Abstract
Objective
To describe the spontaneous evolution of age-related changes affecting knee joint articular cartilage, walking speed and a serum biomarker of cartilage remodeling in C57BL/6-JRj wild-type male mice.
Design
Histological changes were assessed by the Osteoarthritis Research Society International (OARSI) score (0=normal, 6=vertical clefts/erosion to the calcified cartilage extending >75% of the articular surface) in newborn, 1-week- and 1-, 3-, 6-, 9- and 12-month-old C57BL/6-JRj wild-type male mice, walking speed by the Locotronic system, and serum C-terminal telopeptide of type II collagen (CTX-II) content by ELISA in 1-, 3-, 6-, and 9-month-old C57BL/6-JRj wild-type male mice.
Results
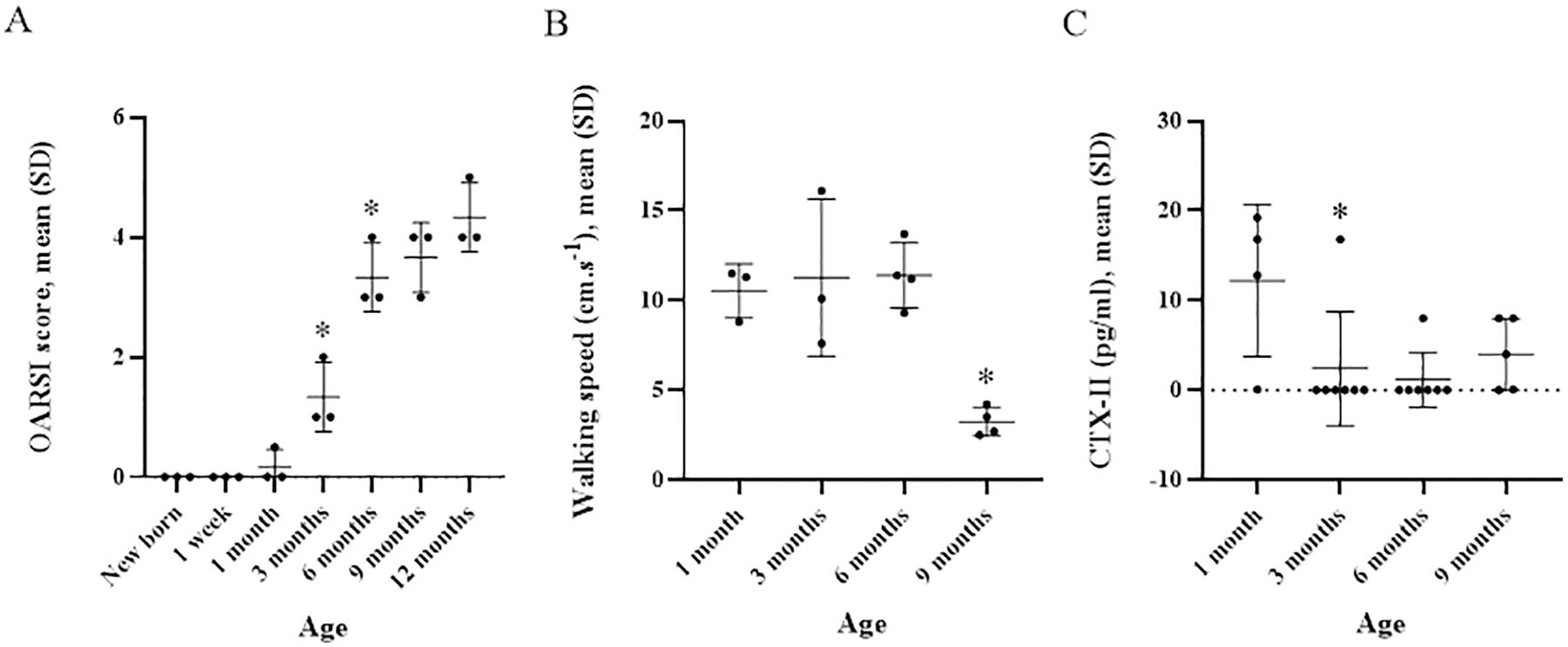
Mean (SD) OARSI score significantly increased from 0.2 (0.3) to 1.3 (0.6) (p=0.03) between 1 and 3 months of age and from 1.3 (0.6) to 3.3 (0.6) (p=0.04) between 3 and 6 months of age. Mean walking speed was stable between 1 and 6 months of age but significantly decreased from 11.4 (1.8) to 3.2 (0.8) cm.s-1 (p=0.03) between 6 and 9 months of age. Serum CTX-II content was maximal at 1 month of age, then decreased from 12.2 (8.5) to 2.4 (8.4) pg/ml (p=0.02) between 1 and 3 months of age, remaining low and stable thereafter.
Conclusions
C57BL/6-JRj wild-type male mice showed continuously increasing osteoarthritic changes but delayed decreasing walking speed with age. These variations were maximal between 3 and 9 months of age. Maximal serum CTX-II content preceded these changes.
Animal models of osteoarthritis (OA) have been used to study the pathogenesis of cartilage degeneration and the potential of anti-OA drugs.1,2 In mice, maximal OA changes have been reported from 9 to 12 months of age 2 but actually occur as early as 2 months of age. 3 However, a comprehensive quantitative assessment of age-related evolution of OA-type cartilage lesions is lacking. In 2010, the Osteoarthritis Research Society International (OARSI) grading score, developed to describe histopathogical changes in human OA cartilage, was adapted to OA animal models. 4 We aimed to describe the spontaneous evolution of age-related changes affecting knee joint articular cartilage, walking speed, and a serum biomarker of cartilage remodeling in C57BL/6-JRj male mice. Histological changes were assessed by the OARSI score in newborn, 1-week-old and 1-, 3-, 6-, 9-, and 12-month-old mice, walking speed by the Locotronic system, and serum C-terminal telopeptide of type II collagen (CTX-II) content by ELISA (enzyme-linked immunosorbent assay) in 1-, 3-, 6-, and 9-month-old mice. We found that C57BL/6-JRj male mice had continuously increasing OA changes, but delayed decreasing walking speed with age. Variations were maximal between 3 and 9 months of age. Maximal serum CTX-II content preceded these changes.
For the purpose of our study, a total of 40 C57BL/6-JRj male mice were obtained from the Janvier lab (France, Le Genest-Saint-Isle) and housed in cages at our animal facility (BioMedTech Facilities INSERM US36 | CNRS UMS2009 | Paris Descartes, Université de Paris, 45, Rue des Saints-Pères, Paris, France) under conventional conditions (light = 12/24 hours, dark = 12/24 hours; room temperature =23°C), with standard food and water ad libitum. Animal handling was restricted to individuals from the animal facility staff. In order to reduce the effects of hormonal status on our prespecified outcomes, we restricted our study to male mice. All experiments were performed by the same 2 examiners (JA and KT). For a given timepoint, the same batch of mice was used to collect the 3 prespecified outcomes (i.e., histology, walking speed, and CTX-II). Because the batch of mice had to be euthanized for the purpose of histological analyses for a given timepoint, we had to use different batches of mice for each timepoint. At the end of the study, all mice were euthanized by cervical dislocation. The present study was approved by our institutional review board (CEEA no. 34, Université Paris Descartes). Experiments were reported in accordance with the ARRIVE checklist (Supplemental Appendix).
Murine knee joint tissues were obtained from newborn, 1-week-old and 1-, 3-, 5-, 6-, 9-, and 12-month-old C57BL/6-JRj male mice (n = 3 per timepoint). Left knee joints were dissected and fixed in 4% paraformaldehyde solution for 24 hours, at 4°C, then decalcified with 10% EDTA (ethylenediaminetetraacetic acid) phosphate buffered saline without CaCl2 and MgCl2 for 1 month at 4°C. The solution was renewed every 4 days. Samples were stored in 70° ethanol at 4°C. Knee joints were dehydrated in successive ethanol baths of 70°, 95°, 100° and isopropanol, then embedded in paraffin blocks following a prespecified orientation to provide standardized sagittal sections starting from the medial part of the knee joint. Seven-µm thick section were cut by using a microtome (2155, Leica) to obtain 10 to 13 serial sections from the knee region of interest, defined as the region where the medial tibia plateau, medial meniscus, and medial femur condyle were visible. Slides were stained by safranin-O/fast green. 4 For each sample, images of one sagittal section of the stained knee region ( Fig. 1 ) of interest were obtained by using an optical microscope with objective 40 (Olympus, BH-2, Q imaging RETIGA 2000 R Camera). Three independent readers assessed all images in a blinded manner and scored histological changes affecting the articular cartilage of the knee joint according to the OARSI score (0 = normal, 6 = vertical clefts/erosion to the calcified cartilage extending >75% of the articular surface). 4 Expression of Netrin-1, a guidance molecule, and its receptor UNC5b were studied by immunohistochemistry. The correlation between Netrin-1 and UNC5b expression and OARSI score was studied using the Spearman’s rank correlation coefficient.
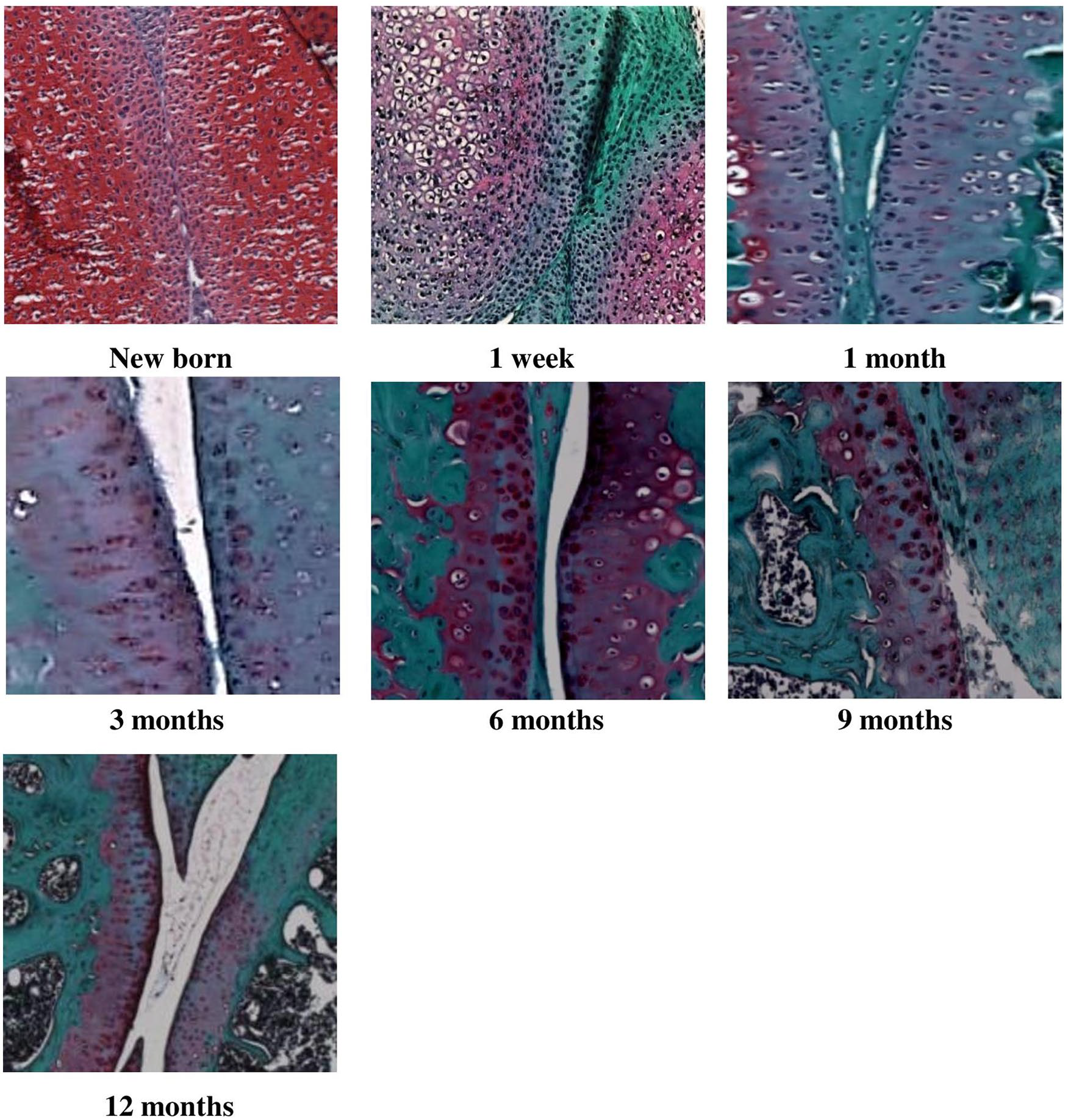
Histology of left knee joint from newborn, 1-week-old and 1-, 3-, 6-, 9-, and 12-month-old wild-type C57BL/6-JRj male mice stained with safranin-O/fast green (×40). Safranin-O is a marker of cartilage matrix proteoglycans. Fast green counterstains nonspecific collagen proteins.
Walking speed of 1-, 6-, and 9- month-old mice was measured by using a locomotor system (Locotronic). Because of technical issues, we were unable to collect walking speed at 12 months. Briefly, this system provides an inner space of 75-cm length, 8-cm width, and 12-cm height. The system consists of 77 sensor bars that record the movements of the mouse in real time. The detector is connected to a computer, and data are analyzed by using the Locotronic software (v2.3.0.0). Mice were placed in the testing room for 30 minutes before testing. Mice were tested between 10 a.m. and 2 p.m. An initial back-and-forth walk served as “practice” and was excluded from analysis. For each mouse, the walking speed was recorded 3 times at 30-minute intervals. If a mouse changed direction midway through the recording, the walk was repeated until the mouse walked the full length of the corridor without changing directions. We did not observe any failed run during the experiment. The reported walking speed corresponds to the mean (SD) of the 3 measures.
Blood samples were collected from the venous sinus of 1-, 3-, 6-, and 9-month-old mice (n = 4, 7, 7, and 5, respectively) under general anesthesia. Because of technical issues, we were unable to collect blood samples at 12 months. Blood samples were centrifuged at 10,000 rpm for 10 minutes at 4°C. Plasma was collected and stored at −80°C. Serum CTX-II content was measured for each timepoint by using Serum Pre-clinical Cartilaps ELISA (Artialis Group, Belgium). The Serum Pre-clinical Cartilaps ELISA results are linear in the range of 3.7 to 300 pg/mL. The detection limit is 3.7 pg/mL. The coefficient of variation (CV) was 5.8%.
Quantitative variables were expressed with mean (SD) and were compared by nonparametric Mann-Whitney U test. All analyses involved using SYSTAT 13 for Windows v13.00.05. Graphs were designed by using GraphPad Prism 5 v5.01. Interobserver correlation coefficient for OARSI scoring was calculated by using MedCalc v16.8.4. A P value <0.05 was considered statistically significant.
Mean OARSI score significantly increased from 0.2 (0.3) to 1.3 (0.6) (P = 0.03) between 1 and 3 months of age and from 1.3 (0.6) to 3.3 (0.6) (P = 0.04) between 3 and 6 months of age ( Table 1 and Fig. 1 ). Mean OARSI score continuously increased from 3.3 (0.6) to 4.3 (0.6) but not significantly between 6 and 12 months of age (P = 0.10) ( Table 1 and Fig. 2A ). For OARSI scoring, interobserver correlation coefficient was 0.97 (95% confidence interval 0.96-0.99). The Spearman’s rank correlation coefficient with OARSI score was −0.623 for Netrin-1 and −0.193 for UNC5b.
Evolution of Cartilage Changes, Walking Speed, and Serum C-Terminal Telopeptide of Type II Collagen (CTX-II) Content in C57BL/6-JRj Male Mice. a
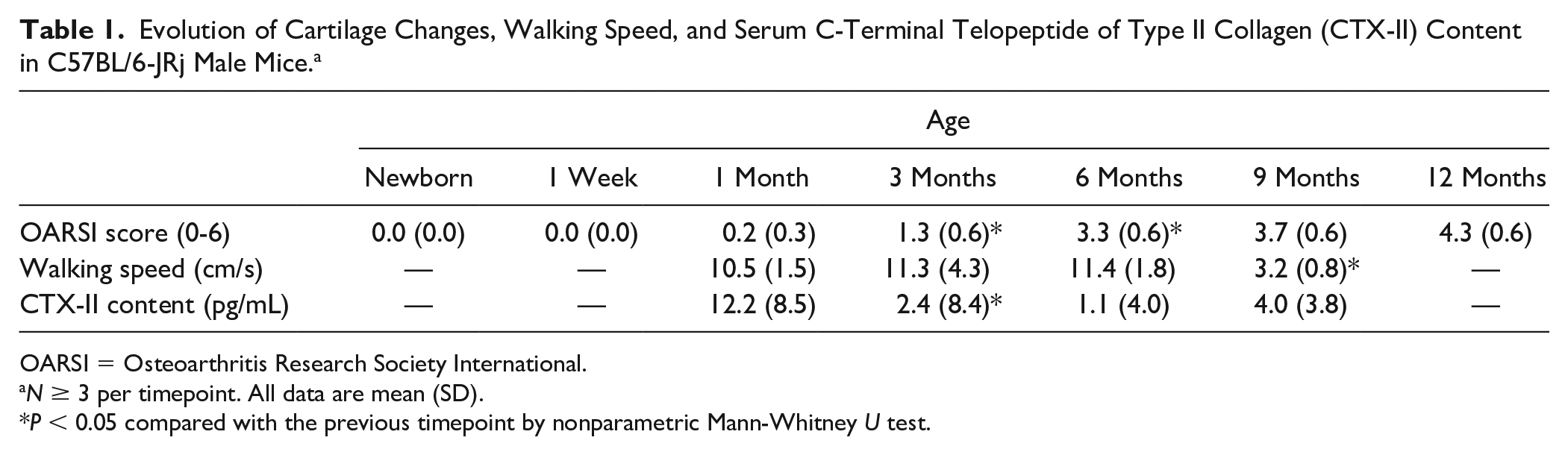
OARSI = Osteoarthritis Research Society International.
N ≥ 3 per timepoint. All data are mean (SD).
P < 0.05 compared with the previous timepoint by nonparametric Mann-Whitney U test.
(
Mean walking speed was stable between 1 and 6 months of age. It significantly decreased from 11.4 (1.8) to 3.2 (0.8) cm/s (P = 0.03) between 6 and 9 months of age ( Table 1 and Fig. 2B ). We did not observe any failed runs during the experiment. Mean serum CTX-II content was maximal at 1 month of age, then decreased from 12.2 (8.5) to 2.4 (8.4) pg/mL (P = 0.02) between 1 and 3 months of age and remained low (<5 pg/mL) and stable thereafter ( Table 1 ).
In summary, we found that C57BL/6-JRj male mice had continuously increasing OA changes and delayed decreasing walking speed with age. Variations were maximal between 3 and 9 months of age. Serum CTX-II content was maximal at 1 month, then decreased and did not vary between 3 and 9 months of age.
Our study is the first to describe the spontaneous evolution of knee joint cartilage changes with ageing by using the semiquantitative OARSI score in wild-type mice. In 1956, Sokoloff and colleagues showed spontaneous development of OA in wild-type C57BL/6 mice. 5 Yamamoto and colleagues further assessed spontaneous OA lesions by using light ultrastructural-histochemical analyses in wild-type C57 black mice. 3 With the classification system described by Okabe, 6 the authors found that the incidence of OA increased from 25% to 80% between 2 and 16 months of age. 3 Our findings, along with those from Yamamoto et al., 3 suggest that tissue-detectable OA-like changes occur spontaneously as early as 2 months of age in wild-type mice. The evolution of OA changes has also been described in induced-OA mouse models. 7 In 2010, Glasson and colleagues 4 specifically designed the OARSI score to semiquantify, in a standardized manner, OA lesions in mouse models of induced OA or spontaneous OA in genetically modified animals. In our study, we used the OARSI score to semiquantify spontaneous OA lesions with age in wild-type mice. Our findings suggest a continuous and evolving process of cartilaginous lesions from 1 to 12 months of age in wild-type mice.
Because walking speed is considered a clinical biomarker of lower-limb disease, 8 we further assessed whether histological lesions were associated with decreased walking speed. Although the maximal increase in OARSI score was between 3 and 6 months of age, the maximal decrease in walking speed was from 6 to 9 months of age. This finding suggests that like in human OA, cartilage lesions in mice precede clinical impairment and could remain “asymptomatic” for a while.
CTX-II is a biological biomarker of cartilage remodeling. In humans, increased serum CTX-II content is associated with end-stage OA in menopausal women 9 and is correlated with clinical and radiological changes in people at different OA stages. 10 CTX-II content was also shown to predict the development of OA. 11 In a longitudinal study of STR/ort OA mice, Sarukawa and colleagues 12 found that urinary CTX-II content was maximal at 2 months, then decreased significantly between 2 and 3 months of age. Consistently, we found that serum CTX-II content was maximal at 1 month, then decreased between 1 and 3 months of age. Surukawa et al. 12 hypothesized that high CTX-II content could precede detectable joint lesions and therefore could serve as a biological biomarker to predict disease progression at early stages of OA. Because type II collagen is restricted to articular cartilage, CTX-II content increases with increasing cartilage damage, until most of the cartilage is destroyed, then CTX-II production starts to decline, 13 as we observed from 1 month of age in our model. Another plausible hypothesis is that once a certain level of cartilage destruction has been reached, CTX-II production also reaches a plateau and possibly starts to decline. This could indicate failure of cartilage repair mechanisms in the advanced stages of OA.
Our study has limitations. Our sample size was small, and we did not perform a power calculation. Therefore, our results should be considered only exploratory. Increasing the number of mice would have enhanced the power of our study. However, in order to comply with the ethical recommendations from our local ethical committee, we had to limit the number of mice used in the present study. To draw definitive conclusions, further research using larger samples are necessary. Our findings were restricted to a specific mouse strain and to male mice only. OA development in C57BL/6 mice can be highly variable due to in cage behavior/activity. We cannot exclude that patterns of OA evolution would have been different depending on the mouse model and gender assessed. Extending the description to female mice would be highly relevant. Finally, histological assessment of synovial inflammation and of osteophyte formation, radiological assessment as a noninvasive technique, the use of modern sequencing and proteomic techniques and longer follow-up could have added relevant information regarding joint changes over time.
In conclusions, OARSI score spontaneously and continuously increases along with delayed decrease in walking speed in C57BL/6-JRj wild-type male mice. Changes are maximal between 3 and 9 months. Maximal serum CTX-II content precedes these changes. Our description could serve to schedule interventions and assessment timepoints in mouse model of spontaneous OA. However, interpretation of our results is restricted to C57BL/6 male mice and limited by the small sample size. Definitive conclusions can be drawn only from larger studies.
Supplemental Material
(4)_Appendix_1 – Supplemental material for Aging Cartilage in Wild-Type Mice: An Observational Study
Supplemental material, (4)_Appendix_1 for Aging Cartilage in Wild-Type Mice: An Observational Study by Joulnar Akoum, Khadija Tahiri, Marie-Th�r�se Corvol, Didier Borderie, Fran�ois �tienne, Fran�ois Rannou and Christelle Nguyen in CARTILAGE
Footnotes
Supplemental Material
Authors’ Note
The work reported was done at INSERM UMRS 1124, Toxicité Environnementale, Cibles Thérapeutiques, Signalisation Cellulaire et Biomarqueurs (T3S), UFR Sciences Fondamentales et Biomédicales, Centre Universitaire des Saints-Pères, 75006 Paris, France.
Acknowledgments and Funding
The authors thank Céline Becker for technical assistance with the Locotronic system, staff members of the animal facility (Plateforme d’hébergement et d’élevage, BioMedTech Facilities INSERM US36 | CNRS UMS2009 | Paris Descartes, Université de Paris, 45, Rue des Saints-Pères, Paris, France) for animal handling, staff members of the Plateforme Histologie, Immunomarquage, Microdissection laser (HistIM, Institut Cochin Paris, France) for technical assistance with histology and Mrs Laura Smales for professional copyediting. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our work was supported by the Research on OsteoArthritis Diseases Network (ROAD) funded by the Fondation ARTHRITIS. Joulnar Akoum was supported by the Agence Universitaire de la Francophonie and CNRS-L (appel d’offres régional 2016). The funding sources were not involved in the study design, collection, analysis and interpretation of data; in the writing of the manuscript; or in the decision to submit the manuscript for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The present study was approved by our institutional review board (CEEA no. 34, Université Paris Descartes).
Animal Welfare
Experiments were reported in accordance with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) checklist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
