Abstract
Objective
MicroRNAs (miRNAs) play a key role in the differentiation of bone marrow–derived mesenchymal stem cells (BMSCs) into chondrocytes. Our previous study found that novel-miR-81 can relieve osteoarthritis, but its role in chondrogenic differentiation of BMSCs remains unclear. The purpose of this study was to explore the role of novel-miR-81 in chondrogenic differentiation of BMSCs.
Methods
We used a model in which transforming growth factor (TGF)-β3-induced BMSCs differentiation into chondrocytes. We detected the expression Sox9, Collagen Ⅱ, Aggrecan, novel-miR-81, and Rac2 by real-time reverse transcription-quantitative polymerase chain reaction (RT-qPCR). Western blot was performed to detect the expression of Sox9, Collagen Ⅱ, and Rac2. Dual-luciferase reporter gene assay confirmed that the association between novel-miR-81 and Rac2. In addition, the ectopic chondrocyte differentiation of BMSCs was performed subcutaneously in nude mice. The effect of novel-miR-81 and Rac2 on ectopic chondrogenic differentiation of BMSCs was determined by immunohistochemical staining.
Results
Novel-miR-81 upregulated in chondrogenic differentiation of BMSCs. Rac2 was a key target of novel-miR-81. Mimic novel-miR-81 and siRac2 upregulated the expression of Sox9, Collagen Ⅱ, and Aggrecan.
Conclusion
Novel-miR-81 promotes the chondrocytes differentiation of BMSCs by inhibiting the expression of target gene Rac2, which provides potential targets for BMSCs transplantation to repair cartilage defects.
Introduction
Articular cartilage has a unique physiological structure that lacks blood vessels and nutrient supply. Cartilage defects caused by natural degeneration or trauma can lead to irreversible damage, which are often accompanied by pain and loss of function, placing a heavy burden on society and families. Implants consisting of bone marrow–derived mesenchymal stem cells (BMSCs) that seeded on scaffolds are considered as a promising therapy for repairing these defects. However, the differentiation ability of BMSCs is poor, which limits the further application of BMSCs. Therefore, it is very important to explore the mechanism of BMSCs differentiation into chondrocytes.
MicroRNAs (MiRNA), about 21-25 nucleotides in length, is a highly conserved endogenous non-protein-coding RNA. 1 MiRNAs can induce translational silencing by binding to the 3′-untranslated region (3′-UTR) of target messenger RNAs (mRNAs), thereby affecting cell proliferation, differentiation, apoptosis, and ontogeny. 2 Increasing evidence suggests that miRNAs play important regulatory roles in the differentiation of BMSCs into chondrocytes.3-6 With advancing in next-generation sequencing or massively parallel DNA sequencing technologies and plummeting costs, miRNA sequencing methods are widely used for genome-wide discovery of known and novel miRNA expression. 7 It is urgent to explore the function of novel miRNAs. Therefore, further studies on the molecular mechanism of novel miRNAs regulating the chondrogenic differentiation of BMSCs can provide new ideas for the treatment of cartilage defects.
Our group discovered some new miRNAs that have not been recorded in miRbase by BGISEQ-500 sequencing technology, including novel-miR-81. Our previous study found that novel-miR-81 can relieve osteoarthritis. 8 However, the association of novel-miR-81 with chondrogenic differentiation of BMSCs has not been established. This study aimed to investigate the role and mechanism of novel-miR-81 in the chondrogenic differentiation of BMSCs.
Materials and Methods
Culture and Differentiation of BMSCs into Chondrocytes
Sprague-Dawley rats (60-80 g) were purchased from the Institute of Medical Biology Chinese Academy of Medical Sciences. License number: SCXK 2019-0008, quality certificate number: 110322220100770286. Sprague-Dawley rats were euthanized by injection of sodium pentobarbital (200 mg/kg body weight). The tibia and femur were separated under sterile conditions in the biosafety cabinet, and the bone marrow was flushed out with DMEM basic containing 1% double antibody. BMSCs were collected through centrifugation at 1,200 rpm for 10 min at room temperature. BMSCs were cultured with SD rat BMSCs complete medium in an atmosphere of 5% CO2 at 37°C. P3 and P4 BMSCs can be used in the following experiments. The induction group was cultured for 7 days with induction medium containing 100 nmol dexamethasone, 50 μg/mL
Differentiation of BMSCs into osteoblasts and adipocytes
Osteogenic cell induction medium contained 100 nmol dexamethasone, 10 ng/ml β-glycerophosphate solution, and 50 μM ascorbic acid. BMSCs were differentiated into adipocytes by stimulating BMSCs with 100 μM H2O2 in serum-free medium for 1 hour.
Alkaline Phosphatase Staining (ALP), Alizarin Red Staining (AR), and Oil Red O Staining
BMSCs were induced to osteoblasts for 7 days and stained for alkaline phosphatase with Alkaline Phosphatase Stain Kit (Solarbio, Beijing, China). BMSCs were induced to osteoblasts for 14 days and stained with alizarin red with Alizarin Red S solution (Solarbio, Beijing, China). Using Oil red O staining (Sigma, Darmstadt, Germany) to detect the differentiation of BMSCs into adipocytes.
Flow Cytometry
BMSCs were incubated with CD292 (BD Biosciences, 1:10 dilution, NJ) and CD45 (BD Biosciences, 1:10 dilution) antibodies for 30 minutes in the dark, washed and resuspended with phosphate-buffered saline (PBS), and detected by BD C6 flow cytometer.
Real-Time Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR)
Total RNA was extracted using EZ-press RNA Purification Kit (EZBioscience, Roseville). RNA was reverse transcribed into complementary DNA (cDNA) using Color Reverse Transcription Kit (EZBioscience). Quantitative polymerase chain reaction (qPCR) was performed using a 2 x Color SYBR qPCR Master Mix (EZBioscience). The 2−ΔΔCq method was used to analyze the RT-qPCR data. U6 was used as a reference for the detection of novel-miR-81, and GAPDH was used as a reference for Sox9, Collagen Ⅱ, and Aggrecan. Specific primers were used for quantitative PCR as follows: Sox9 (TGGCAGAGGGTGGCAGACAG and CGTTGGGCGGCAGGTATTGG); Collagen Ⅱ (GGAGCAGCAAGAGCAAGGAGAAG and GGAGCCCTCAGTGGACAGTAGAC); Aggrecan (GCTACGACGCCATCTGCTACAC and ATGTCCTCTTCACCACCCACTCC); GAPDH (ATGGCTACAGCAACAGGGT and TTATGGGGTCTGGGATGG); novel-miR-81 (CGTTGGGGATGGTAGGGTCT and AGTGCAGGGTCCGAGGTATT); Rac2 (CGATGAGGCAATCCGAGCAGTC and GGACCGTGGGAGTGGGAAGAC); Fgr (CCTGGAATGGGCTGTGTGTTCTG and TGAGGCGAGATGGACGAAGACC); Lat2 (TGTTGCTGCTGCTGCTCTTGG and CTTGCTGGTTCCTCTGCTCGTAG); Calcr (CTCCTCGTGCTGTAGCCTTTGC and TCGCCTTCGTTGTTGCTGACTG); Cd4 (CCACGGTTTCTCTGCCTCTGTTC and GCTGGGTGTGCTTGAGTGTCTG); Impad1 (GCTCCTGTCCCTACCTGCTCTTAG and GGTTGGCACTCCACTGTACTTCTG).
Western Blot
The total protein was extracted on ice by RIPA Lysate (Beyotime, Shanghai, China) containing protease inhibitor cocktail (Thermo Fisher Scientific, MA). Protein concentration was determined by the BCA Protein Assay reagent (Beyotime). The total protein (30 µg/lane) was separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE; FUDE, Hangzhou, China) and then transferred to a polyvinylidene difluoride (PVDF) membrane (Merck Millipore, Darmstadt, Germany). Membranes were blocked with 5% non-fat milk for 1 hour at room temperature. After the incubation with primary antibodies Sox9 (Abcam, 1:4,000 dilution, Cambridge, UK), Collagen Ⅱ (Abcam, 1:1,000 dilution), and GAPDH (Abcam, 1:10,000) at 4°C for 12 hours, they were eluted them with TBST and incubated with a secondary antibody (Abcam, 1:10,000 dilution) for 1 hour. Proteins were visualized using horseradish peroxidase–linked secondary antibodies and enhanced chemiluminescence.
Cell Transfection
When BMSCs reached to 70%-80% confluence, novel-miR-81 mimic, mimic NC, novel-miR-81 inhibitor, inhibitor NC, siRac2, and siNC were transfected into BMSCs at a final concentration of 100 nmol by using the Lipofectamine™ 3000 (Invitrogen, Carlsbad, CA). siRac2 and siNC were purchased from Sangon Biotech (Shanghai, China). Novel-miR-81 mimic, mimic NC, novel-miR-81 inhibitor, and inhibitor NC were obtained from GenePharma (Suzhou, China). The sequence of novel-miR-81 has been patented, and the patent number is 202011581026.1.
siRac2-001 sense: 5′-GCUGGACCUUCGAGAUGACAATT-3′
siRac2-001 antisense: 5′-UUGUCAUCUCGAAGGUCCAGCTT-3′
siRac2-002 sense: 5′-GGCCAAGGAUAUUGAUUCAGUTT-3′
siRac2-002 antisense:5′-ACUGAAUCAAUAUCCUUGGCCTT-3′
siRac2-003 sense: 5′-CCUUUGUCAGAAAAACACATT-3′
siRac2-003 antisense: 5′-UGUGUUUUUCUGACAAAGGTT-3′
siRNAs negative control sense: 5′-UUCUCCGAACGUGUCACGUTT-3′
siRNAs negative control antisense: 5′-ACGUGACACGUUCGGAGAATT-3′
Novel-miR-81 mature miRNA sequence: 5′-UUGGGGAUGGUAGGGUCUCUGAGG-3′
Novel-miR-81 mimic sense: 5′-UUGGGGAUGGUAGGGUCUCUGAGG-3′
Novel-miR-81 mimic antisense: 5′-UCAGAGACCCUACCAUCCCCAAUU-3′
Novel-miR-81 inhibitor: 5′-CCUCAGAGACCCUACCAUCCCCAA-3′
Mimic NC sense: 5′-UUCUCCGAACGUGUCACGUTT-3′
Mimic NC antisense: 5′-ACGUGACACGUUCGGAGAATT-3′
Inhibitor NC: 5′-CAGUACUUUUGUGUAGUACAA-3′
Dual-Luciferase Reporter Assay
BMSCs (2 × 104 cells/well) were seeded in 24-well plates. After cultured for 24 hours, novel-miR-81mimic (100 nM) with plasmids of Rac2 WT 3′UTR (100 ng) or MUT 3′UTR (100 ng), novel-miR-81inhibtor (100 nM) with plasmids of Rac2 WT 3′UTR (100 ng) or MUT 3′UTR (100 ng) were co-transfected into cells with Lipofectamine 3000 (Thermo Fisher Scientific, Guangzhou, China) according to the manufacturer’s instructions. After 48 hours transfection, firefly and renilla luciferase activities were calculated by Dual-Luciferase Reporter Assay System Kit (Promega Corporation, Madison, WI).
Ectopic Chondrogenesis of BMSCs
Normal saline was used to configure 20%-30% gelatin and 2%-3% sodium alginate, respectively. Next, the gelatin and sodium alginate are mixed at A ratio of 10 to 1 to make liquid A. Autoclave liquid A for later use. Animal groups (n = 6 per group) were rno-miR-81 mimic group, mimic NC group, rno-miR-81 inhibitor group, inhibitor group NC group, siNC group, siRac2 group. rno-miR-81 mimic, rno-miR-81 inhibitor, siRac2 and their corresponding NC were transfected into BMSCs for induction culture. Each group of cells was counted after Trypsin-EDTA digestion. Cell suspension of 75 μl was obtained by 7.5 × 105 cells. Cell suspension of 75 μl is mixed with 75 μl liquid A to form liquid B. Liquid B of 150 μl is the volume of each graft pack, and 150 μl of liquid B was absorbed with a 1-mL syringe and placed in a refrigerator at 4°C for 15 minutes. Solution B is slowly squeezed into 3%-5% calcium chloride solution and left for 5 minutes. The solidified liquid B was cleaned by PBS and placed in the medium for transplantation. Nude mouse (BABL/c-nu/nu), 18-22g, purchased from the Laboratory Animal Center of Guangzhou University of Chinese Medicine. License number: SCXK (Guangdong) 2018-0034, quality certificate number: 44005800012630. The solidified liquid B was transplanted into the back of nude mice subcutaneously. After 14 days, the graft mass was removed.
Alcian Blue Staining
Sections were dewaxed under the conditions of xylene I for 10 minutes, xylene II for 10 minutes, absolute ethanol for 10 minutes, 95% alcohol for 5 minutes, 85% alcohol for 5 minutes, and 75% alcohol for 10 minutes. After washing with PBS, alcian blue staining solution (Cyagen Biosciences Inc, Guangzhou, China) was added dropwise for 30 minutes. Rinse with running water for 3-5 minutes. Sections were dehydrated under conditions of 75% alcohol for 10 minutes, 85% alcohol for 5 minutes, 95% alcohol for 5 minutes, absolute ethanol for 10 minutes, xylene II for 10 minutes, and xylene I for 10 minutes. Neutral gum was added dropwise for mounting. After the neutral gum was completely solidified, film was collected under a microscope.
Immunohistochemistry (IHC)
Perform immunohistochemical staining according to the instructions of Histostain-Plus Kits (Bioss, Beijing, China). primary antibody included Sox9 (Abcam,1:250 dilution) and Collagen II (Abcam, 1:250 dilution).
Statistical Analyses
Statistical analysis was performed using GraphPad Prism 8.0 and SPSS 23.0. Quantitative data were expressed as the mean ± standard deviation. The statistical analysis of difference between groups was performed by unpaired two-tailed Student’s t test. P < 0.05 was considered to indicate a statistically significant difference.
Results
Identification of BMSCs
BMSCs gradually changed from round, irregular polygonal, short fusiform to long fusiform ( Fig. 1A ). After the BMSCs were induced into chondrocytes for 7 days, alcian blue staining showed the formation of blue acid mucopolysaccharides ( Fig. 2A ). After BMSCs were induced into osteoblasts for 7 days, ALP staining showed the formation of brown alkaline phosphatases ( Fig. 1B ). After BMSCs were induced into osteoblasts for 21 days, AR staining showed the formation of red calcium nodules ( Fig. 1B ). After BMSCs were induced into adipocytes for 1 day, Oil red O staining showed the formation of red lipid droplets ( Fig. 1B ). The above results indicated that the obtained BMSCs have multiple differentiation potential and can differentiate into chondrocytes, osteoblasts, and adipocytes. Flow patterns showed that the cells were positive for CD29 and negative for CD45 ( Fig. 1C ), indicating that these cells showed the characteristics of BMSCs. In conclusion, the obtained BMSCs can be used for subsequent research.
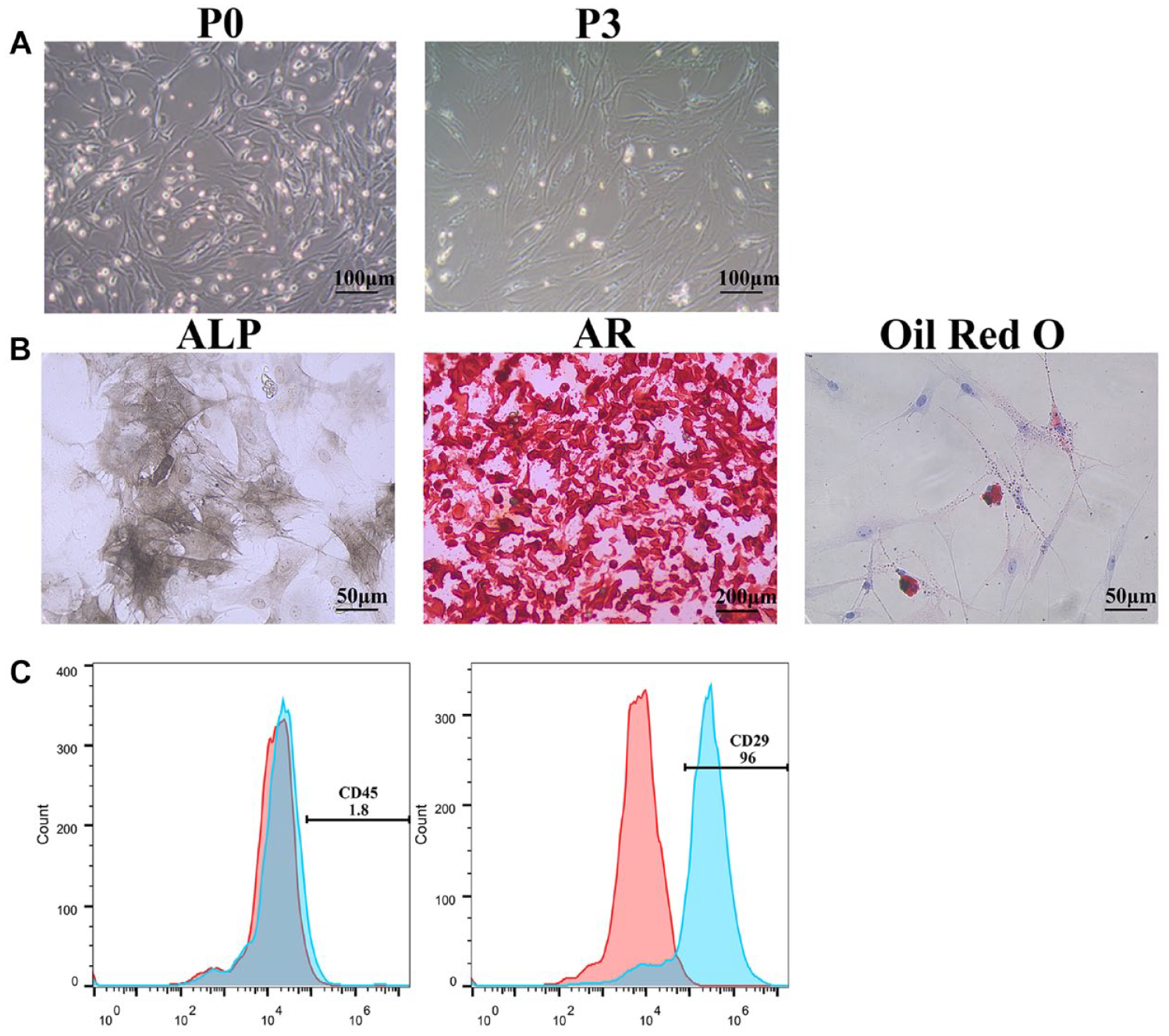
Identification of BMSCs. (
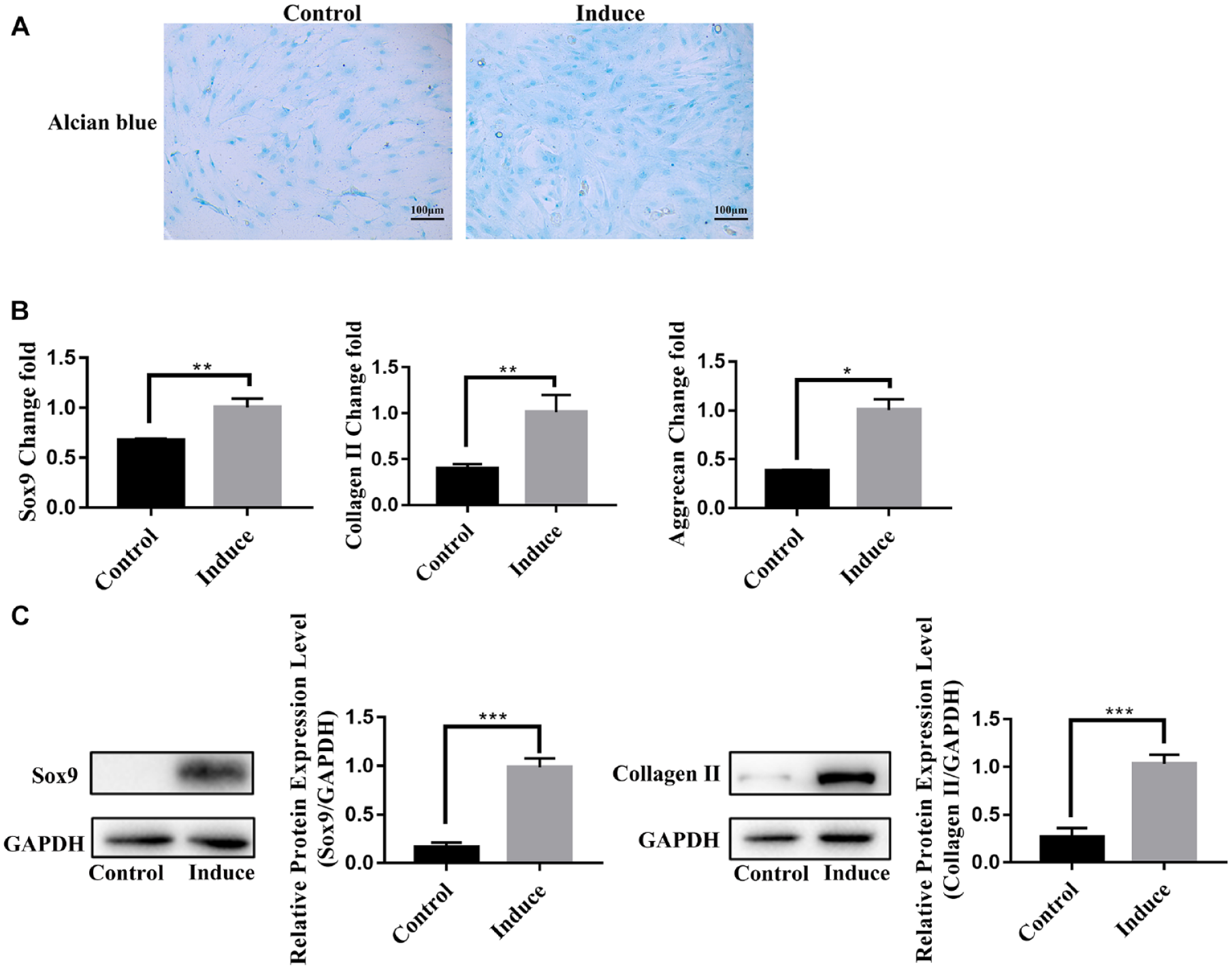
TGF-β3 promoted the differentiation of BMSCs into chondrocytes in vitro. (
TGF-β3 Promoted the Differentiation of BMSCs into Chondrocytes In Vitro
TGF-β3 is a key factor in the differentiation of BMSCs into chondrocytes. 9 In this study, BMSCs were treated with 10 ng/ml TGF-β3 for 7 days. Alcian blue staining showed that the acid mucopolysaccharide in the induced group was significantly higher than that in the control group ( Fig. 2A ). The mRNA and protein levels of Sox9 and Collagen II in the induced group were significantly higher than that in the control group by RT-qPCR ( Fig. 2B ) and Western blot ( Fig. 2C ). In addition, Aggrecan was also upregulated in the induced group by RT-qPCR ( Fig. 2B ). Based on these results, TGF-β3-induced chondrocyte differentiation model of BMSCs was successfully established.
Novel-miR-81 Played a Positive Regulatory Role in Chondrocyte Differentiation of BMSCs In Vitro
Novel-miR-81 was significantly increased in the induced group by RT-qPCR ( Fig. 3A ). Next, we transfected novel-miR-81 mimic and inhibitor into BMSCs for 24 hours, and then added TGF-β3 to culture for 7 days. The expression of novel-miR-81 in the mimic group was more than 2,654,770 times higher than that in the m-NC group ( Fig. 3B ). Alcian blue staining showed that the acid mucopolysaccharide in mimic group was significantly higher than that in m-NC group ( Fig. 3C ). The mRNA and protein levels of Sox9 and CollagenⅡ in mimic group were significantly higher than that in m-NC group by Western Blot ( Fig. 3D ). These results indicated that novel-miR-81 plays a positive regulatory role in chondrocyte differentiation of BMSCs in vitro.
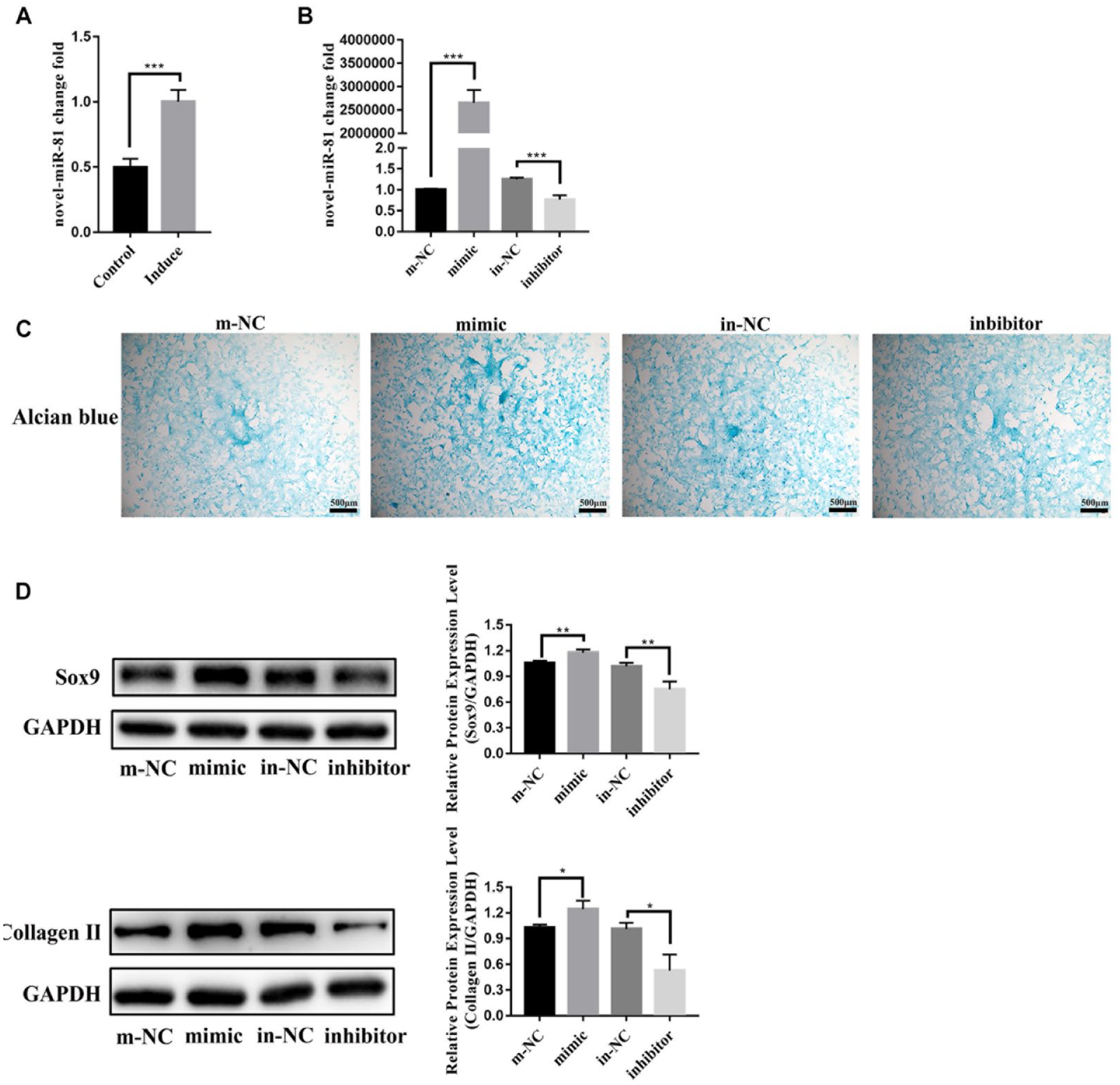
Novel-miR-81 played a positive regulatory role in chondrocyte differentiation of BMSCs in vitro. (
The Direct Target of novel-miR-81 was Rac2 In Vitro
Beijing Genomics institution predicted the potential target genes of novel-miR-81 through the bioinformatics website, including Fgr, Cd4, Lat2, Calcr, Rac2, Impad1, and so on ( Fig. 4A ). RT-qPCR showed that the expression of Rac2, Fgr, Lat2 was decreased in the mimic group compared with the m-NC group, while the expression of Rac2, Fgr, Lat2 was increased in the inhibitor group compared with the in-NC group ( Fig. 4A ). Compared with Fgr and Lat2, Rac2 decreased most significantly in the mimic group, so the follow-up experiments mainly focused on Rac2. Rac2 were significantly reduced by novel-miR-81mimic by Western blot ( Fig. 4B ). More importantly, compared with the NC group, relative luciferase activity of WT-Rac2 was significantly inhibited by novel-miR-81mimic and significantly increased by novel-miR-81 inhibitor. At the same time, there was no difference in luciferase activity of MUT groups ( Fig. 4C ). The results confirmed that the direct target of novel-miR-81 is Rac2.
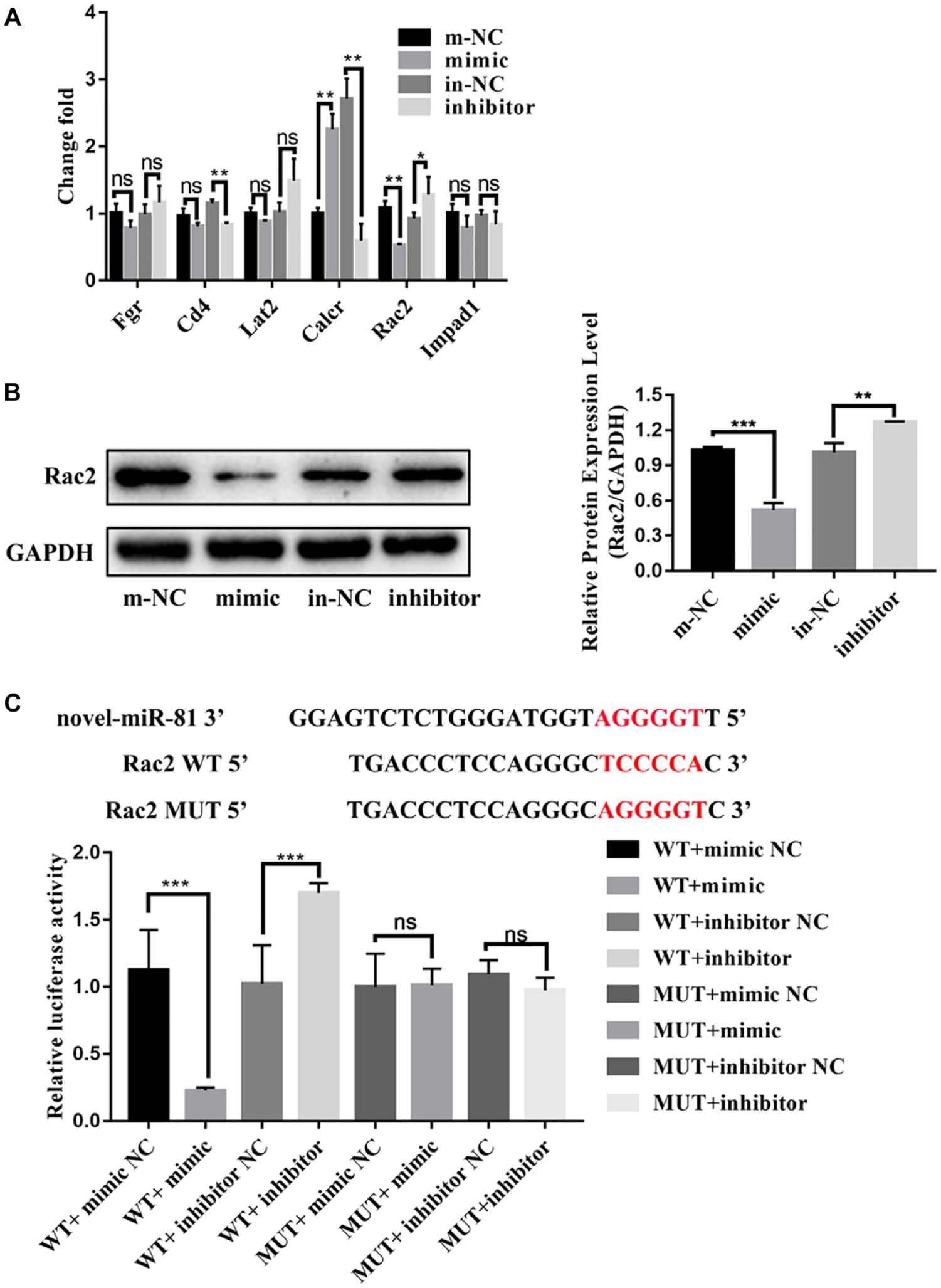
The direct target of novel-miR-81 was Rac2 in vitro. (
SiRac2 Positively Regulated Chondrocyte Differentiation of BMSCs In Vitro
The study found that the target gene of novel-miR-81 is Rac2. To explore the effect of Rac2 on chondrocyte differentiation of BMSCs, we constructed an interfering fragment of Rac2. RT-qPCR analysis showed that the expression of Rac2 mRNA was obviously downregulated after siRac2-002 transfection into BMSCs ( Fig. 5A ). siRac2-002 is the best interference fragment, so it was used for subsequent experiments. In addition, siRac2 promoted the expression of novel-miR-81 ( Fig. 5B ). Alcian blue staining showed that the acid mucopolysaccharide in siRac2 group was significantly higher than that in siNC group ( Fig. 5C ). RT-qPCR results showed that the expression of early cartilage markers Sox9, Collagen Ⅱ, and Aggrecan in siRac2 group was significantly higher than that in siNC group ( Fig. 5D ). Western blot showed that the expression of Sox9 and Collagen Ⅱ in siRac2 group was significantly higher than that in siNC group ( Fig. 5E ). The results confirmed that Rac2 negatively regulated chondrocyte differentiation of BMSCs.
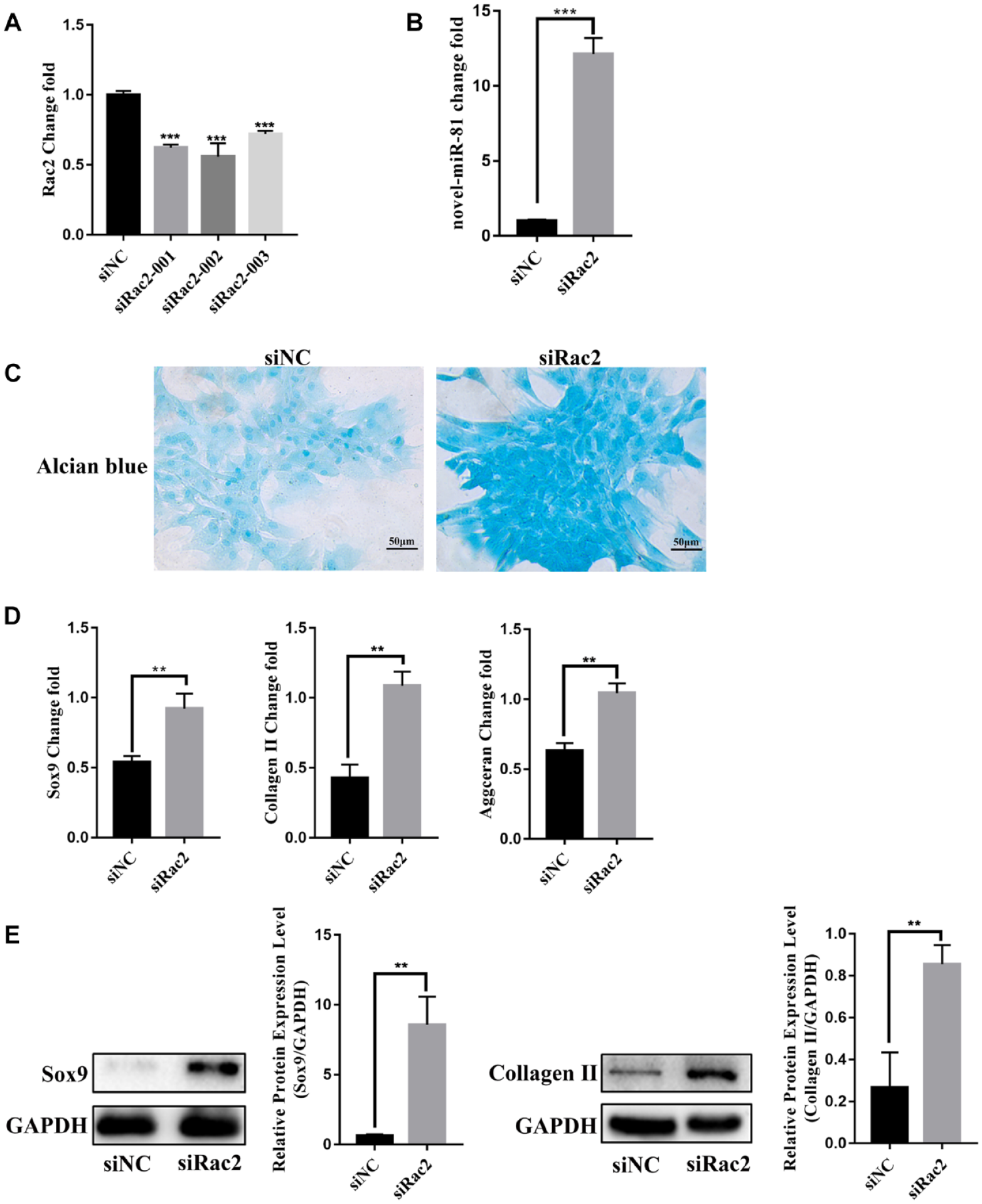
siRac2 positively regulated chondrocyte differentiation of BMSCs in vitro. (
Novel-miR-81 Promoted Chondrogenic Differentiation of BMSCs In Vivo
In order to further determine whether novel-miR-81 can also promote the cartilage differentiation of BMSCs in vivo, the experiment of ectopic cartilage differentiation was carried out in nude mice. The sodium alginate gel containing BMSCs was transplanted into the back of nude mice, and the samples were collected 14 days later. A small mass can be seen at the transplant site ( Fig. 6A ). Alcian blue staining showed that the blue product at the mass site was significantly increased compared with the surrounding tissue ( Fig. 6B ). IHC results showed that the expressions of Sox9 and Collagen II in the mimic group were significantly higher than those in the m-NC group, indicating that novel-miR-81 mimic could also promote the cartilage differentiation of BMSCs in vivo. Compared with the in-NC group, the expressions of Sox9 and Collagen II in the inhibitor group were significantly decreased, indicating that novel-miR-81 inhibitor inhibited the chondrogenic differentiation of BMSCs ( Fig. 6C ). As shown in Suppl. Figure S1, the expression of collagen X in the novel-miR-81 mimic group was significantly lower than that in the m-NC group. These results suggested that novel-miR-81 could promote chondrogenic differentiation of BMSCs in vivo.
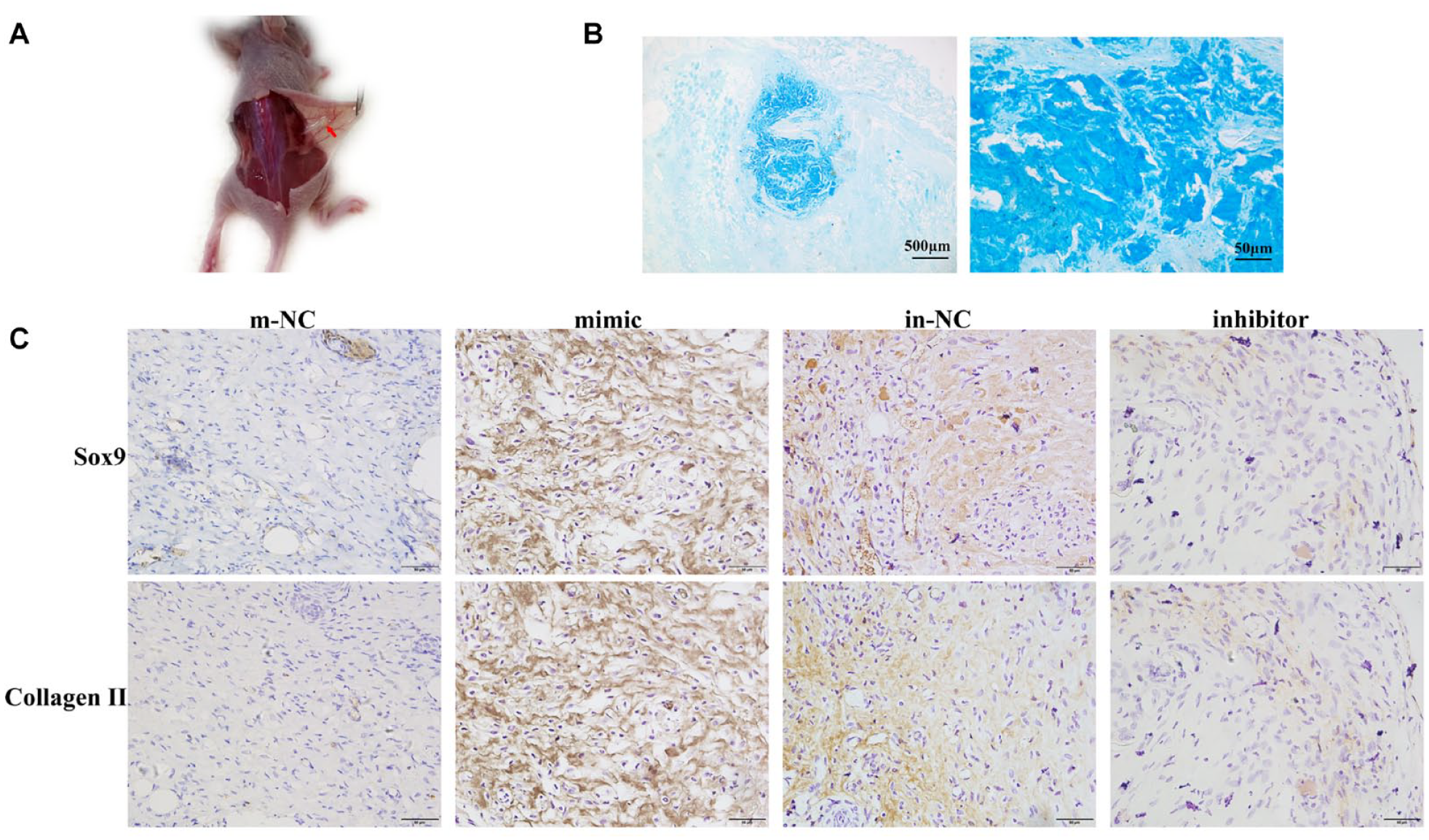
Novel-miR-81 promoted chondrogenic differentiation of BMSCs in vivo. (
SiRac2 Played a Positive Regulatory Role in Chondrocyte Differentiation of BMSCs In Vivo
Rac2 was significantly decreased in the siRac2 group compared with the siNC group by IHC ( Fig. 7A ). Sox9 and Collagen II were significantly increased in the siRac2 group ( Fig. 7A ). Taken together, our results demonstrated that siRac2 plays a positive regulatory role in chondrocyte differentiation of BMSCs in vivo.
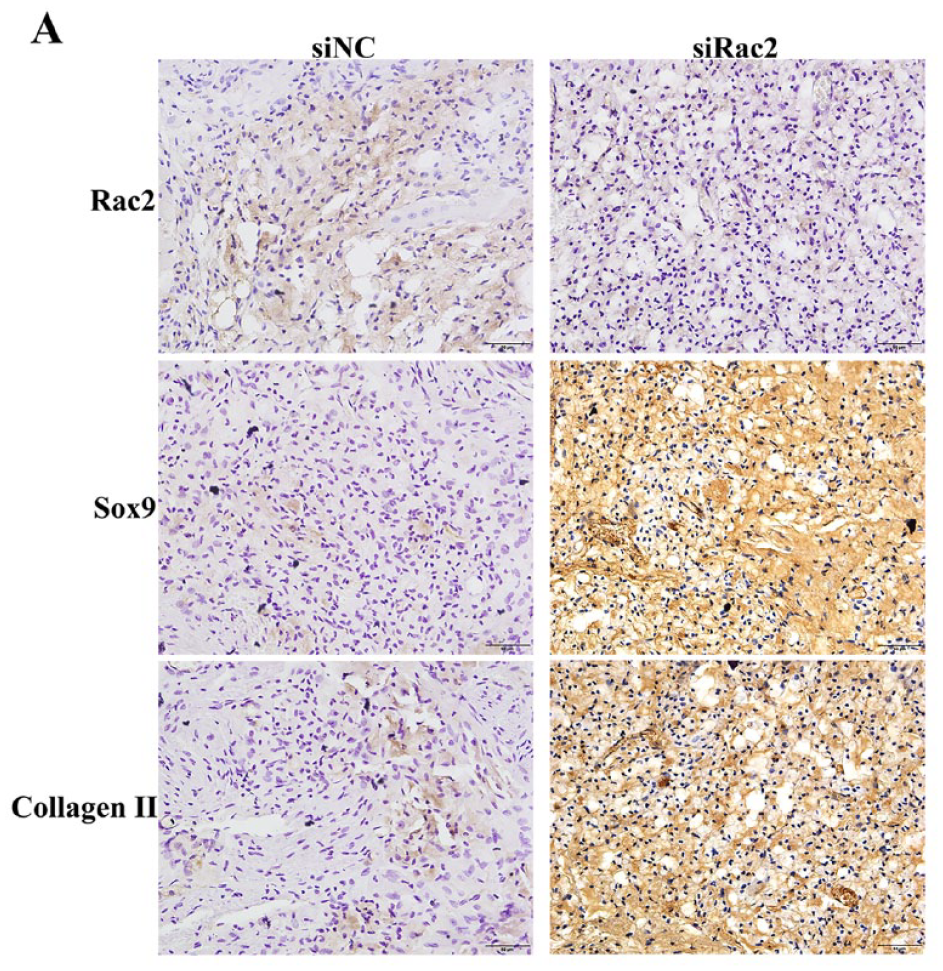
siRac2 played a positive regulatory role in chondrocyte differentiation of BMSCs in vivo. (
Discussion
Differentiation of BMSCs into chondrocytes is an important strategy for the treatment of cartilage defects. Therefore, it is very important to study the regulatory mechanism of BMSCs differentiation into chondrocytes. In in vitro models, this study used classical induction of TGF-β3 to induce BMSCs to differentiate into chondrocytes. 9 Interestingly, we found that novel-miR-81 expression was significantly increased during chondrogenic differentiation of BMSCs. Next, lipofection experiments were used to determine that novel-miR-81 promoted chondrogenic differentiation of BMSCs. Dual-luciferase reporter genes revealed that Rac2 was a target gene of novel-miR-81. Furthermore, silencing Rac2 expression promoted chondrogenic differentiation of BMSCs. More importantly, we further confirmed that novel-miR-81 and siRAC2 promoted chondrogenic differentiation of BMSCs in ectopic chondrogenic differentiation experiments in nude mice. Therefore, we concluded that novel-miR-81 promoted the chondrogenic differentiation of BMSCs by inhibiting the expression of Rac2, which may be a therapeutic target for cartilage defects.
Cartilage lacks the ability to repair itself due to the relative avascular nature of the tissue and the limited ability of cells to migrate to the tissue for repair. 10 If the cartilage defect is not repaired in time, it will soon lead to degenerative changes and even disability of the joint. 11 Stem cells repair damaged articular cartilage has become the most effective way to treat cartilage damage. 12 BMSCs can differentiate into chondrocytes in a suitable induction environment in vitro. 13 In addition, BMSCs have the advantages of convenient access, low immunogenicity, and the ability to secrete trophic factors. 14 Therefore, BMSCs are ideal seed cells for the treatment of cartilage defects. 15 The differentiation ability of BMSCs is poor, which limits the further application of BMSCs. Therefore, it is very important to explore the mechanism of BMSCs differentiation into cartilage.
MiRNAs are essential regulators in the process of cartilage formation and development, 16 and also play an important role in regulating the differentiation of BMSCs into chondrocytes. Some miRNAs positively regulate the differentiation of BMSCs into chondrocytes, such as miR-140 and miR-410,3,4 while others inhibit the differentiation of BMSCs into chondrocytes, such as miR-30a and miR-203a-3p.5,6 The role of novel-miR-81 in chondrogenic differentiation of BMSCs is unknown. In this study, we found that the expression of novel-miR-81 was increased during the differentiation of BMSCs into chondrocytes in vitro. This suggests that novel-miR-81 may be involved in the chondrogenic differentiation of BMSCs in vitro. Furthermore, we confirmed that novel-miR-81 positively regulated the chondrogenic differentiation of BMSCs.
The small GTPase Rac2 is a member of the Rho family of Ras-like small GTPases. 17 Rac2 regulates multiple functions, including cell growth and proliferation, chemotaxis, superoxide production, phagocytosis, transcription, and cytoskeletal organization and rearrangements. 18 Rac2 knockdown has significantly increased bone mass. 19 In addition, miRNAs regulate Rac2 expression. 20 In this study, Rac2 was a target gene of novel-miR-81 and Rac2 knockdown significantly promoted the expression of cartilage markers Sox9 Collagen Ⅱ and Aggrecan. The above results indicated that novel-miR-81 promotes the chondrocytes differentiation of BMSCs by inhibiting the expression of target gene Rac2 in vitro.
To further determine whether novel-miR-81 and siRac2 also promoted cartilage differentiation of BMSCs in vivo, ectopic chondrogenic differentiation experiments were carried out in nude mice. Sodium alginate gelatin not only has superior mechanical properties but also supports cartilage regeneration. 21 As a scaffold material, it can achieve a uniform cell mixture and ideal shape, which makes it easier to fill cartilage defects in a minimally invasive way. Therefore, sodium alginate gelatin was used as scaffold material in this experiment. Our study found that the expression of Sox9 and Collagen II in the tissues of the novel-miR-81 mimic group and siRac2 group was significantly higher than that of the respective NC groups. This indicated that novel-miR-81 and siRac2 promoted cartilage differentiation of BMSCs in vivo, which is consistent with the cellular results.
This study has some limitations. First, we selected BMSCs of rats and monolayer culture and did not select BMSCs of human and pellet or micromass culture. It is well known that human BMSCs and pellet or micromass culture are more responsive to the human environment. Second, we performed ectopic chondrogenic differentiation experiments on nude mice, but did not perform cartilage defect repair experiments. The ultimate significance of our study on the differentiation of BMSCs into cartilage is to repair cartilage defects. Therefore, the mechanism of novel-miR-81 in cartilage defects is a question that we need to further explore.
Conclusion
Novel-miR-81 targeted Rac2 to promote chondrogenic differentiation of BMSCs. Accordingly, novel-miR-81/Rac2 may represent a potential target for the future treatment for bone defect or bone loss. Further studies are needed to explore how to apply novel-miR-81/ Rac2 to clinical treatment to repair bone defect.
Supplemental Material
sj-docx-1-car-10.1177_19476035231207778 – Supplemental material for Novel-miR-81 Promotes the Chondrocytes Differentiation of Bone Marrow Mesenchymal Stem Cells Through Inhibiting Rac2 Expression
Supplemental material, sj-docx-1-car-10.1177_19476035231207778 for Novel-miR-81 Promotes the Chondrocytes Differentiation of Bone Marrow Mesenchymal Stem Cells Through Inhibiting Rac2 Expression by Ziwei Luo, Jinqi Xie, Haoxiang Ye, Jie Zhang, Yangping Liu, Chunmei Ma, Jiahui Cao, Hao Pan, Xiaosheng Liu, Xianxi Zhou, Jiechen Kong, Dongfeng Chen and Aijun Liu in CARTILAGE
Supplemental Material
sj-tif-2-car-10.1177_19476035231207778 – Supplemental material for Novel-miR-81 Promotes the Chondrocytes Differentiation of Bone Marrow Mesenchymal Stem Cells Through Inhibiting Rac2 Expression
Supplemental material, sj-tif-2-car-10.1177_19476035231207778 for Novel-miR-81 Promotes the Chondrocytes Differentiation of Bone Marrow Mesenchymal Stem Cells Through Inhibiting Rac2 Expression by Ziwei Luo, Jinqi Xie, Haoxiang Ye, Jie Zhang, Yangping Liu, Chunmei Ma, Jiahui Cao, Hao Pan, Xiaosheng Liu, Xianxi Zhou, Jiechen Kong, Dongfeng Chen and Aijun Liu in CARTILAGE
Footnotes
Acknowledgment and Funding
The authors thank Xu Tong and Liu Xinchao for polishing the manuscript. Thanks to the Experimental Teaching Center of Basic Medical School of Guangzhou University of Chinese Medicine for the help of this study. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China, Grant/Award Numbers: 82174368, 32000551, 82004026, 82205159; Natural Science Foundation of Guangdong Province, Grant/Award Number: 2017A030312009; Shenzhen Natural Science Foundation: JCYJ20220530150213031.
Declaration of Conflicting Interests
The author(s) declare that they have no competing interests. The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Approval
The experimental procedures were in accordance with the Guangzhou University of Chinese Medicine Laboratory Animal Ethics Committee Regulations. Experimental animals were used following the guidelines set by the National Institute of Health Guide for the care and use of laboratory animals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
