Abstract
Objective
To compare substantial clinical benefit (SCB) of a hydrogel-based, matrix-associated autologous chondrocyte implantation (M-ACI) method versus microfracture (MFx) in the treatment of knee cartilage defects.
Design
Propensity score matched-pair analysis, using the MFx control group of a phase III study as comparator for M-ACI treatment in a single-arm phase III study, resulting in 144 patients in the matched-pair set.
Results
Groups were comparable regarding baseline Knee Injury and Osteoarthritis Outcome Score (KOOS), sex, age, body mass index, symptom duration, smoking status, and previous knee surgeries. Defect sizes in the M-ACI group were significantly larger than in the MFx group (6.4 cm2 vs. 3.7 cm2). Other differences concerned location, number, and etiology of defects that were not considered to influence the interpretation of results. At 24 months, significantly more patients in the M-ACI group achieved SCB in KOOS pain (72.2% vs. 48.6%; P = 0.0108), symptoms (84.7% vs. 61.1%, P = 0.0039), sports/recreation (84.7% vs. 56.9%, P = 0.0008), and quality of life (QoL; 72.2% vs. 44.4%, P = 0.0014). The SCBs for KOOS activities in daily living and International Knee Documentation Committee score were higher for M-ACI but not significantly different from MFx. The SCB rates consistently favored M-ACI from 3 months onward. The highest improvements from baseline at 24 months in patients with SCB were observed for KOOS sports/rec. (M-ACI: 60.8 points, MFx: 55.9 points) and QoL (M-ACI: 58.1, MFx: 57.4).
Conclusion
Hydrogel-based M-ACI demonstrated superior SCB in KOOS pain, symptoms, sports/rec., and QoL compared with MFx in patients with knee cartilage defects through 2 years follow-up.
Introduction
Focal chondral or osteochondral defects of the knee joint, caused by trauma or other reasons, are a risk factor for the development of premature osteoarthritis (OA) in the spontaneous course (if left untreated) or after unsuitable defect treatment.1,2
Different methods are available for the surgical treatment of localized full-thickness cartilage defects. In this context, restorative procedures, such as matrix-assisted autologous chondrocyte implantation (M-ACI) or osteochondral autograft transplantation (OAT), have been reported to be associated with better mid- to long-term clinical outcomes, including lower complication and failure rates when compared with reparative marrow stimulation techniques, such as microfracture (MFx), provided that the appropriate treatment indications including manageable defect size of the respective restorative procedure are observed.3,4
To assess the outcome of cartilage repair procedures, patient-reported outcome measures (PROMs) are commonly used that typically focus on assessing patient’s symptoms, physical function, and quality of life (QoL). 5 The threshold for what is considered a successful treatment outcome assessed by PROMs depends on several factors, including the specific PROM being used, the patient population being treated, and the goals of the treatment.
The Knee Injury and Osteoarthritis Outcome Score (KOOS) and International Knee Documentation Committee (IKDC) score are widely used PROMs validated for articular cartilage repair with good psychometric properties and are recommended as a primary endpoint for studies in knee articular cartilage repair by the International Cartilage Regeneration and Joint Preservation Society (ICRS).6,7
Although clinically relevant improvement is often used as a benchmark to evaluate the effectiveness of cartilage repair,5,8 there has been increasing recognition that substantial clinical benefit (SCB), that is, an improvement in clinical outcome that is needed for a patient to feel substantially better, is a more relevant endpoint for evaluating treatment success. This is particularly important in the context of knee cartilage repair, where patients often suffer from chronic pain and disability during or after physical activities or even at rest, which is perceived as a significant reduction in QoL, similar to patients scheduled for knee arthroplasty.9,10 In addition, these patients frequently avoid or reduce sports and exercise, which, in turn, may increase the risk of cardiovascular disease and other conditions.9,11
The purpose of this study was to assess the SCB achieved in the KOOS and IKDC score as defined by Ogura et al. 12 for a minimally invasive, hydrogel-based matrix-associated autologous chondrocyte implantation (M-ACI) in comparison with MFx in a propensity score matched-pair (PSM) analysis.
The authors hypothesized that superior outcomes would be demonstrated with hydrogel-based M-ACI.
Methods
Study Participants
Patients in the M-ACI group originated from the single-arm phase III study with NOVOCART Inject plus (TETEC—Tissue Engineering Technologies AG, Reutlingen, Germany; EudraCT no. 2016-002817-22, referred to as the NInject trial) and patients in the MFx group from the control group of the randomized phase III study comparing NOVOCART 3D plus and MFx (EudraCT no. 2011-005798-22, referred to as the N3D trial).
Both trials were conducted in full compliance with the principles laid down in the Declaration of Helsinki, the Guideline E6 for Good Clinical Practice of the International Conference on Harmonization (ICH GCP), and relevant local laws and regulations. After approval by the local ethics committees and federal authorities, patients who consented in writing to participate in the trials were enrolled and treated between October 2017 and February 2019, in the NInject trial, and between May 2013 and February 2018, in the N3D trial.
In both trials, men and women aged 18 to 65 years (or minors in the age of at least 14 years with closed epiphyseal growth plate) with localized articular cartilage defects of the knee (defect grade of III or IV according to the ICRS classification; maximum of 2 defects) were eligible for enrollment.
Relevant differences in eligibility criteria included defect size (4-12 cm² in the NInject trial and 2-6 cm2 in the N3D trial), defect location (patellar and tibial defects could be treated in the NInject trial but were excluded in the N3D trial), and prior cartilage repair (allowed in the NInject trial but not in the N3D trial). Detailed inclusion and exclusion criteria for both trials were reported previously. 13
Propensity Score Matching
One hundred patients were treated with M-ACI in the NInject trial and 85 patients received MFx in the N3D trial. Eight patients from the NInject trial with prior failed cartilage repair on the target knee were excluded from this analysis as this was an exclusion criterion in the N3D trial. Thus, 92 M-ACI patients and 85 MFx patients were available for PSM analysis. In the prespecified PSM procedure, each patient in the treatment group was provided with 1 patient from the comparison group (1:1 matching). A greedy algorithm was used for matching patients from both trials. The width required for matching patients was set at 0.2 times the pooled standard deviation estimate of the logit of the propensity score.
A logistic regression model was used for the 2 treatment groups to estimate the propensity score. Influencing variables, which were expected to have an impact on the therapy effect included in the model, were overall KOOS at baseline, time since first symptoms, prior target knee surgery, age, and sex. These variables were selected based on their statistical significance in the primary models and models of covariate effects for the KOOS score in the N3D trial. As defect sizes differed significantly in the 2 studies (4-12 cm2 in the NInject trial vs. 2-6 cm2 in the N3D trial), defect size was not included as a variable in the model as this would have resulted in an insufficient number of expected matched pairs for meaningful statistical analyses.
Surgical Techniques and Rehabilitation
M-ACI was performed as described earlier. 14 Briefly, in the first step, osteochondral biopsies were harvested from patients during arthroscopic surgery from a non-weightbearing area of the knee joint and were in vitro-culture expanded. In the second step, M-ACI was performed either arthroscopically or through a mini-arthrotomy approach using NOVOCART Inject plus, a two-component hydrogel-based M-ACI system, consisting of an autologous articular chondrocyte suspension (2-8 Mio. cells per mL) and a crosslinker solution. During injection using a dual-chamber syringe application system, the cell containing component and the crosslinker solution are mixed, resulting in the formation of the cell-seeded hydrogel at the site of administration.
MFx, a one-step surgical procedure, was performed according to Steadman and colleagues. 15
After surgery, patients in both groups followed a defined rehabilitation protocol based on Hirschmüller and colleagues.13,16 Limitations on weightbearing for 6 weeks were recommended with stepwise increase to full weightbearing between 7 and 8 weeks after surgery. Continuous passive motion with increasing range of motion was started a day after surgery for 6 weeks. Within the first 6 weeks, physical therapy was aimed primarily at the reduction of swelling, isometric quadriceps activity, and mobilization. The phase from week 12 to 26 was characterized by strength training, maximum sensorimotor stimulation, and low-impact sports, such as cycling or Nordic Walking. After week 26, a stepwise return to sports was allowed, depending on the persisting differences between treated and untreated leg and clinical symptoms. Criteria for unrestricted sports performance included being completely pain-free and having normal findings in the clinical examination of the knee joint as well as an almost complete return of strength and sensorimotor control. High-impact sports were not recommended within the first year after treatment.
Assessment Criteria and Statistical Analysis
Outcome assessment was based on the KOOS and IKDC scores, collected in both studies preoperatively and at 3, 6, 12, 18, and 24 months after treatment, respectively.
The SCB for the individual KOOS subscores and the IKDC score was defined as published by Ogura et al. 12 Accordingly, SCBs for the KOOS subscores were ≥27.7 points improvement compared with the preoperative level for pain, ≥14.28 points for symptoms, ≥29.4 points for ADL, ≥30.0 points for sports/rec., ≥37.5 points for QoL, and ≥34.4 points for the IKDC score.
The rate of patients achieving SCB (SCB responder rate) was assessed at all time points. Patients with missing assessments and patients classified as treatment failures before or during the respective visit were to be handled as SCB nonresponders, regardless of KOOS or IKDC response.
A sensitivity analysis was performed using a complete case approach. Only non-missing KOOS subscores or IKDC scores collected at the respective visit were analyzed. Patients classified as treatment failures before or during the respective visit were to be handled as SCB nonresponders, regardless of KOOS or IKDC response.
The SCB responder rates (i.e., the proportion of patients achieving SCB) at month 24 were compared by means of the Cochran-Mantel-Haenszel χ2 test stratified by region (Western Europe vs. Central and Eastern Europe).
The changes in KOOS subscores and IKDC score from baseline to the 3-, 6-, 12-, 18- and 24-month assessments are summarized separately for SCB responders and nonresponders.
All statistical analyses were performed using the software package SAS, Version 9.4 or higher.
Results
Propensity Score Analyses
Of the 92 patients eligible in the M-ACI group and 85 in the MFx group, a total of 72 matched pairs were selected by the predefined matching procedure.
The mean propensity scores in the eligible (i.e., prior to matching) M-ACI and MFx patients were 0.5543 and 0.4824, respectively, with a mean difference of 0.0718. In the matched-pairs population, the mean propensity scores in the M-ACI and MFx groups were 0.5327 and 0.5166, respectively, with a reduction in mean difference to 0.0161.
For most variables, a marked decrease in the mean difference was observed between the eligible population and the matched pairs (baseline KOOS 81.7%, time since symptoms 44.3%, age 60.3%, sex 100%, and logit propensity score 77.9%).
The only variable with no reduction in mean differences was “prior surgeries on the target knee,” which is explained by the eligible population in both groups having optimally similar proportions of patients with prior surgeries on the target knee. The variance ratios for all variables in the matched-pairs population were well within the commonly used bounds of 0.5 to 2.0. 17
The standardized mean differences between M-ACI and MFx groups in the eligible and matched populations are displayed in Figure 1 , which includes a cutoff of 0.1 for negligible differences, based on recommendations by Normand and colleagues. 18 The standardized differences of all variables were in the range of negligible differences, except for time since symptoms and logit propensity score, which were slightly above the cutoff point (0.1062 and 0.1226), but considered insignificant.
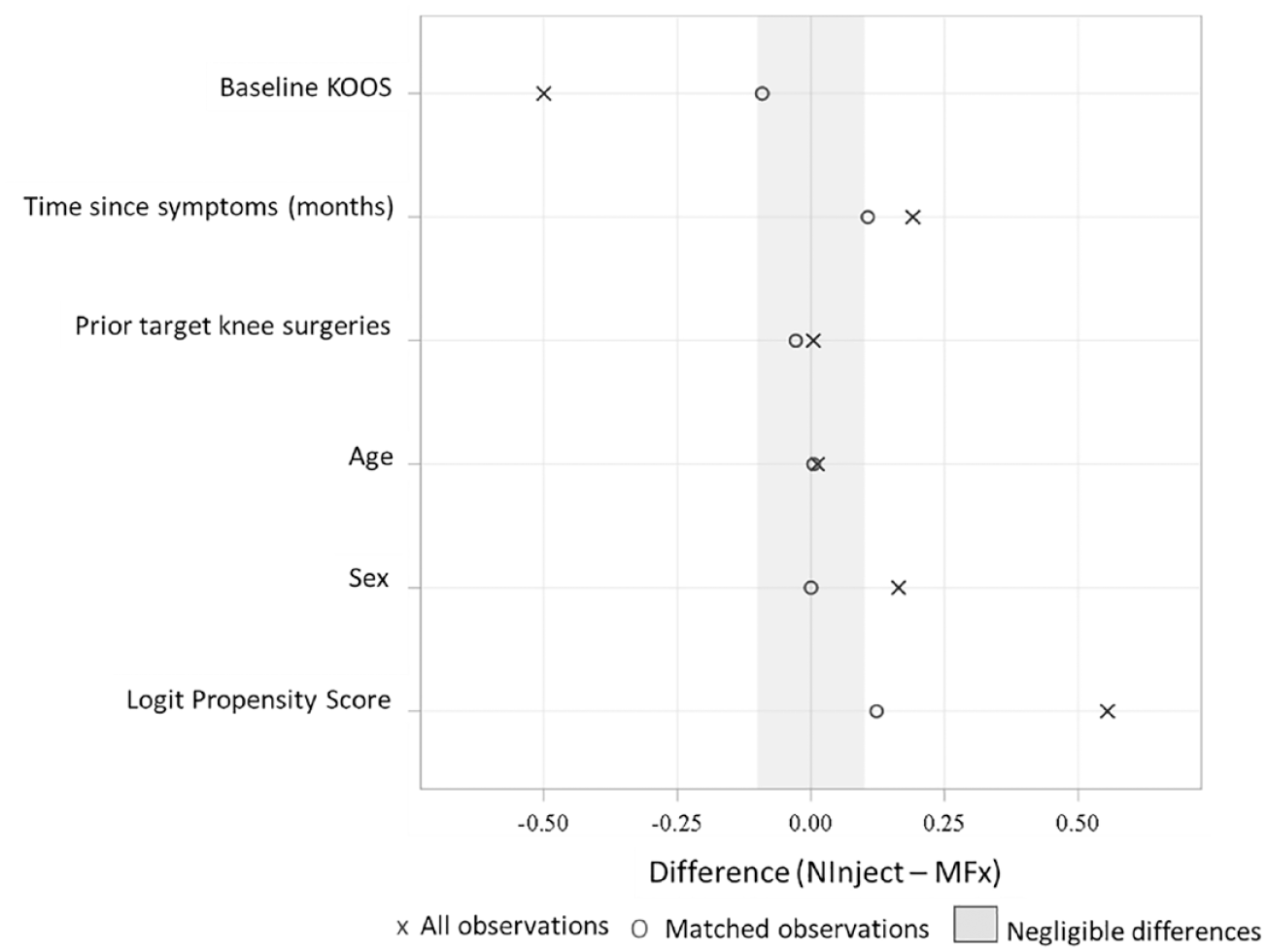
Standardized mean differences before and after matching. KOOS = Knee Injury and Osteoarthritis Outcome Score; MFx = microfracture.
Patient Population
The matching procedure resulted in a total of 144 patients in the matched-pair set (72 patients in the M-ACI group and 72 patients in the MFx group).
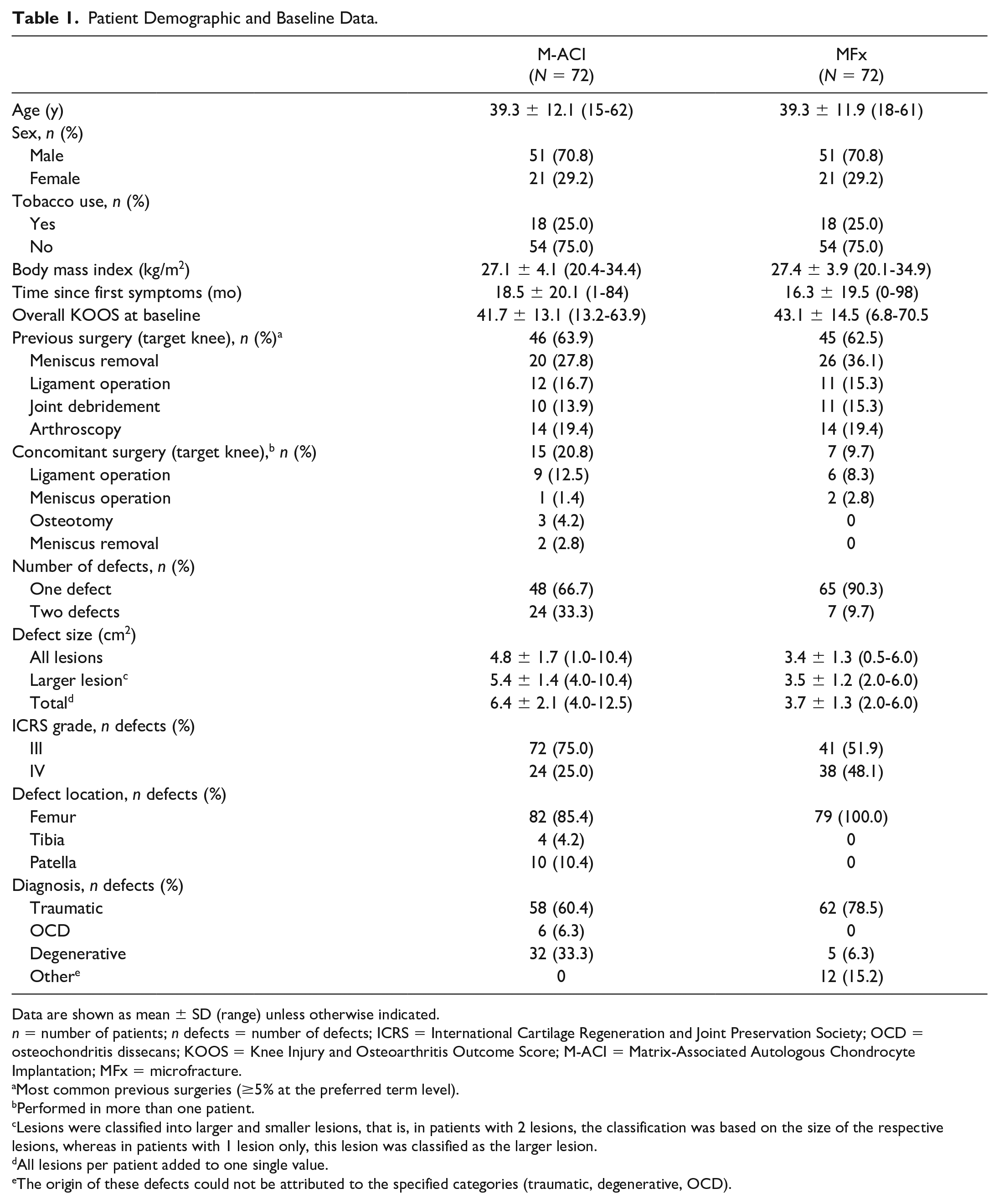
Patient demographic and baseline characteristics are summarized in Table 1 . The treatment groups were well-balanced with respect to overall KOOS at baseline, time since first symptoms, prior target knee surgery, age, and sex (i.e., the matched variables) as well as body mass index and tobacco use. A slight imbalance in the number of patients with concomitant surgeries (i.e., surgeries other than M-ACI transplantation or MFx) was considered irrelevant.
Patient Demographic and Baseline Data.
Data are shown as mean ± SD (range) unless otherwise indicated.
n = number of patients; n defects = number of defects; ICRS = International Cartilage Regeneration and Joint Preservation Society; OCD = osteochondritis dissecans; KOOS = Knee Injury and Osteoarthritis Outcome Score; M-ACI = Matrix-Associated Autologous Chondrocyte Implantation; MFx = microfracture.
Most common previous surgeries (≥5% at the preferred term level).
Performed in more than one patient.
Lesions were classified into larger and smaller lesions, that is, in patients with 2 lesions, the classification was based on the size of the respective lesions, whereas in patients with 1 lesion only, this lesion was classified as the larger lesion.
All lesions per patient added to one single value.
The origin of these defects could not be attributed to the specified categories (traumatic, degenerative, OCD).
There were some statistically significant (P < 0.01) differences in defect characteristics between treatment groups in terms of defect location (no patellar or tibial defects in the MFx group), number of defects (more patients with more than 1 defect in the M-ACI group), and defect etiology (more patients with degenerative lesions in the M-ACI group; no osteochondritis dissecans [OCD] lesions in the MFx group). A further difference between treatment groups was defect size, which was significantly larger in the M-ACI group compared with the MFx group (total defect size: 6.4 cm2 vs. 3.7 cm2, P < 0.0001).
Clinical Efficacy
At 24 months, significantly more patients in the M-ACI group compared with the MFx group achieved SCB in KOOS pain (72.2% vs. 48.6%; P = 0.0108), symptoms (84.7% vs. 61.1%, P = 0.0039), sports/rec. (84.7% vs. 56.9%, P = 0.0008), and QoL (72.2% vs. 44.4%, P = 0.0014; Table 2 ). The SCB responder rates for KOOS ADL and IKDC were also higher in the M-ACI group than in the MFx group (KOOS ADL: 58.3% vs. 44.4%, IKDC: 58.3% vs. 47.2%); however, the differences were not statistically significant (KOOS ADL: P = 0.2197, IKDC: P = 0.3969; Table 2 ). The corresponding results of the sensitivity analysis indicated higher SCB responder rates in the M-ACI group for all KOOS subscores and the IKDC score, with statistical significance achieved for KOOS sports/rec. (P = 0.0336) and QoL (P = 0.0251).
Substantial Benefit Responder Rates at 24 Months (Main Analysis).
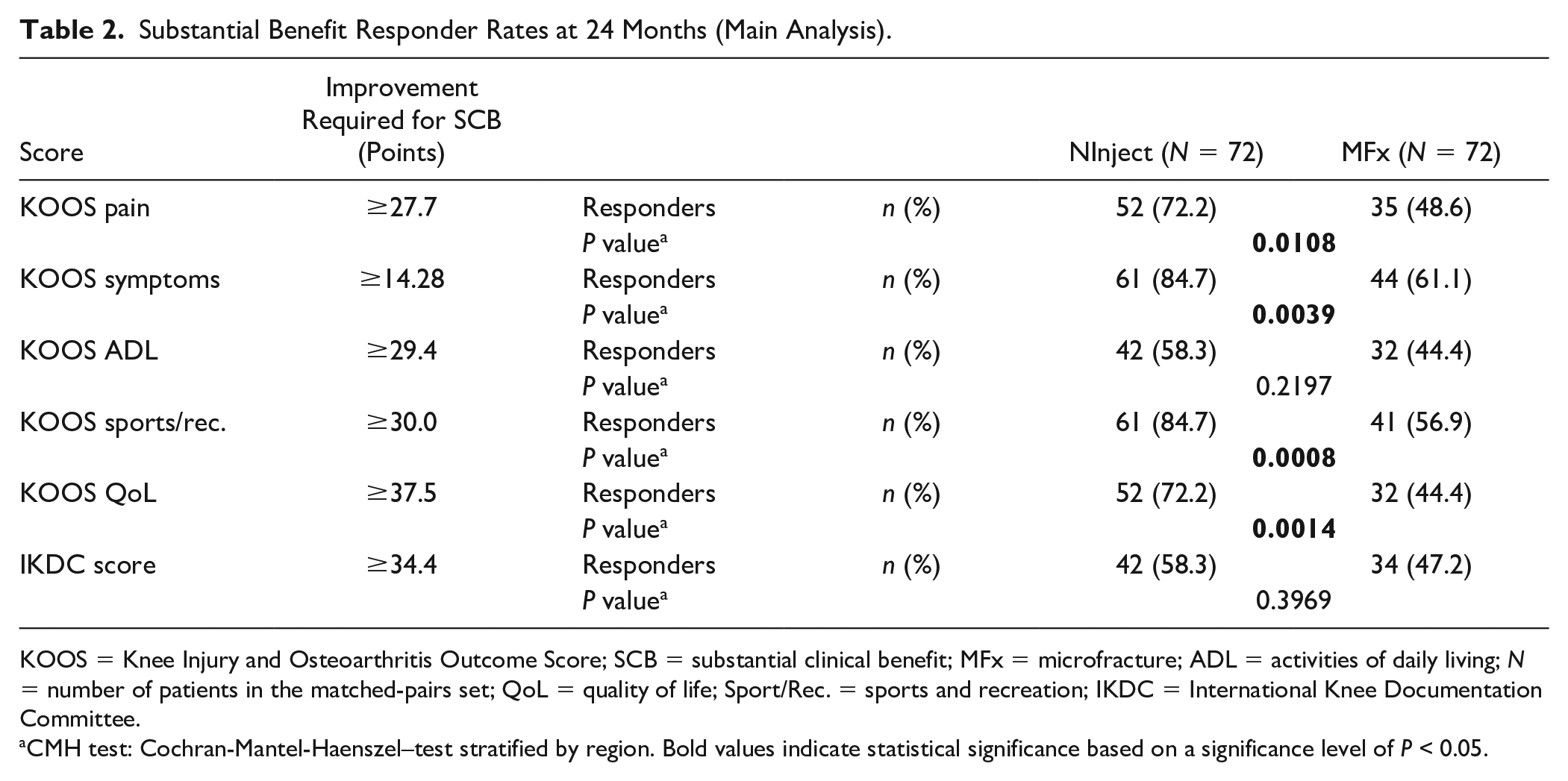
KOOS = Knee Injury and Osteoarthritis Outcome Score; SCB = substantial clinical benefit; MFx = microfracture; ADL = activities of daily living; N = number of patients in the matched-pairs set; QoL = quality of life; Sport/Rec. = sports and recreation; IKDC = International Knee Documentation Committee.
CMH test: Cochran-Mantel-Haenszel–test stratified by region. Bold values indicate statistical significance based on a significance level of P < 0.05.
With respect to earlier time points, SCB responder rates in the main and sensitivity analyses were all in favor of the M-ACI group compared with MFx from 3 months onward ( Fig. 2 ).
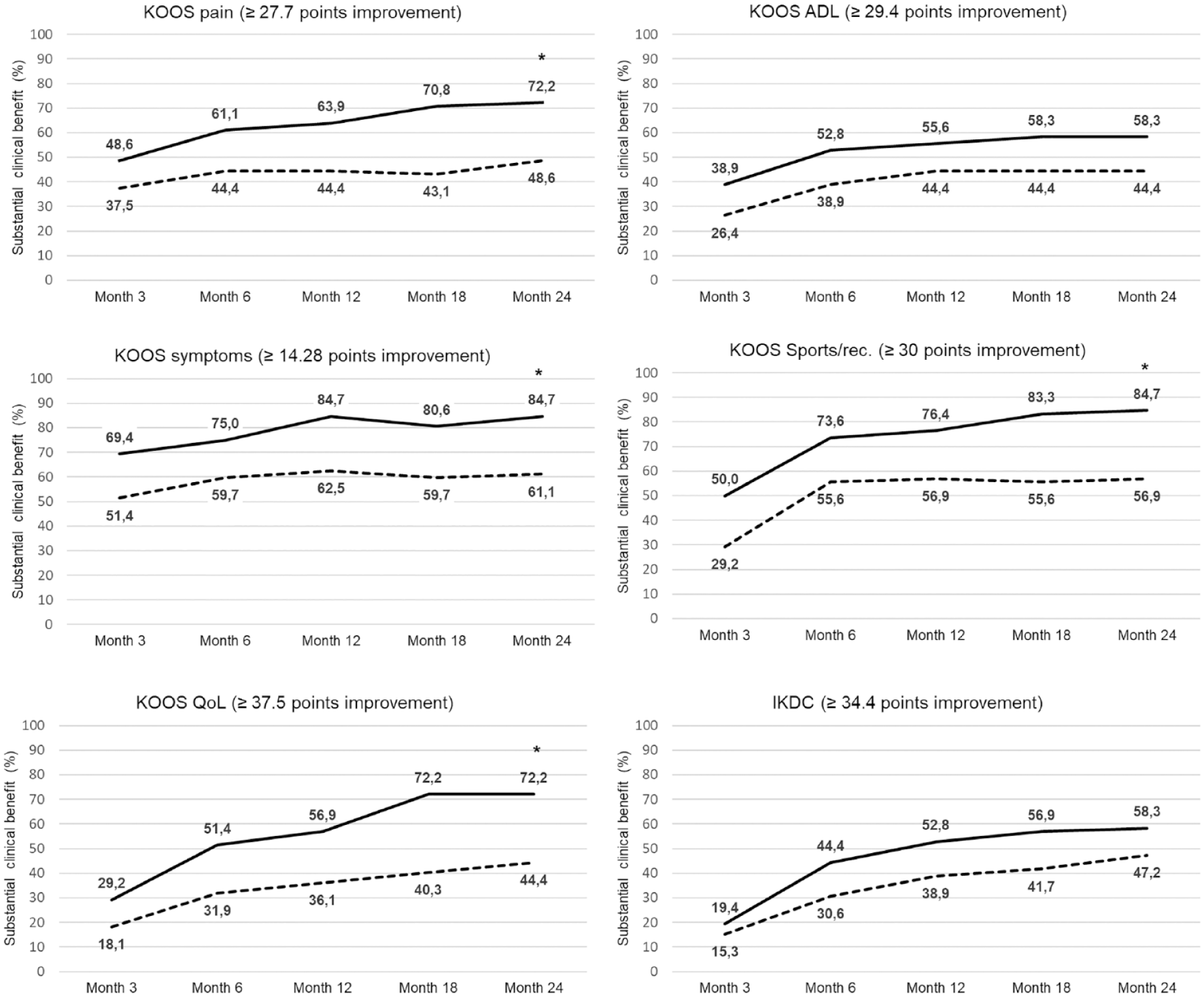
Substantial clinical benefit rates for KOOS subscores and IKDC score over time (main analysis). An asterisk indicates significant differences between treatment groups (significance testing performed at 24 months only). The solid lines refer to the M-ACI group and dashed lines to the MFx group. The numbers represent the percentage of patients achieving substantial clinical benefit in the respective KOOS subscores and IKDC score at each time point. KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = activities of daily living; QoL = quality of life; Sports/rec. = sports and recreation; IKDC = International Knee Documentation Committee score.
The KOOS subscore and IKDC score changes from baseline at 24 months in patients with SCB were overall comparable between treatment groups ( Fig. 3A ). The highest improvements were observed for sports/rec. (M-ACI: 60.8 ± 17.2 points, MFx: 55.9 ± 20.8 points) and QoL (M-ACI: 58.1 ± 16.5, MFx: 57.4 ± 15.4).
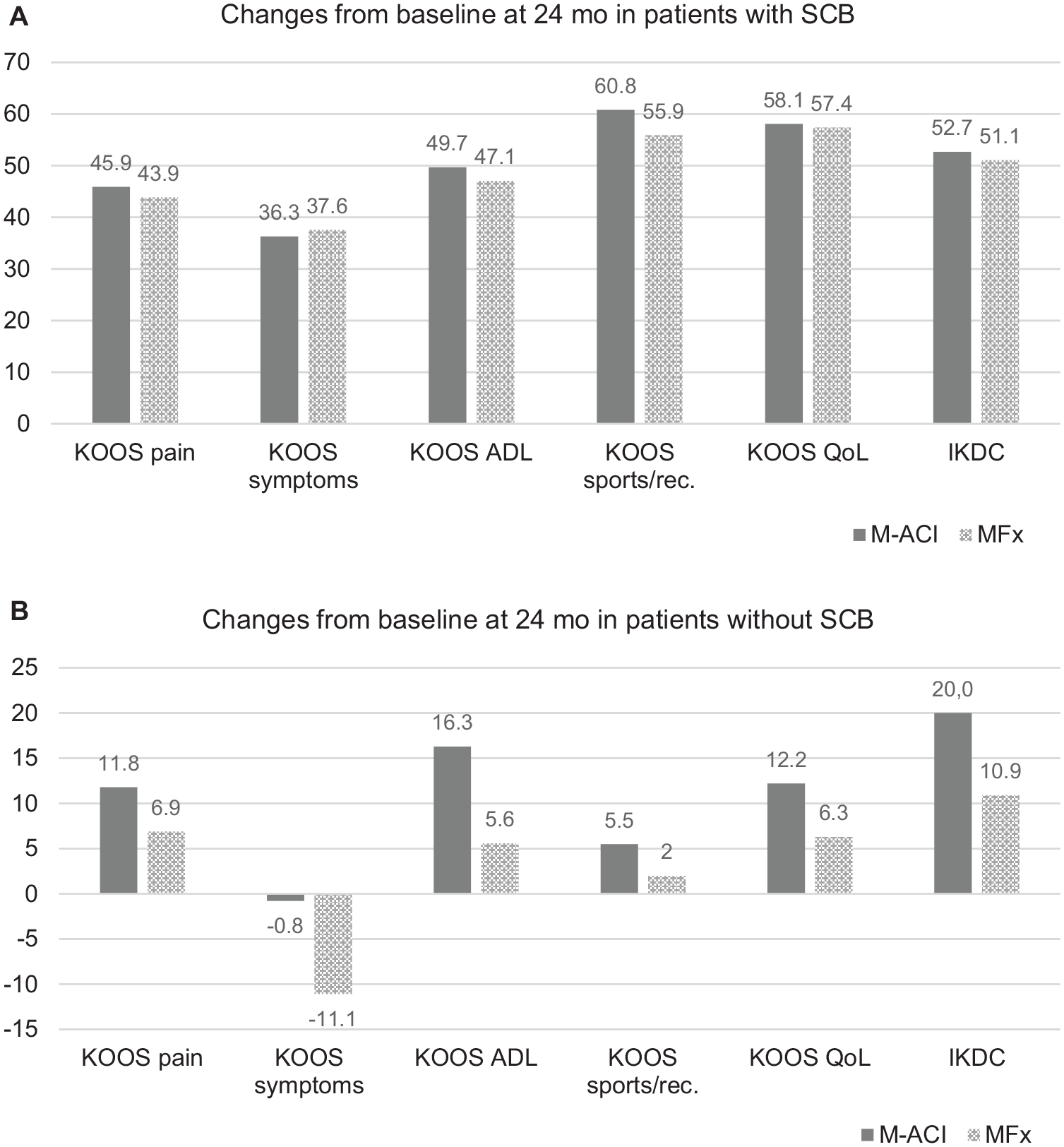
Changes from baseline at 24 months in KOOS subscores and IKDC score in patients with (A) and without (B) substantial clinical benefit (SCB). The numbers represent the mean change from baseline in the respective KOOS subscores and the IKDC score. KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = activities of daily living; QoL = quality of life; Sports/rec. = sports and recreation; IKDC = International Knee Documentation Committee score.
For patients who did not achieve an SCB, the improvement in both groups was by definition not as pronounced (
Treatment Failures and Unplanned Surgeries
No treatment failures (defined as surgical re-interventions affecting the covered surface of the transplant area) were reported in either the M-ACI or MFx group. Unplanned surgeries (mainly concerning the meniscus) were performed in 6 patients (8.3%) in the M-ACI group and 3 patients (4.2%) in the MFx group. In the MFx group, none of the unplanned subsequent surgeries were assessed as treatment related. In the M-ACI group 1 patient experienced a surgery-related lateral patellar compression syndrome that could have been caused by overtightened sutures of the knee joint capsule during transplantation surgery.
Discussion
In the present PSM, significantly more patients in the M-ACI group achieved SCB compared with patients treated by MFx in the KOOS subscores pain (P = 0.0108), symptoms (P = 0.0039), sports/rec. (P = 0.0008), and QoL (P = 0.0014) after 24 months of follow-up. The SCB responder rates for KOOS ADL and IKDC were also higher in the M-ACI group, but not significantly different from MFx.
There is an increasing focus on SCB in cartilage repair research, reflecting the growing understanding that treatment success should be evaluated from the perspective of patients, who typically expect optimal treatment results rather than minimal improvements.
Clinical benefit, including overall KOOS responder rates based on a threshold of a >10-point improvement and IKDC score based on a threshold of a >20.5-point improvement, has recently been published in the same matched-pair patient population presented here. 13 These thresholds were based on publications suggesting that an 8- to 10-point increase in KOOS and an improvement of more than 20.5 points in the IKDC score represent a minimal clinically relevant improvement (minimal clinically important difference [MCID]).5,8 In the abovementioned analysis, the overall KOOS and IKDC responder rates were significantly higher for hydrogel-based M-ACI compared with MFx (KOOS: 94.4% vs. 65.3%, P < 0.0001; IKDC: 83.3% vs. 61.1%, P = 0.0126). 13
The focus of this analysis was to compare the SCB, that is, an improvement significant enough to make a patient feel substantially better, of hydrogel-based M-ACI with MFx after a follow-up of 2 years. There is no universally agreed-upon threshold for what constitutes a substantial clinical improvement following cartilage repair procedures. The thresholds for SCB used in the current PSM analysis were based on a publication by Ogura et al., 12 who investigated SCB for KOOS and IKDC after a minimum follow-up of 2 years (mean 2.3 years) in 92 patients who had received ACI (either collagen membrane covered ACI or M-ACI) for the treatment of knee cartilage defects. The patient population was comparable to that in the present matched-pair analysis in terms of follow-up duration, defect size (total defect area: 6.9 cm2 vs. 6.4 cm2), and number of defects (1.6 vs. 1.2), but patients were younger (mean age 31.4 vs. 39.3 years), concomitant procedures were performed more frequently (67% vs. 20.8%), and more defects were located at the patella (43.4% vs. 10.4%).
Applying the SCB thresholds proposed by Ogura et al. to the present PSM analysis, changes from baseline in the individual subscores were clearly different in patients with and without SCB in both treatment groups, supporting the validity of the proposed SCB thresholds. Hydrogel-based M-ACI has shown statistically significant superiority compared with MFx in terms of the percentage of patients with SCB in KOOS pain (72.2% vs. 48.6%; P = 0.0108), symptoms (84.7% vs. 61.1%, P = 0.0039), sports/rec. (84.7% vs. 56.9%, P = 0.0008), and QoL (72.2% vs. 44.4%, P = 0.0014). The SCBs for KOOS ADL (58.3% vs. 44.4%, P = 0.2197) and IKDC score (58.3% vs. 47.2%, P = 0.3969) were not significantly different. Overall, the proportion of patients achieving SCB in each KOOS subscore and IKDC score in the M-ACI group was clearly higher than reported in the study by Ogura et al. 12 (pain: 45.8%, symptoms: 37.6%, ADL: 37.3%, sports/rec.: 58.3%, QoL: 50.6%, and IKDC: 47.6). 12
The strongest improvements from baseline in patients with SCB in both treatment groups of the matched-pair analysis were observed for KOOS sports/rec. (M-ACI: 60.8 points, MFx: 55.9 points) and QoL (M-ACI: 58.1, MFx: 57.4). This is in accordance with a systematic review of randomized controlled trials (RCTs) by Abraamyan et al. 19 investigating KOOS subscores in response to various cartilage restoration procedures, where the authors consider KOOS sport/rec. and QoL the two most responsive KOOS subscores. Moreover, the differences in SCB rates between the 2 groups in the present matched-pair analysis were most pronounced in these subscores, which is consistent with the systematic review by Abraamyan et al., in which ACI/M-ACI treatment resulted in clinically superior outcomes compared with microfracturing in KOOS sport/rec. The mean delta in KOOS QoL after ACI/MACI was 6.6 points greater than after microfracturing; however, statistical significance was narrowly missed (P = 0.06). 19 Of note, KOOS sport/rec. and QoL are also considered the most relevant domains for determining patient satisfaction.20,21
The inferior outcome after MFx compared with other cartilage repair techniques could be due to the inferior quality of the repair tissue. MFx is known to result in primarily fibrocartilage with poor biomechanical properties and inferior degree of defect fill, particularly in larger lesions.22,23 In this regard, in vitro studies have shown that mesenchymal stromal cells from bone marrow or other origins produce less extracellular matrix per seeded cell in relation to chondrocytes isolated from articular cartilage and more proteins associated with fibrous cartilage, hypertrophic cartilage, and bone formation.24-27 In a meta-analysis comparing histological outcomes after different cartilage repair techniques, OAT repair tissue primarily comprised hyaline cartilage, followed closely by repair tissue formed by cell-based techniques, whereas MFx was found to produce primarily fibrocartilage. The percent change in clinical outcome was also significantly associated with the percent of repair tissue biopsies showing only hyaline cartilage (R2 = 0.24, P = 0.024). 28 For hydrogel-based M-ACI, significantly higher magnetic resonance observation of cartilage repair tissue (MOCART) scores were reported for the M-ACI group compared with MFx after 24 months (MOCART score least-squares mean: 86.9 points vs. 69.1 points for MFx, P = 0.0096) based on the subgroup of patients with magnetic resonance imaging (MRI) examination (M-ACI: 21 patients with 25 lesions, MFx: 28 patients with 31 lesions). 13 Of note, in the M-ACI group, knee cartilage defects in the MRI subpopulation were considerably larger (5.3 cm2) than in the MFx group (3.3 cm2). In addition, there is evidence from gray-level co-occurrence matrix (GLCM) texture analysis of quantitative T2 maps that the damaged perilesional cartilage also benefited from hydrogel-based M-ACI treatment in terms of tissue structure approximation to healthy cartilage. 29
An inferior repair tissue structure characterized by fibrocartilage formation and/or lower MOCART scores might also be an explanation for worsening of clinical outcome after cartilage repair over time. In a meta-analysis from Jones et al. including 3,894 patients, MFx (mean defect size: 3.17 cm2) lost its ability to provide clinically relevant pain relief during midterm follow-up (5-9 years). This was not the case with ACI (mean defect size: 3.98 cm2) even in long-term follow-up (≥10 years), where a further decrease in pain was observed. 30
The superior SCB of hydrogel-based M-ACI compared with MFx in this PSM analysis might be attributed to the beneficial cartilage restoration properties of the product NOVOCART Inject plus. Both in vitro and in vivo experiments have shown that in the three-dimensional crosslinked hydrogel, human chondrocytes have a chondrocyte-typical, spherical cell morphology, similar to that in hyaline cartilage, with high viability, and that the hydrogel stabilizes the phenotype of chondrogenic cells or significantly improves the phenotype of expanded chondrocytes, respectively.31-33 Furthermore, due to its physical properties, the hydrogel provides a barrier function for inflammatory and endothelial cell invasion, resulting in anti-inflammatory, anti-angiogenic, and thus anti-osteogenic effects, thereby inhibiting the adverse consequences of these cartilage-destructive pathways.31,33,34 Even low-grade inflammation, for example, following knee injury, can not only promote cartilage degeneration but also negatively alter the joint environment for chondrogenesis and impair the outcome of cartilage repair.35-38
Furthermore, unlike membrane-covered or scaffold-seeded M-ACI, hydrogel-based M-ACI allows for arthroscopic or minimally invasive application without the need for additional implant manipulation or fixation. The associated reduction of surgical comorbidity and the shortened cut-to-suture time for product implantation reduces the risk of adverse events (surgical-site infection, sepsis, extended length of hospital stay, and readmission) and protects against detrimental structural, biochemical, and metabolic changes of articular cartilage correlated with increased operative times and suture or fibrin glue fixation.39-44
Limitations
A limitation of this analysis is that covariates for propensity score matching were limited to include as many patients as possible in the matched-pair analysis to achieve meaningful results. This led to some imbalances mainly in defect characteristics (e.g., size, location, etiology, and number of lesions). The impact of these differences in patient characteristics remains unclear; however, it can be assumed that they do not represent a bias in favor of the M-ACI group as discussed previously by Niemeyer et al. 13 Nonetheless, it is acknowledged that MFx does not represent a suitable comparator for M-ACI in larger defects. Future studies may focus on comparing hydrogel-based M-ACI with other methods of treating larger cartilage defects. Another limitation is that the definition of SCB thresholds is affected by a multitude of factors, such as patient characteristics, study size, pathology, intervention, length of follow-up, and calculation methods for SCB. The thresholds for SCB used in the current PSM analysis were based on a publication by Ogura et al. 12 Some parameters, such as pathology (cartilage defects of the knee), intervention (either collagen membrane covered ACI or M-ACI), length of follow-up (2.3 years), and defect characteristics, such as defect size and number of defects in this study were overall comparable to the M-ACI group of the present matched-pair analysis. Other parameters were not comparable, including age, concomitant procedures, and defect location. The impact of these differences in patient characteristics on the KOOS subscore SCB thresholds is unknown.
Conclusions
In this PSM analysis, M-ACI using an in situ crosslinked hydrogel has demonstrated SCB in large cartilage defects of the knee (4-12 cm2) that is superior to MFx in smaller defects (2-6 cm2) based on KOOS pain, symptoms, sports/rec., and QoL. Whereas the traditional benchmark for evaluating cartilage repair is clinically relevant improvement, this study demonstrates the need and value in further evaluating SCB as a measure of success more important to the patient. This endpoint is particularly relevant for knee cartilage repair as patients often suffer from chronic pain and disability.
Footnotes
Acknowledgments and Funding
The authors acknowledge the following investigators for contributing a significant number of patients to the phase III studies used for matching: Martin Hanus (Department of Orthopedics and Traumatology, 2nd Faculty of Medicine, Charles University in Prague and Motol University Hospital, Praha, Czech Republic), Rimtautas Gudas (Hospital of Lithuanian University of Health Sciences, Kaunas Clinics), Juozas Belickas (Lithuanian University of Health Sciences, Ortopedijos Technika, Kaunas, Lithuania), Markas Fiodorovas and Algimantas Cebatorius (University Hospital Klaipeda, Klaipeda, Lithuania), Milan Pastucha (Department of Orthopedics, Hospital Hořovice, Hořovice, Czech Republic), Petr Hoza (Department of Orthopedics, Hospital Pardubice, Pardubice, Czech Republic), and Krisztián Magos (Kastélypark Clinic Hajdú, Tata, Hungary). This study was funded by TETEC—Tissue Engineering Technologies AG, Reutlingen, Germany.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.G., A.Kö., and A.Ki. are employees of TETEC AG. R.S. is an employee of Aesculap Biologics LLC. P.A. and P.N. received consulting or advisory, and speaking and lecture fees from TETEC AG. P.A. holds a professorship sponsored by Aesculap AG.
Ethical Approval
Ethical approvals for the studies used for propensity score matching were obtained from the independent ethics committee (IEC) of the University Medical Center Regensburg, Germany (lead IEC of the N3D study) and the ethics committee of the Bavarian State Medical Association, Germany (lead IEC of the NInject study).
Trial Registration: NInject trial: NCT03319797; EudraCT no. 2016-002817-22; N3D trial: NCT01656902; EudraCT no. 2011-005798-22
