Abstract
Objective
The zonal properties of articular cartilage critically contribute to the mechanical support and lubrication of the tissue. Current treatments for articular cartilage have yet to regenerate this zonal architecture, thus compromising the functional efficacy of the repaired tissue and leading to tissue degeneration in the long term. In this study, the efficacy of zonal cartilage regeneration through bilayered implantation of expanded autologous zonal chondrocytes was investigated in a porcine chondral defect model.
Design
Autologous chondrocytes extracted from articular cartilage in the non-weight bearing trochlea region of the knee were subjected to an expansion-sorting strategy, integrating dynamic microcarrier (dMC) culture, and spiral microchannel size-based zonal chondrocyte separation. Zonal chondrocytes were then implanted as bilayered fibrin hydrogel construct in a porcine knee chondral defect model. Repair efficacy was compared with implantation with cell-free fibrin hydrogel and full thickness (FT) cartilage-derived heterogenous chondrocytes. Cartilage repair was evaluated 6 months after implantation.
Results
Sufficient numbers of zonal chondrocytes for implantation were generated from the non-weight bearing cartilage. Six-month repair outcomes showed that bilayered implantation of dMC-expanded zonal chondrocytes resulted in substantial recapitulation of zonal architecture, including chondrocyte arrangement, specific Proteoglycan 4 distribution, and collagen alignment, that was accompanied by healthier underlying subchondral bone.
Conclusion
These results demonstrate that with appropriate expansion and isolation of zonal chondrocytes, the strategy of stratified zonal chondrocyte implantation represents a significant advancement to Autologous Chondrocyte Implantation-based cartilage regeneration, with the potential to improve the long-term integrity of the regenerated tissues.
Keywords
Introduction
The zonal organization of articular cartilage is critical for the biphasic mechanical properties of the tissue to provide lubrication and load transmission during the joint motion. Articular cartilage can be separated into superficial zone (SZ), middle zone (MZ), and deep zone (DZ).1,2 Across the zones, they are different in terms of chondrocyte morphology and size, collagen organization, and extracellular matrix composition. In SZ, the chondrocyte is relatively small and has an elongated morphology. The collagen in the SZ is tightly packed and aligned parallel to the articulating surface, providing high tensile strength at the articular surface, 3 and act as a permeability barrier facilitating generation of fluid load support in the underlying zones.4,5 Proteoglycan 4 (PRG4), secreted specifically by SZ chondrocytes, acts as lubricant to provide frictionless joint motion. 6 Going deeper into the articular cartilage, the chondrocytes increase in size and have rounded morphology. MZ has randomly organized collagen fibrils, while the collagen alignment in DZ is perpendicular to the articulating surface and this alignment was shown to be important in resisting the compressive force in the deeper zone.7,8 The collagen fibrils in deep zone are anchored to the subchondral bone, securing the cartilage to the underlying bone to maintain the structural integrity of osteochondral unit.3,9 The high concentration of Type II Collagen (Col2) and Aggrecan (Agg) in the MZ and DZ endows the tissue with a high osmotic pressure to withstand the compressive force. Minor proteins such as cartilage oligomeric matrix protein (COMP) and Type IX Collagen (Col9) are also present specifically in the deeper zones to regulate the collagen fiber size, inter-fiber cross linking and interaction with Agg.10,11
The avascular nature of articular cartilage has limited its self-healing capability. Autologous Chondrocyte Implantation (ACI) is the only cell-based therapy currently approved by FDA for treatment of injury-induced articular cartilage lesions. Although the short-term outcomes of ACI treatment showed improved tissue regeneration, the integrity of the regenerated tissues degenerated in the long-term. 12 One of the possible reasons could be the implantation of a heterogeneous mixture of chondrocytes which may not have the ability to reproduce the zonal architecture of normal cartilage. Zonal chondrocyte subpopulations were shown to crosstalk and influence each other’s phenotypes. The presence of SZ chondrocytes increased the proliferative and biosynthetic activities of DZ chondrocytes.13 -15 On the other hand, the presence of DZ chondrocytes decreased the GAG secretion of SZ, 15 while upregulating the secretion of PRG4 in SZ chondrocytes. 13 Recapitulation of zonal architecture in regenerated tissues has thus been hypothesized to improve the quality of regenerated cartilage.16,17
Various efforts have been attempted to recreate the zonal architecture of articular cartilage employing heterogenous chondrocytes with the use of multi-layered composite scaffold that mimic the structural design, chemical cues, and mechanical characteristics of mature articular cartilage to guide the morphology, orientation, and phenotypic state of chondrocytes. Polymer scaffolds fabricated with pore-size gradients was shown to promote the anisotropic cell distribution and matrix contents similar to that in superficial, middle, and deep zones of articular cartilage. 18 Hybrid hydrogel scaffold with electrospun nanofiber mesh of different orientation was employed to stimulate cellular morphology and ECM contents assembling zonal cartilage. 19 A multi-zonal nanocomposite scaffolds that mimics the distinct collagen orientation and matric components of zonal cartilage support the growth of tissue that are reminiscent of the natural analogue. 20 3D bio-printed scaffold with different cell densities mimicking cell gradient of the human articular cartilage zones were also studied. 21 On the other hand, formation of stratified tissue constructs with zonal chondrocytes require a relatively simpler scaffold design due to their intrinsic zonal phenotypes.16,17 Zonal chondrocytes were utilized to form multi-layered cartilage constructs via layer by layer seeding of zonal chondrocytes at high density on the hanging well. 22 The multilayer architecture of the constructs was able to be maintained after 4 weeks of culture in vitro.23,24 Multilayer agarose hydrogel of different concentrations14,25 and photo-crosslinkable hydrogel such as poly(ethylene oxide) diacrylate (PEODA)15,26 containing zonal chondrocytes were also investigated to create multi-layered cartilage constructs. The tissue constructs formed depth-dependent ECM content and mechanical strength,14,25 as well as a stronger mechanical property than single layered hydrogel contained only DZ chondrocytes. 15 However, the lack of an efficient zonal chondrocyte isolation protocol has hindered the clinical implementation of stratified zonal chondrocyte implantation. 16 Current methods of zonal tissue segregation are manual, labor-intensive, and lack specificity. Furthermore, the limited number of autologous chondrocytes available from patients has entailed prolonged chondrocyte expansion that has led to zonal phenotype and functional loss. 27 A more efficient zonal chondrocyte isolation method, couples with a chondrocyte expansion protocol that maintain the zonal phenotype of the expanded cells is thus critical to realize the regeneration of articular cartilage zonal architecture through stratified implantation of zonal chondrocytes.
Toward this end, an inertial spiral microchannel device that performed high throughput label-free size-based cell separation was demonstrated to segregate and enrich zonal chondrocytes, derived from full thickness (FT) weight bearing (WB) articular cartilage tissue.
17
Delivery of the unexpanded zonal chondrocytes as bilayered fibrin hydrogel constructs in a proof-of-concept rat osteochondral defect study resulted in superior cartilage regeneration with mechanically enhanced tissues, in comparison to the implantation of FT cartilage-derived heterogenous chondrocytes.
17
A post-expansion cell sorting protocol was further developed that coupled dynamic microcarrier (dMC) expansion of FT chondrocytes followed by spiral microchannel sorting (
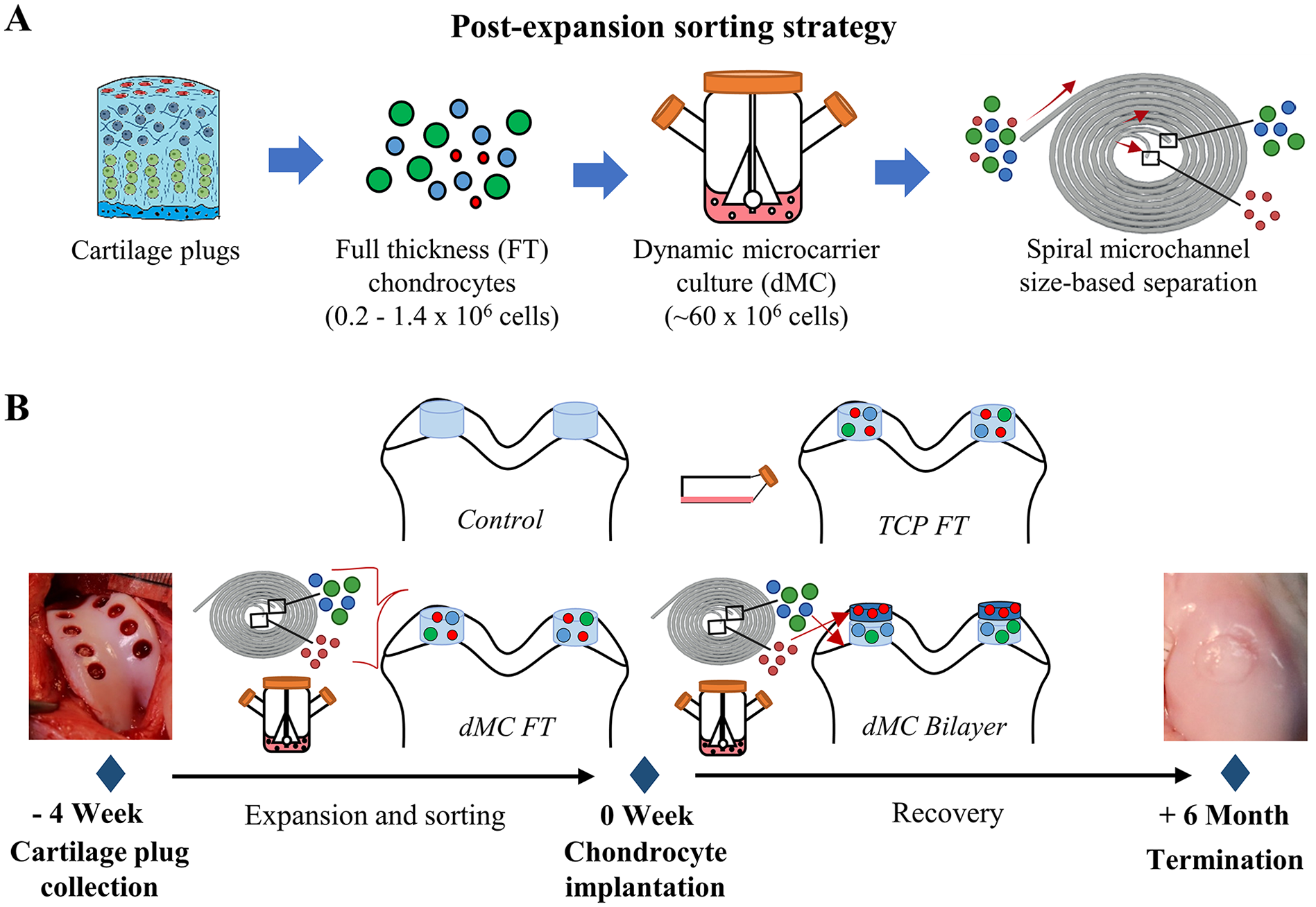
(
Methods
Primary Chondrocyte Isolation and Culture
WB and NWB chondrocytes were isolated from the femoral condyle and trochlea of articular cartilage, respectively, of 11 to 13 months old micropigs (PWG Genetics Pte Ltd, Singapore). Diced cartilage was digested in 0.25% (w/v) type-II collagenase overnight at 37°C. Isolated chondrocytes were sorted using the inertial spiral microchannel device, or expanded on tissue culture plate (TCP) for 1 passage. FT chondrocytes were seeded with an initial cell density of 5000 cells/cm2 in a complete media containing low glucose Dulbecco’s modified Eagle Medium (DMEM) supplemented with 10% (v/v) fetal bovine serum (FBS), 1% GlutaMAX and 1% (v/v) penicillin-streptomycin at 37°C in 5% CO2 atmosphere. All the reagents mentioned were purchased from Thermo Fisher Scientific.
Dynamic Microcarrier Culture
Cytodex® 1 microcarriers (Sigma-Aldrich) were prepared according to the manufacturer’s protocol. Microcarriers were added to Corning® ProCulture® 125 ml glass spinner flask (Sigma-Aldrich) at a density of 10,000 microcarriers/ml in a total volume of 50 ml. TCP-passage 1 chondrocytes were seeded on microcarriers at an initial cell seeding density of 5000 cells/cm2. In the first 6 hours, microcarriers and chondrocytes were cultured under intermittent stirring at 25 rpm for 2 minutes for every 30 minutes. The culture was kept static in the next 18 hours before continuous stirring regime at 60 rpm. The dynamic microcarrier culture was maintained for 2 passages at 8 days each. Chondrocytes were harvested with 0.1% (w/v) pronase solution (Sigma-Aldrich) and separated from microcarriers by passing through a 100 µm strainer (BD Bioscience, USA). Cell preparation was observed under microscope to ensure all the microcarriers were removed.
Chondrocyte Sorting With Inertial Spiral Microchannel Device
The schematic diagram of microchannel device and the illustration of the zonal chondrocyte distribution in the microchannel is shown in
Chondrocyte Re-Differentiation in Fibrin Hydrogel
Chondrocytes were suspended in 50 mg/ml fibrin provided in Fibrin Sealant Tisseel Kit (Baxter International Inc., USA). An equal volume of 10 IU thrombin solution was added to initiate gelation. The final cell density was 10 million cells/ml. The cell-hydrogel constructs were cultured for 3 weeks in the chondrogenic medium made of high glucose DMEM supplemented with 10-7 M dexamethasone, 1% ITS+ premix, 50 µg/ml ascorbic acid, 1 mM sodium pyruvate, 0.4 mM proline and 10 ng/ml of TGF-β3 (R&D System, USA).
Real-Time Polymerase Chain Reaction (qPCR) Analysis
Total RNA was extracted with the RNeasy® Mini Kit (Qiagen, Germany) and reverse transcription was performed using iScriptTM cDNA synthesis kit (Bio-Rad, USA). Real-time PCR was carried out using POWER SYBR® green PCR master mix on ABI 7500 Real-time PCR System (Applied Biosystem, USA) at 95°C for 10 minutes and 40 cycles of amplification composed of 10 minutes of denaturation at 95°C and 1 minute extension at 60°C. The gene expression level was normalized to the commonly used reference gene, glyceraldehyde-3-phosphate dehydrogenase (GAPDH)30,31 and was calculated using the formula, 2-∆∆Ct, with reference to undifferentiated porcine mesenchymal stem cells.
Histological and Immunohistochemical Staining
Hydrogel constructs were fixed in 10% neutral buffered formalin (Sigma-Aldrich) overnight at 4°C followed by graded ethanol dehydration and paraffin embedding. Cartilage tissues were decalcified before graded ethanol dehydration. Decalcification was performed at room temperature in 30% formic acid for 4 weeks with weekly change of decalcification solution. Samples were cut into 5 µm section using microtome (Leica, USA). Proteoglycan was stained with 0.1% Safranin O Solution (Acros Organics, USA) and counterstained with 0.02% Fast green solution (Sigma-Aldrich) and Accustain® Harris hematoxylin (Sigma-Aldrich). Immunohistochemical staining was performed to identify Col2, Type I Collagen (Col1), PRG4 and Col9 using Type II collagen mouse monoclonal antibodies (Clone 6B3 at 1:500 dilution, Chemicon, USA), Type I Collagen mouse monoclonal antibodies (1:500 dilution, Sigma-Aldrich), Lubricin rabbit polyclonal antibodies (ab-94933 at 1:100 dilution, Abcam, UK) and Type IX Collagen mouse antibodies (D1-9, B3-1 at 1:1000 dilution, Chondrex Inc., US). For Col2, Col1 and Col9 immunohistochemical staining, tissue sections were subjected to pepsin digestion for antigen retrieval. For PRG4 immunohistochemical staining, heat antigen retrieval was performed in sodium citrate buffer (10 mM sodium citrate, 0.05% Tween 20, pH 6.0). The Col9 and PRG4-positive cell were quantified manually from 3 hydrogel samples per group and 3 areas in each sample. The data were presented as the percentage of the positively stained cell.
Polarized Light Microscopy
Cartilage tissue sections were digested with 1 µg/ml of pepsin at 37°C for 30 mins and 4°C for the next 12 hours. The sections were then dehydrated and mounted. Visualization of collagen fiber orientation was carried out with Phoenix 5.0 MP Polarization camera (Lucid Vision Labs Inc., Canada) using circular polarization and filtered red light as input light.
Compression Test
Compression test was carried out using Instron tester 5567 (Singapore) at a compression rate of 0.01 mm/s with a 5 mm indenter to 50% strain for hydrogel constructs, or a 1 mm indenter to 30% strain for cartilage tissues. The compression tests were performed at 3 different locations on the cartilage defects. The strain was calculated using the formula, e = 1—L/L0 where L0 and L was the thickness of the hydrogel constructs before and after compression. The thickness of the cartilage before compression was 1 mm. Youngs’ modulus was calculated using the formula, E = r/e, where r and e represent the stress and strain of the samples, respectively, derived from the stress-strain curve generated from the Bluehill software.
Tribology Test
The frictional properties of hydrogel constructs and cartilage tissues were measured using a nanotribometer (Anton Paar) with a sphere cantilever (spring constant: 7.5 N/m) and a 5 mm spherical borosilicate glass tips (Nova Scan). The measurement was performed with 20 cycles of linear movement at 3 different locations. The cantilever contact load was 0.2 mN and the sequence was performed at maximum linear speed of 0.15 mm/s with 1 mN normal load. The coefficient of friction was provided by the Anton Paar software (Version 8.0.20) and was calculated as the slope of friction force over the applied normal force.
Animal Experiments
All the procedures were performed according to the Institutional Animal Care and Use Committee (IACUC) at the National University of Singapore (Protocol number: R17-1410). In all, 11 to 13 months old male micropigs were divided into 4 groups: 1) defect with empty fibrin hydrogel implantation (Control), number of animal (n) = 10; 2) defect with fibrin hydrogel containing autologous TCP expanded FT chondrocytes (TCP FT), n = 10; 3) defect with fibrin hydrogel containing autologous microcarrier expanded FT chondrocytes (dMC FT), n = 12; 4) defect with bilayered fibrin hydrogel (SZ chondrocytes overlaying MZ/DZ chondrocytes) containing autologous zonal chondrocytes from E/S strategy (dMC Bilayer), n = 12. The timeline of animal experiments is shown in
Figure 1B
. In the first surgery, 8 cartilage plugs (3 mm diameter, 1-2 mm depth) were collected from the NWB region of the trochlea in left knee of each animal with a 3 mm biopsy punch. Collected NWB cartilage plugs were processed to isolate chondrocytes and the isolated chondrocytes were expanded with/without size-based sorting. In the second surgery, critical size bicondylar chondral defects (6 mm diameter, 1 mm depth) were created at the WB regions of the lateral and medial femoral condyles in right knee of each animal with a 6 mm biopsy punch, resulted in 2 defects in each animal. 10 defects were created for Control and TCP FT groups while 12 defects were created for dMC FT and dMC Bilayer groups. Penetration to the subchondral bone was avoided to prevent bleeding. Same implantation was performed on both defects of each animal. In each defect, total volume 50-60 µl fibrin hydrogel with or without cells was delivered. For Group 4, 30 µl of fibrin hydrogel encapsulating MZ/DZ chondrocytes was first delivered to the defect and overlaid with 30 µl of fibrin hydrogel encapsulating SZ chondrocytes. The final fibrinogen, thrombin and cell concentrations were 23 mg/ml, 25 IU and 10 million cells/ml, respectively. All animals were allowed to move freely without immobilization after the surgeries. Animals were euthanised 6 months after the second surgery. The quality of cartilage repair was assessed with a modified O’Driscoll scoring system as shown in
Magnetic Resonance Imaging (MRI)
The MRI scanning was performed using a 3T whole body MRI scanner (Magnetom Skyra, Siemens; Erlangen, Germany). The following sequences were used: pd_tse_sag_FS_hi-res and pd_tse_cor_FS_hi-res (Voxel size of 0.3 x 0.3 x 2.0 mm; field of view [FOV] read of 140 mm; FOV phase of 100%; slice thickness of 2 mm; repetition time of 2000 ms; time to echo of 13 ms). Syngo fastView software (Siemens Healthineers; Erlangen, Germany) was used in the MRI scan analysis.
Micro-Computed Tomography (microCT)
The micro-CT imaging was performed using microCT scanner (Bruker micro-CT, Kontich, Belgium). About 500 consecutive slices were obtained from each defect with a voxel size of 40 µm/pixel. An X-ray source was adopted to scan at voltage of 90 kV in 20 mm field of view (FOV). CT-Analyzer (Skyscan) was used for the analysis bone resorption volume. VGSTUDIO MAX (Volume Graphics) was used in 3D reconstruction of the micro-CT scan.
Statistical Analysis
The statistical significance between 3 or more groups was evaluated by one-way ANOVA with Tukey post-hoc test. The statistical significance between 2 groups was evaluated by independent-samples t-test. The significance was set at p<0.05. All statistical analysis was performed with IBM SPSS statistical software (IBM, USA).
Results
Comparison of NWB and WB Derived Zonal Chondrocyte Phenotypes
The feasibility of size-based separation of zonal chondrocytes from FT NWB chondrocytes using inertial spiral microchannel was evaluated, in comparison to WB chondrocytes.
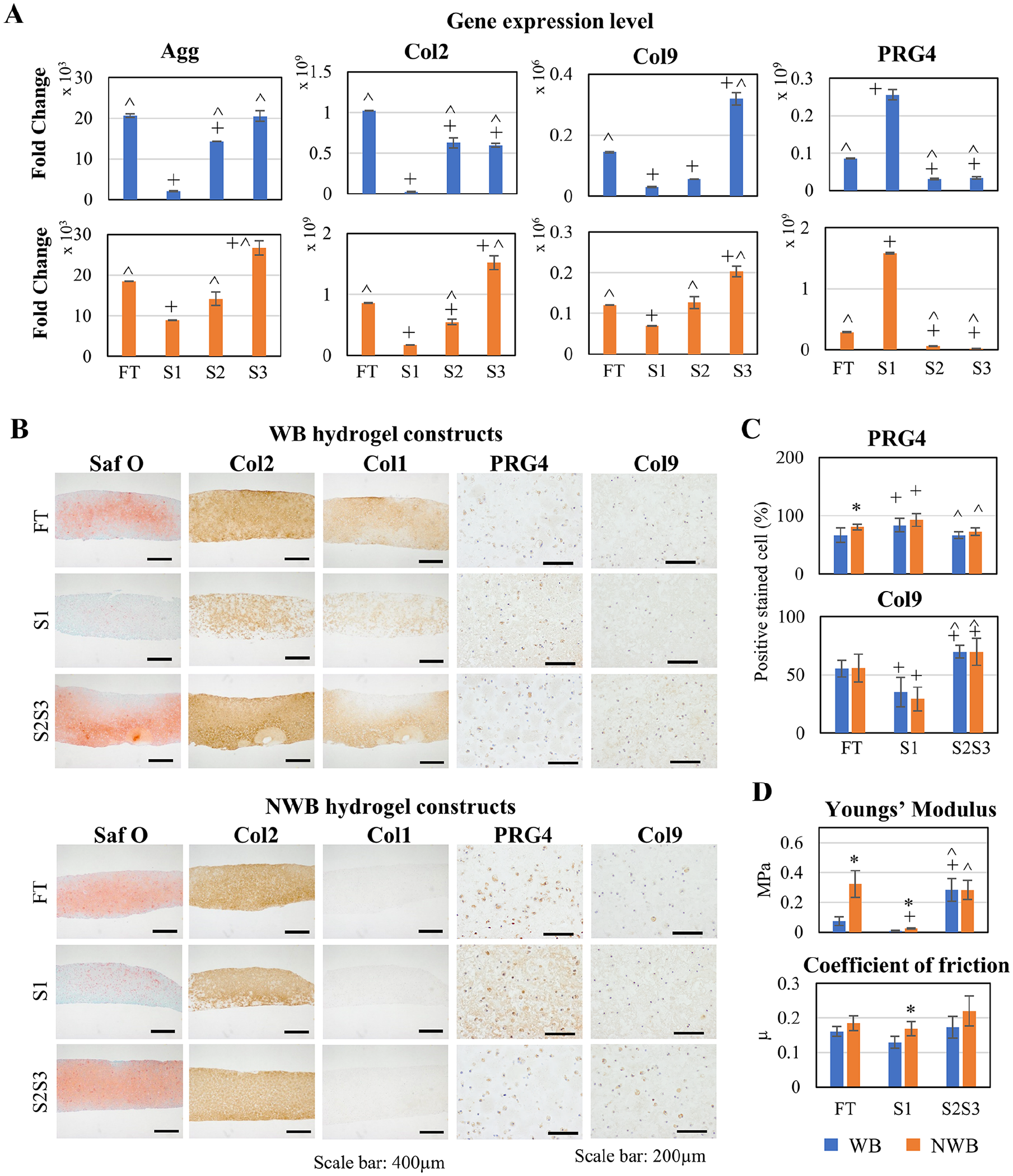
Zonal phenotype characterization of WB and NWB chondrocytes. (
We then compared the efficacy of post-expansion sorting strategy in producing zonal chondrocytes from WB and NWB FT chondrocytes. The strategy involved the expansion of freshly isolated FT chondrocytes first on TCP for one passage (P1, 2 weeks), then on dMC for 2 passages (P2 and P3, 8 days in each passage), followed by zonal chondrocyte size-based separation. The expand-sort cells, as S1 and S2S3 combine, then underwent 3 weeks of tissue formation in 3D hydrogel, as illustrated in
Histological analysis of the tissue constructs showed that zonal cartilage characteristics was observed in both WB and NWB tissue constructs where Saf-O and Col2 were stained stronger in S2S3 compared to S1 tissue constructs (
In Vivo Study: Zonal Phenotype of Regenerated Tissues
In healthy articular cartilage, chondrocytes in the deeper zone form an array of 4 to 5 cells in a columnar organization that aligns perpendicularly to the surface. The columnar arrangement of chondrocytes at DZ was maintained in the native tissue adjacent to the regenerated tissues in all study groups (
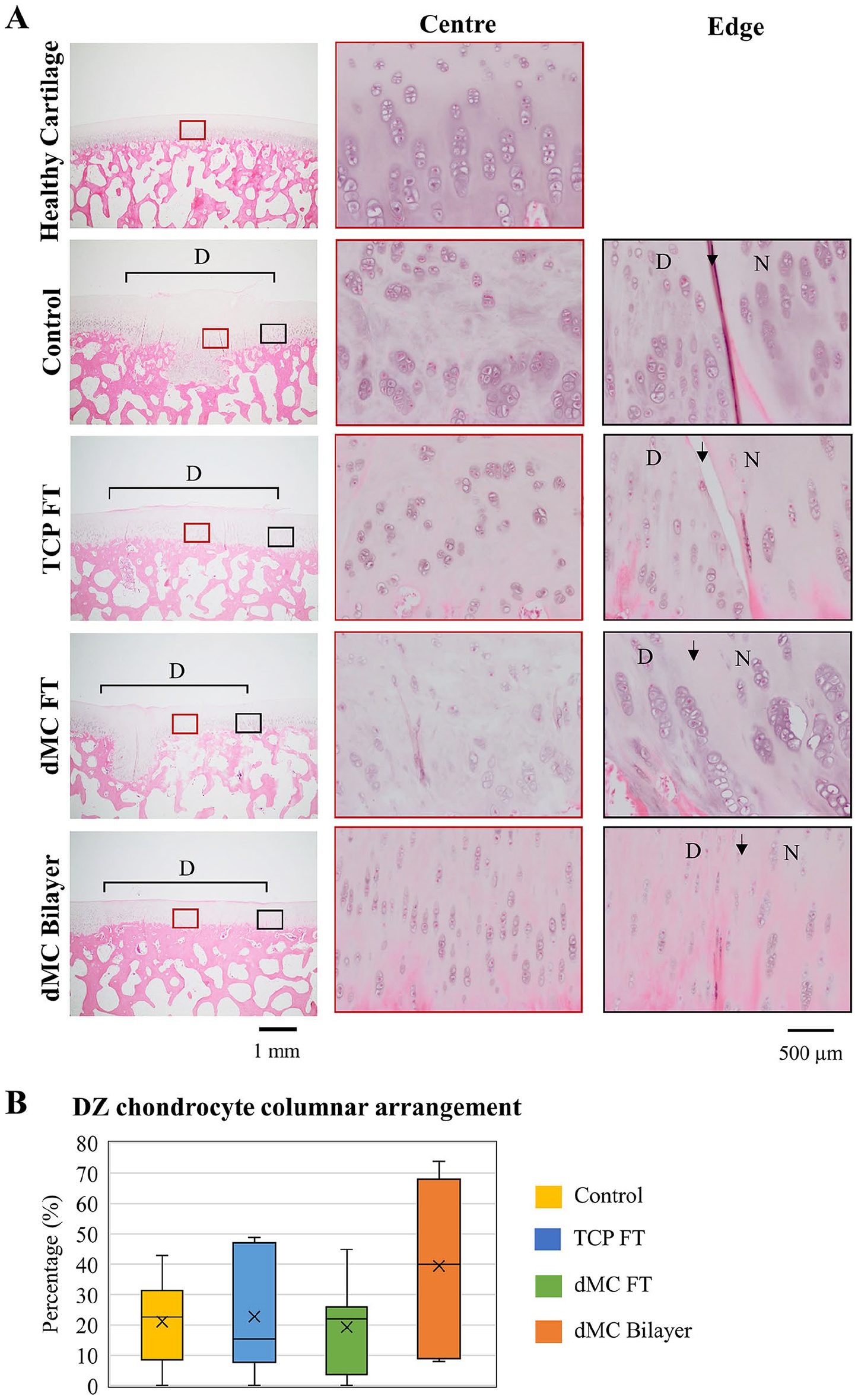
Zonal properties of regenerated tissues. (
Summary on the Observation on Zonal Characteristic of Regenerated Tissues Based on H&E Staining, PRG4 Immunohistology and Polarized Light Microscopy.
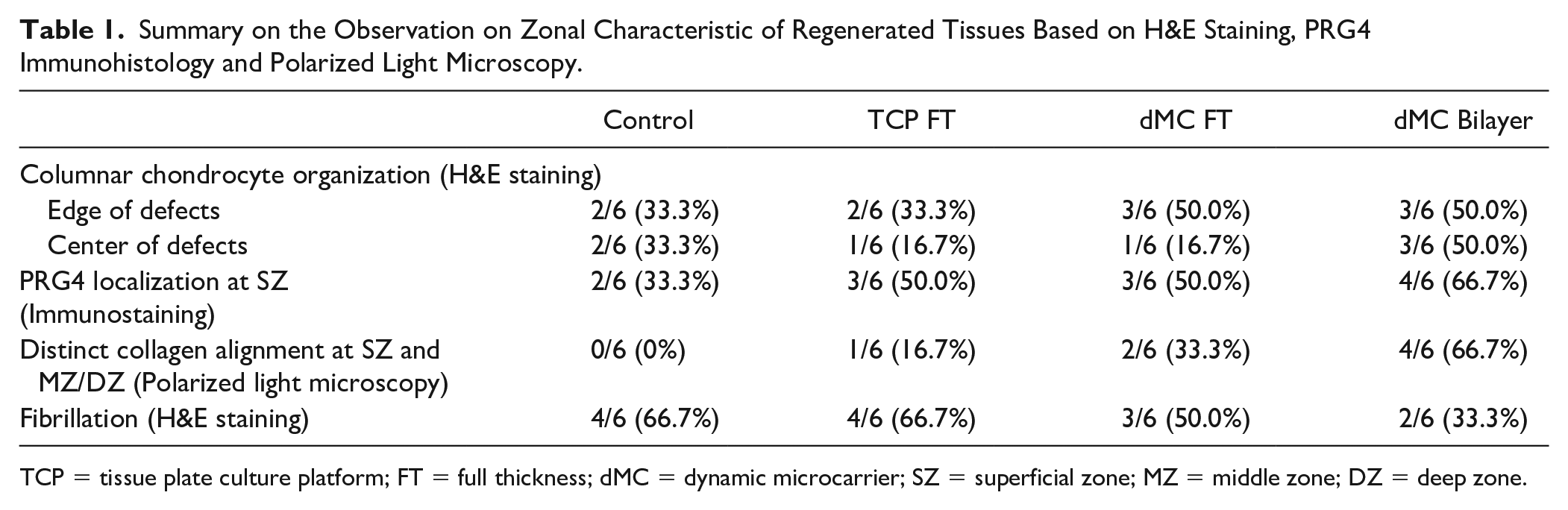
TCP = tissue plate culture platform; FT = full thickness; dMC = dynamic microcarrier; SZ = superficial zone; MZ = middle zone; DZ = deep zone.
PRG4 immuno-staining (
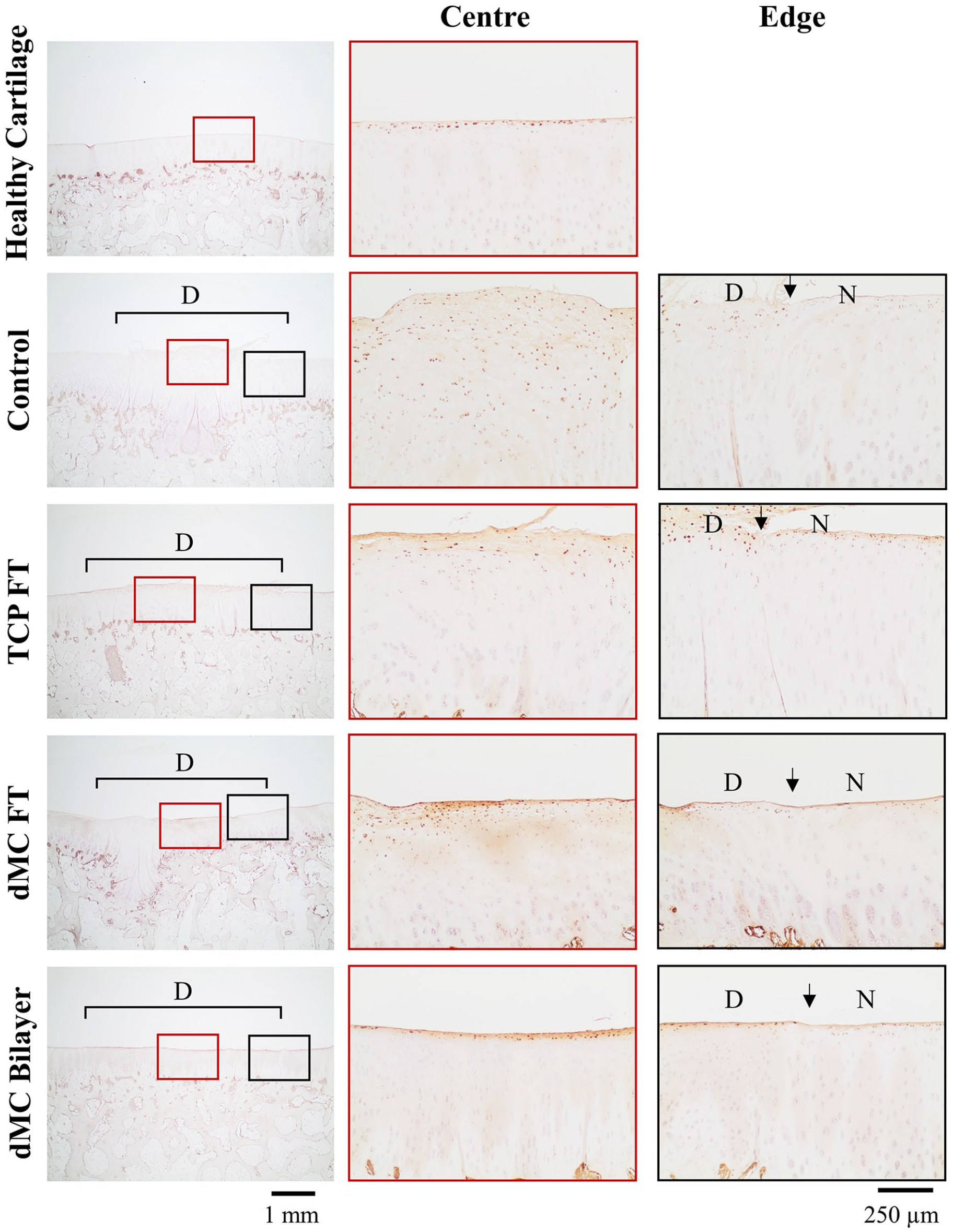
Zonal properties of regenerated tissues. Immunohistochemical analysis of SZ specific protein, PRG4, by immuno-staining. 20x magnification for images on first column. 100x magnification for images on second and third column. Center indicates center of the defects labeled by red boxes; Edge indicates the right edge of defects labeled by black boxes. SZ = superficial zone; N = native tissues; D = defects; TCP = tissue plate culture platform; FT = full thickness; dMC = dynamic microcarrier. Arrows indicate the sites of the defect edge. Images are representative of samples from n = 6 per group.
Summary on the Observation on Structural Integrity of Subchondral Bone Based on MRI Analysis and MicroCT Imaging.
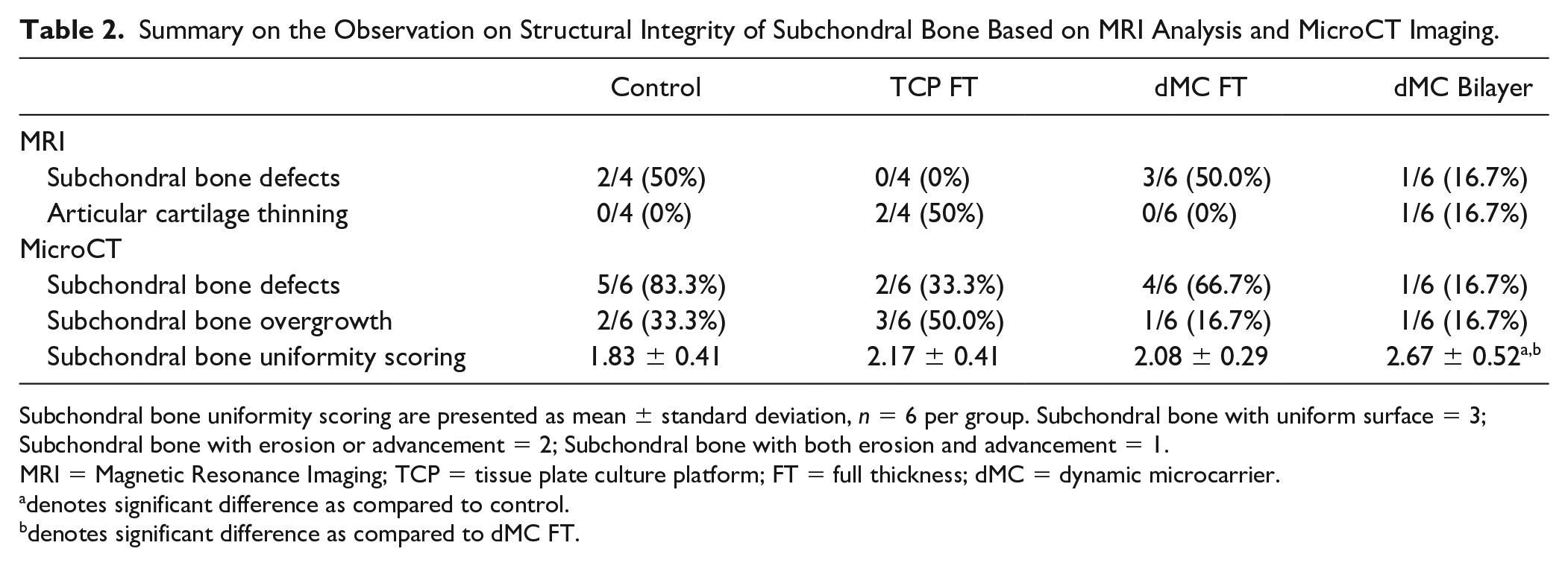
Subchondral bone uniformity scoring are presented as mean ± standard deviation, n = 6 per group. Subchondral bone with uniform surface = 3; Subchondral bone with erosion or advancement = 2; Subchondral bone with both erosion and advancement = 1.
MRI = Magnetic Resonance Imaging; TCP = tissue plate culture platform; FT = full thickness; dMC = dynamic microcarrier.
denotes significant difference as compared to control.
denotes significant difference as compared to dMC FT.
Zonal orientation of collagen fibers in a healthy articular cartilage was demonstrated by polarized light microscopy where distinct optical birefringence of collagen fibers was visible between SZ and MZ/DZ, with a bright thin layer at the SZ and dark scale at the deeper zone (
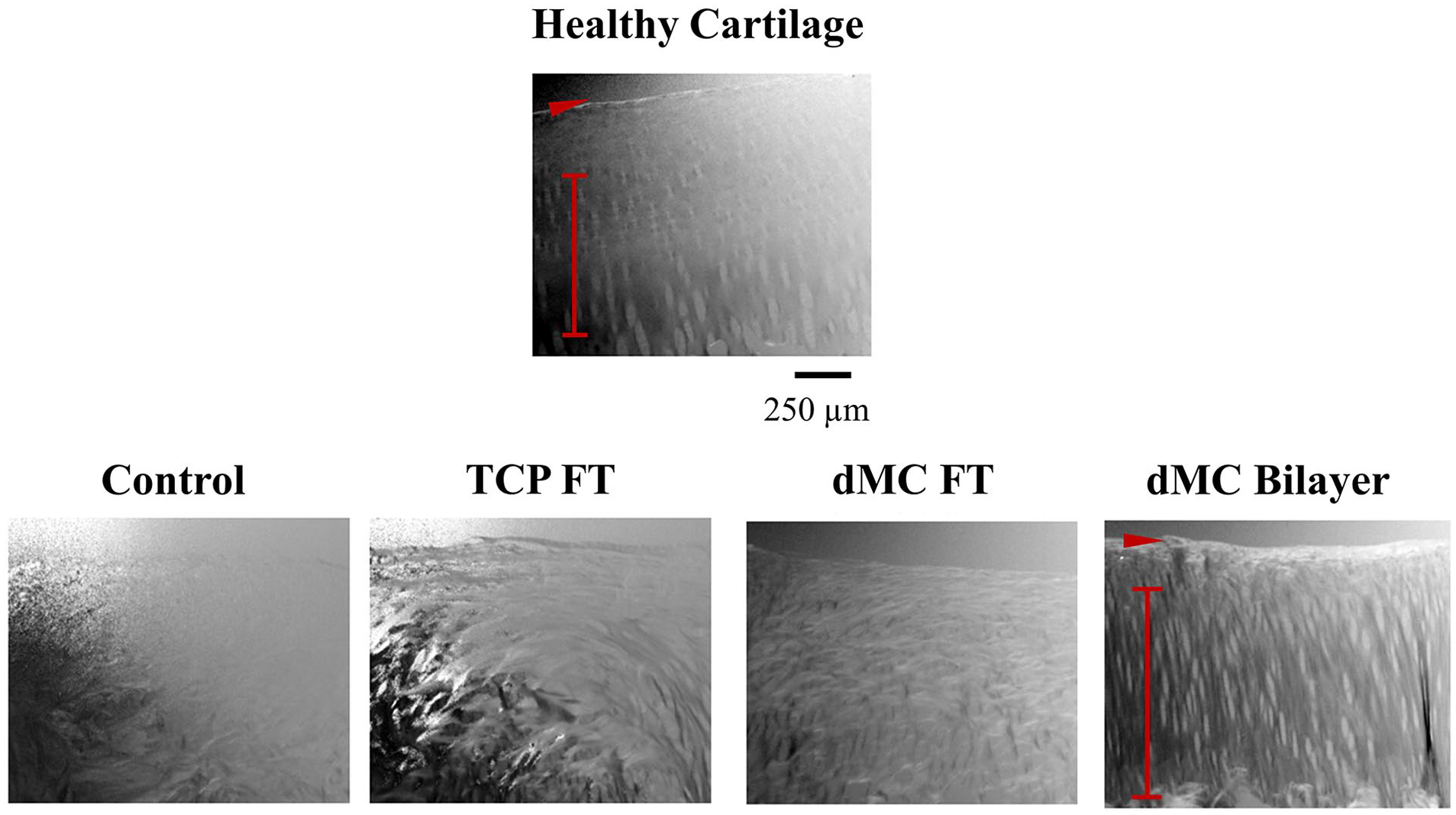
Polarized light microscopy images of healthy articular cartilage tissues and regenerated tissues taken at the center of defects. The collagen fibers arranged parallel to the articular surface are indicated by red arrowhead and the collagen fibers arranged perpendicular to the articular surface are indicated by red line. 100x magnification. Images are representative of samples from n = 6 per group. TCP = tissue plate culture platform; FT = full thickness; dMC = dynamic microcarrier.
In Vivo Study: Cartilage Characteristics of Regenerated Tissues in Porcine Chondral Defect Model
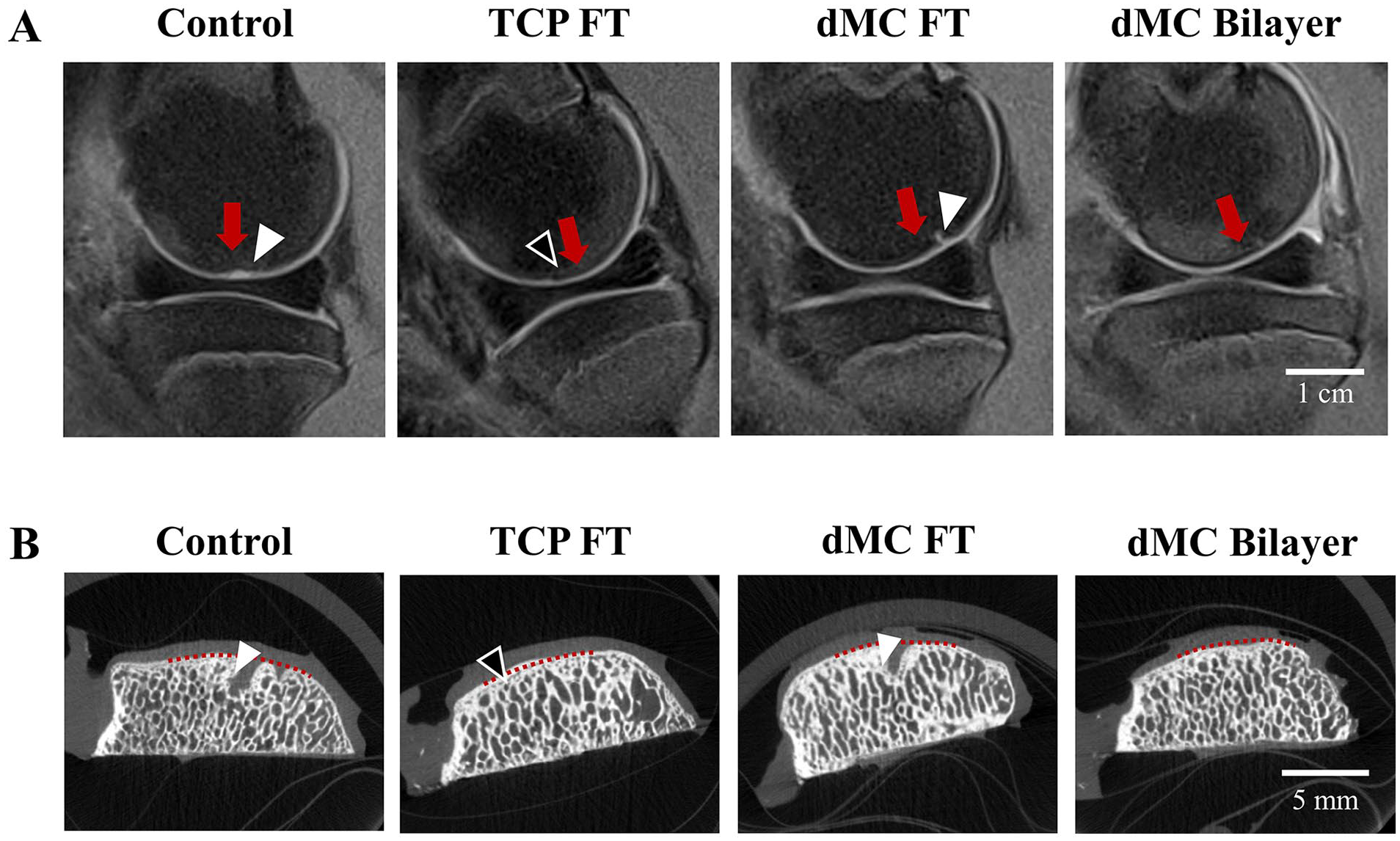
(
Representative microCT sections at the center of defects in each group were demonstrated in
Differences could be observed between groups in terms of sulphated glycosaminoglycans (sGAG) contents. The Saf O staining for 4 out of 6 defects (66.7%) in Control group, 5 out of the 6 defects (83.3%) in each of the TCP FT and dMC FT group, and 3 out of 6 defects (50%) in dMC Bilayer group were unsatisfactory when compared to heathy cartilage tissue (
Discussion
The beneficial effect of zonal chondrocyte bilayered implantation for articular cartilage regeneration has previously been demonstrated using freshly isolated chondrocytes or pre-differentiated MSC-derived chondrocytes on rodent and rabbit osteochondral defect models.17,33 The proof-of-concept studies were however followed up over a short-term recovery period of up to 6 or 12 weeks. Moreover, the cartilage defect sites in both the rat and rabbit models were at the non-weight bearing region of the trochlea. In this study, the regenerative efficacy of zonal chondrocyte bilayered implantation was validated using a porcine model that has anatomically similar knee joints as humans, with cartilage defects created at the femoral condyles that was subjected to similar mechanical microenvironment akin to the weightbearing portion of the human cartilage lesion. In addition, having previously shown the regenerative efficacy of freshly isolated zonal chondrocytes,
17
this study progressed to demonstrate the effectiveness of culture expanded zonal chondrocytes generated using our established dMC post-expansion sorting strategy as illustrated in
We first demonstrated that chondrocytes derived from NWB cartilage tissue can be sorted by size using the spiral microchannel device into zonal chondrocyte subpopulations (
The regenerative efficacy of dMC expanded and spiral microchannel sorted NWB zonal chondrocytes delivered as bilayered zonal chondrocyte implantation in a porcine chondral defect model was compared to TCP-expanded FT chondrocytes (TCP FT, the current ACI cell source) and dMC expanded FT chondrocytes, to provide unequivocal validation to the benefit of the bilayered implantation approach (
The perpendicular arrangement of collagen fibers that were directed against the compressive force served as a damping system to resist lateral fluid flow during compression7,8 and anchored the regenerated cartilage to the underlying subchondral bone.3,9 This would enable the articular cartilage to resist loading force without significant deformation of its solid matrix
8
in the dMC Bilayer group, while the organized SZ collagen fibers would reduce the articular cartilage permeability and enhance the fluid load support to the underlying zone.4,5 On the other hand, the lack of organized 3D structural properties in the regenerated tissues could lead to susceptibility to physical and physiological stresses.
42
Accordingly, a higher prevalence of subchondral bone abnormalities was detected in the Control, TCP FT and dMC FT groups relative to the dMC Bilayer group [
The cartilage-bone interface served to maintain structural integrity of the subchondral bone during articular joint motion. 9 During the creation of critical size chondral defects, disruption to the cartilage-bone interface was inevitable, hence would have subjected the subchondral bone at the defect sites to remodeling. Different paracrine activities of SZ and DZ chondrocytes could have influence the homeostasis of the subchondral bone. DZ chondrocytes has been reported to support mineralization, while SZ chondrocytes, through the action of PTHrP secretion, suppressed mineralization. 43 The positioning of DZ chondrocytes next to the subchondral bone in the dMC Bilayer implantation could have facilitate the restoration of the disrupted cartilage-bone interface induced during surgery. On the other hand, dMC-FT implantation in which proximity of SZ near the subchondral bone in the mixed chondrocyte population might cause inhibition to mineralization, further compounded to bone erosion. It is known that the changes in subchondral bone can affect the long-term survival of the regenerated tissues.44,45 Hence, despite the similar performance between dMC FT and dMC Bilayer in terms of histology scoring and mechanical properties at the 6-month time point of tissue analysis, defect site tissues in dMC Bilayer group could be superior in providing long-term integrity of the regenerated tissue than dMC FT with their capability in recapitulating the zonal characteristic of native articular cartilage tissues and maintaining integral subchondral bone.
Even though a general trend of improvement in bilayered implantation of zonal chondrocytes was observed, the high variability of the treatment outcomes offset the statistical significance between groups (
Conclusion
Current ACI strategies employ a heterogeneous mixture of expanded chondrocytes. The zonal repair of cartilage lesions with stratified implantation of zonal chondrocytes have yet to be realized due to the inefficient zonal chondrocyte isolation process, limited availability of autologous chondrocytes and chondrocyte dedifferentiation after expansion. In this study, we showed that the dynamic microcarrier culture expanded and size-based sorted zonal chondrocytes can overcome the clinical challenges to produce sufficient number of zonal chondrocytes from the limited amount of clinically relevant NWB cartilage plugs to support autologous stratified zonal chondrocyte implantation. This 6-month pre-clinical porcine cartilage repair study showed improved regenerative efficacy of bilayered zonal chondrocyte implantation that encouraged the regeneration of zonal architecture and healthier underlying subchondral bone. Taken together, with appropriate expansion of zonal chondrocytes, the strategy of stratified zonal chondrocyte implantation could represent a significant advancement to current ACI-based cartilage regeneration, with the potential to improve the long-term integrity of the regenerated cartilage tissues.
Supplemental Material
sj-docx-1-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-docx-1-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-TIF-2-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-TIF-2-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-TIF-3-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-TIF-3-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-tif-4-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-tif-4-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-tif-5-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-tif-5-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-tif-6-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-tif-6-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-TIF-7-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-TIF-7-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-jpg-8-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-jpg-8-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Supplemental Material
sj-TIF-9-car-10.1177_19476035221093063 – Supplemental material for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes
Supplemental material, sj-TIF-9-car-10.1177_19476035221093063 for A Pre-Clinical Animal Study for Zonal Articular Cartilage Regeneration Using Stratified Implantation of Microcarrier Expanded Zonal Chondrocytes by Ching Ann Tee, Zheng Yang, Yingnan Wu, Xiafei Ren, Maciej Baranski, Daryl Jimian Lin, Afizah Hassan, Jongyoon Han and Eng Hin Lee in CARTILAGE
Footnotes
Acknowledgments and Funding
This research is supported by National Medical Research Council of Singapore (NMRC/CIRG/1477/2017) and the National Research Foundation, Prime Minister’s Office, Singapore under its Campus for Research Excellence and Technological Enterprise (CREATE) program, through Singapore-MIT Alliance for Research and Technology (SMART): Critical Analytics for Manufacturing Personalized-Medicine (CAMP) Inter-Disciplinary Research Group. C.A.T. was supported by SMART Graduate Fellowship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the procedures in animal experiments were performed according to the Institutional Animal Care and Use Committee (IACUC) at the National University of Singapore (Protocol number: R17-1410).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
