Abstract
Objective
A novel aragonite-based scaffold has been developed. In this study, mid-term clinical and magnetic resonance imaging (MRI) results on 12 patients affected by isolated chondral or osteochondral lesions of the knee treated by the scaffold implantation have been evaluated at a mean follow-up of 6.5 (range: 5-8) years.
Design
The study population consisted of 3 females and 9 males, mean age 34.4 (20-51) years. The lesion was located on the medial femoral condyle, the trochlea, and the lateral femoral condyle in 5, 5, and 2 patients, respectively. In all cases, a single lesion over grade 3 of the International Cartilage Restoration and Joint Preservation Society (ICRS) classification was treated: in 9 cases by implantation of one plug, and in 2 cases with 2 plugs; the mean size of the lesion was 2.5 cm2 (1-7).
Results
One patient failed and was revised with a custom-made metal implant (Episealer). Overall, Knee Injury and Osteoarthritis Outcome Score (KOOS) significantly improved from 45 ± 13 preoperatively to 86 ± 13 at final follow-up. All KOOS subscales improved significantly: pain subscale increased from 48 ± 12 to 92 ± 11; symptoms from 66 ± 13 to 91 ± 13; activity of daily living (ADL) from 60 ± 19 to 90 ± 21; sport from 23 ± 20 to 75 ± 20; finally, quality of life (QoL) increased from 27 ± 14 to 77 ± 19. Long-term MRI MOCART score was 64.
Conclusions
This study shows continued significant clinical improvement and good magnetic resonance imaging (MRI) findings with a minimum 5 years follow-up after implantation of a novel aragonite derived scaffold for the treatment of cartilage lesions of the knee. One patient failed and was revised with a custom-made metal implant (Episealer).
Keywords
Introduction
Chondral and osteochondral lesions of the knee are common and affect young and middle-aged, often active and athletic patients who have functional impairment due to their cartilage damage. 1 If left untreated, cartilage lesions can progress and cause damage to the underlying subchondral bone, leading to chronic pain, stiffness and limitations in daily activities. In addition, untreated cartilage lesions can increase the risk of developing osteoarthritis (OA), a degenerative joint disease characterized by progressive damage to the cartilage, subchondral bone and other joint structures2-4; in fact, according to the Centers for Disease Control and Prevention, an estimated 15% of U.S. adults develop OA. 5
For an International Cartilage Restoration and Joint Preservation Society (ICRS) lesion grade 3 or 4, when more than 50% of the cartilage layer is damaged or the subchondral bone is affected, surgical intervention is justified.
Nowadays, there is a growing awareness of the importance of the osteochondral unit in cartilage repair, and the role of natural or synthetic scaffolds, which are not only vehicles for stem cell delivery but also have an intrinsic ability to promote cartilage repair. 6
The subchondral bone plays a multifaceted role in knee cartilage health. First, it provides structural support; in addition, it aids in nutrient supply to avascular cartilage. It contributes to shock absorption, cysts or sclerosis can hinder this function, increasing stress on cartilage. Injuries or inflammation in the subchondral bone can trigger an inflammatory response affecting adjacent cartilage. When addressing cartilage lesions in the knee, considering the subchondral bone’s state is crucial. Understanding this interplay is pivotal for effective treatment, aiming to restore the osteochondral unit to maintain joint function and prevent cartilage issues. 7
A novel aragonite-based scaffold has been developed for the treatment of chondral and osteochondral lesions of the knee. Outcomes and safety of the new device have been evaluated at short-term follow-up,8-11 and a recent multicenter randomized clinical trial has shown superiority over the standard of care (microfracture and debridment).7,12 The aim of this article is to report on the medium-term outcome results of this scaffold.
Materials and Methods
Clinical and magnetic resonance imaging (MRI) results of 12 patients from a single center, who were enrolled in a prospective multicenter international trial from May 2014 to May 2017, were analyzed. A 3-year follow-up on these same patients was previously published. 9 From the original 13 patients, 2 patients failed between 3 to 5 years. In one case, failure was not related to the surgery: the patient was diagnosed with arthritis due to hemochromatosis after the scaffold implantation. In the second case, a 48-year-old male was revised after 5 years with a focal arthroplasty (Episealer, Episurf, Sweden) due to persisting pain and swelling after an extensive overload of the knee, due to landing on the knee after a high fall.
The clinical trial was conducted according to the Helsinki Declaration of 1964 and its later amendments, good medical practice, and the harmonized standards for clinical investigation of medical devices (ISO 14155). In addition, the trial was approved by an independent Institutional Review Board, recorded in the national register for clinical trials, and registered on clinicaltrials.org (B300201733407). Before entering the study, every patient signed informed consent and agreed to comply with the postoperative rehabilitation protocol.
The headline inclusion criteria were as follows (full inclusion/exclusion criteria on Table 1): patients aged between 18 and 55 years, symptomatic, full or nearly full-thickness (grade 3-4 ICRS classification) chondral or osteochondral isolated lesion of the femoral condyle, trochlea, with a KOOS Pain score between 30 and 65, a defect area up to 7 cm2, primary or secondary articular cartilage repair, stable knee. On the contrary, exclusion criteria were as follows: uncorrected misalignment >5°, patellar cartilage pathology, meniscal resection of more than 50%, bony defect depth over 4 mm, bipolar cartilage lesions, and OA of the operated knee.
Inclusion and Exclusion Criteria.
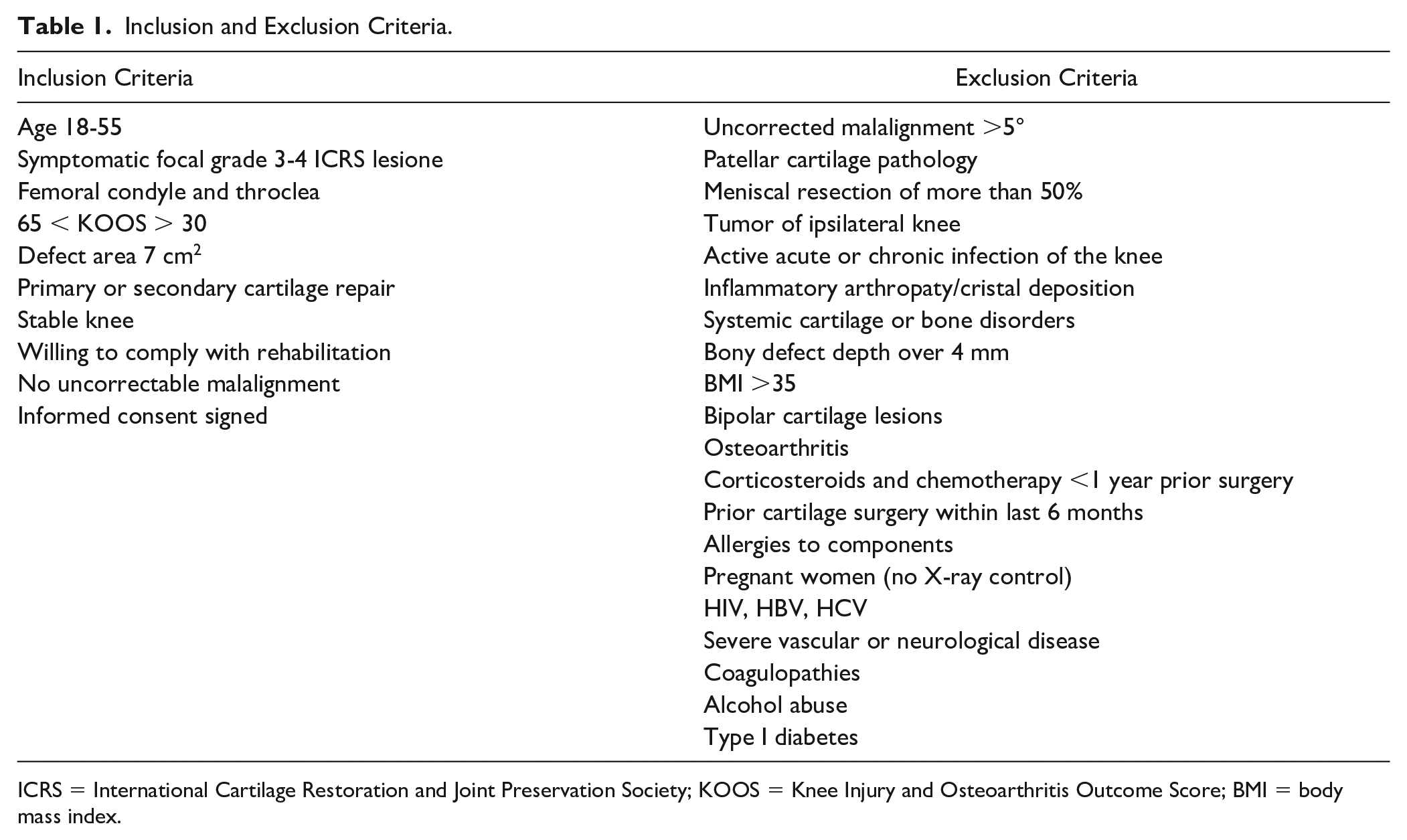
ICRS = International Cartilage Restoration and Joint Preservation Society; KOOS = Knee Injury and Osteoarthritis Outcome Score; BMI = body mass index.
The study population (Table 2) consisted of 3 females 25% and 9 males 75 aged 34.4 (20-51) years with a body mass index (BMI) of 25.0 ± 3.6. Osteoarthritis severity was K/L 1 in 4 patients (33%) and K/L 2 in 8 patients (66%)) and K/L 3 in 1 patient. Lesion size ranged from 1.7 to 7.0 cm2 (average: 2.5 cm2) with 5 defects (41.7%) located on the medial femoral condyle (MFC), 2 (16.7%) on the lateral femoral condyle (LFC), and 5 (41.7%) in the trochlear groove. Acute symptomatic onset was present in 7 patients 58%, whereas, in general, sports was the primary cause of injury. Six patients had prior surgery of the index knee as in debridement, MF, partial meniscectomy (PM), or a combination of these interventions. In all cases, there was a single lesion over grade 3 of the ICRS classification. In 10 cases, they were treated by implantation of a single plug. In only two cases, two plugs were implanted.
Clinical Case of Trochlear Osteochondritis Dissecans (OCD) With Post-Op MRI 7 Years Follow-Up.
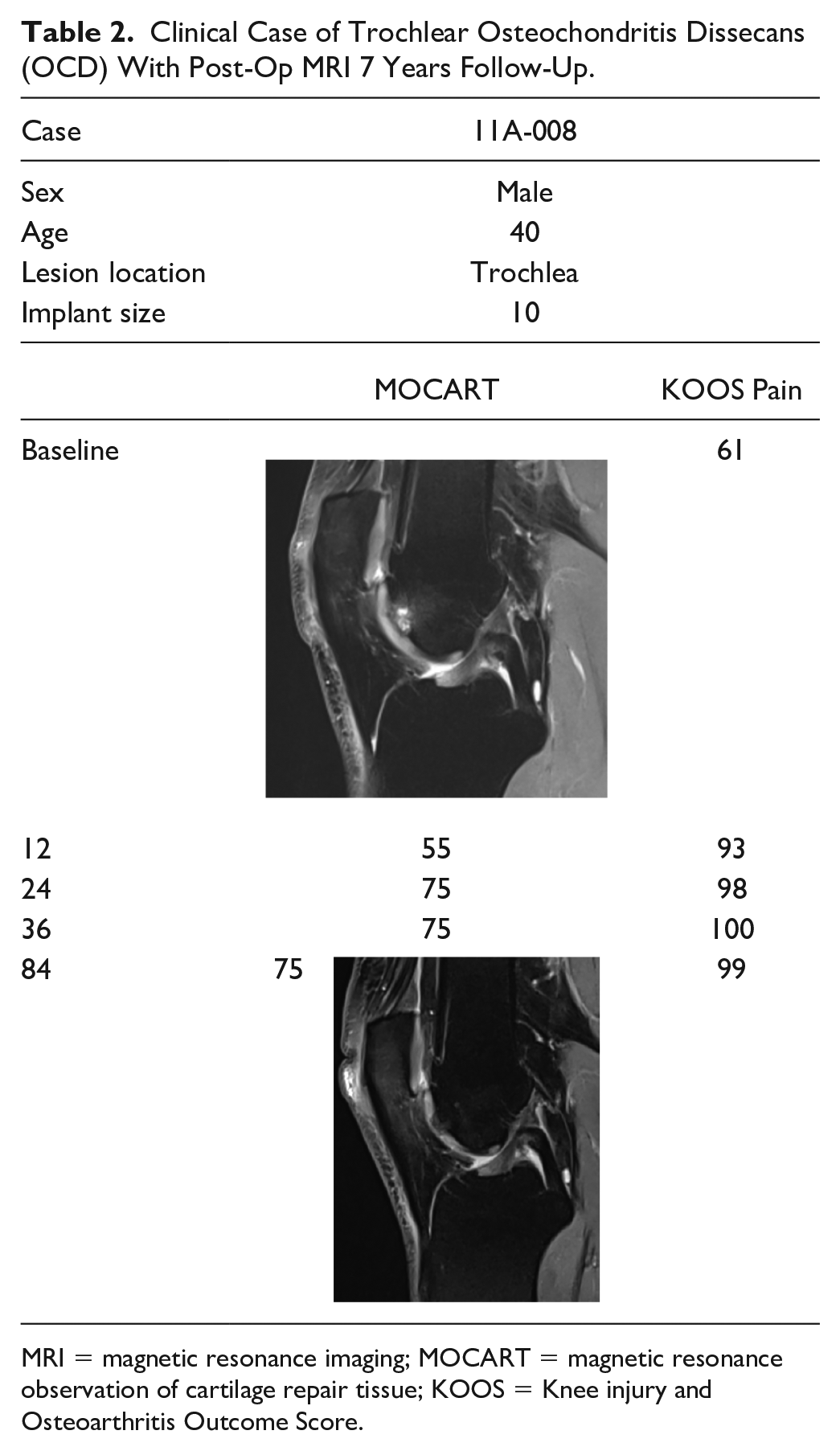
MRI = magnetic resonance imaging; MOCART = magnetic resonance observation of cartilage repair tissue; KOOS = Knee injury and Osteoarthritis Outcome Score.
The mean size of the lesion was 2.5 cm² (1-7cm2). In 5 cases, 1 plug size 10 was used, in 4 cases a plug 12.5 was implanted, in 1 case a size 14 and in the last 2 cases size 15 was used. No small implants were used (diameter 7.5 mm). In the two cases where two plugs were implanted, a 12.5 + 10 and a 12.5 + 12.5 were used.
Clinical Evaluation
Clinical outcomes were evaluated at baseline, 6, 12, 18, 24, and 36 months, and final follow-up (range: 60-96 months) using the KOOS pain subscale, that was the primary endpoint. Secondary measures included other KOOS subscales (symptoms, activity of daily living [ADL], sports/recreation, quality of life [QoL]). Additional analysis assessed potential influences on outcomes, including gender, prior or combined surgery, and injury mechanism.
Imaging Analysis
Magnetic resonance imaging evaluation was performed preoperatively and at 12, 24, 36, 60, 72, 84, and 96 months at latest follow-up following a standardized imaging protocol with 7 special sequences for cartilage analysis on a 1.5-T MR machine (Siemens Healthcare, Magnetom Aera). Magnetic resonance imaging at final follow-up was available for 10/11 patients. Sequences consisted of (1) sagittal T1-weighted fast spin echo sequence, (2) sagittal T2/PD-weighted fast spin echo sequence with fat saturation, (3) axial T2/PD-weighted fast spin echo sequence with fat saturation, (4) coronal PD-weighted fast spin echo sequence with fat saturation, (5) coronal oblique PD-weighted fast spin echo sequence with fat saturation oriented orthogonal to the scaffold, (6) sagittal 3D spoiled gradient echo sequence with water excitation (DESS), and (7) sagittal T2 mapping sequence composed of an 8 echo train. A dedicated 15-channel knee coil was used in every case. Osteochondral defect typing was preoperatively determined according to the ICRS scoring system, whereas preoperative OA severity was scored using the K/L classification on weight-bearing anteroposterior (AP) radiographs of the index knee. The Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) 2.0 knee score (100 points) 25 was used for semiquantitative assessment of osteochondral restoration at the respective time points after surgery. In subjects with 2 implants, both were scored separately. The criterion of “subchondral changes” was applied on the bony region surrounding the scaffold. In addition of the MOCART score, the implant incorporation in the subchondral bone was determined by the scaffold signal intensity (% of scaffold surface) on multidirectional T2 images.
Scaffold in-growth was classified as “66% to 100% incorporation,” “33% to 66% incorporation,” or “0% to 33% incorporation.” All radiographs and MRIs were evaluated by an independent musculoskeletal radiologist and an experienced orthopedic surgeon (FDC).
Study Device, Surgical Procedure, and Rehabilitation Protocol
Agili-C™ scaffold (CartiHeal Ltd, Israel), commercial name of the discussed implant, is a CE-marked (european certified) porous biphasic implant by CartiHeal Ltd. (Israel). It is made of over 98% aragonite calcium carbonate from natural coralline exoskeleton. The top layer has micro-drilled channels.
A single senior knee surgeon performed all procedures. Patients had arthroscopic surgery for lesion evaluation. A mini-arthrotomy was done, lesion dimensions documented, and a guided pin inserted. The defect was prepared using a drill, reamer, and shaper, repeated 3 times. Healthy cartilage edges were contoured. The scaffold was placed beneath the native cartilage and surrounded by vital subchondral bone, maintaining a 5-mm bone bridge for multiple implants. Agili-C surgical tools were used for precise implant placement.
Patients were discharged with a knee brace and followed a specific rehabilitation protocol based on the joint defect. For trochlear lesions, full weight-bearing was allowed after 2 weeks, with limited range of motion for 8 weeks. For condylar lesions, range of motion was restricted for 2 weeks and progressive weight-bearing started after 8 weeks. Soft tissue mobilization, core stability training, and initial strengthening exercises were allowed for the first 4 weeks. Closed chain exercises were introduced at 6 weeks, followed by resistance muscle strengthening at 3 months. At 6 months, patients could gradually resume most sports, excluding jogging until 9 months and contact sports until 12 months.
Statistical Analysis
Descriptive data were displayed with their means and standard deviation (SD). Data were assessed for normal distribution by the Kolmogorov-Smirnov test before proceeding with parametric or nonparametric analysis. Normal data distribution was rejected (P < .05) for at least one time point in every outcome measure. Seeing the small sample size, nonparametric testing (Mann-Whitney U test) was chosen to compare two variables. All statistical tests were performed in JASP (JASP Team [2023]. JASP (Version 0.17.3)[Computer software]).
Results
Overall KOOS score (KOOS5) improved significantly from 45 ± 13 preoperatively to 85 at 2 years 10 and to 86 ± 13 at final follow-up (Figs. 1 and 2). All KOOS subscales improved significantly: the pain subscale increased from 48 ± 12 to 92 ± 11; symptoms from 66 ± 13 to 91 ± 13; ADL from 60 ± 19 to 90 ± 21; sport from 23 ± 20 to 75 ± 20; finally, QoL increased from 27 ± 14 to 77 ± 19.
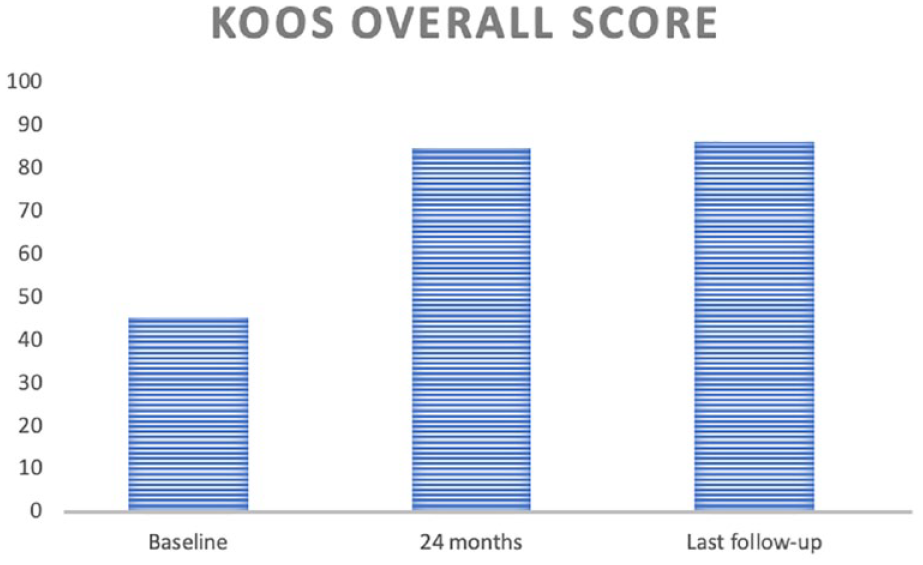
KOOS scores at baseline, 2 years and final follow-up showing a stable improvement. KOOS = Knee Injury and Osteoarthritis Outcome Score.
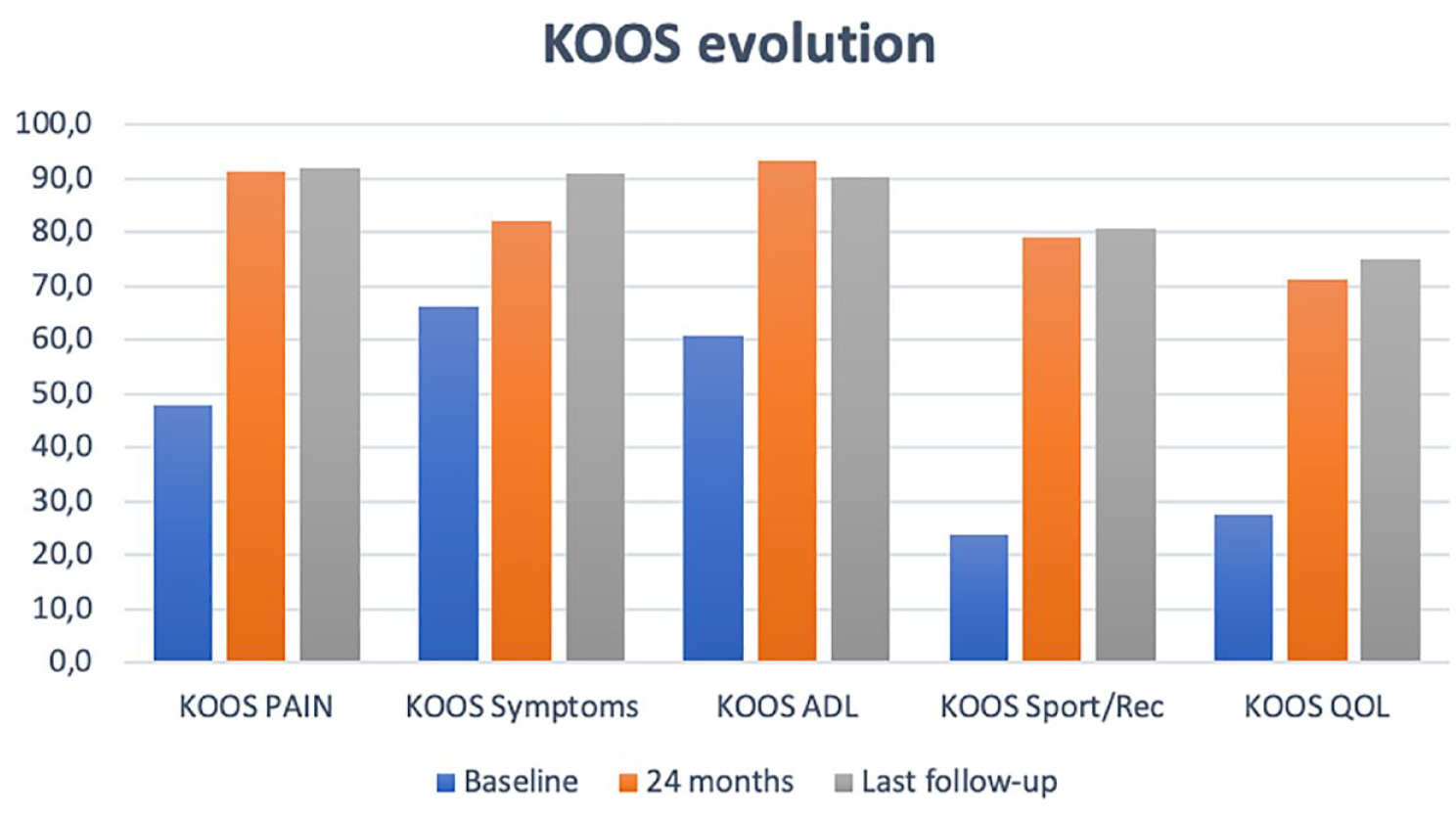
KOOS subscales at baseline, 2 years, 3 years, and final follow-up. KOOS = Knee Injury and Osteoarthritis Outcome Score.
Further analysis was performed to evaluate variables that might influence the clinical outcome: gender, previous or combined surgery, and mechanism of injury. Gender did not significantly influence the clinical outcome. Patients with previous cartilage surgery 6 improved from 36 ± 7 to 78 ± 1 while patients without previous cartilage surgery 6 improved from 49 ± 10 to 94 ± 6 (independent t-test P = .044). Similarly, patients that underwent a previous meniscectomy 4 improved to 77 ± 1while the nonmeniscectomy group 12 improved to 94 ± 6 at latest follow-up (independent t-test P = .019). Finally, acute injuries 7 tended to show superior results (93 ± 6 vs 77 ± 7; independent t-test P = .082) (Figs. 3-5)
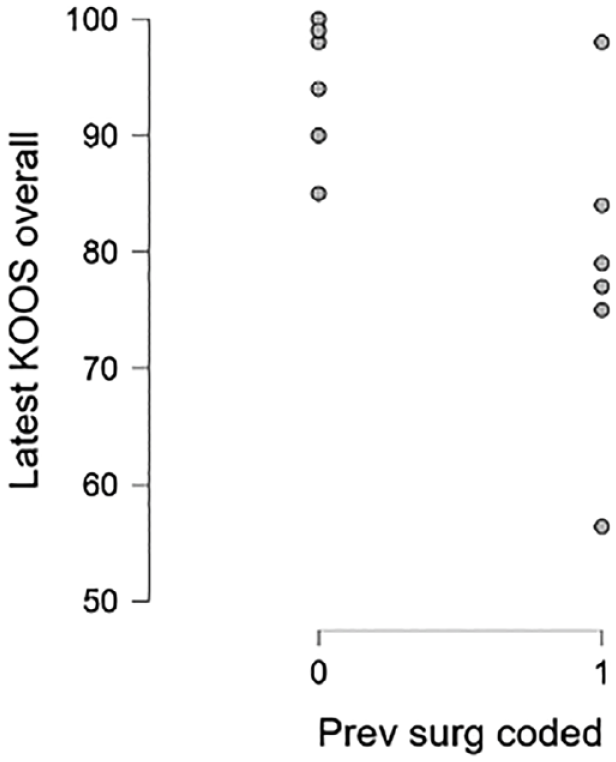
Scatter plot depicts overall KOOS at final follow-up for patients without (0) and with (1) previous cartilage surgery. KOOS = Knee Injury and Osteoarthritis Outcome Score.
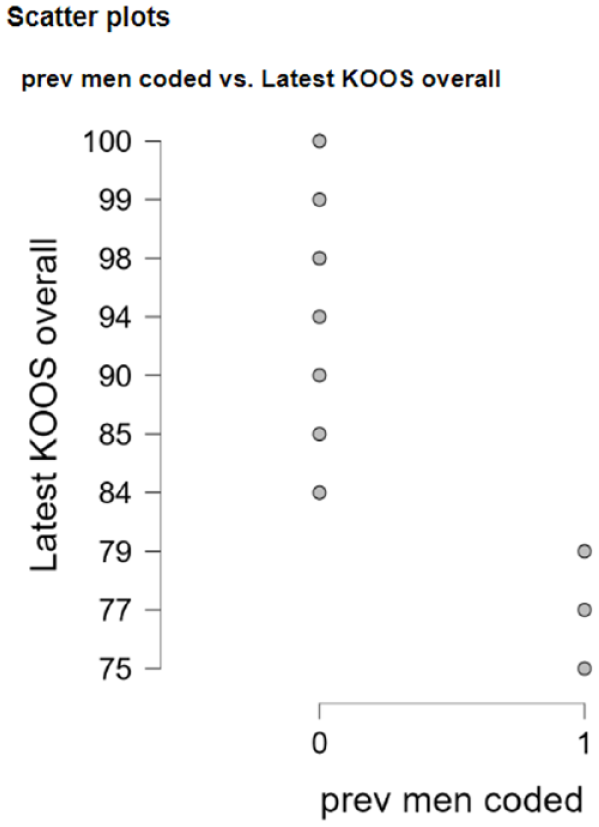
Scatter plot depicts overall KOOS for patients without (0) and with previous meniscectomy (1). KOOS = Knee Injury and Osteoarthritis Outcome Score.
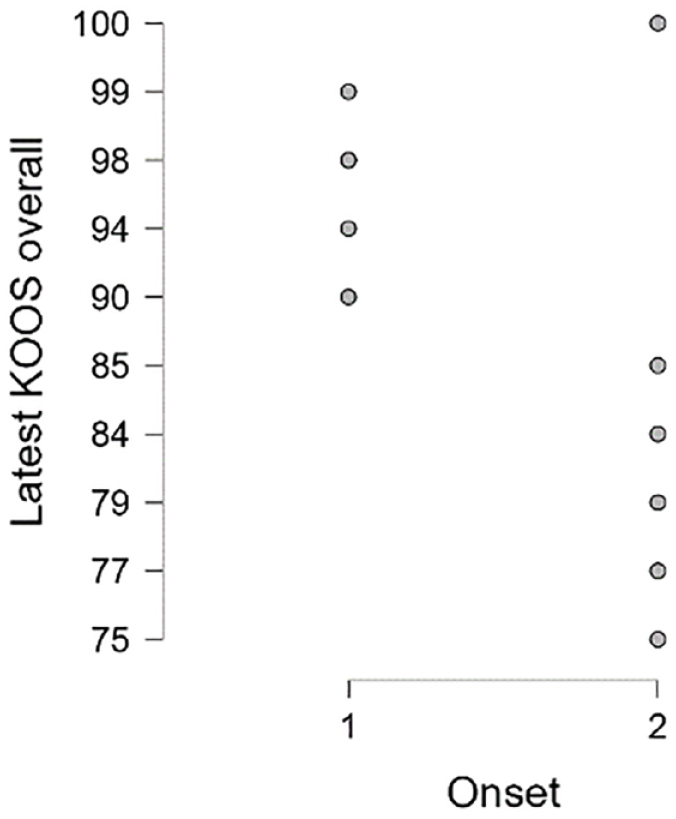
Scatter plot depicts overall KOOS score of patients with an acute onset (1) vs gradual onset (2). KOOS = Knee Injury and Osteoarthritis Outcome Score.
Magnetic resonance imaging at final follow-up was available for 11/12 patients.
The mean Modified Cincinnati (MOCART) score was 64, indicating a moderate level of cartilage repair. Interestingly all patients exhibited a defect filling ranging from 75% to 100%; in 8 cases, a complete integration was found with the surrounding cartilage (Fig. 6A and B), and in the remaining 4 patients, a split-like defect less than 2 mm was present. Moreover, the repair tissue exhibited good signal intensity, although its structure was not homogeneous in all cases. Notably, at the latest follow-up, complete integration of the scaffold, with cartilage formation and bone remodeling was observed, without any significant bony abnormalities, even in a challenging to treat trochlear OCD (Table 2).
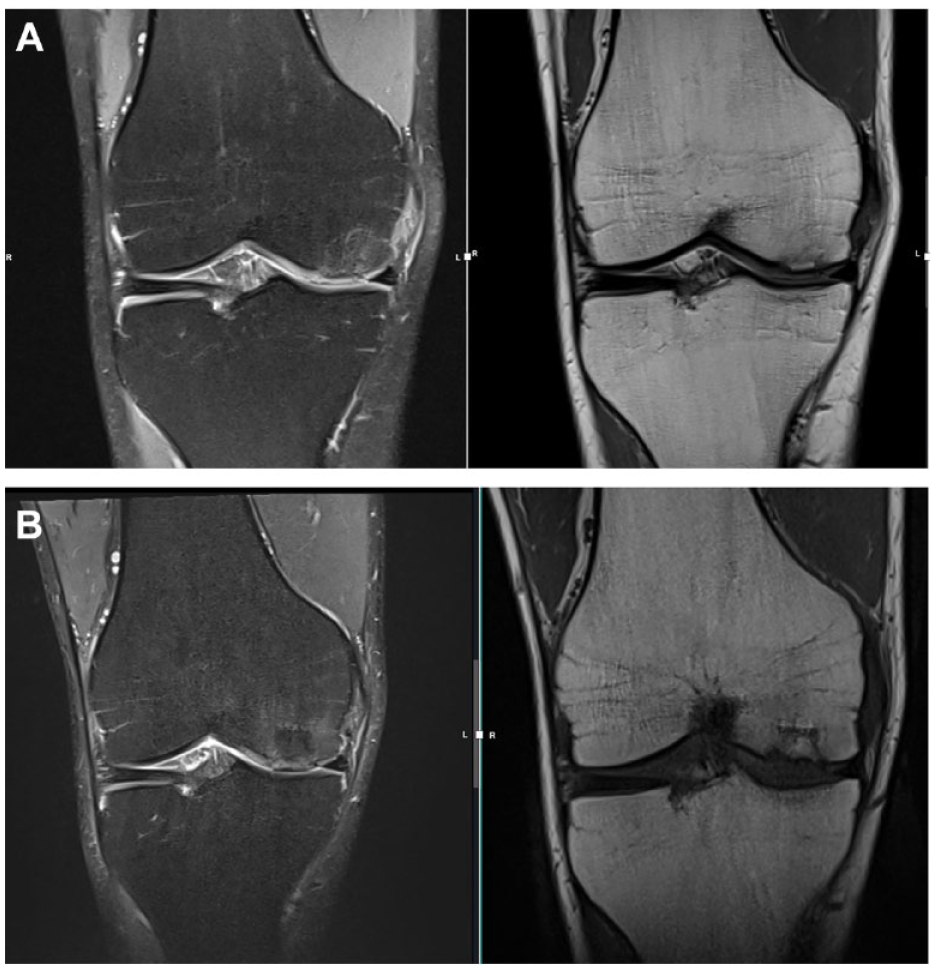
(A) Preoperative MRI. (B) Postoperative MRI 7 years follow-up. MRI = magnetic resonance imaging.
Discussion
This study provides the longest follow-up of the novel aragonite scaffold and illustrates the continued improved clinical outcome demonstrated by the increase of the KOOS scores and the stability of the repair tissue as documented on MRI.
The novel aragonite-biphasic scaffold has been thoroughly investigated; preclinical studies have widely shown promising results for the treatment of knee cartilage lesions. In a sheep model, the scaffold was able to regenerate hyaline-like cartilage. The preclinical studies also showed that the scaffold could integrate well with the surrounding tissue.13,14
Furthermore, the scaffold was able to promote the formation of a durable and functional cartilage layer over a period of 12 months in a preclinical study for the treatment of large cartilage lesions in the goats. 14
This Agili-C™ scaffold has received approval from the U.S. Food and Drug Administration (FDA) for clinical use in the United States, and the results of the randomized clinical trial have been recently published. 8 In this study involving 251 patients from 26 medical centers, the aragonite-based implant outperformed debridement/microfracture (control group) for cartilage defects in the knee. Patients in the implant group showed significantly better outcomes across all evaluation points, with a 2-fold greater mean improvement in KOOS after 2 years. At the 24-month mark, 88.5% of the implant group achieved at least 75% defect fill on MRI, compared to only 30.9% in the control group (P < 0.0001). The implant group also had a lower failure rate (7.2%) compared to the control group (21.4%).
These good results are confirmed at the 3 years follow-up publication on the same population by Van Genechten et al. 9 which analyze the results of 13 patients with a small- to medium-sized lesion in the distal femur treated by scaffold implantation. Primary outcome (KOOS pain) improved with 36.5 ± 14.7 points at 12 months (P = .002) and 41.2 ± 14.7 points at 36 months (P = .002) follow-up. Similar increasing trends were observed for the other KOOS subscales, IKDC, and Lysholm score, which were significantly better at each follow-up time point relative to baseline (P < .05). Activity level increased from 2.75 ± 1.6 to 4.6 ± 2.2 points at final follow-up (P = .07). The MOCART was 61.7 ± 12.6 at 12 months and 72.9 ± 13.0 at 36 months postoperatively. Sixty-six to 100% implant integration and remodeling was observed in 73.3% cases at 36 months.
This is the first study to report minimum 5-year clinical and MRI results of the biphasic aragonite-derived scaffold. One patient failed and was revised with a custom-made metal implant (Episealer). The primary endpoint was the KOOS pain which increased from 45 ± 13 to 86 ± 13 at last follow-up. Secondary endpoints included the other 4 KOOS subscales (symptoms, ADL, sports/recreation, QoL), which were all significantly better compared to baseline. Moreover, comparing KOOS at 24 months follow-up to the latest follow-up KOOS score, it remains stable up to the latest follow-up of 8 years from implantation. These results demonstrate that the initial good results of this osteochondral scaffold, previously reported for the treatment of knee chondral and osteochondral lesions8,9 were confirmed at mid-term follow-up.
The mean MRI MOCART score of the 11 patients that underwent an MRI at latest follow-up was 64, with defect filling from 75% to 100% present in all patients, moreover all the patients presented a split-like defect at repair tissue <2 mm to a complete integration of the scaffold at latest follow-up. No major bony defects or subchondral changes were found, with a good signal intensity of the repair tissue, unlike microfractures.
Comparing the MOCART score to another scaffold commonly used for the treatment of large osteochondral lesions of the knee (Maioregen), there is one study reporting mid-term results 15 at a mean follow-up of 46 ± 17 months, in which only 57% of the patients had a complete filling of the defect and a complete integration in only 78.6% of the patients.
A recent systematic review and meta-analysis reports the results of multilayer cell-free scaffolds for the treatment of knee osteochondral defects. 16 Among the developed osteochondral scaffolds, only three have currently been documented in clinical trials, and one (TruFit) has already been withdrawn from the market for the poor clinical and MRI results achieved.
Dhollander et al. 17 documented a failure rate of the TruFit scaffold of 20% at 1 year of follow-up with the histological analysis showing fibrous vascularized repair tissue. Shivji et al. 18 evaluated this scaffold also at long-term follow-up (121 months), reporting no statistically significant improvement in any score from baseline, while the MRI evaluation showed incomplete or no evidence of plug incorporation and persistent chondral loss.
The Maioregen scaffold has been evaluated in a systematic review from D’Ambrosi et al. 19 Only four studies out of the 16 included in the analysis reported a follow-up longer than 24 months. Significant clinical improvement has been reported in almost all studies with further improvement up to 5 years after surgery. But due to the limited evidence from the studies included in the review, there is no support for the superiority of Maioregen over conservative treatment or other cartilage techniques in terms of clinical improvement during follow-up. Moreover, the authors report that to address the uncertainties surrounding the regeneration of hyaline cartilage and subchondral bone in-growth, it is crucial to conduct well-designed, large-scale, randomized controlled trials in order to assess the potential benefits of future synthetic scaffolds.
If we consider osteochondral autografts for the treatment of large cartilage lesions of the knee, autologous osteochondral transplantation may not be suitable because of the limited graft availability and donor site morbidity that causes pain and stiffness, affecting the overall knee function and recovery. 20
Finally, studies have shown that microfracture can be effective in promoting the growth of a fibrocartilage. This fibrocartilage may not provide the same level of durability and functionality of hyaline cartilage. Over time, fibrocartilage may wear down, leading to the recurrence of symptoms and the need for additional treatments. 21 Thus, microfracture has acceptable short-term clinical results, but results can be expected to decline over time, with clinical and radiological degradation after 2 years of follow-up. 22 Moreover, microfracture is generally recommended for smaller cartilage lesions, and its success rate tends to decrease for larger and more extensive defects.23,24 Considering these factors, alternative treatment options may be more appropriate for large cartilage lesions of the knee. These options included techniques such as autologous chondrocyte implantation (ACI), matrix-based autologous chondrocyte implantation (MACI), or osteochondral allograft transplantation.
Even though autologous chondrocytes transplantation reported good clinical results, 25 the use of this technique (ACI) for treating cartilage lesions of the knee has faced criticism primarily due to its high costs and the requirement for two surgical procedures.
Therefore, based on the literature, current surgical options are not able to sufficiently regenerate the complete osteochondral unit, except in the case of whole cold-stored osteochondral allograft transplants, which are not available in many parts of the world outside the United States. 26
Limitations of this study are the small sample size and the absence of a control “standard of care” group. Moreover, a larger cohort is needed to have stronger data on subanalysis, to evaluate all the variables that might influence the clinical outcome.
Conclusions
This is the first mid-term report demonstrating continued significant clinical improvement and strong MRI findings with a minimum 5 years follow-up, supporting the use of the aragonite-based scaffold as a single-stage procedure for the treatment of isolated focal chondral and osteochondral lesions of the knee. One failure was reported, the patient underwent revision with a small custom-made metal implant.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval 17/3/2014 Belgium Registration Number B300201419853.
