Abstract
Objective: Articular cartilage injury is common after athletic injury and remains a difficult treatment conundrum both for the surgeon and athlete. Although recent treatments for damage to articular cartilage have been successful in alleviating symptoms, more durable and complete, long-term articular surface restoration remains the unattained goal. In this article, we look at both new ways to prevent damage to articular surfaces as well as new techniques to recreate biomechanically sound and biochemically true articular surfaces once an athlete injures this surface. This goal should include reproducing hyaline cartilage with a well-integrated and flexible subchondral base and the normal zonal variability in the articular matrix. Results: A number of nonoperative interventions have shown early promise in mitigating cartilage symptoms and in preclinical studies have shown evidence of chondroprotection. These include the use of glucosamine, chondroitin, and other neutraceuticals, viscosupplementation with hyaluronic acid, platelet-rich plasma, and pulsed electromagnetic fields. Newer surgical techniques, some already in clinical study, and others on the horizon offer opportunities to improve the surgical restoration of the hyaline matrix often disrupted in athletic injury. These include new scaffolds, single-stage cell techniques, the use of mesenchymal stem cells, and gene therapy. Conclusion: Although many of these treatments are in the preclinical and early clinical study phase, they offer the promise of better options to mitigate the sequelae of athletically induced cartilage.
Keywords
Introduction
Although running on an uninjured knee alone has not been shown to increase cartilage wear, high-impact athletic activity such as football and American football have long been associated with knee injury including damage to the ligament, meniscus, and articular cartilage, leading to higher rates of disability in long-time competitors. Cartilage damage in these athletes has long been considered a difficult clinical problem, but through scientific and surgical breakthroughs in the last 20 years, we are beginning to offer athletes ways to mitigate damage and disability.1-4 The first- and second-generation treatments, described elsewhere in this journal, were instrumental in encouraging a burgeoning class of scientists, surgeons, and entrepreneurs to further investigate ways to both prevent injury to this pristine articular surface and to restore the surface to normal enduring function once injured.
The goals of cartilage repair are to restore an articular surface that matches the biomechanical and biochemical properties of normal hyaline cartilage and to prevent the progression of focal cartilage injury to end-stage arthritis. In recent literature, investigators have shone a light on the importance not only of restoring a smooth, gross superficial surface but also of promoting the restoration of a normal bone cartilage interface with acceptable subchondral flexibility.5,6 This, along with the multilayered variability seen in the ultrastructure of normal hyaline cartilage, enables the effective transfer of shear and compressive load from cartilage to bone. Although existing treatment options have been shown to effectively ameliorate symptoms, achieving this more comprehensive restoration of zonal variability with intact tidemark will be necessary to maintain the superficial repair and avoid long-term degenerative disease. In addition, although it is well accepted that any effective surgical treatment must concomitantly address ligamentous instability and malalignment, full restoration after injury will not be achieved without similarly addressing the biochemical and viscoelastic properties of the synovial fluid. This chemical milieu is often altered at the time of injury and interventional surgery.7,8
Glucosamine, Chondroitin Sulfate, and Other Dietary Supplements
Glucosamine (G) 1,500 to 2,000 mg/d and chondroitin sulfate (Cs) 800 to 1,200 mg/d and avocado-soy unsaponifiables (ASU) 300 to 600 mg/d, taken together or alone, are useful as adjunct therapies in cartilage disorders. Each is sold as prescription, over the counter (OTC), or as supplements, depending upon the country. Basic science studies indicate more anticatabolic than anabolic effects, and all 3 probably act as signal modulators of inflammatory and degradative enzyme pathways, perhaps through the partial inhibition of the NF-κB pathway.9-11 Multiple other potential biochemical pathways have been uncovered.12-18 Some animal evidence indicates pretreatment with G, Cs, or ASU might delay the course of osteoarthritis (OA) after traumatic injury,19-21 but without human evidence, prophylactic use of these supplements cannot be recommended. G, Cs, and ASU have nonsteroidal anti-inflammatory drug (NSAID)–sparing effects.15,22,23 In acute pain, coadministration of fast-acting agents such as acetaminophen or NSAIDs with G, Cs, and/or ASU, followed by prompt removal of the former, may be advised. Most of the 40+ human clinical trials on G, Cs, or ASU have shown positive results either for structure-modifying effects or pain/function improvement.24-31 Importantly, structural benefits were independent of symptom relief.32-35 The National Institutes of Health (NIH)–funded GAIT (Glucosamine/Chondroitin Arthritis Intervention Trial) had mixed results and created controversy.36,37 Most of the subjects started with low pain scores, thereby creating a possible floor effect bias. G, Cs, and the combination of G + Cs scored better than placebo, but not significantly so. The active comparator, celecoxib, failed to meet statistical significance in 40 of 42 reported outcomes. In the 2 that were statistically significant, the effect was not clinically meaningful. This suggests a high probability for a false null result for G and Cs. Nevertheless, in the moderate to severely affected subjects, the combination of G + Cs yielded positive clinical results that were highly statistically significant. There have been no separate studies on dosing for high-level athletes, and therefore, we would recommend the following similar doses for athletes who participate regularly: 600 mg ASU, 3,000 mg G, and 1,600 mg Cs once daily.
Viscosupplementation
Viscosupplementation with intra-articular injection of exogenous hyaluronic acid (HA) has an integral role in the treatment algorithm of OA and cartilage damage. The healthy human knee contains approximately 2 mL of synovial fluid. In the osteoarthritic knee, the concentration of HA is reduced to one half to one third of the normal value. 38 Endogenous HA is produced by the type B synoviocytes and fibroblasts of the synovium, and its role is multifactorial. 38 It provides joint lubrication and absorbs shock while also promoting chondrocyte proliferation/differentiation.39-41 HA has also been shown to inhibit tissue nocioceptors and stimulate endogenous hyaluronan formation. 42 Viscosupplements have both chondroprotective and anti-inflammatory effects. 43 Chondroprotection occurs through downregulation of the gene expression of OA-associated cytokines and enzymes. 44 The anti-inflammatory effect occurs by downregulation of TNF-α, IL-8, and iNOS in synoviocytes. 43 In addition to in vitro and animal studies showing chondroprotection, a recent clinical study using noncrosslinked sodium HA has shown human chondroprotection both on radiographic and high magnification arthroscopic evaluation. In our opinion, these findings suggest the use of viscosupplementation for small defects in articular cartilage in the athlete and perhaps as a postinjury treatment, in season, for patients with bone bruises on MRI. Although further study is needed to validate efficacy for these uses, the low morbidity associated with the use of HA supports its use for these potential indications.
These chondroprotective effects have also driven the use of viscosupplementation in the postoperative knee. Pain that persists after arthroscopy can be decreased with the use of HA injection. It has been shown to result in decreased joint swelling and to be NSAID sparing. 45 Additionally, the disease-modifying effects of HA have been shown to reduce cartilage degeneration and promote tissue repair after microfracture in an animal model.46,47 This occurs by inhibiting the production of nitric oxide and by stabilizing proteoglycan structure.48,49
Role of Platelet-Rich Plasma
Platelet-rich plasma (PRP) can be defined as the volume of the plasma fraction from autologous blood with platelet concentration above baseline (200,000 platelets/µL). 50 PRP contains different growth factors, which regulate key processes involved in tissue repair.51,52 The rationale for topical use of PRP is to stimulate the natural healing cascade and tissue regeneration by a “supraphysiological” release of platelet-derived factors directly at the site of treatment. PRP has been successfully used in surgical and outpatient procedures in the treatment of several musculoskeletal problems.53-55
While recent published randomized controlled trials using PRP in the Achilles and rotator cuff tendons have shown little to no statistical improvement, various authors have used PRP to treat chondral defects in athletes and obtained good results.56,57 A prospective study from our authors in Milan followed up 50 patients active in sports with degenerative lesions of the knee. All patients were treated with 2 intra-articular injections (1 monthly) of leukocyte-rich PRP. The study revealed that the use of PRP in patients with chronic degenerative disease of the knee could act to diminish pain and improve symptoms and quality of life. A prospective randomized study comparing PRP to High molecular weight (HMW) HA as well as Low molecular weight (LMW) HA reported superior outcomes at 6 months with PRP injection (3 injections). 58
Role of Pulsed Electromagnetic Fields (PEMFs): I-ONE Therapy
Preclinical studies have shown that pulsed electromagnetic fields (PEMFs), with specific physical signal parameters (I-ONE therapy, IGEA, Carpi, Italy), 59 in vitro favor the proliferation of chondrocytes, stimulate proteoglycan synthesis, and demonstrate A2A adenosine receptor agonist activity.60-65 In vivo, I-ONE therapy prevents degeneration of articular cartilage and downregulates the synthesis and release of proinflammatory cytokines in the synovial fluid.66-68 These findings suggest that I-ONE therapy may be used in humans to control joint inflammation and to stimulate cartilage anabolic activities, finally resulting in chondroprotection. Clinical studies show that I-ONE therapy is an effective chondroprotective treatment for patients, without any negative side effects, that limits inflammation, reduces recovery time, and ultimately preserves a healthy articular cartilage of the knee.69,70 The positive results are maintained also at 3 years’ follow-up. 69 Our authors from Milan prospectively followed up 32 patients treated with I-ONE therapy for 1 year ( Fig. 1 ). Patients showed significant improvement in all scores at final follow-up (P < 0.005). The data of their work further confirm the findings of previous clinical studies, which showed the benefits of using I-ONE therapy—a noninvasive, specific, and local biophysical treatment—in order to control the inflammatory process and to provide faster functional recovery without any side effects.

I-ONE pulsed electromagnetic fields (PEMFs) generator.
Emerging Surgical Technologies
Despite a plethora of new cartilage repair techniques, the vast majority of patients with cartilage injury worldwide are still treated with palliative measures such as debridement, lavage, and anti-inflammatory medication. 71 Therefore, a very large number of patients (over 85%) could benefit significantly from the successful development of more cost-effective, reproducible restorative surgical procedures.
Emerging options for surgical intervention can be grouped into the following categories: third-generation cell techniques (described elsewhere in this journal), off-the-shelf scaffolds, minced cartilage or one-stage techniques, and enhanced autologous mesenchymal stem cell techniques (Table 1).
Options for Treatment of Cartilage Injury
Scaffolds
Various scaffolds have been made of either natural or synthetic products, but all share common traits: They are conductive, biocompatible, and resorbable so they are bioreplaced by healthy normal tissue as bone and chondral ingrowth occurs. The idea of a single-stage, off-the-shelf acellular product that can bring the body’s own cells effectively into an injured area and promote chondrogenesis is quite appealing both for its potential low morbidity and its cost-effectiveness. Many scaffolds have been tested in preclinical studies, but only a few have been used in human subjects to date for a cartilage indication. Currently, several studies are being performed using these scaffolds as an adjunct to microfracture to enhance the effectiveness of the locally derived marrow cells in healing cartilage defects. Potentially, scaffolds could be enhanced further by the addition of certain signal proteins or even chondrocytes to encourage more complete chondrogenesis. Synthetic scaffolds currently under study have been largely made from polylactic acid/polyglycolic acid (PLA/PGA) or synthetic HA.72,73 Natural materials reported include chitosan from arthropod exoskeletons, 74 hyaluronate, 75 aragonite from coral with HA, 76 agarose, alginate, and fibrin glue. 77 Chondromimetic (Orthomimetics (Tigenix: leuven, Belgium)) utilizes a type II collagen scaffold with CaPO4 and GAG. 78 Hydrogels have also been used in preclinical studies. Gelrin (Regentis (Regentis: Or-Akiva, Israel)) uses fibrinogen polyethylene glycol, which is ultraviolet activated in situ to form one such hydrogel to be used as a microfracture adjunct. 79 Demineralized donor-derived cancellous bone matrix, Osteosponge SC (Bacterin International Inc., Belgrade, MT), is another off-the-shelf option currently approved as a bone void filler. It delivers BMP 2, 4, and 7 to the site and is under study to repair osteochondral defects. The only plug-in large-scale clinical use to date in the United States has been the TruFit (Smith & Nephew, London, United Kingdom).80,81 This PLGA CaSO4 biopolymer has no level I or II evidence yet to support efficacy in chondral or bone healing.
One-Stage Minced Cartilage Surface
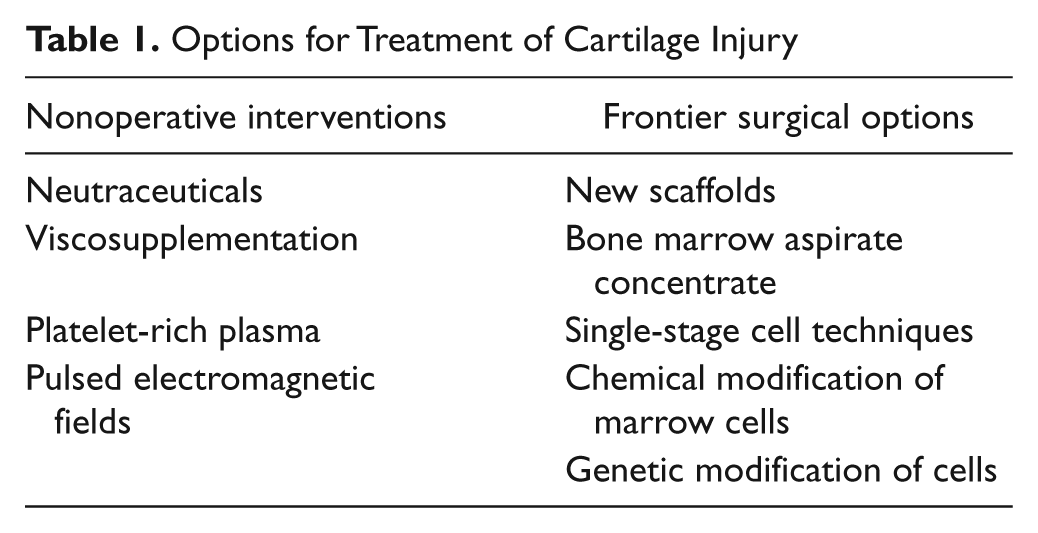
Studies have shown that chondrocytes might migrate to and from small freshly cut pieces of cartilage. 74 Two options exist for one-stage minced cartilage techniques: DeNovo NT (Zimmer, Warsaw, IN) uses human infantile allograft articular cartilage minced into small pieces, which is provided the day of surgery and mixed with fibrin glue to make a putty-like structure to fill the osteochondral defect. No sewing of a patch is needed, and animal studies show both good hyaline-like chondral fill and no immunogenic reaction. 82 CAIS (DePuy Mitek, Raynham, MA) uses minced autogenous cartilage taken the same day as surgery, placed on a PLA/PGA scaffold, and attached to the prepared defect with a PDS/PGA staple. Horse and early human pilot studies show encouraging results (Figure 2). 83
(
Another off-the-shelf technique uses a type I/III collagen membrane (Geistlich, Wolhusen, Switzerland) to help stabilize marrow stimulation clot in larger lesions. This AMIC technique has been described by Benthien and Behrens and recommended by Steinwachs et al. for retropatellar lesions. They have reported promising short-term results in at least one small human pilot study of 32 patients. 84 Longer term study is needed and is currently ongoing.
Role of Bone Marrow Aspirate Concentrate (BMAC)
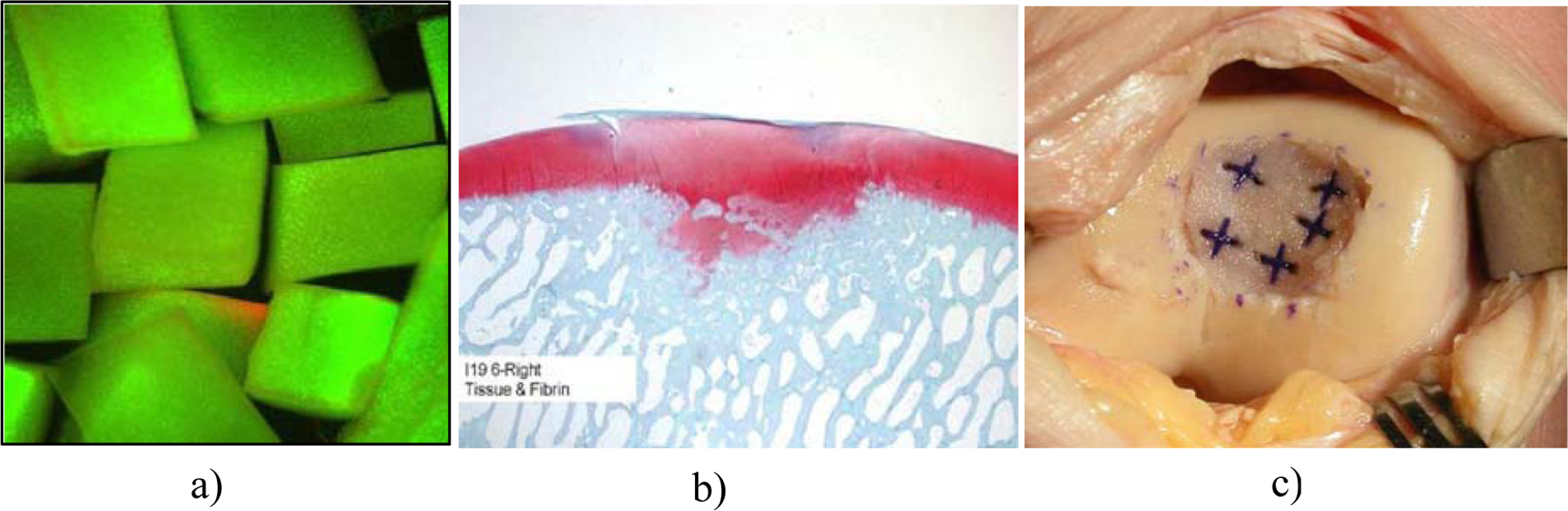
Recent studies demonstrated that marrow-derived stem cells (MSCs) secrete bioactive molecules that stimulate angiogenesis and mitosis of tissue-specific and -intrinsic progenitors and reduce T cell surveillance and inflammation, and some authors have also recognized that the presence of other nucleated cells is able to restore the damaged tissue.85-92 The authors from Milan are performing a single-step surgery utilizing autologous BMAC containing MSCs and growth factors for cartilage repair in large osteochondral lesions measuring even up to 22 cm2 in size. In a prospective study, 93 we followed up a group of 15 athletes operated on for grade IV cartilage lesions. The average size of the lesions was 9.2 cm2 ( Fig. 3A ). We harvested 60 mL of BMAC from the ipsilateral iliac crest using a dedicated aspiration kit and centrifuged using a commercially available system (BMAC Harvest Smart PreP2 System, Harvest Technologies, Plymouth, MA) ( Fig. 3B and 3C ). The cartilage defect was templated and the collagen membrane fashioned according to the defect size. Using Batroxobin enzyme (Plateltex act, Plateltex S.R.O., Bratislava, Slovakia), the BMAC was activated and produced a sticky clot material which was implanted into the prepared cartilage defect ( Fig. 3D ). Finally the defect was covered with a collagen membrane (Chondrogide, Geistlich) in order to protect MSCs ( Fig. 3E ).
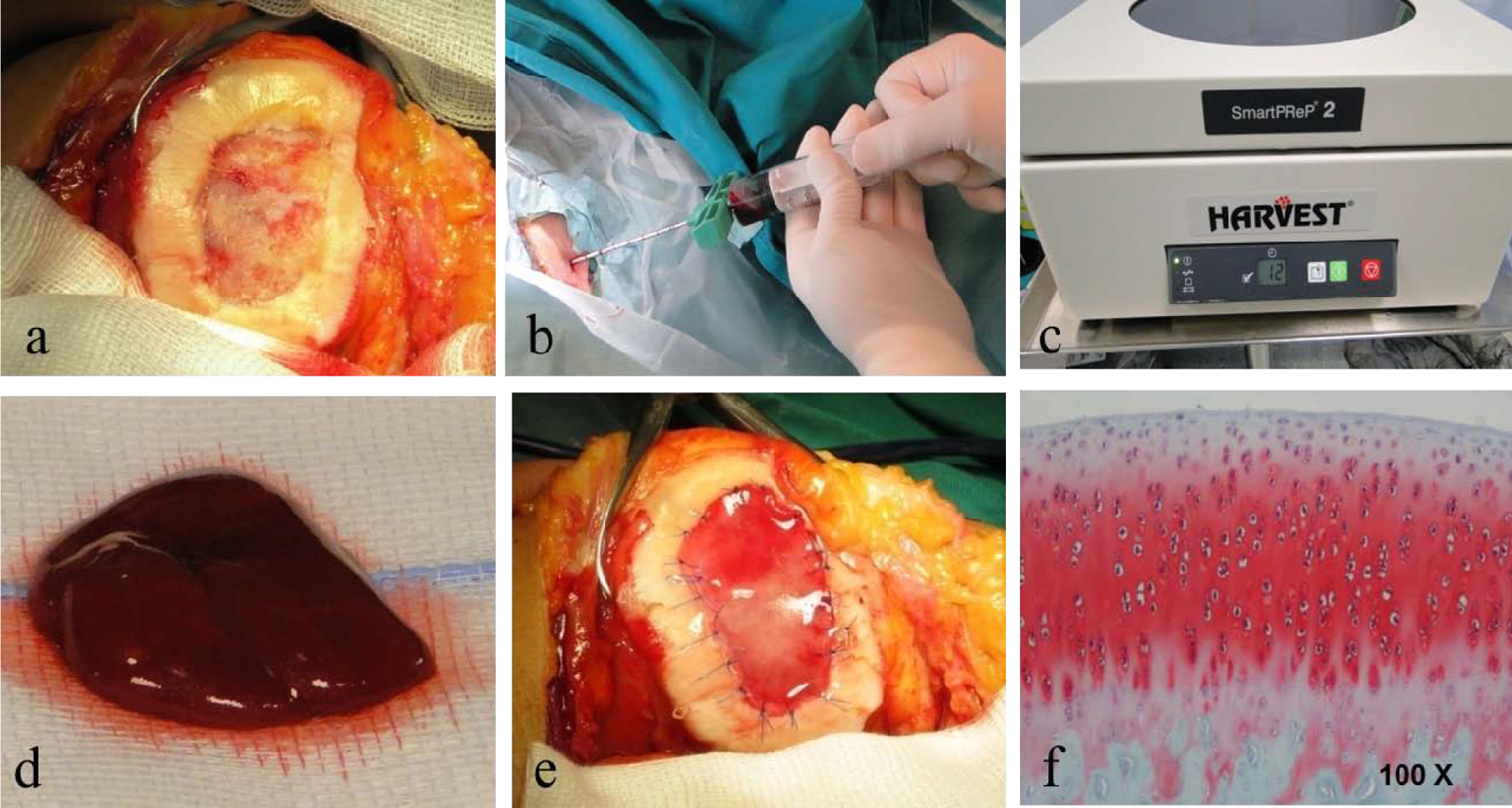
(
All patients showed significant improvement in all scores (visual analog scale [VAS], International Knee Documentation Committee [IKDC], Knee injury and Osteoarthritis Outcome Score [KOOS], Lysholm, Marx, SF-36 [physical/mental], and Tegner) at final follow-up (P < 0.005). Integration with adjacent cartilage was complete in 95% of our patients with restoration of the cartilage layer and subchondral bone. Second-look arthroscopies revealed a smooth, newly formed tissue with continuous intact to the healthy surrounding cartilage; no hypertrophy was identified. Macroscopic evaluation showed normal to nearly normal as classified by the International Cartilage Repair Society (ICRS) visual scoring system. Good histological findings were reported for the specimens analyzed, which presented hyaline-like features ( Fig. 3F ).
Considering that microfracture is usually used to treat lesions smaller than 3 cm2 and that the average size of lesions treated with autologous chondrocyte implantation is also smaller (5.3 cm2 in the last report by Peterson et al. on 224 patients),4,94 data from this study with average lesion size of 9 cm2 suggest a potential for treatment of larger articular cartilage lesions. 93
Manipulating Autogenous MSCs
Several growth factors or BMPs have shown promise in promoting hyalinization of repair tissue when placed in situ in defects. Chubinskaya and others have shown that Op-1 can effectively promote the formation of true hyaline cartilage in articular defects in several animal models. 95 Other signaling proteins including the TGF-B family members and Wnt-related proteins have been shown to be instrumental in adult cartilage homeostasis, while different Wnt and other proteins have been shown to inhibit chondrogenesis of MSCs. 96 In addition, enzymes such as MAP kinases can effect these proteins’ ability to adhere and effect mesenchymal cell development. 97 Richardson has shown that Dkk-1 is a hormone that acts on WNT signaling. In the presence of high levels of this hormone, mesenchymal cells trend toward cartilage formation, while in a low-level milieu, bone is formed. 98 As we further characterize these signal proteins and find ways to present and harness their power as promoters or inhibitors, we should be able to more effectively push the undifferentiated MSC toward normal chondrogenesis.
Gene Therapy
Gene therapy in combination with advanced tissue engineering techniques also offers some appealing options for improving articular cartilage repair. Adenoviral-mediated transfection of cDNA encoding for TGF-β1, insulin-like growth factor 1 (IGF-1), BMP-7, and BMP-2 has been shown to stimulate expression of cartilage-specific extracellular matrix components and decrease chondrocyte dedifferentiation. 99 Genes can be transferred either into mature chondrocytes or into chondroprogenitor cells used for cartilage repair. Pluripotent progenitor cells seem to be more receptive to transduction with recombinant adenoviral vectors and may provide the preferred platform for delivery of genes to enhance cartilage repair. Stem cells have the potential to differentiate into chondrocytes under appropriate conditions, potentially with improved cell viability, and are at the forefront of articular cartilage regeneration investigations. Specifically, mesenchymal stem cells (MSCs), found in bone marrow, skin, and adipose tissue, are capable of differentiating into articular cartilage as well as other cells of mesenchymal origin. 100 Hui et al. compared MSC transplants to cultured chondrocytes, osteochondral autografts, and periosteal grafts in animal models of osteochondritis dissecans. 101
Based on histological and biomechanical evaluation, several studies have found the MSC transplants to be comparable to cultured chondrocytes and superior to periosteum and osteochondral autograft in their ability to repair chondral defects.101-103 Stem cells alone or genetically modified MSCs can be used to augment existing cell-based cartilage repair methods. MSCs derived from adipose tissue alone or transfected with a vector for BMP-2 have been shown to predictably heal cartilage defects, with increased hyaline cartilage quality by genetic engineering.104,105 Experimental studies using implantation of PGA scaffolds seeded with retrovirally transduced periosteal MSC expressing BMP-7 and sonic hedgehog (Shh) genes into osteochondral defects significantly enhanced the quality of the cartilage repair tissue, resulting in much smoother cartilage surfaces and increased hyaline morphology. 106 These techniques have not been confirmed in clinical studies; however, they hold great scientific promise for treating cartilage injuries in athletes in the near future.
Conclusion
Since the time of Hippocrates, cartilage damage in athletes has remained a vexing problem. Current surgical techniques have been successful in restoring gross surface characteristics, but true biomechanical and biochemical restoration of the injured surface remains illusory. Although many of the newer techniques discussed above have not yet been proven in long-term level I or II studies, and some are still in the preclinical phase, they offer us a glimpse into the near future and the exhilarating potential for more resilient long-term joint restoration for our athletic populations.
Footnotes
The authors received no financial support for the research and/or authorship of this article.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
