2).
The median lesion size was 3.0 cm2 [IQR: 2.6, 3.5]. The majority of lesions involved the patella (30 lesions, 88%). Among patellar lesions, the central ridge was most commonly involved (15 lesions, 52%). Eleven chondral lesions (32%) occurred due to patellar instability (Table 3).
Description of Chondral Lesions.
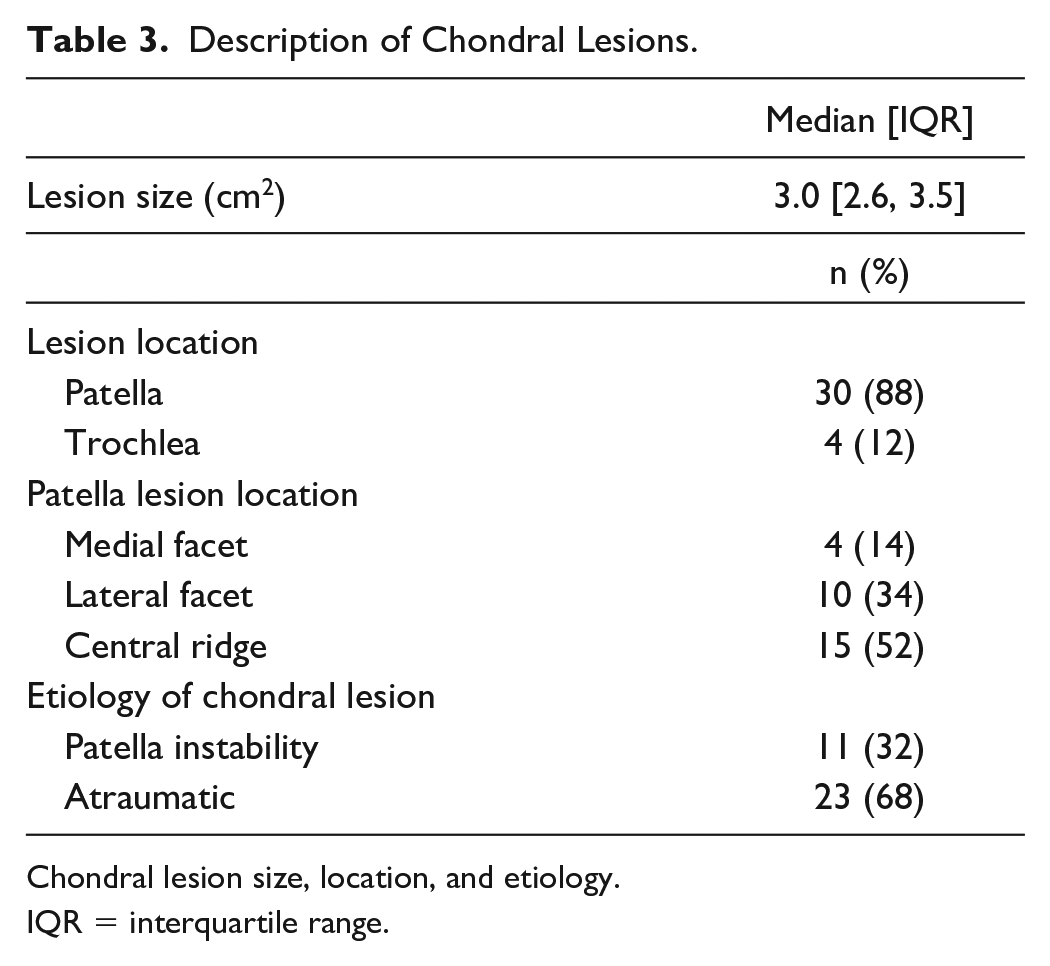
Chondral lesion size, location, and etiology.
IQR = interquartile range.
Twenty-five patients (74%) achieved PASS at minimum 1-year follow-up. Patients who achieved PASS had higher average MOCART scores (61.8 ± 16.0) than those who did not achieve pass (45.0 ± 12.8, P = 0.011, Table 4). Eighteen patients (out of 27 with both preoperative and postoperative KOOS QoL scores, 67%) achieved the MCID for KOOS QoL. However, patients who achieved the MCID for KOOS QoL did not have higher MOCART scores than those who did not achieve the MCID (61.9 ± 18.3 vs. 53.3 ± 17.1, P = 0.25). There were no differences between patients who achieved PASS and MCID for KOOS QoL versus those that did not achieve PASS and MCID for KOOS QoL with regard to age, sex, lesion size, lesion location, smoking status, cardiac history, oral contraceptive use, time to postoperative MRI, time to PASS/KOOS QoL evaluation, time between postoperative MRI and PASS/KOOS QoL evaluation, and follow-up time (P > 0.05 for all, Tables 4 and 5). Patients who achieved the MCID for KOOS QoL had lower average BMI compared to those who did not achieve the MCID (P = 0.01), but there was no difference in BMI among patients who did versus did not achieve PASS (P > 0.05, Tables 4 and 5).
Patient Factors and PASS.
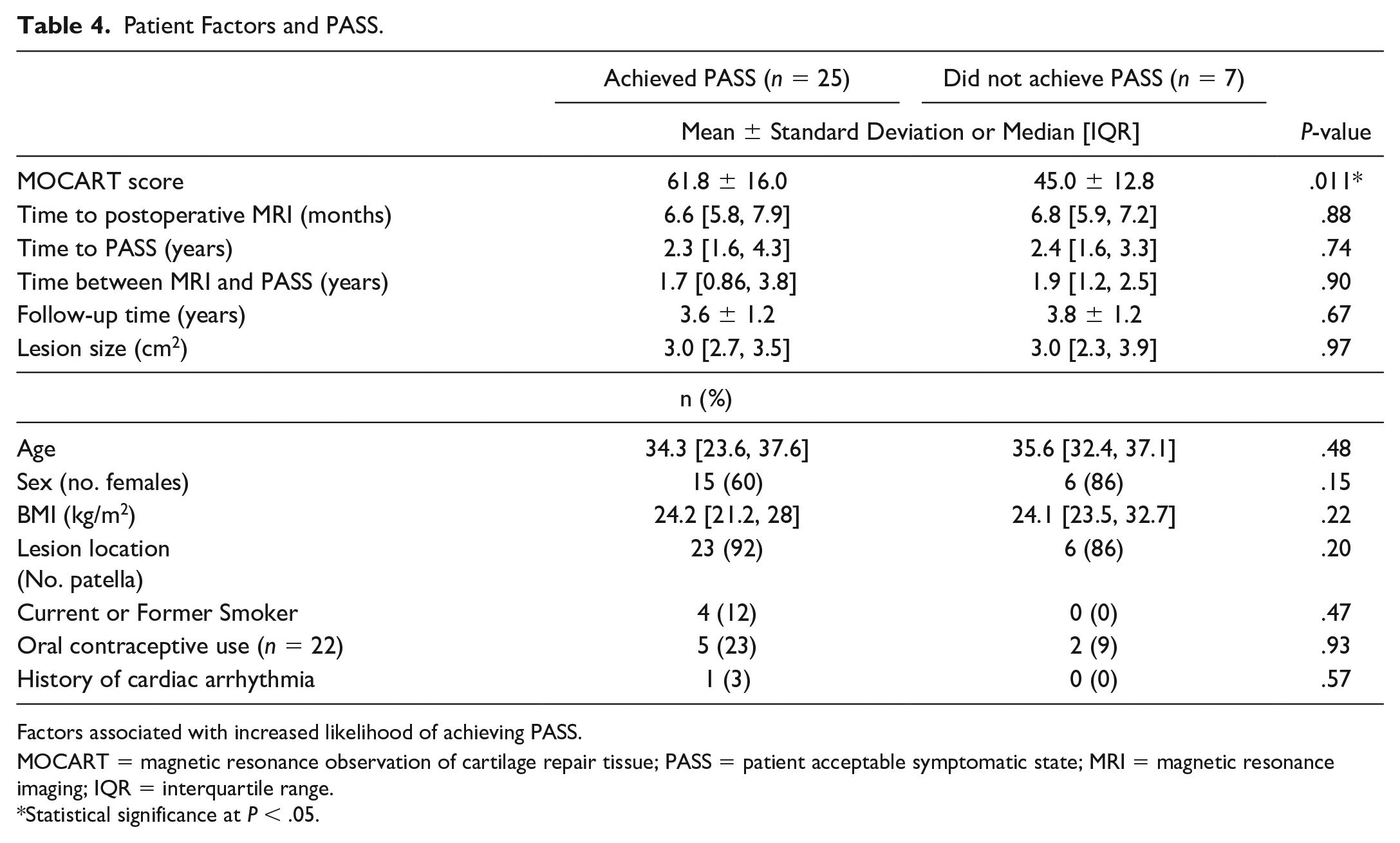
Factors associated with increased likelihood of achieving PASS.
MOCART = magnetic resonance observation of cartilage repair tissue; PASS = patient acceptable symptomatic state; MRI = magnetic resonance imaging; IQR = interquartile range.
Statistical significance at P < .05.
Patient Factors and MCID for KOOS QoL.
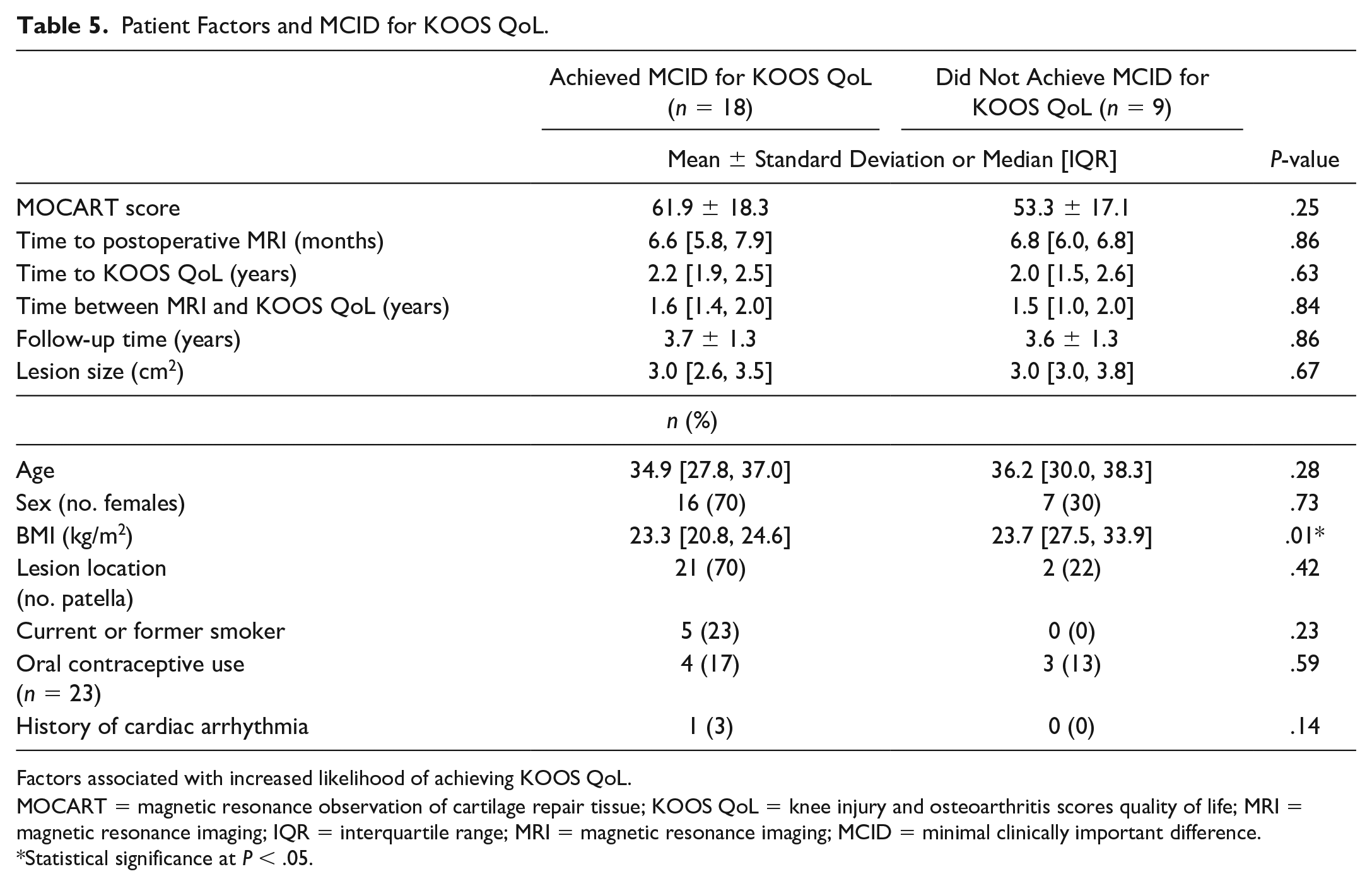
Factors associated with increased likelihood of achieving KOOS QoL.
MOCART = magnetic resonance observation of cartilage repair tissue; KOOS QoL = knee injury and osteoarthritis scores quality of life; MRI = magnetic resonance imaging; IQR = interquartile range; MRI = magnetic resonance imaging; MCID = minimal clinically important difference.
Statistical significance at P < .05.
MOCART scores ranged from 20 to 95 with an average MOCART score of 58.5 ± 17.2. To determine the threshold MOCART score which predicted ability to achieve (1) PASS and (2) MCID for KOOS QoL, area under the curve analysis was performed on receiver operating curves with threshold values ranging from 30 to 80. A threshold MOCART value of 55 was associated with the highest AUC for both PASS (0.778) and MCID for KOOS QoL (0.667) compared with the remaining values tested (Table 6).
ROC AUC Analysis.
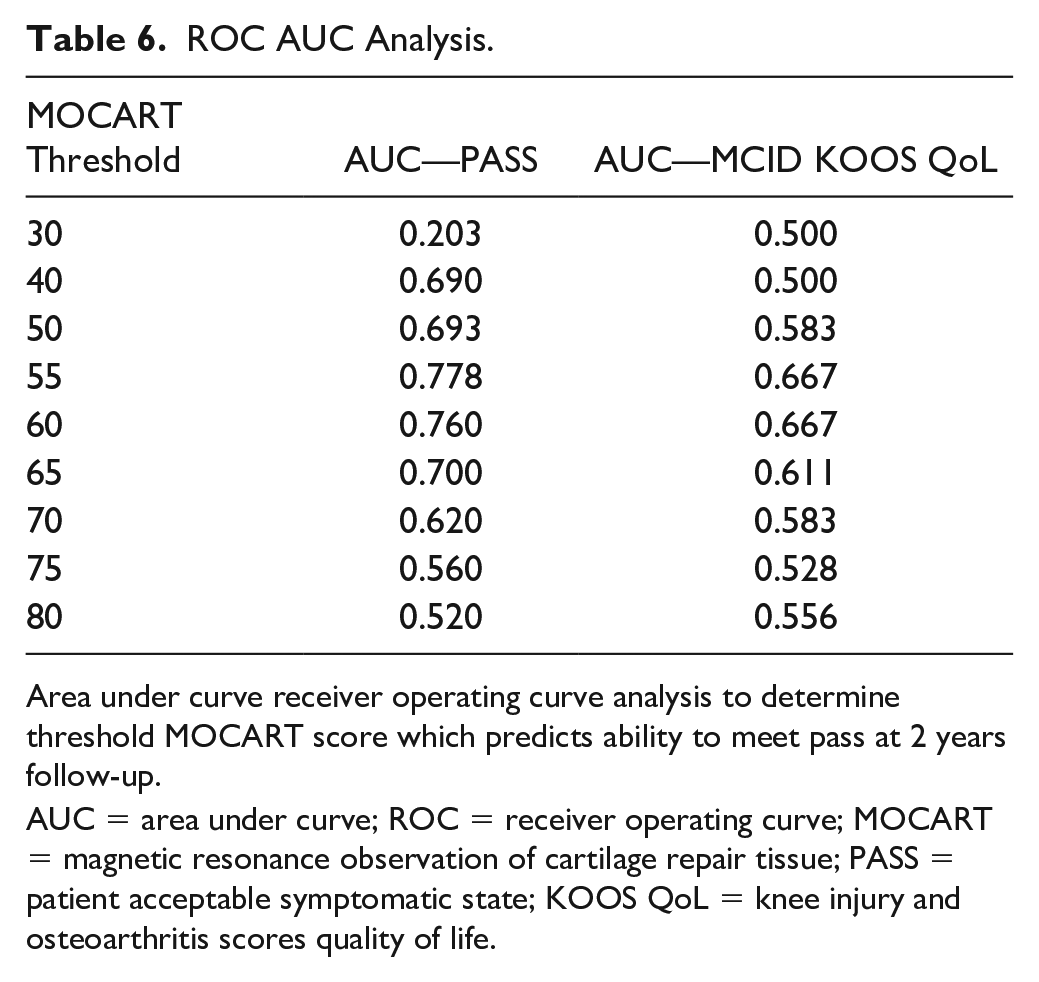
Area under curve receiver operating curve analysis to determine threshold MOCART score which predicts ability to meet pass at 2 years follow-up.
AUC = area under curve; ROC = receiver operating curve; MOCART = magnetic resonance observation of cartilage repair tissue; PASS = patient acceptable symptomatic state; KOOS QoL = knee injury and osteoarthritis scores quality of life.
There were two failures in the cohort, both conversions to patellofemoral joint arthroplasty at an average of 2.3 ± 0.4 years. One of the patients achieved PASS and the MCID for KOOS QoL prior to graft failure, and the second did not achieve PASS or the MCID for KOOS QoL. Although there was a tendency for the group that did not achieve PASS to be more likely to experience failure of the ACI graft, this finding was not statistically significant (P = 0.077, Figure 2).
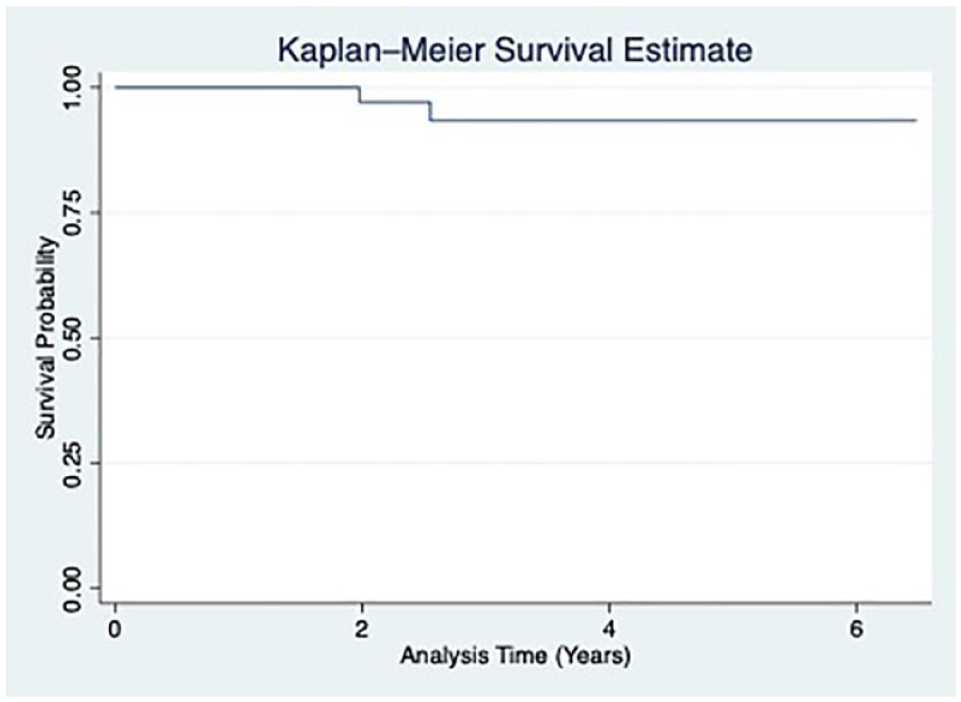
Kaplan-Meier survival curve for all patients included in the study.
Discussion
We found that higher MOCART scores at 6 months postoperative are associated with an increased likelihood of achieving PASS at minimum 1-year follow-up in patients undergoing ACI for Grade IV chondral defects of the patellofemoral joint. Moreover, MOCART scores > 55 are predictive of the ability to achieve PASS and the MCID for KOOS QoL following ACI for chondral defects of the patella or trochlea.
Similar to previous studies, we found that the majority of patients have good outcomes at midterm follow-up following MACI for patellofemoral chondral lesions.1,2 However, previous investigations of the relationship between clinical and imaging outcomes following patellofemoral MACI have only examined correlations between PROMs and individual components of the MOCART score. Ochs et al. 10 saw a concomitant increase in MOCART scores and IKDC scores in patients undergoing ACI for chondral defects about the knee, although the authors did not look for a direct relationship between the two scores. Robertson et al. 13 found a moderate positive correlation between both the MRI appearance of ACI grafts about the knee and KOOS scores and MRI appearance and functional outcome measures, although there was no evaluation of clinically significant improvement in KOOS scores in the aforementioned study. The present study builds on the existing literature by comparing clinically significant improvement in patient symptoms with an objective evaluation of ACI graft maturity on MRI.
There are several limitations to this study. First, our small sample size may indicate that our study is underpowered. Given that this study was retrospective in nature, a priori power analysis was not possible to perform. Due to the inherent limitations of post hoc power analysis, 14 post hoc power analysis was also not performed. However, the size of our cohort is similar to that of previous studies examining outcomes of ACI for patellofemoral chondral lesions.1,2 Given that phone surveys were used, it is possible that the results are susceptible to response and selection biases. Moreover, this study contains single-surgeon data, which has its inherent limitations. There may be variability in surgical technique or postoperative protocols between surgeons which may limit generalizability of the findings from the current study. Twenty seven of the 34 patients included in this study had both pre- and postoperative KOOS QoL Scores, and incomplete KOOS QoL data may also limit generalizability of the findings in the study. The floor and ceiling effects associated with the KOOS QoL score may impact study results as well. Finally, we only have minimum 2-year follow-up data available, although previous studies have shown that ACI are most likely to fail in the first 2 years postoperatively, 15 so we feel as though minimum 2-year follow-up is appropriate for this cohort.
Conclusion
In sum, there appears to be a relationship between graft maturity on imaging and clinical outcome measures in patients undergoing ACI for patellofemoral chondral defects. Perhaps routine surveillance with MRI should be performed on patients following ACI for patellofemoral chondral defects, and closer follow-up should be considered for patients with MOCART scores ≤ 55 at 6-month follow-up. Additional longitudinal studies can evaluate the relationship between imaging and clinical outcome measures in patients undergoing patellofemoral ACI.
Footnotes
Author Contributions
JR: Study conception and design, data analysis, manuscript writing
CF: Data collection, manuscript editing
MR: Data collection, manuscript editing
AB: Image analysis, manuscript editing
SS: Study conception and design, data collection, manuscript editing
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number T32AR078751. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Julia S. Retzky is under the T32 grant.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
IRB study # 2020-2123
Informed Consent
Obtained prior to enrolling patient in study
