Abstract
Objective
A pivotal aspect of cartilage tissue engineering resides in cell culture medium supplementation, in view of maximizing in vitro cell proliferation and preserving cellular functionality. Autologous human serum (aHS) is commonly used as an inducive supplement for safe human articular chondrocyte (HAC) proliferation prior to clinical implantation. However, practical clinical use of aHS is hindered by constraining manufacturing requirements and quality assurance-driven downstream processing. The present study investigated potential alternative use of commercial human platelet lysate (hPL) supplements in HAC manufacturing workflows related to clinical therapeutic pathways.
Design
Differential effects of hPL, aHS, and fetal bovine serum were assessed on primary cultured HAC parameters (viability, proliferative rates, and morphology) in 2-dimensional in vitro systems. A 3-dimensional HAC pellet model served for postexpansion assessment of cellular functionality, by visualizing proteoglycan production (Alcian blue staining), and by using qRT-PCR relative quantification of chondrogenic marker (SOX9, COL2-A1, and ACAN) genetic expression.
Results
We found that monolayer HAC culture with hPL or aHS supplements presented similar characteristics (elongated cell morphology and nearly identical growth kinetics). Chondrogenic activity appeared as conserved in HACs expanded with human or bovine supplements, wherein histologic analysis indicated a progressive sGAG accumulation and SOX9, COL2-A1, ACAN gene expression was upregulated in 3-dimensional HAC pellet models.
Conclusion
This study therefore supports the use of hPL as a functional equivalent and alternative to aHS for cultured HAC batch preparation, with the potential to effectively alleviate pressure on clinical and manufacturing bottlenecks in cell therapy approaches for cartilage regeneration.
Keywords
Introduction
Articular cartilage possesses limited intrinsic healing capabilities, wherein structural wear and defects are often associated with remnant pain, functional loss, and long-term complications.1-3 Limitations of standard therapeutic management of cartilage lesions have prompted the development of specific regenerative approaches and tissue engineering strategies. Promising protocols have been investigated, among them microfracturing, mosaicplasty, or cell therapies, the latter presenting utmost potential to date in cartilage regenerative medicine. 4 Therein, autologous chondrocyte implantation (ACI) was reported as both effective and durable for treatment of knee articular cartilage defects.5-7 However, therapeutic outcomes remain dependent on functional quality of cultured autologous human articular chondrocytes (HAC), biopsied from healthy, nonloaded, and undamaged zones. 8 Therefore, several manufacturing parameters of in vitro HAC isolation and mitogenic propagation require optimization, favoring important endpoint harvest cell yields and postexpansion maintenance of chondrogenic potential. Therein, selection of specific and appropriate culture medium supplementation (e.g., fetal bovine serum, autologous human serum, human platelet lysate) plays a preponderant role in manufacturing workflows for clinical HAC lot preparation. Despite historical widespread use of fetal bovine serum (FBS) in pharmaceutical and biotechnology industries, the use of this animal-sourced ancillary raw material has been increasingly disputed, over several quality and safety concerns (e.g., poor characterization, high variability, or immunogenicity and infectious risks).9,10 Alternatives to FBS comprise autologous human serum (aHS), commonly and effectively used in high concentrations for HAC expansion in clinical protocols. 11 Nevertheless, specific burdens are introduced with the use of aHS, such as patient blood harvest (i.e., sufficient for production of 50 mL of aHS), onerous and constraining material processing under current good manufacturing practice (cGMP) conditions, and high interindividual variability among donors, related to metabolic changes. In view of the aforementioned limitations of FBS and aHS, human platelet lysate (hPL) was investigated as a functional alternative for HAC expansion preceding clinical transplantation.12-14 Derived from platelet-rich plasma (PRP) by chemical lysis or mechanical disruption, hPL yields many bioactive components (e.g., platelet-derived growth factor [PDGF], transforming growth factor-β1 [TGF-β1], basic fibroblast growth factor [bFGF], vascular endothelial growth factor [VEGF]), which synergistically act in promoting cell proliferation, migration, and synthesis of extracellular matrix proteins (e.g., collagen, glycans).15,16 Thoroughly investigated for culture of various mesenchymal stem cells, commercial hPL has displayed positive functional effects over FBS in several in vitro studies on chondrocyte proliferation and differentiation.12-14,17-22 Despite specific inherent advantages and to our best knowledge, no studies have clearly demonstrated the potential of hPL as a substitute to aHS for HAC manufacturing within clinical cell therapy protocols. Therefore, the objective of this study was to determine the specific and differential effects of hPL (i.e., vs. aHS or FBS) supplementation on morphology, proliferation rates, and evolutive chondrogenic expression potential of cultured HACs to be clinically applied for cartilage repair.
Materials and Methods
Primary HAC Isolation
Clinical cartilage samples were anonymized, stored, and logged in a departmental biobank, complying with internal regulations thereof and following an approved protocol (i.e., Vaud Cantonal State Ethics Committee Reference No. 264/12). Human articular cartilage biopsies were obtained from 26 patients aged 23 to 66 years (i.e., 52.73 ± 11.26 years; mean ± SD). Cartilage was harvested from posterior regions of femoral condyles in patients undergoing partial or total knee arthroplasty, or from large osteochondral fragments in patients suffering from osteochondritis dissecans and undergoing arthroscopic ablation. Tissue samples were placed in a sterile container with conserved transport medium (i.e., 1% penicillin, 1% streptomycin [Gibco, USA], Dulbecco’s modified Eagle medium [DMEM; Gibco, USA]) and transferred to the laboratory at room temperature within less than 2 to 4 hours for processing. The samples were washed twice in 100 mm Petri dishes with 10 mL of sterile 1X phosphate-buffered saline (PBS, Bichsel, Switzerland) supplemented with 1% penicillin, 1% streptomycin (Gibco, USA). Subsequently, small cartilage fragments were obtained after mechanical disruption by using a sterile scalpel and forceps to produce less than 1-mm fragments. The equivalent of 200 mg fragmented tissue was digested overnight at 37 °C in 10 mL of DMEM supplemented with 0.8 % pronase and 0.8% type II collagenase under gentle shaking. The resulting HAC suspensions were filtered using 70-µm nylon cell strainer (Falcon, USA) and were washed once with sterile 1X PBS. Then, HACs were seeded with a minimum density of 4 × 103 cells/cm2 and expanded in standard monolayer culture. Culture media were composed of Ham’s F-12 Nutrient Mix (Gibco, USA) and DMEM (Gibco, USA) in 1:1 proportion, with 2 mM
HAC Proliferation Assays
For a short-term proliferation assay, primary HAC types (n = 10) were seeded at a relative viable density of 2.5 × 103 cells/cm2 in 6-well plates (Corning, USA). Culture medium was supplemented with 10% v/v hPL (Stemulate, Sexton Biotechnologies, USA), 10% v/v aHS, or 10% v/v FBS (Life Technologies, USA). HAC proliferation was evaluated daily by microscopic enumeration between days 3 and 7 after culture initiation. Experimental triplicates were performed for each time point and serum supplementation (i.e., FBS, aHS, hPL).
For a long-term proliferation assay, primary HAC types (n = 16) were seeded at a relative viable density of 2.5 × 103 cells/cm2 in culture flasks (75 cm2, TPP, Switzerland). Culture medium was supplemented with 10% v/v hPL or 10% v/v FBS. Comparison with aHS was not performed as available aHS quantities were not sufficient for more than 2 passages in this configuration. HAC proliferation was evaluated by determining endpoint (i.e., 85% confluency) cell yields as described above between passages 2 and 9. Population doubling times (PDT) were determined for each passage using an ad hoc formula (Supplementary Material).
Chondrogenic Potential Evaluation Assay
Subsequent to serial cell expansions for each test group (i.e., FBS, aHS, hPL), specific HAC chondrogenic potentials were investigated in a 3-dimensional pellet culture system. To this end, suspensions containing 5 × 105 cells were centrifuged at 230 × g for 5 minutes in 15 mL Falcon tubes. The Falcon tubes were not completely closed in order to allow gas exchange. Resulting pellets were cultured for 7 and 14 days in 2 mL of chondrogenic medium without serum supplementation, consisting of Ham’s F12 and DMEM in 1:1 proportion, with 2 mM
Histology
Pellets obtained after 7 and 14 days of culture were embedded in paraffin after 4% formaldehyde fixation. Thin 5-µm sections were cut and placed onto microscope slides (Superfrost Plus, Menzel-Gläser, Germany), deparaffinized, stained 30 minutes with Alcian blue for sulfated glycosaminoglycan (sGAG) detection, and counterstained 1 minute with Nuclear Fast Red (Vector Laboratories, USA). Histology slides were microscopically assessed and imaged.
Gene Expression
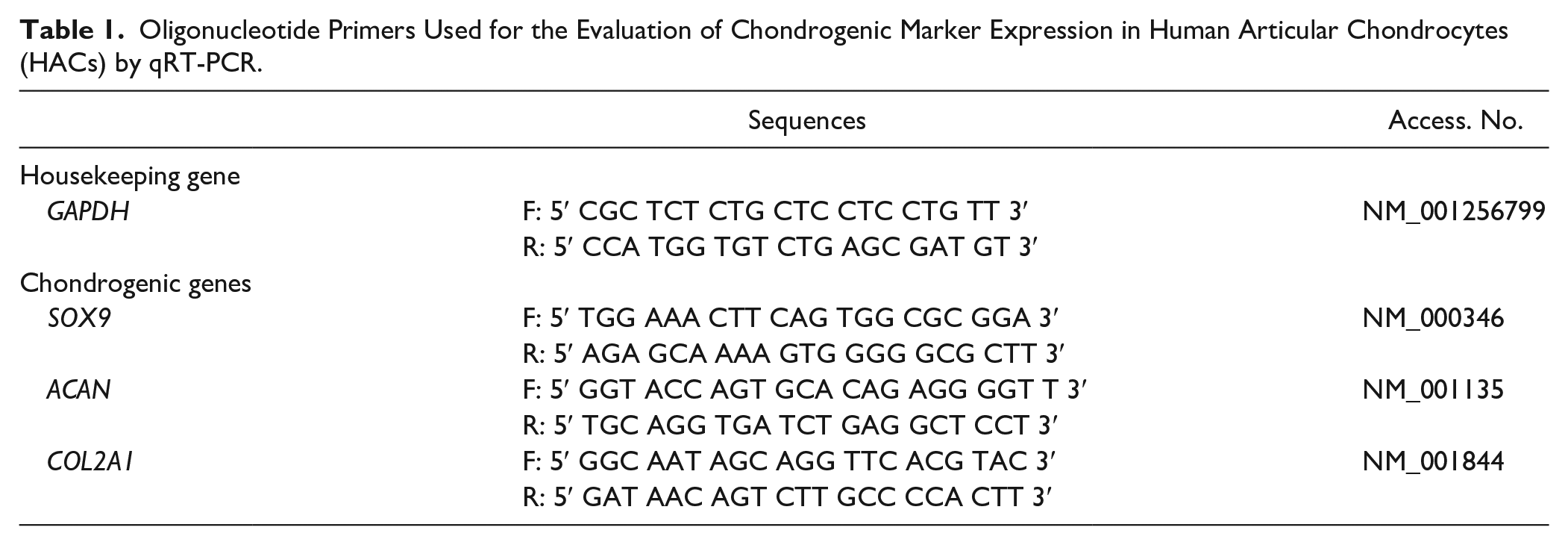
Expression patterns of chondrogenic markers (i.e., SOX9, ACAN, COL2A1) were analyzed by qRT-PCR using a StepOnePlus Real-time PCR system (Applied Biosystem, USA). Total RNA was extracted from 2 or 3 pooled pellets obtained at each time point for each cell type. Pellets were mechanically disrupted using a 18G needle and 500 µL of TRIzol reagent (Invitrogen, USA) according to the manufacturer’s protocol. RNA concentration and purity levels were measured using a Nanodrop system (Thermo Fisher Scientific, USA) and 500 ng of total RNA were reverse transcribed into cDNA using a random hexamer mix and a Superscript III First-Strand Synthesis System Supermix (Invitrogen, USA), according to the manufacturer’s instructions. The reverse transcription cycle conditions using PCR Biometra Tpersonal were as follows: 25 °C for 10 minutes, 48 °C for 30 minutes, and 95 °C for 5 minutes. The qRT-PCR was performed with 10 ng of cDNA using KAPA SYBER Fast (Kapa Biosystems, Roche, Switzerland), according to the manufacturer’s instructions. Fluorescence was acquired using the following cycling conditions: 95 °C for 3 minutes (enzyme activation) and 40 amplification cycles (95 °C for 3 seconds and annealing extension at 60 °C for 30 seconds). Each sample was run in triplicate, and the relative expression level for each gene was normalized to the GAPDH expression levels. Sequences of the primers used for this study are listed in Table 1 . Gene expression levels were quantified using the 2−ΔΔCt method.
Oligonucleotide Primers Used for the Evaluation of Chondrogenic Marker Expression in Human Articular Chondrocytes (HACs) by qRT-PCR.
Human Sera Growth Factor Analysis
ELISAs (enzyme-linked immunosorbent assays) were performed according to manufacturer recommendations (R&D System, USA) to analyze sera compositions for human bFGF, TGF-β1, and PDGF-BB. Samples of aHS were analyzed for bFGF and PDGF-BB contents (n = 10), and for TGF-β1 contents (n = 6). Seven batches of hPL from the same supplier were analyzed (i.e., all 3 growth factors).
Sample Groups and Statistical Analyses
Proliferation assay results were expressed as mean ± standard deviation, with n = 10 for the short-term proliferation assay and n = 16 for the long-term proliferation assay. Data from qRT-PCR assays were presented as box-and-whisker plots, wherein box plots represented medians and quartiles, and whiskers represented minima and maxima, with n = 8 for hPL and n = 4 for aHS and FBS, respectively. To evaluate statistical significance of differences between each group in proliferation and qRT-PCR assays, Mann-Whitney tests were applied using P < 0.05. In the analysis of sera composition, differences in growth factor concentration variances were examined using Levene’s test, wherein significant differences were assessed using P < 0.05.
Experimental Results
Short-Term Preservation of HAC Proliferative Morphology and Rates in aHS- and hPL-Containing Media
Morphological features of proliferating HACs at P0 were similar for the 3 test groups (i.e., aHS, hPL, FBS), wherein cells adopted polygonal shapes and were present in a uniform size distribution. However, differences in cell density and yields were clearly identified between the aHS or hPL groups, as compared with the FBS group (
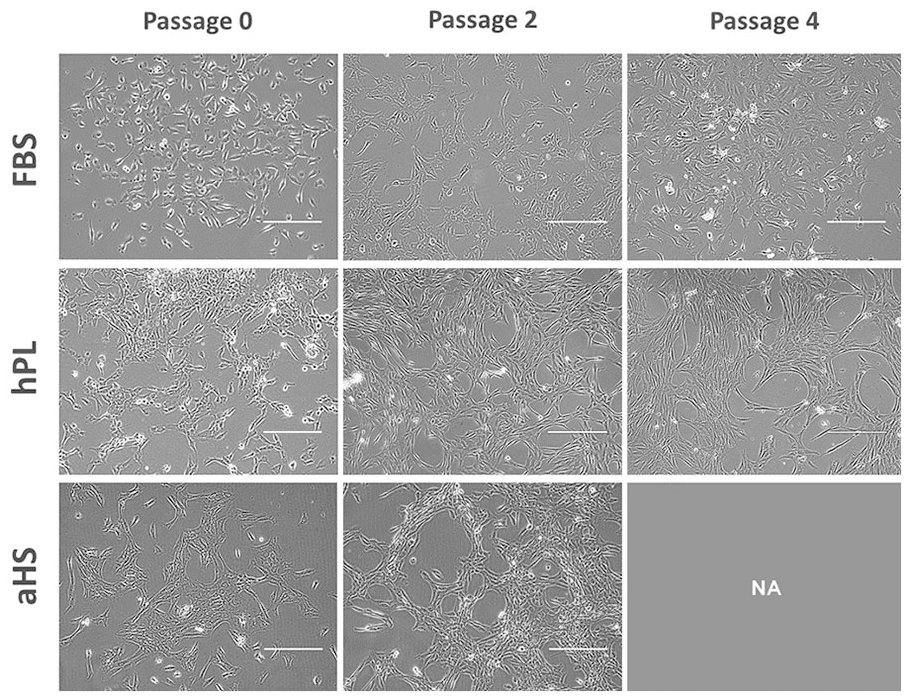
Morphology of HACs expanded in monolayer culture. HACs were isolated from articular cartilage samples and cultured in F12:DMEM supplemented with 10% FBS, 10% hPL, or 10% aHS. Scale bars represent 0.2 mm. HAC, human articular chondrocyte; F12:DMEM, Ham’s F12–Dulbecco’s modified Eagle medium; FBS, fetal bovine serum; hPL, human platelet lysate; aHS, autologous human serum.
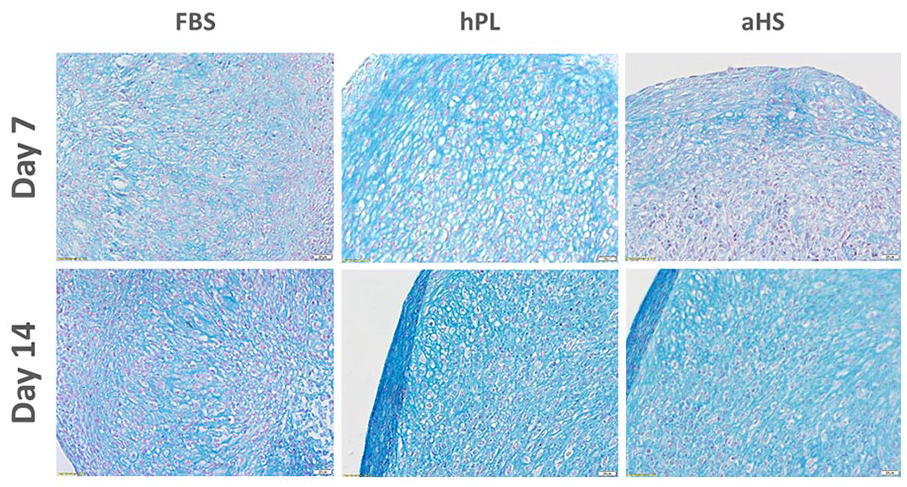
Chondrogenesis assessment in 3-dimensional pellets. Chondrogenic differentiation was histologically assessed at day 7 and day 14 of induction. Three-dimensional HAC pellets were stained with Alcian blue and counter-stained with Nuclear Fast Red. Scale bars represent 0.02 mm. HAC, human articular chondrocyte.
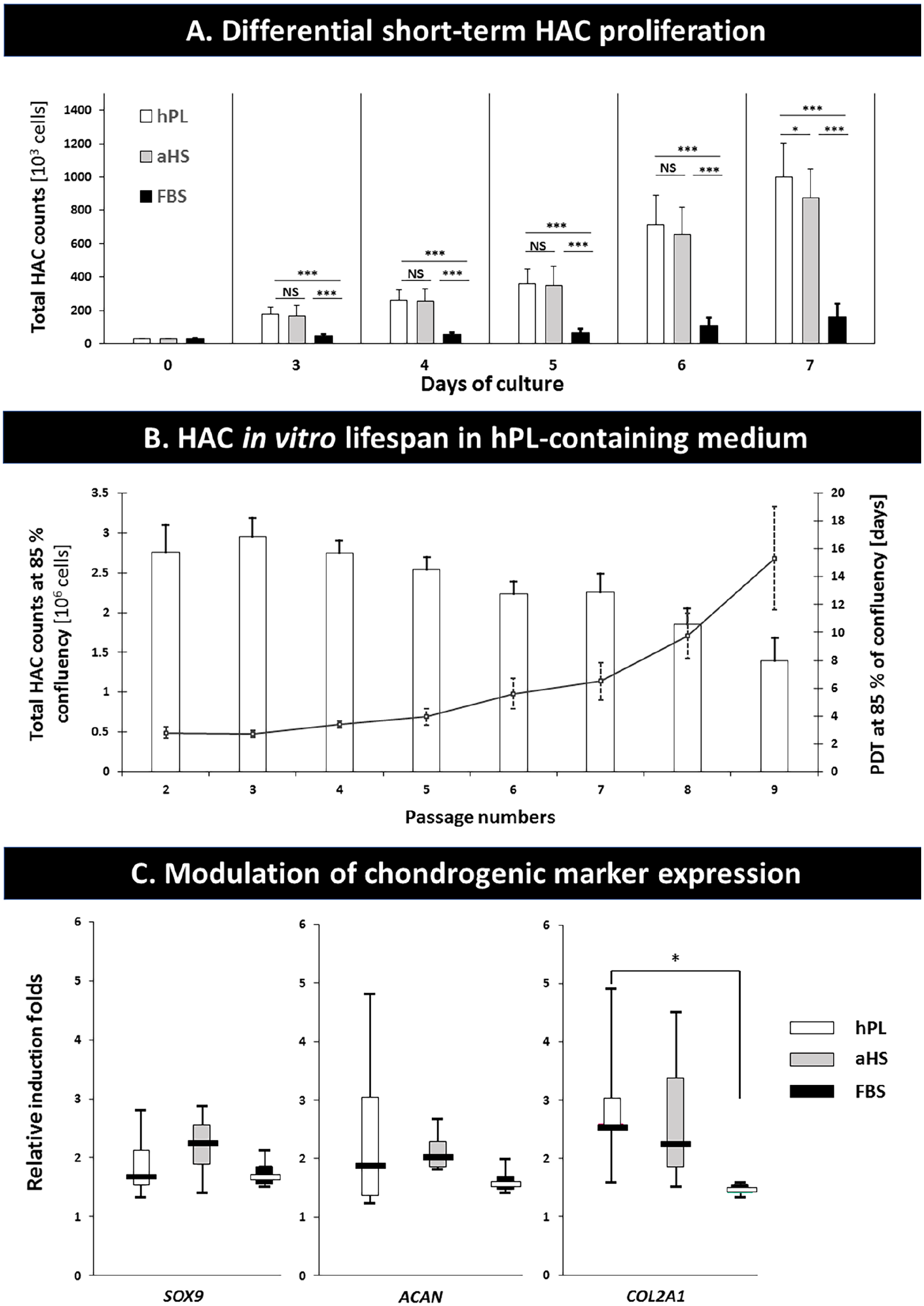
(
Long-Term Preservation of HAC Proliferation Capacity in Monolayer Cultures
As long-term homogenous primary HAC growth maintenance constitutes a major challenge, hPL-containing medium was assessed with regard to proliferation rate stimulation in serial sub-cultures. Results indicated that HAC yields were conserved up to P5, with a mean total of 2.75 × 106 cells/flask (
Optimal Maintenance of HAC Chondrogenic Potential after Monolayer Expansion in Presence of hPL
During monolayer expansion, HACs transiently lose their specific chondrogenic phenotype, wherein extracellular matrix production capabilities and potency may be recovered on culture in 3-dimensional conformations and under biochemical induction. Histological data indicated that sGAGs progressively accumulated over the course of the pellet stimulation assay (i.e., 14 days), wherein qualitative assessment of endpoint deposited sGAGs did not distinguish either test-group from the others (
Differential Growth Factor Quantification in HAC Culture Supplements
Specific growth factor contents were found to be relatively similar between hPL and aHS samples for TGF-β1 (i.e., 29.37 ng/mL and 23.06 ng/mL, respectively) and for PDGF-BB (i.e., 12.34 ng/mL and 11.68 ng/mL, respectively) ( Table 2 ). Regarding bFGF, superior quantities were observed for hPL as compared with aHS (i.e., 2.21 ng/mL and 0.94 ng/mL, respectively) ( Table 2 ). Except for TGF-β1, results of this assay emphasized important variability between different batches of hPL or aHS. Compared with aHS, a significantly higher variance of bFGF levels (P = 0.007) and conversely, a lower variance of PDGF-BB levels were noted for hPL ( Table 2 ).
Growth Factor Contents in hPL and aHS Medium Supplements.
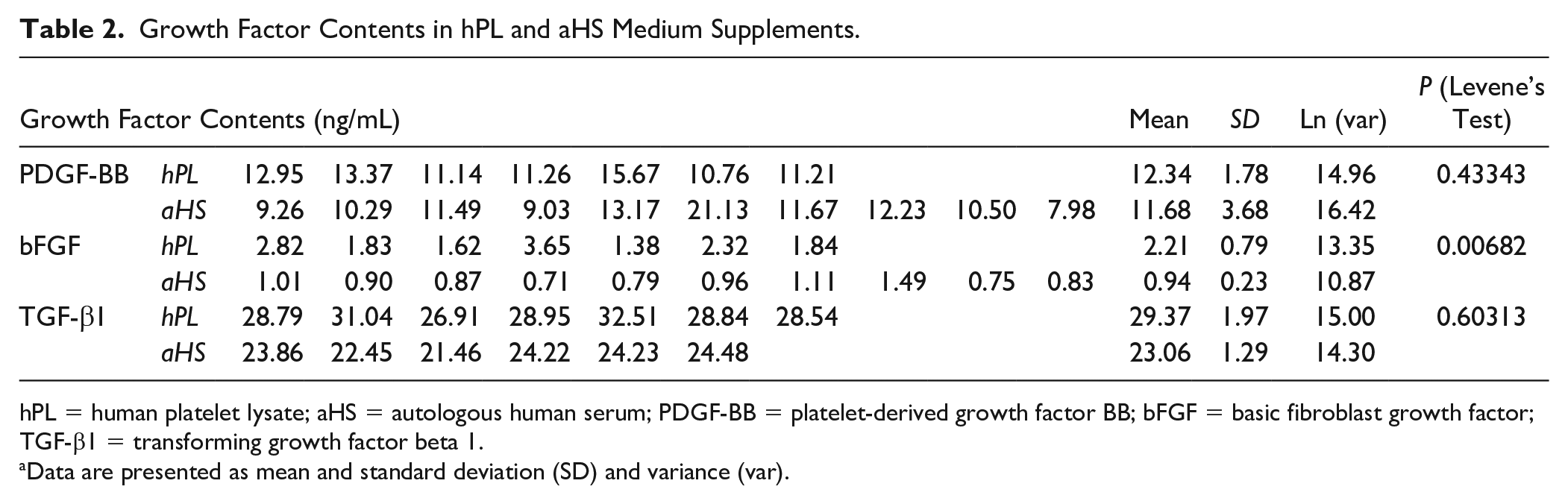
hPL = human platelet lysate; aHS = autologous human serum; PDGF-BB = platelet-derived growth factor BB; bFGF = basic fibroblast growth factor; TGF-β1 = transforming growth factor beta 1.
Data are presented as mean and standard deviation (SD) and variance (var).
Discussion
In Vitro Proliferative HAC Morphology
Proliferation assays revealed that hPL and aHS induced similar morphological modifications in primary HAC monolayer cultures, which were constituted by fibroblast-like cells with high proliferation potential. In 3-dimensional pellet conformations, cultured HACs adopted spherical morphologies and were preferentially located in lacunae, approaching observed cellular behaviors in native cartilage. HACs in healthy tissues present homeostatic quiescence and high differentiation states, in order to maintain the equilibrium of matrix composition. However, initiation of in vitro monolayer HAC expansion results in extensive morphological adaptations, often related to loss of chondrogenic phenotypes. On culture initiation, freshly isolated primary HACs displayed round-to-polygonal cell morphologies, whereas subsequent subcultures exhibited elongated fibroblast-like phenotypes, commonly associated to high proliferation (
Differential HAC Monolayer Proliferation Rates
Clinical cellular therapy protocols require manufacturing of large and consistent batches of therapeutic materials. Results in this study confirmed the functional equivalence of hPL and aHS for effective support of primary HAC isolation and monolayer culture, as well as preservation of extensive in vitro life spans (i.e., >5 passages). Concurringly, Spreafico et al. 13 had compared mitogenic properties of various platelet releasates to aHS or FBS for HAC culture medium supplementation. Strong effects of PRP-based supplements on HAC proliferation were observed. Hildner et al. 12 had reported that 5% to 10% hPL medium supplementation significantly increased HAC proliferation, as compared with FBS. Sykes et al. 14 had reported rapid growth of HACs in presence of hPL (i.e., identical product reference to the one used in this study). From a further mechanistic point of view, Van Thi Nguyen et al. 29 had shown that hPL induced activation of proliferation-related pathways and cell cycle reentry in quiescent human chondrocytes, through expression of cyclin D1 and activation of ERKs, AKT, and HIF-1 pathways. Additionally, Barbero et al. 30 had studied the effects of donor ageing on HAC yields, proliferation, and re-differentiation abilities, revealing that culture medium supplementation with growth factor combinations (i.e., TGF-β1, FGF2, and PDGF) optimally supported proliferation and postexpansion chondrogenic activity resurgence in donors younger than 40 years. However, despite extensive life spans in selected culture systems, HACs are to be clinically transplanted at early passages (i.e., <P4), as soon as technically available for release. Furthermore, interindividual variability of aHS lots should be investigated, for evaluation of additional potential for manufacturing lot standardization with the use of hPL.
Postexpansion Maintenance of HAC Chondrogenic Potential
Despite the transient phenotypic shift on monolayer isolation and culture, HACs were shown to revert to a functional chondrogenic phenotype once properly induced (i.e., pellet culture and biochemical stimulation), irrespective of the nature of human-sourced medium supplement (i.e., hPL vs. aHS). Concurringly, Spreafico et al. 13 had described that gene expression involved in chondrogenic differentiation processes was maintained in 3-dimensional culture on fibrin scaffolds. Hildner et al. 12 had reported that re-differentiation of HACs occurred up to 3.3 population doublings, while suggesting that serial passaging affected functional phenotype recovery more importantly than the use of hPL. However, Sykes et al. 14 had reported that HACs expanded in presence of hPL presented a delay in recovery of their chondrogenic phenotype (i.e., versus FBS supplementation). Conversely, Hildner et al. 12 had also reported that presence of hPL led to enhanced sGAG production in pellet culture (i.e., vs. FBS supplementation). Such conflicting results were discussed in light of differential hPL manufacturing workflows, introducing high variability between products. Subsequently, Sykes et al. 14 investigated differential relative gene expression of SOX9, COL2A1, and ACAN in HAC pellets at days 0, 21, and 28 using a comparative Ct method and cell count normalization, indicating significant effects of FBS supplementation over hPL in preliminary monolayer expansion. Therein, basal expression of SOX9, COL2A1, and ACAN were significantly higher in cells expanded in presence of FBS (i.e., vs. hPL), adding complexity to the analysis. Therefore, genetic analyses in the present study were conducted using induction fold values rather than relative expression, with results marginally favoring the use of human-sourced supplements with regard to chondrogenic marker expression. Overall, integration of results and literature gatherings allowed to conclude that hPL supplementation in preliminary monolayer culture maintained HAC chondrogenic potential, but that several alternative processing parameters (e.g., cell isolation, culture media composition, analysis timepoints, hPL manufacturing process) could modulate endpoint functional outcomes.
Implementation of hPL Supplements in Clinical Cell Therapy Manufacturing Workflows
Current HAC clinical batch manufacturing workflows comprise aHS culture medium supplementation, inherently burdened with important interindividual variability, potentially negatively affecting proliferation rates and overall batch quality, eventually hindering therapeutic efficacy. 31 Relatively important autologous blood volume requirements (i.e., >200 mL) and constraining quality-driven cGMP processing of HACs add complexity to clinical protocols and heighten stress burdens on patients. Therefore, alternative culture supplements such as platelet-derived factors were investigated since the 1980s as promising sources of growth factors for cell culture. 32 Lucarelli et al. 33 and Doucet et al. 34 had underlined the promising potential of platelet-derived extracts for clinical mesenchymal stem cell (MSC)–based cellular engineering products. Subsequently, high potentials of platelet-derived by-products and hPL were highlighted, with strong promotion of in vitro proliferation for various cell types (e.g., MSCs and HACs).12,35-43 Manufacturing processes (i.e., freeze-thawing) for hPL derivation result in the non-selective release of all bioactive platelet factors, most of which (e.g., PDGFs, TGF-β, bFGF) have been described as crucial for chondrogenesis regulation in healthy cartilage16,44. Concurringly, individual exogenous growth factors (i.e., bFGF, TGF-βs, IGF-1, and PDGFs) or combinations thereof had been identified as key for support of chondrocyte expansion and chondrogenic potential maintenance45-48. Such results were in line with the data obtained in the present study.
Furthermore, growth factor quantification assays underlined high variations in PDGF-BB and bFGF contents in hPL and aHS batches. This variability did not appear to drastically impact primary HAC proliferation. Conflicting published information exists related to standardization of human-sourced supplements (e.g., hPL and aHS) using specific growth factors as markers. In alignment with the results presented herein, high interbatch or interproduct variability were reported for growth factors quantified in hPL or aHS, probably due to manufacturing or experimental specificities, without significantly impacting in vitro HAC proliferation characteristics.49,50 Complex compositions of such supplements potentially result in synergistic actions of various bioactive molecules, positively influencing HAC proliferation and/or differentiation.
Therapeutic success of cartilage cell therapy remains largely dependent on cell batch quality, and thereby on composition of human-sourced medium supplements. Although TGF-β1, PDGF-BB, and bFGF remain essential for HAC in vitro proliferation, systematic quantification thereof constitute poor quality control markers. Henschler et al. 51 had underlined important considerations for hPL clinical use, including manufacturing method standardization, quantitative composition determination, and variability reduction by batch pooling. Therefore, it appears highly advisable to validate each new batch of culture supplements based on cell morphology and proliferation characteristics of reference cell types, before any clinical use is implemented.
Conclusion
Main findings of this study comprised the observed functional equivalence between hPL and aHS in terms of primary HAC morphology, proliferation rates, and optimal maintenance of HAC chondrogenic potential after monolayer expansion in presence of hPL. Specific limitations were identified for the study and optimization of HAC clinical batch manufacturing workflows. Namely, culture medium supplement composition variability and high dependance of functional outcomes on processing materials and methodologies were outlined. Overall, results supported the substitution of aHS by hPL for standardized therapeutic HAC in vitro expansion for clinical workflows of cartilage repair interventions. Such practices shall therefore appropriately alleviate patient burdens, diminish manufacturing costs, and ensure therapeutic product quality, thereby maximizing the therapeutic potential of considered interventions by enhancing clinical outcomes.
Supplemental Material
sj-pdf-1-car-10.1177_19476035211035433 – Supplemental material for Human Platelet Lysate as an Alternative to Autologous Serum for Human Chondrocyte Clinical Use
Supplemental material, sj-pdf-1-car-10.1177_19476035211035433 for Human Platelet Lysate as an Alternative to Autologous Serum for Human Chondrocyte Clinical Use by Virginie Philippe, Alexis Laurent, Philippe Abdel-Sayed, Nathalie Hirt-Burri, Lee Ann Applegate and Robin Martin in CARTILAGE
Footnotes
Acknowledgments and Funding
We are grateful to Mrs Caroline Concaro for her valuable scientific advice, and Jerome Pasquier for his statistical help. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Orthopaedic and Traumatology Unit and the Lausanne Orthopedic Research Foundation (LORF).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Vaud Cantonal State Ethics Committee (Reference No. 264/12).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
