Abstract
Objectives
While substantial progress has been made in engineering cartilaginous constructs for animal models, further research is needed to translate these methodologies for human applications. Evidence suggests that cultured autologous chondrocytes undergo changes in phenotype and gene expression, thereby affecting their proliferation and differentiation capacity. This study was designed to evaluate the expression of chondrogenic markers in cultured human articular chondrocytes from passages 3 (P3) and 7 (P7), beyond the current clinical recommendation of P3.
Methods
Cultured autologous chondrocytes were passaged from P3 up to P7, and quantitative polymerase chain reaction (qPCR) was used to assess mRNA expression of chondrogenic markers, including collagen type I (COLI), collagen type II (COLII), aggrecan (AGG), bone morphogenetic protein 4 (BMP4), transcription factor SOX-9 (SOX9), proteoglycan 4 (PGR4), and transformation-related protein 53 (p53), between P3 and P7.
Results
Except for AGG, no significant differences were found in the expression of markers between passages, suggesting the maintenance of chondrogenic potential in cultured chondrocytes. Differential expression identified between SOX9 and PGR4, as well as between COLI and SOX9, indicates that differences in chondrogenic markers are present between age groups and sexes, respectively.
Conclusions
Overall, expression profiles of younger and male chondrocytes exhibit conversion of mature cartilage characteristics compared to their counterparts, with signs of dedifferentiation and loss of phenotype within-group passaging. These results may have implications in guiding the use of higher passaged chondrocytes for engineering constructs and provide a foundation for clinical recommendations surrounding the repair and treatment of articular cartilage pathology in both sexes.
Introduction
With limited repair capacity, articular cartilage is susceptible to damage from trauma, and it can progress to long-term diseases such as osteoarthritis (OA) that may require knee replacement. 1 Brittberg et al. 2 initiated the use of cultured autologous chondrocytes from the knee joint of patients for the treatment of deep cartilage defects. Their results indicated that cultured chondrocytes behaved as mesenchymal stem cells (MSCs) and have the potential to repair cartilaginous injuries. 3 In fact, autologous MSCs for cartilage repair are an expanding topic in tissue engineering literature, particularly successful in animal models including rabbits4,5 and sheep.6,7 Recently, the chondrogenic potential markers (CPMs) of passaged autologous bone marrow–derived MSCs were tested. It demonstrated greater genomic instability and indicators of senescence at P3 when compared to bone marrow mononuclear cells. 8 Markers of cell cycle, DNA replication, and mismatch repair properties, were downregulated in bone marrow MSCs at P3. As a result, researchers and clinicians recommend the implementation of autologous MSCs to be used before P3. However, these observations need to be verified in human cartilage samples, one of the objectives of this study.
During the process of MSC recruitment, migration, proliferation, and condensation, there are a plethora of transcriptional factors that play an important role in regulating chondrogenesis (
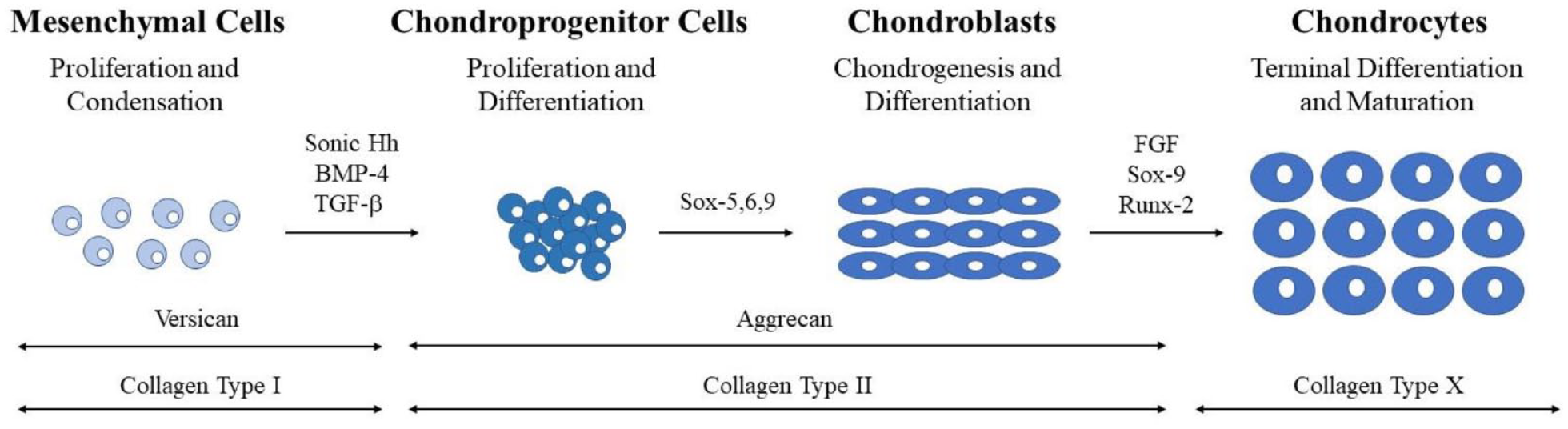
Sequence of events during chondrocyte development. Stages categorized by cell type and represented schematically. Transcription and differentiation factors are listed above the arrows between the histological representations of the cells. ECM proteins are listed above double-headed arrows at the base of the figure.
It is well known that type II collagen is a main component of cartilage. 11 More specifically, Valcourt et al. 12 demonstrated that transforming growth factor (TGF)-β1 is associated with the expression of procollagen form COLII alpha-1. Acting as one of the earliest markers of chondrogenesis, Sox9 is integral for the expression of the type II collagen (COLII) gene, and the suppression of runt family transcription factor 2 (Runx2).13,14 Two additional Sox family members, L-Sox5 and Sox6 are involved in the initial regulation of chondrogenesis. Working in concert, the 3 Sox transcription factors activate the expression of several cartilage-specific genes in addition to COLII, with the goal of achieving mature chondrocytic phenotype. 15 As chondrogenesis is also an active process in the formation of bones, articular cartilage bound chondrocytes must also be under strict regulation by positive and negative feedback factors. Specifically, Sox9 negatively regulates maturation of early chondrocytes toward osteogenic differentiation, whereas L-Sox5 and Sox6 will prevent premature hypertrophy. 10
Terminal differentiation of chondrocytes marks the final, crucial step for mature cartilage development. A leucine zipper protein, cMaf, is expressed in late chondrocytes. 16 At this stage, hypertrophy of chondrocytes is common, marking terminal differentiation and causing cells to secrete angiogenic factors such as vascular endothelial growth factor (VEGF). 17 Primarily, these secretions are restricted to chondrocytes that will become ossified, or the few that lay close to subchondral bone. Deviations in angiogenic factors or their receptors typically result in a deficit in the growth and replacement of both cartilage and bone, tagging angiogenic factors as a key component of normal development and ossification. 17
In addition, the extracellular matrix (ECM) plays an integral role in determining and regulating chondrocyte development. The compositional changes of the stem cells’ extracellular environment, known as the stem cell niche, provide differing external signals for gene expression determining cell fate.18,19 Signaling pathways within the ECM such as BMP and fibroblast growth factor (FGF) are activated to dynamically control their composition. Specifically, the bioactivity of growth factors such as transforming growth factor β (TGF-β) and tumor necrosis factor α (TNF-α) are modulated by ECM components, resulting in proteolytic changes to the environment and phenotype. 20 The presence of COLII and aggrecan (AGG) characteristically mark differentiated chondrocytes, with deviations in expression or content acting as a viable measure of degradation or alterations in differentiation. 21
The objective of this research was to refine the isolation protocol of chondrocytes and further test the CPMs of these human articular cells for tissue engineering. Isolated from injured human knee joints, healthy chondrocytes from varying participants were cultured, passaged, and tested for markers of differentiation with the aim of enhancing clinical recommendations and determining differences in age and sex.
Materials and Methods
Participants and Surgical Procedures
The experimental protocol for this project involved participants from the Division of Orthopaedic Surgery at Kingston General Hospital, and it was approved by the Queen’s University Health Sciences Affiliated Teaching Hospitals Research Ethics Board (Protocol #: SURG-343-16).
Six participants were included in the analysis of this study, stratified by both sex and age. Specifically, 2 female and 1 male participants under the age of 40 were included in the one category, and conversely, 2 male and 1 female over the age of 40 in another. All participants underwent surgery to treat an articular cartilage surface injury of the knee joint.
Chondrocyte Isolation, Culture Condition, Passaging, and Freeze–Thaw Procedure
After injured edges were removed, cartilage samples were collected from healthy articular cartilages. The trimmed cartilage tissue was diced into 2 to 4 mm3 cubes ( Figure S-1 , Supplemental data). Tissue blocks were treated with 100 μg/mL streptomycin, 100 units/mL penicillin, 0.25 μg/mL amphotericin B, 25 mM HEPES, and 0.5% (w/v) protease in 20 mL of chondrocyte growth medium (Lonza Walkersville Inc., Walkersville, MD, USA) for 90 minutes. Growth medium was used to wash the samples 3 times before an 18-hour digestion in 15 mL of 0.15% collagenase A (w/v; Roche Diagnostics Canada, Laval, QC, Canada) in growth medium. Digestion protocols were carried out at 37°C with 95% humidity and 5% CO2. Once the digestion sequence was completed, tissues were passed through a 200-mesh filter (Sigma-Aldrich) to remove undigested material, and the filtrate centrifuged at 600g to collect pelleted chondrocytes. Trypan blue exclusion test was used to evaluate cell viability (~90%-95%).
Isolated chondrocytes were cultured and passaged according to the published protocol in calves 22 and humans 23 with minor modifications. Briefly, isolated chondrocytes were cultured in T25 flask at density of 5 × 105 cell/mL of high glucose Dulbecco’s Modified Eagle’s Medium (DMEM), containing 10% fetal bovine serum (FBS; HyClone Lab Inc., Logan, Utah, USA) and 1× antibiotic-antimycotic solution. Sample lots of FBS were evaluated for the support of human chondrocyte growth prior to purchase. Cell adhesion was assessed 24 hours later. Confluent monolayer chondrocytes were passaged, frozen, and thawed using previously published methods.24,25
Cell Collection, RNA Isolation, and cDNA Transcription
Cultured chondrocytes were maintained to either P3 or P7, and harvested at confluence for further evaluation (
Phase contrast microscopy illustrating chondrocytes in culture. Panel
Total RNA was isolated using a combination of Trizol (Tri Reagent, Molecular Research Center, Inc., Burlington, ON, Canada) and a high pure RNA isolation kit (Roche Scientific Co., Laval, QC, Canada). All wash and elution steps were carried out according to manufacturer’s instructions. RNA was measured using Nano-Drop 2000 spectrometer (Thermo Scientific, Wilmington, DE, USA).
Total RNA (100 ng) was reverse transcribed into cDNA using qPCRBIO cDNA synthesis kits (PCR Biosystems Inc., Wayne, PA, USA) with minor modifications. Briefly, sample RNA was pipetted to 15 µL of water before the addition of 4 µL of a 5× cDNA mix, containing oligo dT primers, and random hexamer deoxynucleotide triphosphate (dNTP) then used for the reaction. One µL of 20× RTase was added, containing a mixture of avian myeloblastosis virus (AMV) and RNase inhibitors. The reaction mixture solution was incubated at 42°C for 30 minutes.
Primer Design and qPCR Protocol
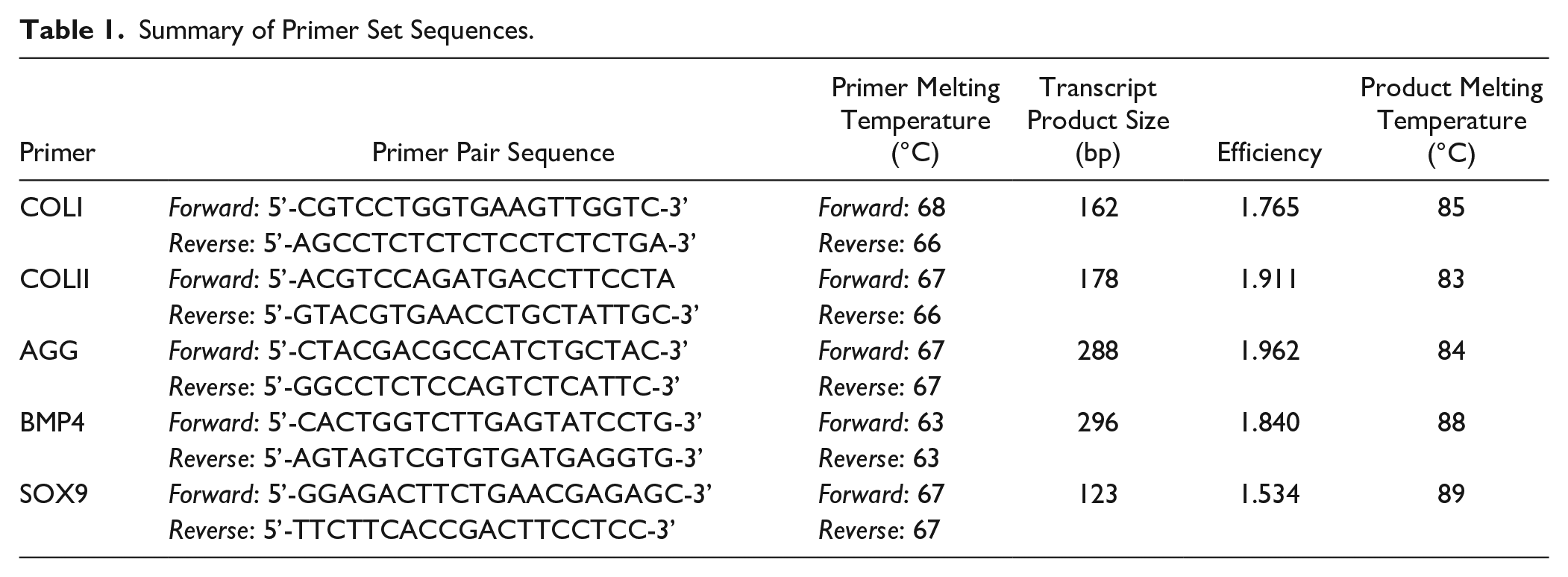
Oligonucleotide primers for collagen type I (COL1), collagen type II (COLII), and AGG genes were previously designed by Robinson 23 ( Table 1 ). In addition, new oligonucleotide primers were designed for BMP4 and SOX9 using the Primer Design 2.01 software (Scientific & Educational Software, Cary, NC, USA) with published mRNA sequences from the NIH GenBank (National Center for Biotechnical Information, Bethesda, MD, USA; www.ncbi.nlm.nih.gov/Genbank). The remaining primers for p53, PGR4, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were purchased as prevalidated primer sets from Integrated DNA Technologies, Inc. (Coralville, Iowa, USA).
Summary of Primer Set Sequences.
Levels of mRNA expression were measured using the standard-curve method for each gene with GAPDH as a reference gene. 23 All qPCR was performed using the LightCycler® 480 system II (Roche Scientific, Laval, QU, Canada), and all samples were run in triplicates.
Primer Verification
To ensure a single product was amplified during PCR, melt curve analyses were conducted for each primer set. The first derivative of the fluorescence versus temperature melt curves displays a Gaussian distribution; exhibiting a single peak that falls above 80°C. Typically, the primer dimers melt at temperatures between 70°C and 75°C, making them easily identifiable in the curve. All primer sets produced a Gaussian melt curve; however, COLII fluorescent expression levels were below the threshold needed for curve analysis.
Statistical Analyses
Data were standardized by taking a ratio of the target gene expression over the expression of GAPDH. All statistical analyses were performed, and graphs plotted using Prism 6.0 Software (GraphPad Software Inc., La Jolla, CA, USA). Target gene mRNA expressional data were compared by 2-way analysis of variance (ANOVA) with multiple comparisons using Tukey’s post hoc test. Data are presented as mean ± standard error of the means (SEM). Values of P ≤ .05 were considered statistically significant.
Results
Passage Comparison of CPM Gene Expression
Each of the primer sets designed and implemented in the study were grouped by passage and tested for differences in relative gene expression. Figure 3A shows a 7.4-fold decrease in AGG expression from P3 to P7.
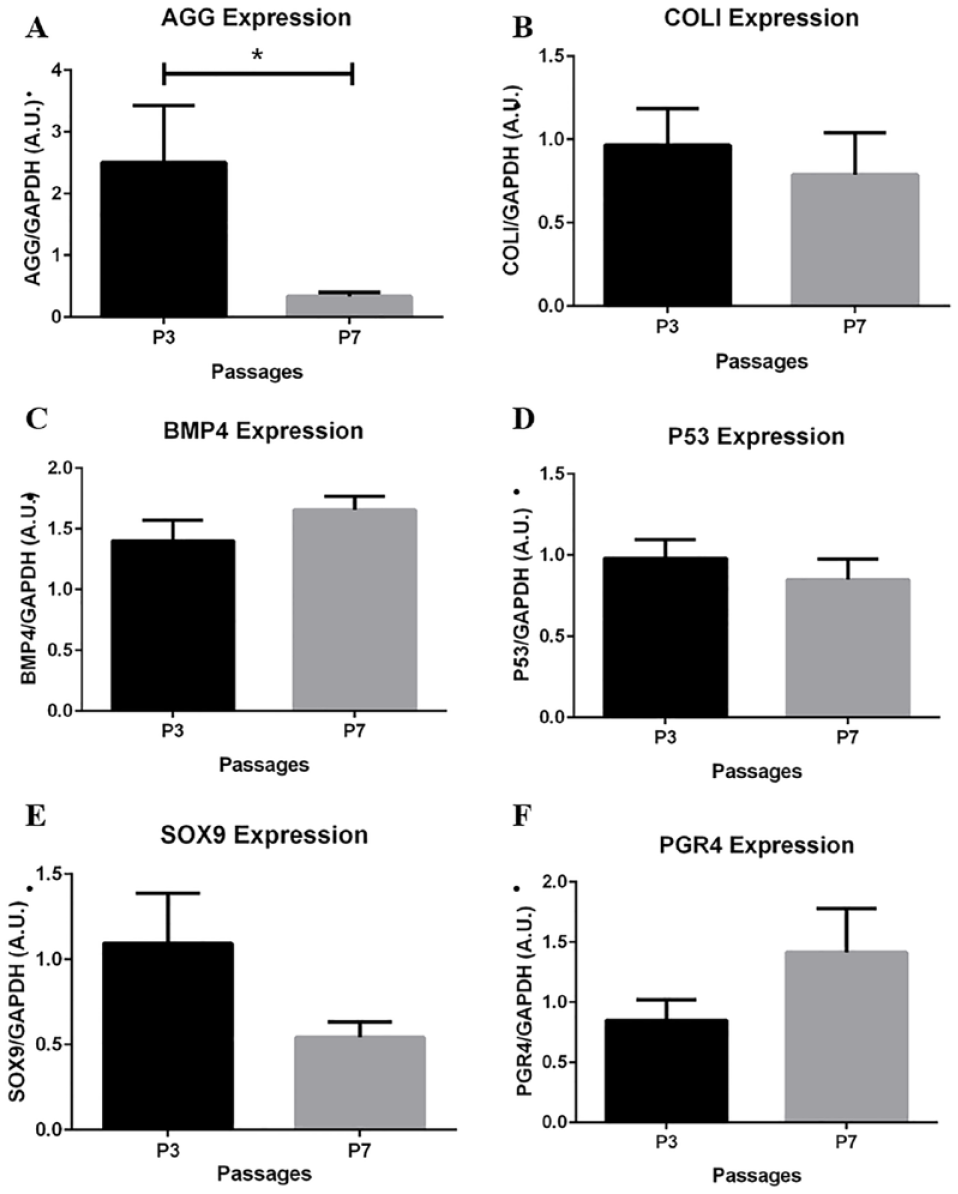
Passage comparison of chondrogenic potential marker (CPM) gene expression. (
The remaining genes maintained a relative amount of expression from P3 to P7, with no significant differences. Specifically, comparing P3 to P7, a 2-fold decrease in SOX9 expression (
Age Comparison for CPM Gene Expression
Each chondrocyte culture and resulting gene expression profile underwent subgroup analysis by age comparing under (<40) or over (>40) 40 years. Significant changes in AGG, SOX9 and PGR4 expression demonstrated dedifferentiation of chondrocytes from patients <40 compared to >40.
No significant difference was found between age groups after analysis of pooled chondrocytes. However, Figure 4B shows a significant 23-fold decrease in AGG expression within the <40 group between P3 and P7 (P < .01). No significant difference was found within the >40 group.
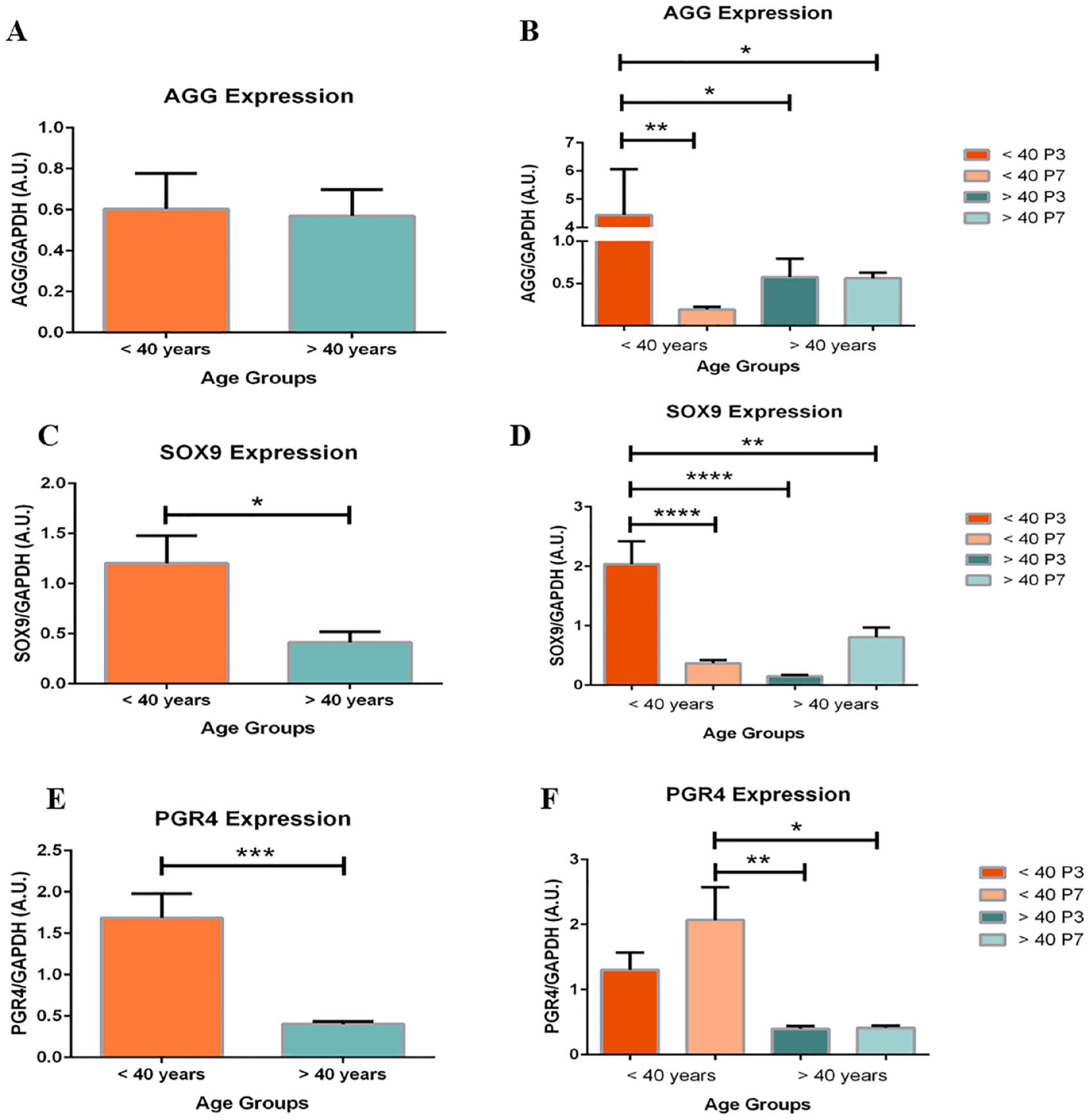
Age-based expression of aggrecan (AGG) expression (
Chondrocytes in the >40 groups expressed significantly less SOX9 than chondrocytes in the <40 age group (
PGR4 expression also differed between and within age groups.
Figure 4E
and
COL1 and P53 Expression, and Maintenance of BMP4 Expression between Age Groups
COLI and P53 expression demonstrated an inverse relationship between age groups. In the <40 age group, COLI (33-fold change; P < .01) and P53 (2.8-fold change; P < .001) expression significantly decreased when comparing P7 to P3 chondrocytes (
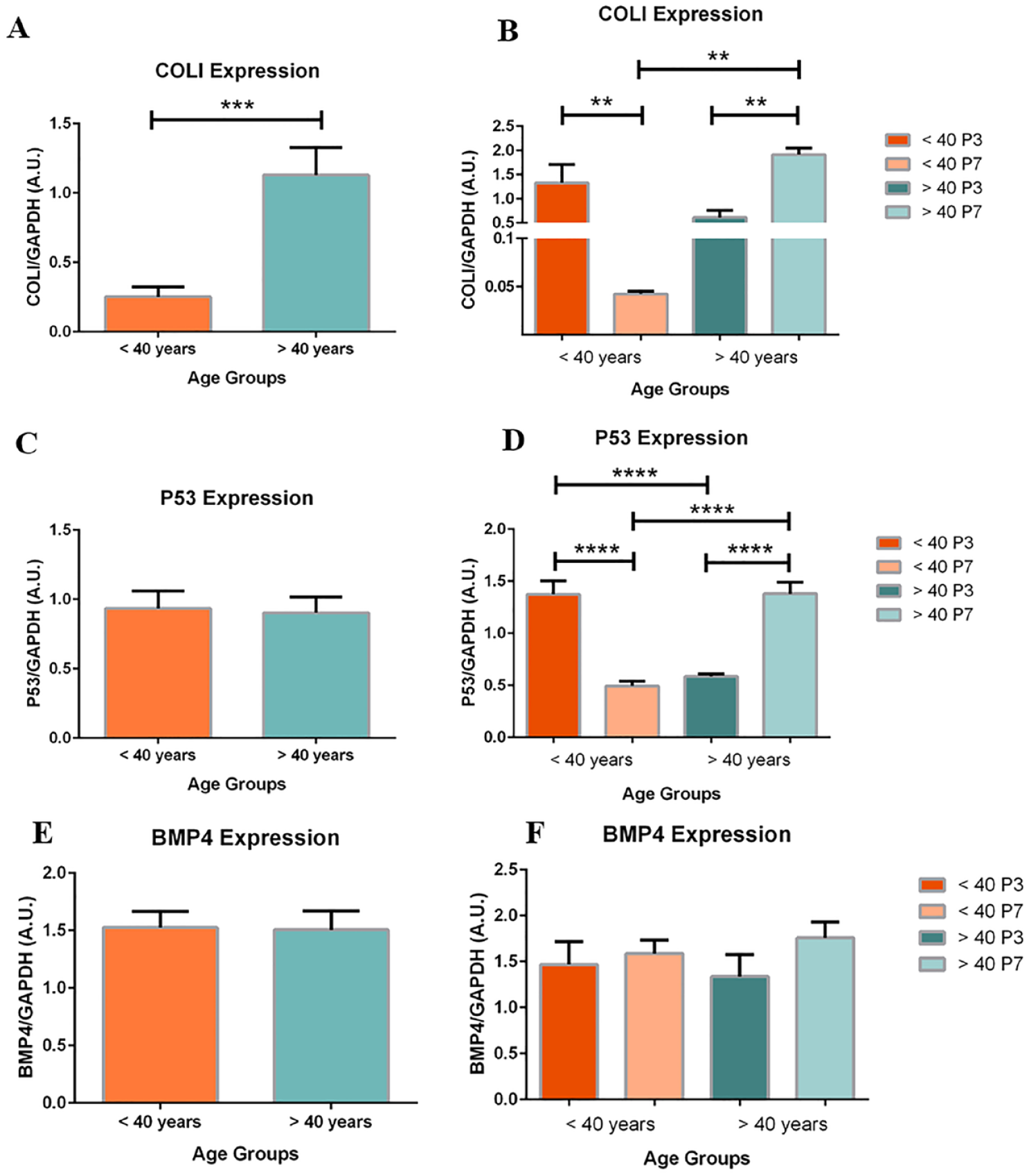
Age-based expression of COLI (
Subdivided by passage, changes in the expression of COLI and P53 in the <40 group were similar to profiles of dedifferentiating chondrocytes, while changes in expression in the >40 group were resembling to profiles of mature articular chondrocytes.
When stratified by passage number and pooled between groups, no significant differences were found in BMP4 expression (
Sex Comparison for CPM Gene Expression
When assessed by sex, significant changes in COLI, AGG, and SOX9 expression demonstrate dedifferentiation of chondrocytes from male patients compared to female.
COLI expression in pooled male chondrocytes was 1.6-fold greater than pooled female chondrocytes (
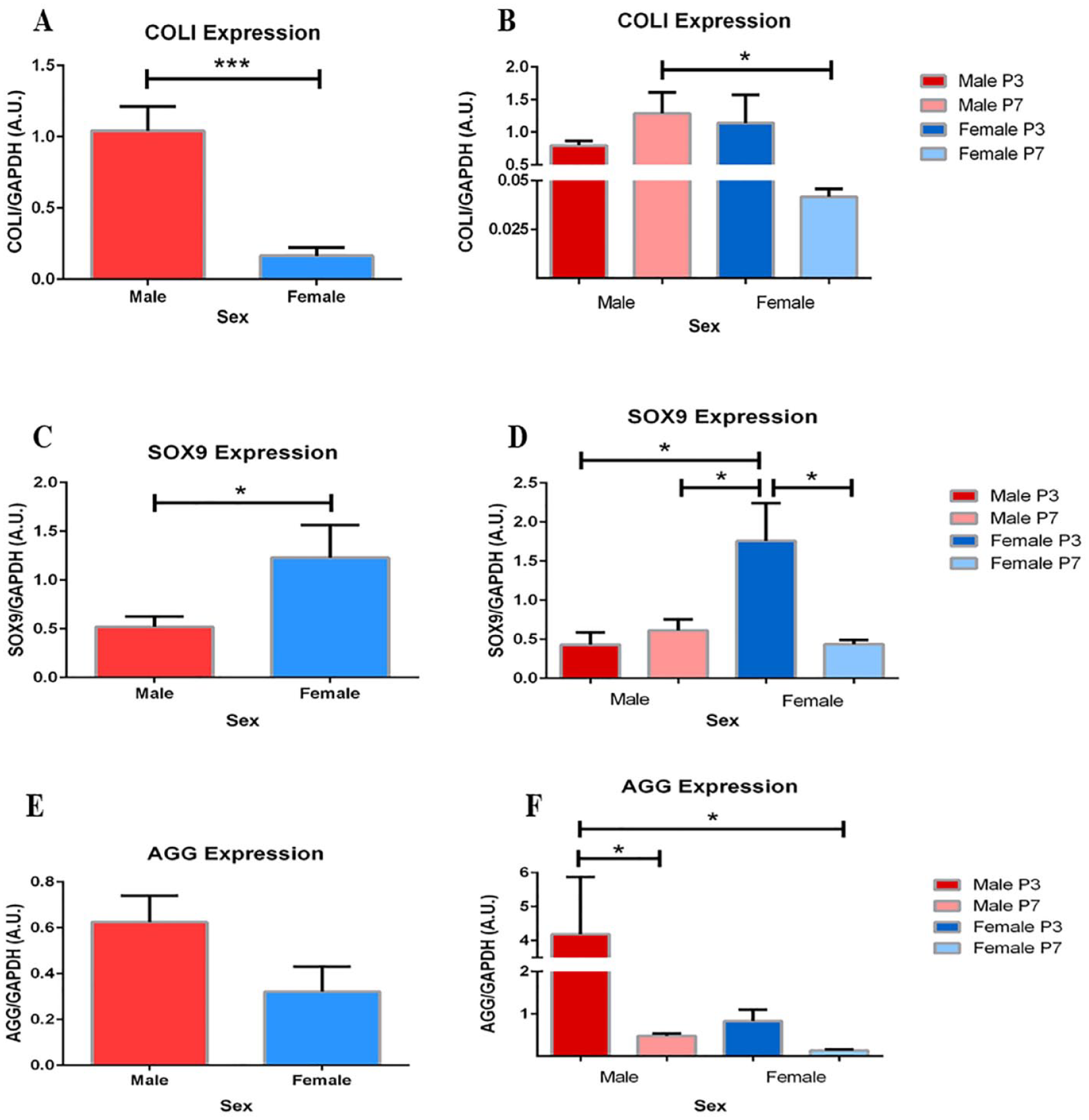
Greater expression of COLI in male chondrocytes. (
Similarly, pooled samples of male chondrocytes express significantly less SOX9 than pooled samples of female chondrocytes (
Finally, observable and significant differences in AGG expression exist between and within sex groups, respectively (
Expression of BMP4, P53, and PGR4 between Sexes
Comparisons between sexes for pooled and subdivided data did not reach significance in expression of BMP4, P53, or PGR4. From a passage perspective, expression of BMP4, P53, and PGR4 were maintained from P3 to P7 (
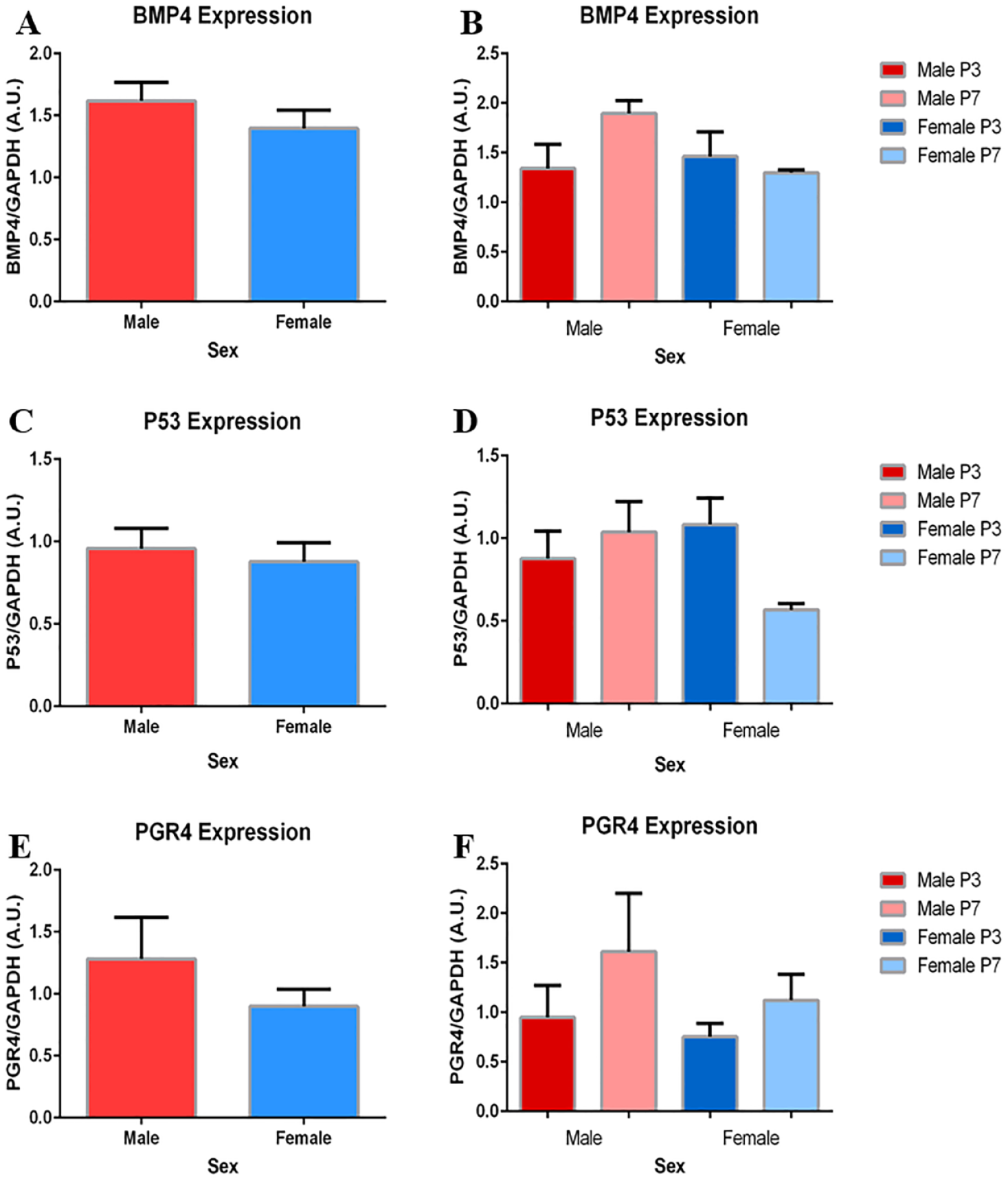
No significant differences in BMP4, P53, or PGR4 expression exist between sexes. (
Discussion
General Comments
Based on guideline of the Primer Verification listed in the Materials and Methods section, COLII measurements were excluded from analysis. This is considered to be acceptable for 3 reasons: (1) there are accepted, alternative CPMs for mature chondrocytes including AGG which was studied, (2) all fluorescent expressions were standardized to GAPDH and the resulting limited expression of COLII would not allow for significant analysis, and (3) considering the relationship between SOX9 and COLII, the limited expression of COLII may be a factor of the limited SOX9 expression and therefore, a potential correlation.
Comparison of CPM Gene Expression between Passages
AGG is one of many proteoglycans critical for proper functioning of articular cartilage. The function of AGG specifically is characterized by its ability to link hyaluronan, forming large proteoglycan aggregates. These aggregates fill the intrafibrillar space, providing mature articular cartilage with the osmotic pressure needed to resist compressive forces.
26
Of the 6 CPM genes tested, significant differences were found only in expression of AGG between P3 and P7 (
AGG gene expression decreased 7.4-fold in P7 cells compared to P3, suggesting lesser proteoglycans in P7 cultures and possibly the dedifferentiation of chondrocytes. As chondrocytes grow to confluence and progress through passages, changes in phenotypic gene expression can be accepted mainly as a result in change of the extracellular environment or niche.18,19 As a result, proteoglycan gene expression is higher in early chondrocyte passages in efforts to secrete more matrix and establish a suitable environment for growth and function. Thus, the literature has demonstrated the presence of AGG, and measurements of a high ratio of AGG to versican, characteristic of mature articular chondrocytes. 21
In addition to AGG, non-significant yet observable differences in ECM factors and components may provide support for chondrocyte dedifferentiation, although contradictions exist. Beginning with collagen fibers, the expression of COLII/COLI is an alternative, accepted ratio characterizing mature articular cartilage.21,27 In this study, COLII measurements were excluded due to indiscernible fluorescence expression and no significant changes in COLI expression were observed between passages (
As mentioned earlier, SOX9 is a transcription factor required for the expression of the COLII gene, characterizing condensation and maturation of articular cartilage chondrocytes.28,29 Although not significant, the 2-fold decrease in SOX9 expression in P7 chondrocytes may suggest a decrease in COLII production and a relatively lower ratio of COLII/COLI (
Conversely, the maintenance of P53 and BMP4 expression suggests that dedifferentiation of chondrocytes throughout passaging is much less likely. P53 is a tumor suppressor gene functioning to halt the induction of pluripotent cells. 29 Without significant change in P53 gene expression, relative suppression levels of stem-like cell potential should remain constant, limiting the reversion of chondrocytes. Contrary to the interpretation of P53, BMP4 expression marks the acceleration of chondrocyte differentiation and maturation.
In addition, observable increases in PGR4 gene expression contradict the interpretation of AGG expression. Referring to a group of proteoglycans, the SZP is a major component of the PGR4 group. Produced exclusively by specialized chondrocytes in the superficial zone of articular cartilage, measurements of SZP/PGR4 expression suggest the presence of mature, superficial articular chondrocytes. 30 A 1.6-fold increase in PGR4 expression may suggest the increase in SZP protein content and the maturation of P7 chondrocytes instead of their dedifferentiation. Overall, the results suggest that cultured articular chondrocytes maintain CPM gene expression and reserve their phenotype up to P7.
Comparison of CPM Gene Expression between Age Groups
Each gene expression profile was compared strictly between age groups, then subdivided by passage to account for significant or influential differences such as AGG gene expression. Similar to passage comparisons, interpretation of CPM genes alluded to similar signs of dedifferentiation, contradicting expression, or relative levels of expression between age groups.
Although not significant in pooled data, in the <40 group, P7 chondrocytes expressed 23-fold less AGG than P3 chondrocytes (
However, age-based expression of COLI and P53 present contradicting results, limiting the interpretation of individual gene expression as markers of differentiation. In the <40 group, COLI expression decreased 33-fold, and P53 decreased 2.8-fold from P3 to P7 (
Finally, BMP4 gene expression did not change within, nor was there a significant difference between age groups (
Comparison of CPM Gene Expression between Sexes
Stemming from the prior discussion on cartilage disease and injury repair, OA is known to affect significantly more females than males over the age of 60.34,35 Considering age as the primary risk factor for OA, and sex as secondary, there are 2 important factors to acknowledge before interpreting sex results. First, it is important to note and integrate the prior relationships discovered in age-based analyses in disease interpretation of sex results. Second, sex differences should also be viewed as an independent variable, with summed results minimizing the effect of both passage and age.
The established ratios expressing mature chondrocytes such as COLII/COLI, and the relationship between SOX9 and COLII expression are highlighted in sex-based comparisons of chondrocytes. Male chondrocytes express significantly more COLI (
On the contrary, significant changes in AGG and lack of change in remaining genes further limit the evaluation of chondrogenic potential between sexes. Building from the expression of COLI and SOX9, male chondrocytes would be expected to have less AGG than female chondrocytes, further supporting dedifferentiation. In Figure 6F , male chondrocyte expression of AGG was shown to significantly decrease from P3 to P7 (8.9-fold change). In addition, summed data show a noticeably greater, though not significant, expression of AGG in males than females. Decreased expression of AGG in higher passaged, older chondrocytes may help to explain the initial drop off in AGG expression seen in P7, though the overall higher mean remains contradictory.
Finally, the lack of significant difference between sexes in BMP4, P53, and PGR4 expression can be interpreted as chondrogenic potential similarities between sexes. Taken together, the differences presented in COLI and SOX9 between sexes may suggest the retention of chondrogenic potential in male chondrocytes. In relation to function and disease, the ability of male chondrocytes to dedifferentiate and alter phenotype may present a compensatory mechanism. Having such a mechanism in place may aid in the injury repair cycle, the restoration of lost proteoglycans, and the production of COLII and SZP, preserving cartilage function. Thus, in association with age results, older female chondrocytes may be more prone to dysfunction and disease due to their loss of CPMs.
Limitations and Future Directions
Small sample size is one factor that may pose limitation to these study results. In addition to being subdivided into small group comparisons (n = 3) based on age and sex, the limited number of samples overall may skew the finding of significance. Although a total of 6 cell cultures is a suitable number for analysis, an increase in overall sample number or employment of alternative techniques may help to confirm the findings.
The overall extent of the analysis performed should be extended to confirm cell type and composition, as well as specific protein levels. Expression of selected CPMs present only in chondrocyte populations is a crucial aspect of this study, suggesting the isolation of chondrocytes in culture. However, a histological examination of chondrocytes paired with immunohisto-chemically staining techniques would provide foundational support in addition to the collagenase isolation for the purity of the chondrocyte cultures.
Despite these limitations, there are some potential future applications for these results. Focused primarily on cartilage tissue engineering, the characterization of passage, age, and sex differences may provide an aid for optimizing chondrocyte isolation, freezing, and culturing techniques. Prior studies have attempted to optimize cartilage tissue engineering, culturing autologous chondrocytes up to 4 weeks before successful implementation. 5 In addition, the same research group identified the approximate number of cells needed to engineer specifically sized tissue. 4 Together, the culturing of only 20,000 cells was efficient in producing a 3 cm2 cartilage construct, expressed mature cartilage genes, and became biomechanically sound.
Integrating results of this study, passage-based analysis would suggest that isolated chondrocytes could grow and passage up to 7 times without expressing signs of dedifferentiation or loss of crucial cartilage phenotype. The maintenance of chondrogenic potential may allow researchers and clinicians more variability and growth time, optimizing the type of tissues for implantation regardless of cell count or final size. However, younger, male chondrocytes are more likely to dedifferentiate compared to older, female chondrocytes, favoring their use in the culturing, engineering, and implantation process. Taken together, researchers and clinicians can make more sound judgments in isolating chondrocytes, opting to freeze younger chondrocytes for future implantation.
Conclusion
The results and subsequent discussion suggest there is maintenance of CPMs throughout passages, and loss of chondrogenic potential in female and older chondrocytes. After analysis of 6 CPM genes, only AGG gene expression decreased from P3 to P7, suggesting the maintenance of chondrogenic potential up to P7. In addition, changes in AGG, SOX9, and PGR4 expression between age groups suggest that chondrocytes will lose their chondrogenic potential with age. Although significant differences in remaining genes were not found, the interplay between AGG, SOX9, and PGR4 align with known changes in aging and osteoarthritic cartilage. Finally, sex-based analysis suggests that chondrogenic potential is not conserved between sexes. Specifically, significant changes in COLI and SOX9 expression present a profile of dedifferentiation in cultured male chondrocytes.
Despite the limitations of the study, interpretations of the results reflect careful consideration for confounding factors and provide a step forward in the characterization of chondrogenic potential in culture. Researchers and clinicians could use resulting considerations of passage, age, and sex differences to better inform and optimize methods for isolation, culturing, and engineering of autologous cartilage structures.
Supplemental Material
sj-pptx-1-car-10.1177_19476035241241930 – Supplemental material for Expression of Chondrogenic Potential Markers in Cultured Chondrocytes from the Human Knee Joint
Supplemental material, sj-pptx-1-car-10.1177_19476035241241930 for Expression of Chondrogenic Potential Markers in Cultured Chondrocytes from the Human Knee Joint by John-Peter Bonello, M. Yat Tse, Trevor J. G. Robinson, Davide D. Bardana, Stephen D. Waldman and Stephen C. Pang in CARTILAGE
Footnotes
Acknowledgements and Funding
DDB, SDW, and SCP acknowledged the financial support on this research project through the Physician Services Inc. (PSI) Foundation at the Kingston General Hospital, Kingston, Ontario, Canada. The Canada Foundation for Innovation (CFI) provided funding for the purchase of the PCR equipment to Drs. Brian Amsden, Stephen D. Waldman, and Stephen C. Pang. JPB and TJGR thanked the Queen’s Graduate Award (QGA) and the Teaching Assistantship programs for financial support on their MSc program at Queen’s University.
Contributions of Authors
Experimental Design: JPB, MYT, DDB, SDW, SCP.
Primer Design: JPB, TJGR, MYT.
Sample Collection and Cell Isolation: DDB, SCP.
Cell Culture and Passaging: JPB, SCP.
Sample Processing and qPCR Analyses: JPB, MYT.
Manuscript Preparation and Editing: JPB, MYT, TJGR, DDB, SDW, SCP.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This research was approved by the Queen’s University Health Sciences Affiliated Teaching Hospital Research Ethics Board; Protocol #: SURG-343-16.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
