Abstract
Objective
The study aimed to evaluate the clinical outcome and repair capacity of a cell-free aragonite-based scaffold in patients with an isolated symptomatic joint surface lesion (JSL) of the knee.
Design
Thirteen patients (age 33.5 ± 8.9; female 23%; body mass index 25.3 ± 3.4, K/L [Kellgren-Lawrence] 1.8) with a JSL (2.6 ± 1.7 cm2 [1.0-7.5 cm2]) of the distal femur were enrolled in a single-center prospective case series. Safety and clinical outcome was assessed by the KOOS (Knee Injury and Osteoarthritis Outcome Score), IKDC (International Knee Documentation Committee), Lysholm, and Tegner activity scale at baseline and 6, 12, 18, 24, and 36 months follow-up. The MOCART 2.0 and scaffold integration were evaluated on magnetic resonance imaging at 12, 24, and 36 months postoperatively.
Results
Primary outcome (KOOS pain) improved with 36.5 ± 14.7 points at 12 months (P = 0.002) and 41.2 ± 14.7 points at 36 months (P = 0.002) follow-up. Similar increasing trends were observed for the other KOOS subscales, IKDC, and Lysholm score, which were significantly better at each follow-up time point relative to baseline (P < 0.05). Activity level increased from 2.75 ± 1.6 to 4.6 ± 2.2 points at final follow-up (P = 0.07). The MOCART was 61.7 ± 12.6 at 12 months and 72.9 ± 13.0 at 36 months postoperatively. Sixty-six to 100% implant integration and remodeling was observed in 73.3% cases at 36 months. No serious adverse events were reported.
Conclusion
The study demonstrated that the biphasic aragonite-based scaffold is a safe and clinically effective implant for treating small-medium sized JSLs of the distal femur in a young and active patient cohort. The implant showed satisfying osteointegration and restoration of the osteochondral unit up to 3 years postimplantation.
Introduction
Localized (osteo)chondral joint surface lesions (JSLs) are commonly seen among all age categories in both arthritic and nonarthritic knees, causing pain and limiting quality of living.1-3 Unfortunately, injured mature cartilaginous tissue owns poor healing potential, due to its avascularity, slow cellular proliferation, and poor cell migration through the densely packed collagen matrix. 4 Depending on defect size, the depth and the location of the injury, some JSL may remain asymptomatic. This suggests an overall underestimation of the cartilaginous injury rate in the knee, especially in the physically active population, with currently unknown consequences regarding early osteoarthritis (OA) development.5,6 When >50% of the cartilage layer is damaged or the subchondral bone is affected (ICRS [International Cartilage Restoration and Joint Preservation Society] lesions grade 3 or 4), surgical intervention is justified to bring symptomatic relief and reduce the risk for OA onset and progression.4,7 Surgical options for isolated JSL in the knee joint are roughly categorized as either indicated for chondral lesions (i.e., microfracture [MF], autologous chondrocyte implantation [ACI], and matrix-induced ACI [MACI]) or for osteochondral defects (i.e., osteochondral autologous transplantation [OAT] and osteochondral allograft [OCA] transplantation). 4 Some surgical techniques might be applicable in both types and be chosen based on lesion size and location, the availability of graft material, and a cell expansion core facility or by the competence of the surgeon. 8 However, each of these surgical options have their respective strengths and weaknesses but none of them succeeded so far in complete restoration of the (osteo)chondral unit in the knee.4,8,9 Therefore, the quest continues in order to find optimized surgical applications which are ideally high in availability, inexpensive, and, most important, consistent in providing predictable outcomes.
Recently, some authors have advocated multiphasic acellular scaffolds for JSL management.10-13 The approach can potentially meet the healing requirements of the subchondral bone and articular cartilage simultaneously, given that the intrinsic healing mechanism of an osteochondral unit is layer-dependent.10,11 When focusing on bony integration of scaffolds, natural aragonite, derived from coralline exoskeleton, owns an extensive history of research as bone graft substitution option.14-16 Coral has shown to resemble the structural and mechanical properties of human cancellous bone and has demonstrated to stimulate the osteoconductive potential, revascularization, and osteogenic differentiation and proliferation of bone marrow–derived mesenchymal stromal cells.16,17 However, in a clinical setting, the use of coral is only positively evaluated for contained bone defects. 18 Now, in the context of osteochondral defect repair of the knee, Kon and colleagues found excellent restoration outcomes of both the articular cartilage and the subchondral bone in a goat model using a modified aragonite-based scaffold.19,20 Recently, the same implant was tested for its osteochondral regenerative potential in an human ex vivo model. 21 Histologically, migration of explant chondrocytes into the scaffold was observed with formation of extracellular matrix high in type 2 collagen and aggrecan but low in type 1 collagen. 21 After 2 months of incubation, progenitor-like cells appeared on the surface of the articular implant, which is one of the features of native hyaline articular cartilage.21,22
To date, limited human clinical data on the biphasic aragonite-based scaffold has been published, nevertheless showing encouraging results.23,24 Therefore, the current study primarily aimed to evaluate the safety, clinical outcome, and cartilage repair performance of the biphasic scaffold in isolated JSL of mild-moderate arthritic knees up to 3 years after implantation.
Methods
A prospective, open-label, clinical case series was conducted at a single hospital, as part of a multicenter study. From May 2014 to May 2017, 13 patients were enrolled. Study’s inclusion criteria were the following: patient’s age 18 years or older, up to 3 treatable cartilage lesions ICRS III–IV on the femoral condyles or the trochlea, a symptomatic treatable lesion area of 1 to 8 cm2, and baseline Knee Injury and Osteoarthritis Outcome Score (KOOS) for pain not less than 30 or more than 65 points. Study’s exclusion criteria were the following: articular cartilage lesions in the tibia or the patella, ligamentous repair or malalignment correction in the index knee within the last 3 months, significant instability of the index knee, lack of functional remaining meniscus, malalignment more than 5° varus or valgus, any known tumor, infection, inflammatory arthropathy or crystal-deposition arthropathy of the index knee, body mass index (BMI) > 35, severe OA of the index knee (Kellgren-Lawrence [K/L] 4), any previous surgical cartilage treatment in the index knee within the last 6 months, diabetes type 1, systemic infections, coagulopathies, vascular or neurologic diseases, chemotherapy within the last year, and substance or alcohol abuse. All patients were approved by an independent adjunction committee prior to implantation based on radiographs and magnetic resonance imaging (MRI) of the index knee. The clinical trial was approved by an independent institutional review board and was recorded in the national register for clinical trials (#B300201419853) and on clinicaltrials.org (NCT02423629). Every patient signed informed consent before entering the study and agreed to comply with the postoperative rehabilitation protocol. Study was aligned and conducted according to the Helsinki Declaration of 1964 and its later amendments, good medical practice, and the harmonized standards for clinical investigation of medical devices (ISO 14155).
The study population ( Table 1 ) consisted of 3 females (23%) and 10 males (77%), aged 33.5 ± 8.9 with a BMI of 25.3 ± 3.4. OA severity was K/L 1 in 31% (4/13), K/L 2 in 61% (8/13), and K/L 3 in 8% (1/13). Lesion size ranged from 1.0 to 7.5 cm2 (average 2.6 cm2) with 6 defects (46.2%) located on the medial femoral condyle (MFC), 2 (15.4%) on the lateral femoral condyle (LFC), and 5 (38.5%) in the trochlear groove. Acute symptomatic onset was present in 5 patients (38.5%), whereas, in general, sports was the primary cause of injury. Seven patients (53.9%) had prior surgery of the index knee as in debridement, MF, partial meniscectomy (PM), or a combination of these interventions.
Patient Basic Parameters.
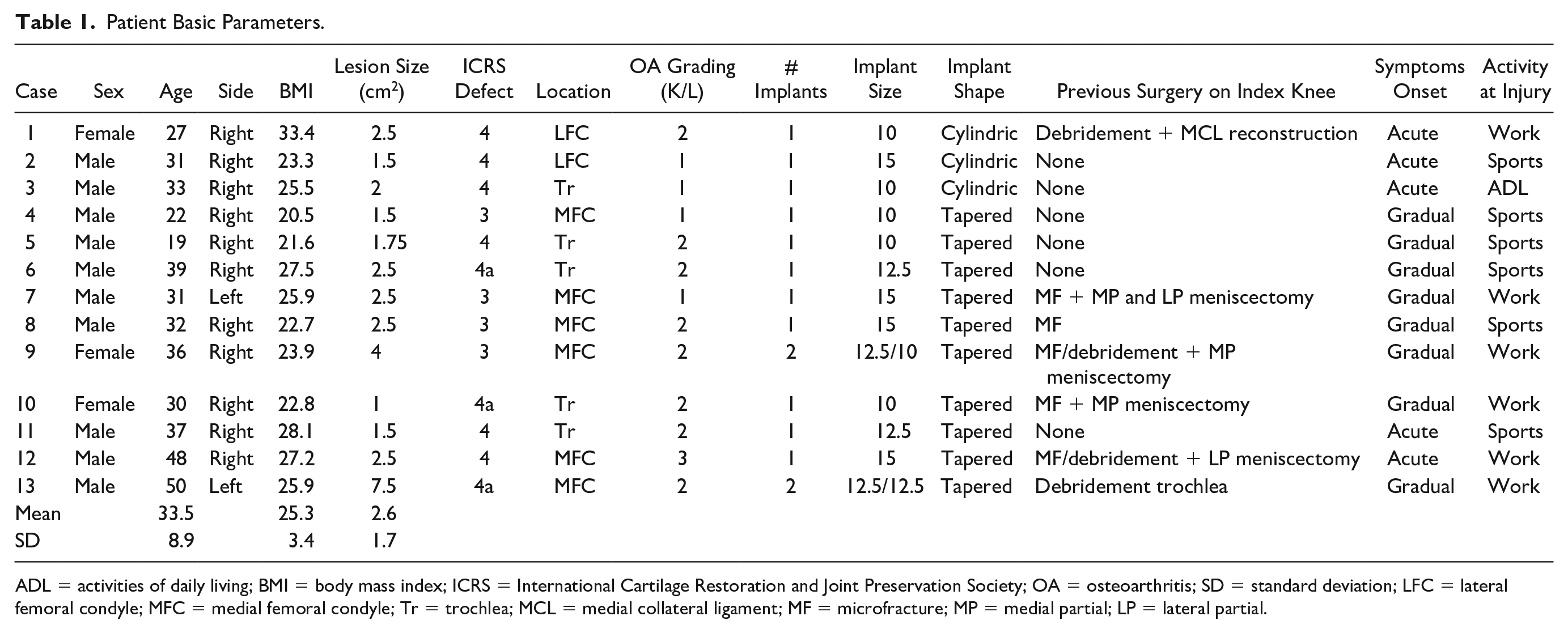
ADL = activities of daily living; BMI = body mass index; ICRS = International Cartilage Restoration and Joint Preservation Society; OA = osteoarthritis; SD = standard deviation; LFC = lateral femoral condyle; MFC = medial femoral condyle; Tr = trochlea; MCL = medial collateral ligament; MF = microfracture; MP = medial partial; LP = lateral partial.
Safety
Intraoperative and postoperative adverse events (AEs) were recorded in relation to the study device. AEs were reported on frequency, time after implantation (months), severity (mild-moderate-severe-serious), and final outcome (resolved-improved-ongoing-death).
Clinical Evaluation
Patients were initially screened by the surgeon to confirm the type of cartilage lesion and to document any significant medical history by the ICRS knee history registration form. Clinical outcome was assessed at baseline and 6, 12, 18, 24, and 36 months after surgery using the KOOS pain subscale as primary endpoint. Other KOOS subscales (symptoms, activities of daily living [ADL], sports/recreation, quality of life [QoL]), the International Knee Documentation Committee (IKDC) subjective knee evaluation form, the Lysholm score, and the Tegner activity scale were secondary endpoint measures.
Imaging Analysis
MRI evaluation was performed preoperatively and at 12, 24, and 36 months after surgery following a standardized imaging protocol with 7 special sequences for cartilage analysis on a 1.5-T MR machine (Siemens Healthcare, Magnetom Aera). Sequences consisted of (1) sagittal T1-weighted fast spin echo sequence, (2) sagittal T2/PD-weighted fast spin echo sequence with fat saturation, (3) axial T2/PD-weighted fast spin echo sequence with fat saturation, (4) coronal PD-weighted fast spin echo sequence with fat saturation, (5) coronal oblique PD-weighted fast spin echo sequence with fat saturation oriented orthogonal to the scaffold, (6) sagittal 3-D spoiled gradient echo sequence with water excitation (DESS), and (7) sagittal T2 mapping sequence composed of an 8 echo train. A dedicated 15-channel knee coil was used in every case. Osteochondral defect typing was preoperatively determined according to the ICRS scoring system whereas preoperative OA severity was scored using the K/L classification on weight-bearing anteroposterior (AP) radiographs of the index knee. The Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) 2.0 knee score (100 points) 25 was used for semiquantitative assessment of osteochondral restoration at the respective time points after surgery. In subjects with 2 implants, both were scored separately. The criterion of “subchondral changes” was applied on the bony region surrounding the scaffold. In extension of the MOCART score, the implant incorporation in the subchondral bone was determined by the scaffold signal intensity (% of scaffold surface) on multidirectional T2 images. Scaffold ingrowth was classified as “66% to 100% incorporation,” “33% to 66% incorporation,” or “0% to 33% incorporation.” All radiographs and MRIs were evaluated by an independent musculoskeletal radiologist and orthopedic resident.
Study Device, Surgical Procedure, and Rehabilitation
Agili-C (CartiHeal Ltd., Israel) is a biphasic CE-marked porous implant indicated for treating articular cartilage lesions and osteochondral defects ( Fig. 1 ). It consists of over 98% calcium carbonate in the crystalline form of aragonite, originating from natural coralline exoskeleton. The top layer of the implant is mechanically modified with micro-drilled channels.
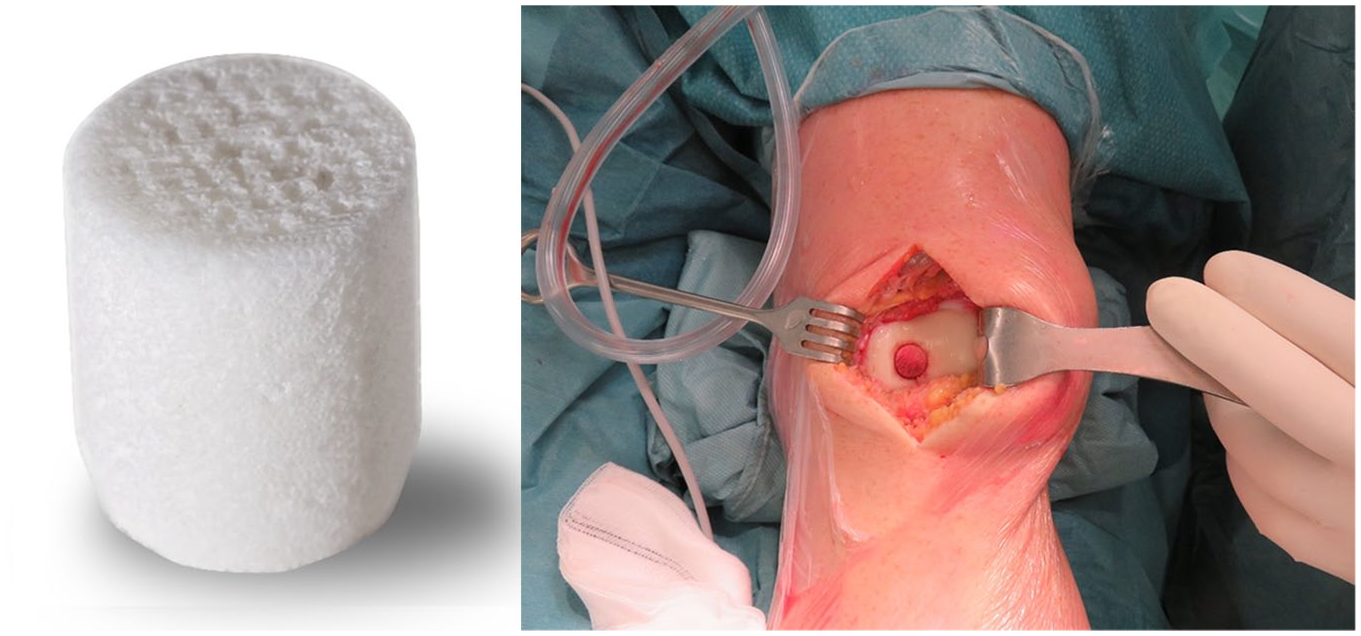
Illustration of the biphasic scaffold before (left) and after (right) implantation in the trochlear groove. The top phase of the implant needs to be gently finger-pushed below the surrounding cartilage edges.
All procedures were performed tourniquet-controlled by a single senior knee surgeon. First, every patient underwent arthroscopic surgery with standard anteromedial and anterolateral portals to macroscopically evaluate the (osteo)chondral lesion. Next, depending on the lesion side, a medial or lateral parapatellar mini-arthrotomy was performed. The lesion dimensions were documented using a sizer and a guidance pin was inserted into the defect centre. This pin was subsequently overdrilled with a dedicated drill bit and protector. The drill was removed and a reamer was introduced to prepare the bottom and depth of the previously made drill hole. A tapered shaper was then inserted to prepare the wall of the defect. The above-mentioned steps were repeated 3 times with thorough rinsing of the shaped defect in between. Finally, the surrounding healthy cartilage edges were contoured with a dedicated cartilage cutter. Once the defect preparations were finalized, the scaffold (sizes 10/12.5/15 mm) was gently introduced with finger pressure until the top phase of the implant was below the native cartilage and completely surrounded by vital subchondral bone ( Fig. 1 ). In case multiple implants were introduced for larger defects, a bone bridge of at least 5 mm between the 2 scaffolds was maintained to provide sufficient implant stability. Implantation was conducted using the Agili-C designated surgical tool set (CartiHeal Ltd., Israel), which is designed for precise site preparation and press fit positioning of the scaffold in load bearing joints.
The day after surgery, patients were discharged from the hospital with a motion-limiting knee brace (0-90° flexion for condylar lesions and 0-30° flexion for trochlear lesions) and received a standardized postoperative rehabilitation protocol specified based on the defect location in the joint. For trochlear lesions, full weight-bearing was allowed already after 2 weeks but range of motion was restricted for the first 8 weeks (gradually increased per 2 weeks). For condylar lesions, range of motion was restricted for only 2 weeks (0-90°) and progressive weight-bearing was initiated after 8 weeks. Passive soft tissue mobilization with assisted range of motion exercises, core stability training and initial strengthening exercises were allowed in both rehabilitation protocols the first 4 weeks after surgery. After 6 weeks, closed chain exercises were introduced, followed by resistance muscle strengthening exercises at 3 months. At 6 months patients were allowed to progressively resume sport activities except for jogging (9 months) and contact sports (12 months).
Statistical Analysis
Descriptive data were displayed with their means and standard deviation (SD). Data were assessed for normal distribution by the Kolmogorov-Smirnov test before proceeding with parametric or nonparametric analysis. Normal data distribution was rejected (P < 0.05) for at least one time point in every outcome measure. Therefore, the Wilcoxon signed Ranks test, a more conservative nonparametric test, was opted for paired analysis of clinical improvement over time. Single model missing data imputation was applied as in “last observation carried forward” or as the average between 2 bordering outcomes in time. The significance level was set at α = 0.05. All statistical tests were performed in Software Package for Social Sciences (SPSS) Statistics version 25.0 (IBM Co., Armonk, NY, USA).
Results
Thirteen study subjects with a knee JSL were successfully treated with 1 or 2 aragonite-based scaffolds ( Table 1 ). In subjects with a cartilage defect size >3.5 cm2 (n = 2), it was preferred to insert 2 devices at a discrete distance, instead of a single larger sized implant. Two subjects had concomitant surgery; an MPFL (medial patellofemoral ligament) reconstruction and an open-wedge HTO, respectively.
Safety
The adverse events are summarized in Table 2 . No serious AEs occurred during surgery or in the follow-up period of 36 months. AEs that were possibly related to the study device were joint arthralgia (50%) and joint effusion (33%), which was resolved by joint aspiration. Implant loosening, failure or infection were not observed during the study course. Hardware was initially removed in the subject with concomitant MPFL reconstruction (7.5 months) followed by arthrolysis (15.5 months) and bloody fluid aspiration (16 months). Another patient had 2 consecutive traumatic accidents of the index knee (at 7 and 31 months), which caused abrasion of the MCL, temporary joint arthralgia, and joint effusion. This subject was completely recovered at final follow-up.
Adverse Events (AEs) during Study Follow-up a .
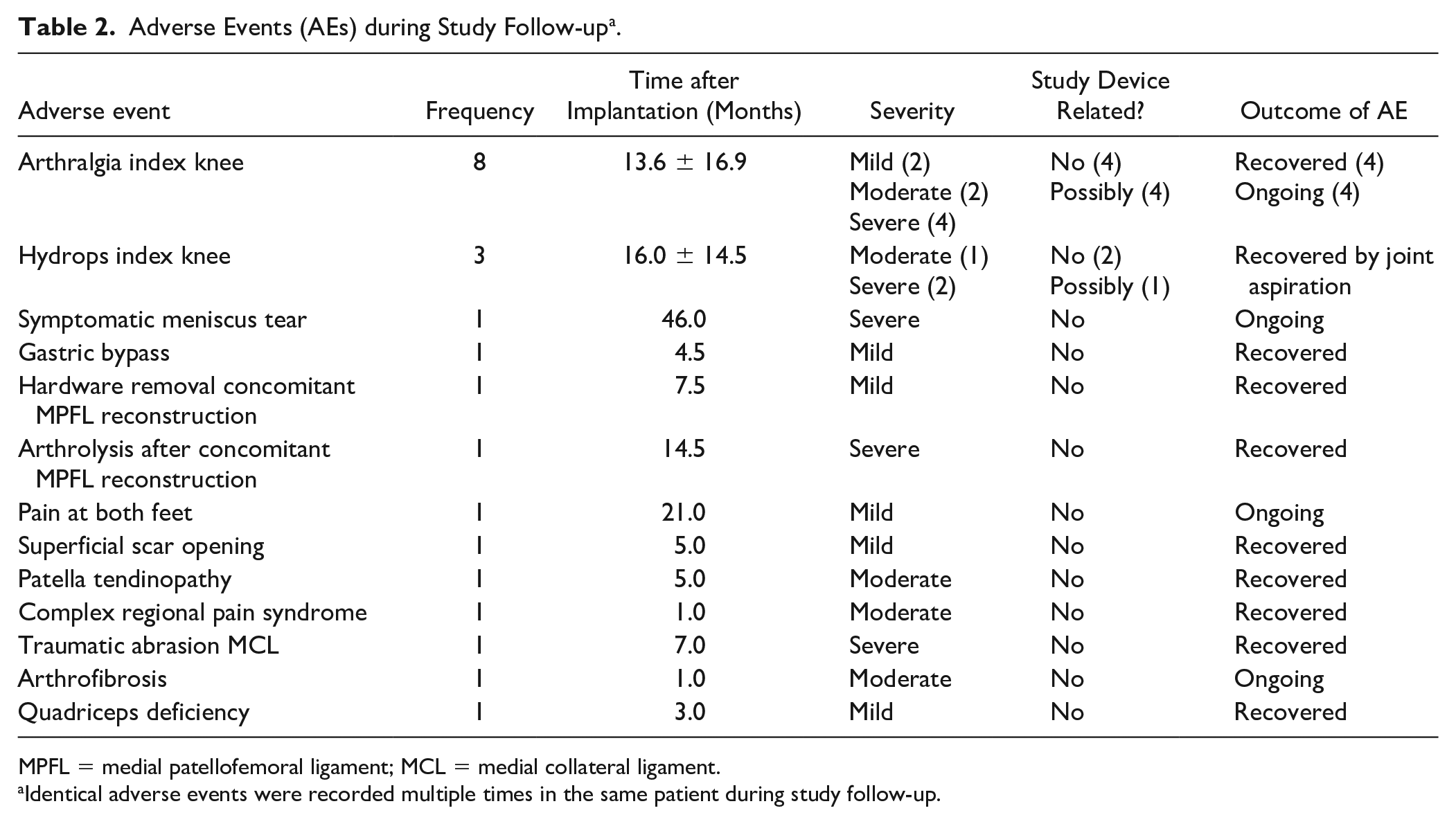
MPFL = medial patellofemoral ligament; MCL = medial collateral ligament.
Identical adverse events were recorded multiple times in the same patient during study follow-up.
Patient-Reported Outcomes
Considering the KOOS pain as primary study endpoint, a significant difference (P < 0.002) was observed for all time points compared to baseline with an absolute improvement of 36.5 ± 14.7 points at 12 months, 41.9 ± 12.2 points at 24 months, and 41.2 ± 14.7 points at 36 months after surgery ( Fig. 2 ). Secondary endpoints (KOOS subscales: Symptoms, ADL, Sports/Recreation, and QoL) showed significantly better outcomes at each follow-up time point relative to baseline (P < 0.050). For the IKDC and Lysholm score, a similar trend of clinical improvement was observed with significantly different outcomes at all time points compared to the preoperative baseline values (P < 0.005; Table 3 ). The Tegner activity scale revealed significantly increased scores from baseline at 18 and 24 months postoperatively (P < 0.050). Despite this improvement, the pre-injury activity level (6.9 ± 1.8) was not restored at 36 months follow-up (4.6 ± 2.2; P = 0.015). One subject missed the complete PROMs at 36 months follow-up and another one missed the KOOS questionnaire at this time point. Further one subject (7.7%) was unable to complete the Tegner activity scale at 18 and 24 months.
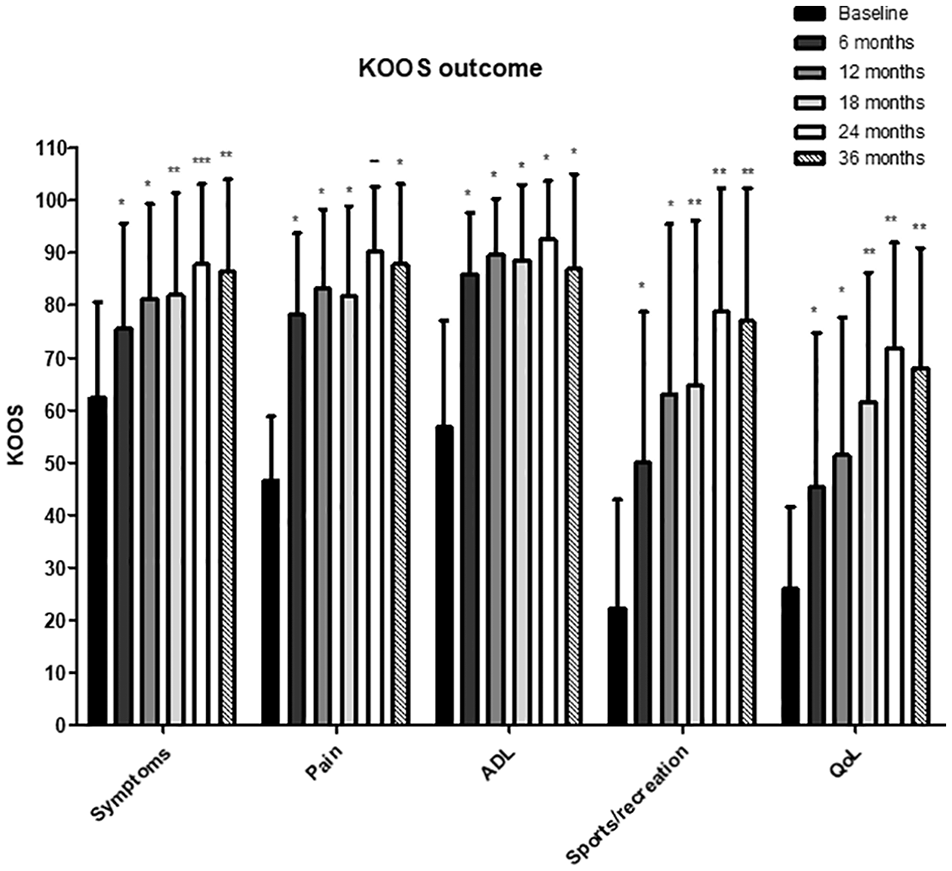
KOOS outcomes for the study population. (Mean + SD).
Secondary Outcome Measures IKDC, Lysholm, and Tegner Score up to 3 Years after Implantation.
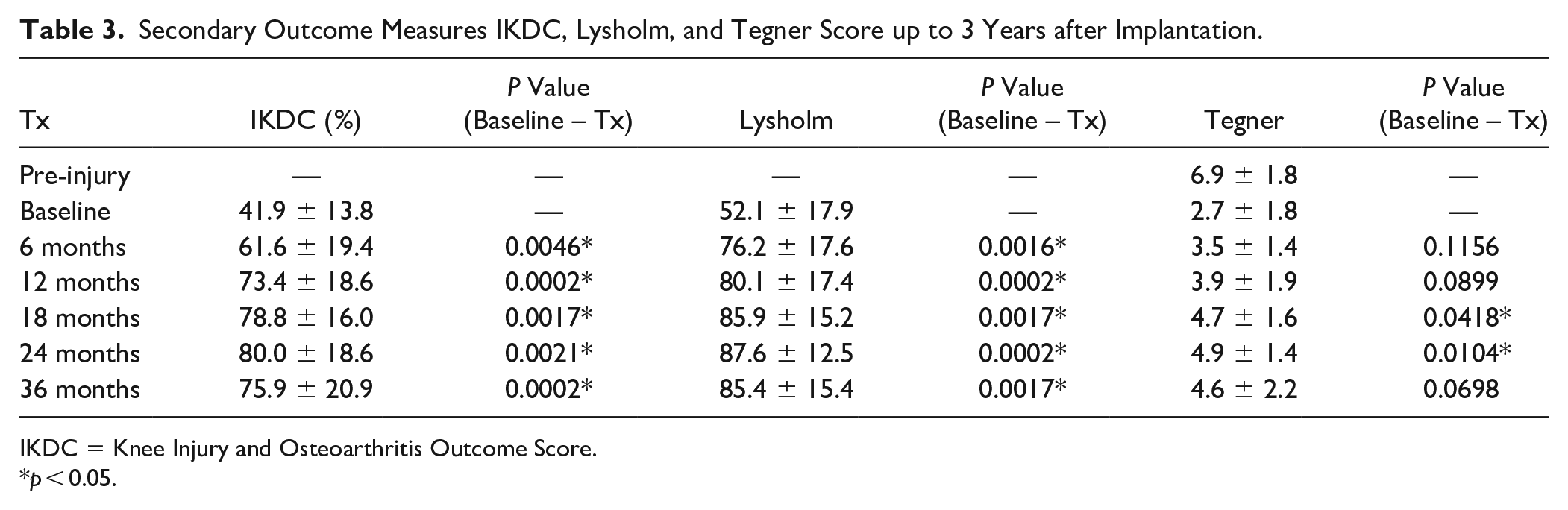
IKDC = Knee Injury and Osteoarthritis Outcome Score.
p < 0.05.
Imaging Outcome
Preoperative defect typing revealed that 4 patients (31%) were suffering from an ICRS grade 3 chondral defect (>50% loss of cartilage depth) and 10 subjects suffering from an ICRS grade 4 defect (penetration into the subchondral bone) out of which 3 had a dislocated osteochondral fragment. Postimplantation, the MOCART 2.0 was 61.7 ± 12.6 at 12 months, 69.0 ± 11.8 at 24 months, and 72.9 ± 13.0 at 36 months. MOCART evolution per implant is outlined in Figure 3 . Bony integration and remodeling of the scaffold was for 66% to 100% completed in 6 implants (40%) at 12 months and in 11 implants (78.7%) at final follow-up ( Table 4 ). A typical pit-like healing pattern of the implant was frequently observed on MRI with peripheral ingrowth of newly formed subchondral bone with subsequent implant biodegradation and formation of a cartilaginous layer ( Fig. 4 ). Examples of excellent and poor scaffold healing with the respective clinical outcome are illustrated in Figure 5 . One patient (one scaffold, 7.7%) was lacking MRI imaging at 36 months follow-up.
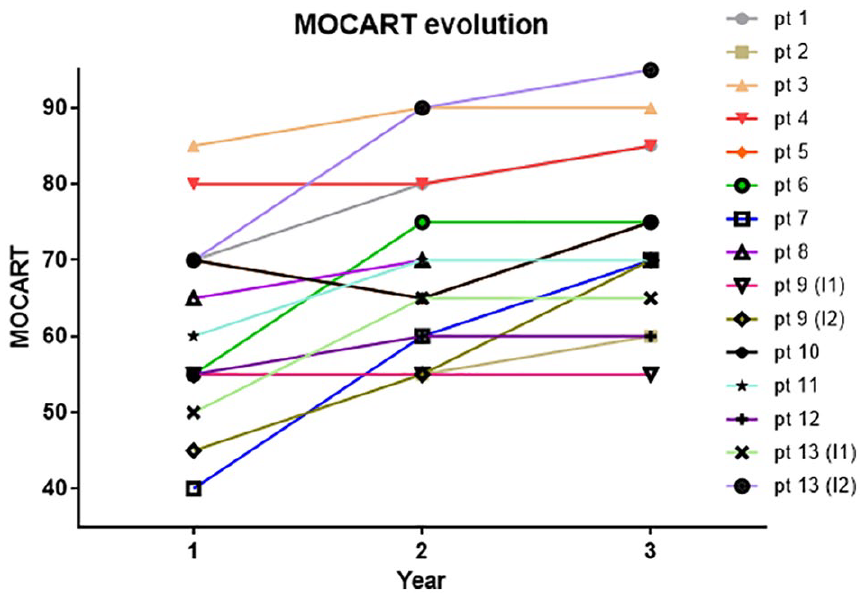
MOCART evolution per patient up to 36 months after scaffold implantation (pt = patient; I = implant).
Scaffold Osteointegration and Remodeling Assessment based on the Signal Intensity Relative to the Surrounding Subchondral Bone of the 15 Study Implants a .
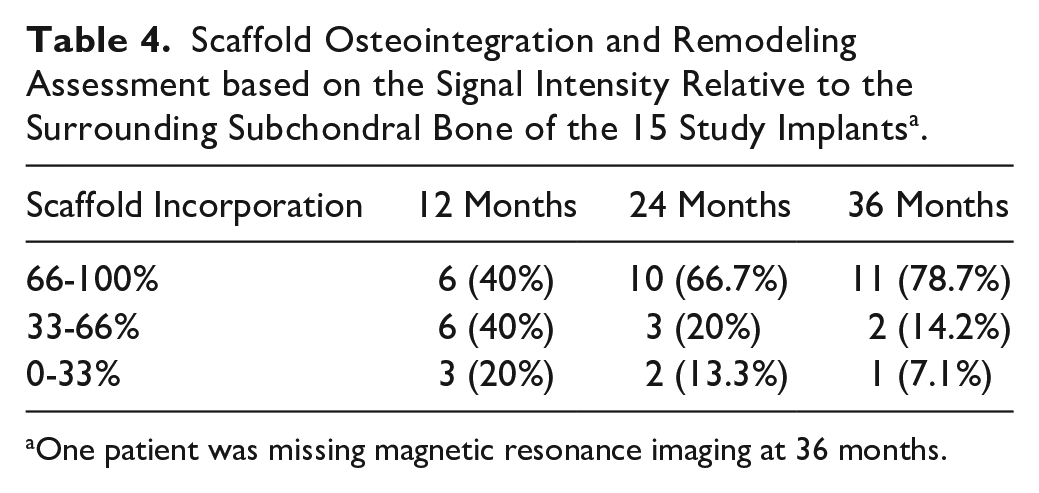
One patient was missing magnetic resonance imaging at 36 months.
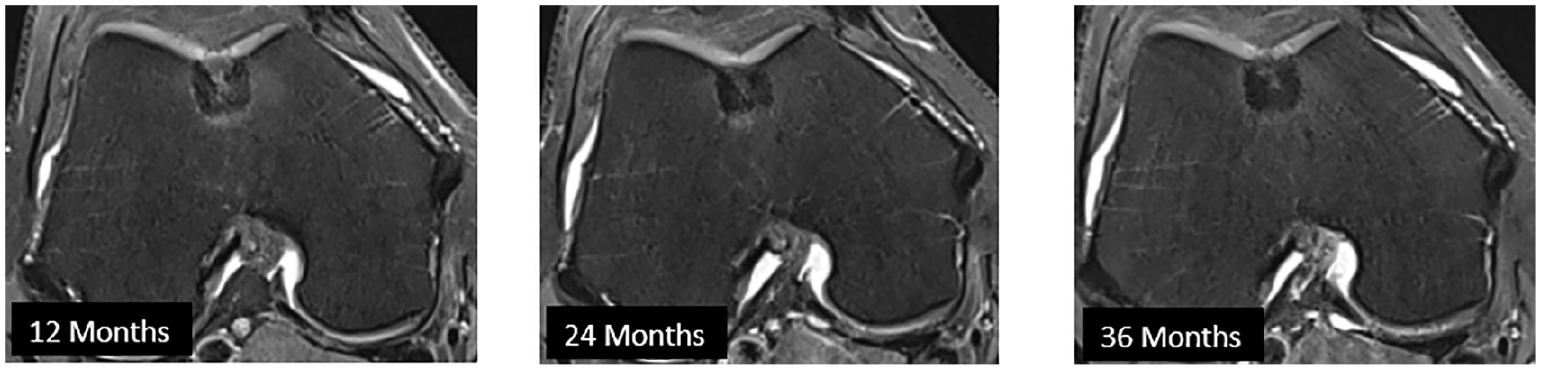
Illustration of the pit-like healing pattern over time, commonly observed after scaffold implantation: axial view on the trochlea.
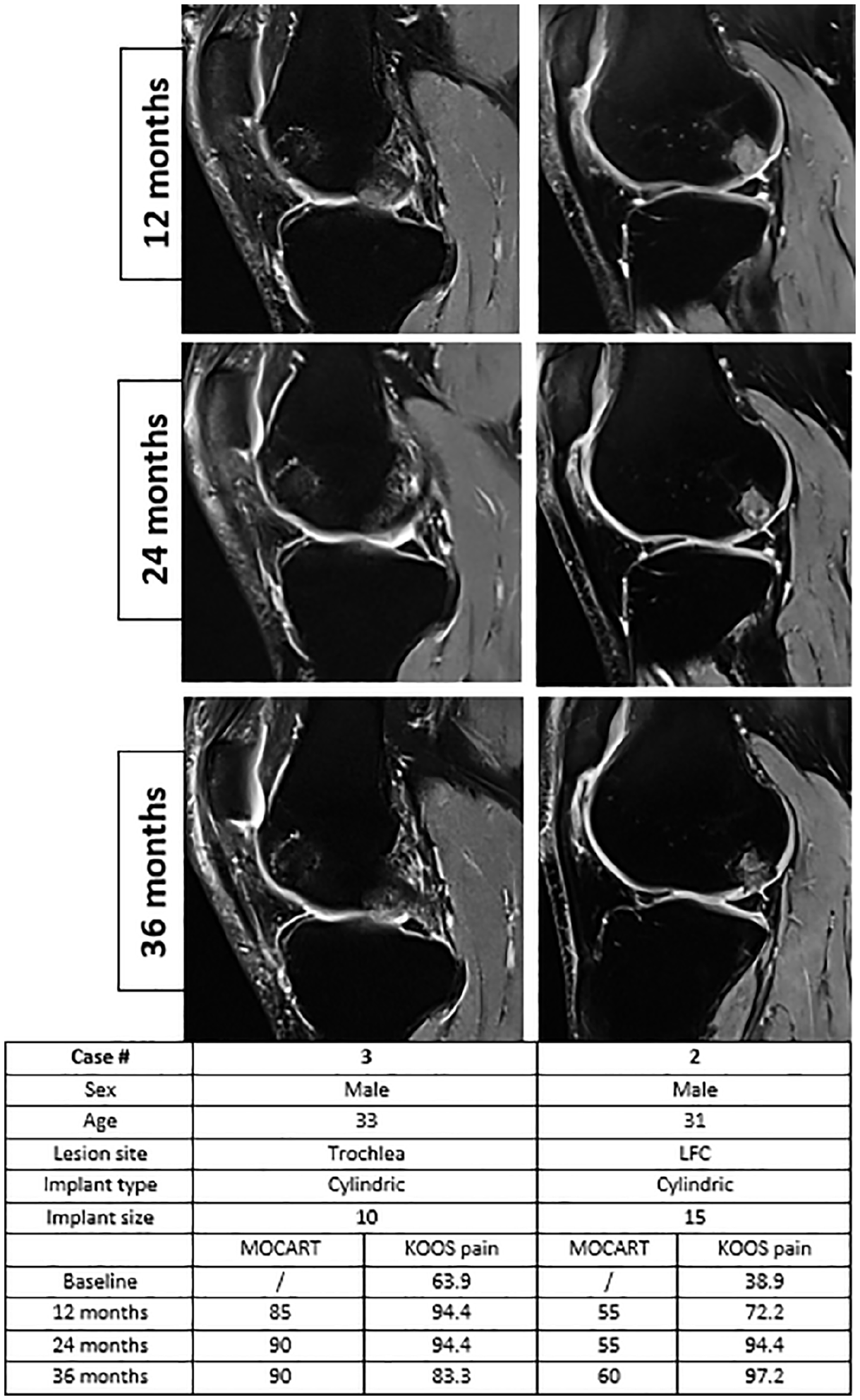
Example of excellent (left) and poor (right) scaffold healing with associated clinical outcome.
Discussion
The study investigated the safety, clinical response, and MRI imaging outcomes after implantation of a biphasic aragonite-based scaffold to treat JSL in mild-moderate arthritic knees. The primary endpoint was the KOOS pain which increased by 36.5 ± 14.7 points at 12 months and 41.2 ± 14.7 points at 36 months after surgery. Secondary endpoints included the other 4 KOOS subscales (symptoms, ADL, sports/recreation, QoL), IKDC, Lysholm, and the Tegner scale, which were all significantly better compared to baseline and stabilized around 2 to 3 years after surgery. The relatively high pre-injury activity level (Tegner score 6.9 ± 1.8) was not restored 3 years postoperatively, but was nevertheless significantly better at 18 and 24 months compared to baseline. The MOCART 2.0 score was 72.9 ± 13.0 after 36 months while 11/14 implants (78.7%) showed 66% to 100% integration with the surrounding subchondral bone.
The current study is the first case series that performed a clinical and imaging evaluation of the novel aragonite-based scaffold in the human knee with outcomes up to 3 year after surgery. Only a single clinical report by Kon et al. investigated the same implant, but primarily focused on outcomes of implant shape difference (tapered vs. cylindrical) at 6 and 12 months postoperatively. 23 Their group found a superior safety profile for the tapered implant (0% vs. 10.5% failure rate) but observed no relevant differences in clinical or imaging outcomes. In the current study, the majority of cases (77%) were operated with a tapered implant and no failures or serious AEs occurred. The AEs that could possibly be related to the implant were arthralgia (4) and hydrops (1) of the index knee and resolved spontaneously or by joint aspiration. A defect fill >75% was present in 11/15 implants (73.3%) at 12 months, which is in line with the reported 84% from the previous cohorts studied by Kon et al. 23 Furthermore, the clinical improvement (IKDC and Lysholm score) at 1 year follow-up was slightly better in the current study, but overall comparable to their outcomes. 23
Since the symptomatic role of subchondral bone has long been an underestimated factor in cartilage defects, focus is trending toward complete osteochondral unit restoration rather than only repairing the macroscopically damaged cartilage.12,13,26 With the availability of multilayered scaffolds, both healing requirements for bone and cartilage can potentially be met by selecting dedicated layer-specific biomaterials.11,12 Because native coral singly does not lead to sufficient cartilage-like tissue formation, 27 the top layer of the studied implant was mechanically modified (drill holes) and enriched with hyaluronic acid that has previously shown to improve the regenerative capacity for a cartilaginous matrix. 27 In the study, the majority of lesions were small-medium sized (1-3 cm2; 11/13, 85%) for which 3 different implant sizes (10-12.5-15 mm) were used. From a biological perspective, assuming slower central osteointegration in large acellular scaffolds (pit-like healing pattern), it was preferred to treat large lesions (>3.5 cm2) with 2 small implants instead of a single large implant. In these cases, a bony bridge of at least 5 mm was maintained to stabilize both implants and to provide complete surrounding with native osteochondral tissue for enhancing scaffold incorporation.
Other multilayered scaffolds such as the Maioregen osteochondral substitute are additionally targeting the calcified tide mark with an outbalanced collagen type 1 (60%)/hydroxyapatite (40%) transition layer (triphasic implant). 10 Although histological analysis have not yet shown the clear advantage of this additional layer, complete scaffold reabsorption has been observed with formation of hyaline-like tissue as confirmed on MRI. 10 To date, few studies have reported outcomes longer than 2 year of follow-up of this triple-layered scaffold, but a low failure rate (3.4%) and stable clinical improvement up to 5 years is suggested.10,28 Another acellular biphasic plug (Trufit) consists of polylactide-coglycolide copolymers (calcium sulfate) and has extensively been studied in the human knee. 29 Conflicting results were found for plug integration with several cases showing only partial bony incorporation with formation of a cyst-like structure of unknown relevance.29,30 For patellofemoral lesions, the reoperation rate with Trufit plugs was found to be 70% at 2 year. 31 In a retrospective study comparing Trufit with OAT, Hindle et al. found a significantly higher clinical improvement in favor of OAT with no difference in re-operation rate. 32 Altogether, high-level clinical evidence is currently lacking to consider one of these cell-free bio-mimetic scaffolds as standard of care for osteochondral repair in the knee.10,29 In the meantime, OAT and fresh OCA transplantations (if available) remain benchmark interventions when it comes to osteochondral unit restoration in the knee with established survival rates averaging 78% at 10 years.9,33
The correlation between clinical improvement and morphological cartilage repair on MRI is controversial. 34 At 12 and 24 months after implantation, respectively, 6 and 3 scaffolds remained below 60 points on the MOCART while showing slow osteointegration in the surrounding subchondral bone. Potential reasons for the slower and incomplete integration process are unknown. Case 2 (1.5 cm2, 1 implant, LFC) and case 9 (4.0 cm2, 2 implants, MFC) scored the lowest on MOCART at 36 months follow-up, but this was not reflected in the clinical improvement, shown by a respectively 58.3 and 41.7 points increase on KOOS pain. In a systematic review and meta-analysis, de Windt et al. concluded that strong evidence is lacking for predicting clinical outcomes after cartilage repair based on morphological MRI (including MOCART). 34 Moreover, similar trends were found for the Maioregen implant during a 5 years follow-up with MRI. 28 In general, this suggests that incomplete scaffold incorporation (not failure) does not necessarily provide worse clinical outcomes at midterm follow-up.
Primary study limitations were the small sample size and the absence of a control “standard of care” group. Since the study specifically aimed to evaluate the initial safety, clinical outcome and the quality of the osteochondral repair on MRI, low sample size without controls appeared to be a responsible choice respecting study objectives. The safety profile and clinical outcomes after implantation of such scaffold were only confirmed once in the human knee joint, 23 which made the authors cautious in the number of patients to include. Next, the implant was introduced for isolated small to medium sized femoral lesions, which is in fact the most common site of focal cartilage damage in the knee. Clinical and imaging outcomes can therefore not be translated to patellar, tibia plateau, or bipolar (osteo)chondral lesions. Finally, the study population was represented by young (33.5 ± 8.9 years) and active patients with minor diffuse OA changes and an acceptable BMI (25.3 ± 3.4), which is an almost ideal population for examining novel techniques in (osteo)chondral repair. 35 In daily clinical practice, however, concomitant injuries of the index knee (meniscus and ACL tears) and patient-specific comorbidities (alignment, diffuse OA, obesity) might temper clinical improvement after implantation of this implant.
To conclude, many questions remain to be answered about the biphasic aragonite-based implant including long-term clinical outcomes, different defect location and size, subsequent OA development, and potential therapeutic superiority to microfracture and OAT, but current short-term clinical results look nevertheless promising in the few cases described. If tested more extensively, the scaffold might become relevant for osteochondral unit restoration with key factors being its high availability and straightforward surgical implementation. Ultimately, this might solve more expensive storage problems and deal with the risk of disease transmission and donor-site comorbidities that are still associated with standard of care procedures such as fresh OCA and OAT.
Conclusion
The study demonstrated that the biphasic aragonite-based scaffold is a safe and clinically effective implant for treating small-medium sized JSLs of the distal femur in a young and active patient cohort. The implant showed satisfying osteointegration and restoration of the osteochondral unit up to 3 years postimplantation.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CartiHeal Ltd. (Tel Aviv, Israel).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Peter Verdonk is consultant for CartiHeal Ltd.
Trial Registration
Agili-C™ implant performance evaluation. https://clinicaltrials.gov/ct2/show/NCT03299959?term=agili-c&cntry=BE&draw=2&rank=1 (trial registration number: NCT02423629).
Ethical Approval
Ethical approval for this study was obtained from the Antwerp university hospital ethical committee (#B300201419853).
Informed Consent
Written informed consent was obtained from all subjects before the study.
