Abstract
Objective
Functional polymorphisms of interleukin 16 (IL16) have been reported to be closely related to the risk of osteoarthritis (OA). However, how IL16 affects OA remains unclear. In this study, the role of IL16 in OA and the possible mechanisms were examined.
Methods
We established a meniscal/ligament injury (MLI) post-traumatic OA model in Sprague Dawley rats and an IL1β-induced ADTC5 cells OA model. We detected the expression of IL16, novel-miR-81, MMP3, and MMP13 by quantitative real-time polymerase chain reaction. Western blot was performed to detect the expression of IL16, MMP3, and MMP13. The association between IL16 and novel-miR-81 was confirmed by luciferase reporter assay. Hematoxylin and eosin staining, Safranin O and Fast Green staining, and immunohistochemical staining were performed to clarify the effect of intra-articular injection of novel-miR-81 agomir in rats OA model.
Results
IL16 was upregulated in OA model. Knockdown of IL16 and overexpression of novel-miR-81 downregulated the expression of MMP3 and MMP13. Importantly, IL16 was a key target of novel-miR-81. Intra-articular injection of novel-miR-81 agomir could attenuate OA progression in rats OA model.
Conclusion
Novel-miR-81 targeted IL16 to relieve OA, suggesting that novel-miR-81and IL16 may be new therapeutic targets for OA.
Copyright © 2022 Ziwei Luo et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Osteoarthritis (OA) is a chronic inflammatory disease caused by the degeneration of articular cartilage, which is characterized by articular cartilage destruction, synovial inflammation, and bone remodeling, 1 often involving the knees, hips, spine, and fingers. 2 Anti-inflammatory therapy is one of the main treatments for OA, such as topical non-steroidal anti-inflammatory drugs (NSAIDs) or oral NSAIDs. 3 Despite the development of some new anti-inflammatory drugs,4 -6 OA still lacks effective treatments, and the patient’s condition usually worsens over time.
Studies have shown that cartilage degeneration is induced by immune system cytokines. 7 Innate immune activation in OA leads to the production of cytokines such as IL1β, IL6, IL8, and tumor necrosis factor (TNF)-α, and the complement system activates and produces matrix-degrading enzymes such as matrix metallopeptidase (MMP) and ADAMTS4/5. 8 Importantly, recent studies have shown that IL16 gene polymorphisms are associated with the risk of knee OA.9,10 However, how IL16 affects OA remains unclear. Therefore, studying the role of IL16 in the occurrence and development of OA will help to discover new therapeutic targets.
In the present study, we found that IL16 was significantly increased in rats OA model. Given that microRNA (miRNA) regulates the target genes involved in OA, 11 we hypothesized that IL16 might be involved in OA as an miRNA target gene. We further aimed to determine the mechanism of IL16 and miRNA in OA, which will provide new targets for OA treatment.
Materials and Methods
Cell Culture
Mouse chondrocyte (ADTC5) was purchased from KeyGEN (Jiangsu, China) and grown in a 1:1 mixture of DMEM and Ham’s F12 (Gibco, Massachusetts, United States) containing 5% fetal bovine serum (Gibco, Massachusetts, United States), 2 mM Glutamine (Gibco, Massachusetts, United States), 100 U/ml penicillin and 100 μg/ml streptomycin (Gibco, Massachusetts, United States) at 37 °C in an atmosphere with 5% CO2. ADTC5 cells were stimulated with IL1β (10 ng/ml) (Novoprotein, Suzhou, China) for 24 hours to mimic OA in vitro and then harvested for further studies. 12 Sprague Dawley (SD) rats (60-80 g) were purchased from the Laboratory Animal Center of Guangzhou University of Chinese Medicine (License number: SCXK 2018-0047, quality certificate number: 44005900002750). Animals were euthanized by injection of sodium pentobarbital (200 mg/kg body weight). Tibia and femur were isolated under sterile conditions, and the bone marrow was flushed out with DMEM basic containing 1% double antibody. Cells were collected through centrifugation at 1000 rpm for 10 minutes at room temperature. Bone marrow mesenchymal stem cells (BMSCs) were cultured with SD rat BMSCs basal medium (Cyagen, Shanghai, China) in an atmosphere of 5% CO2 at 37 °C. P3 and P4 generations of BMSCs can be used for subsequent experiments.
Cell Transfection
When cells population reached to 70% to 80% confluence, siIL16, siNC, novel-miR-81 mimic, mimic NC, novel-miR-81 inhibitor, and inhibitor NC were transfected into cells at a final concentration of 100 nmol by using the Lipofectamine™ 3000 (Invitrogen, Carlsbad, United States). siIL16 and siNC were purchased from Sangon Biotech (Shanghai, China). Novel-miR-81 mimic, mimic-NC, novel-miR-81 inhibitor, and inhibitor-NC were obtained from GenePharma (Suzhou, China). We found some new miRNAs that miRBase has not yet recorded by BGISEQ-500 sequencing technology (Beijing Genomics institution, Beijing, China), including novel-miR-81 (
siIL16-01 sense: GACAGCAUUUACGGCCCUAUUTT
siIL16-01 antisense: AAUAGGGCCGUAAAUGCUGUCTT
siIL16-02 sense: CGGUUCACAGAGUGUUUCCAATT
siIL16-02 antisense: UUGGAAACACUCUGUGAACCGTT
novel-miR-81 mature miRNA sequence: UUGGGGAUGGUAGGGUCUCUGAGG
novel-miR-81 mimic sense: UUGGGGAUGGUAGGGUCUCUGAGG
novel-miR-81 mimic antisense: UCAGAGACCCUACCAUCCCCAAUU
novel-miR-81 inhibitor: CCUCAGAGACCCUACCAUCCCCAA
siNC sense: UUCUCCGAACGUGUCACGUTT
siNC antisense: ACGUGACACGUUCGGAGAATT
mimic NC sense: UUCUCCGAACGUGUCACGUTT
mimic NC antisense: ACGUGACACGUUCGGAGAATT
inhibitor NC: CAGUACUUUUGUGUAGUACAA
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
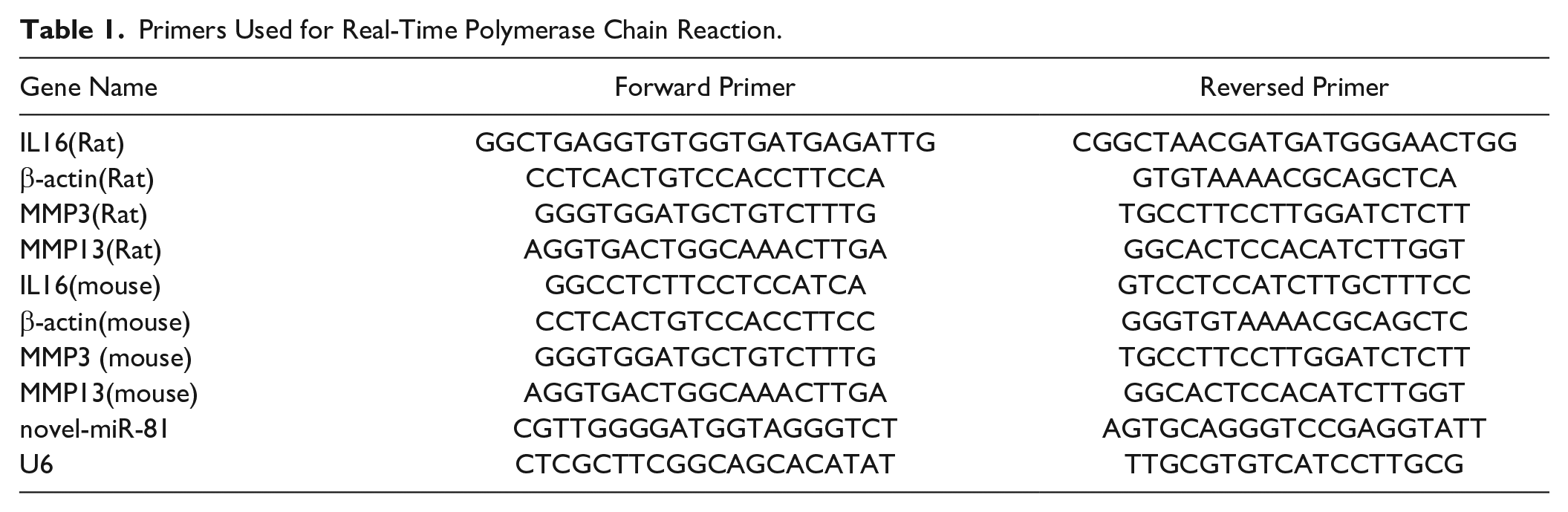
Total RNA was isolated from ADTC5 cells and cartilage tissue of the knee joint of SD rats using the EZ-press RNA Purification Kit (EZBioscience, Roseville, United States) according to the manufacturer’s instructions. The concentration and purity of RNA were tested using an ultraviolet spectrophotometer (Shimadzu Corporation, Kyoto, Japan) with a wavelength of 260/280 nm. Reverse transcription of RNA to complementary DNA by Color Reverse Transcription Kit (EZBioscience, Roseville, United States) and quantitative polymerase chain reaction (qPCR) was performed by using a 2x Color SYBR qPCR Master Mix (EZBioscience, Roseville, United States). The reverse transcription conditions were as follows: 37 °C for 15 minutes, then 85 °C for 5 seconds, and finally 4 °C cooling. The specific reaction conditions, using the CFX96 ™Real-Time PCR Detection System (Bio-Rad Laboratories, Shanghai, China) according to the user’s manual, were as follows: An initial step at 95 °C for 30 seconds, followed by 40 cycles at 95 °C for 5 seconds, 60 °C for 30 seconds and a final extension step at 60 °C for 30 seconds. The 2 -ΔΔCq method was used to analyze the qRT-PCR data. 13 U6 was used as an internal reference for the detection of novel-miR-81, and β-actin was used as an internal reference for IL16, MMP3, and MMP13. The primer sequences used in this study are shown in Table 1.
Primers Used for Real-Time Polymerase Chain Reaction.
Western Blot (WB)
The protein was extracted from ADTC5 cells by RIPA Lysate (Beyotime, Shanghai, China) containing protease inhibitor cocktail (Thermo Fisher Scientific, Massachusetts, United States) at 4 °C and protein concentrations were quantified by bicinchoninic acid (BCA) protein assay kit (Beyotime, Shanghai, China). Loading buffer was added into protein (CWBIO, Beijing, China) and boiled it for 5 minutes. The protein of ADTC5 cells (30 µg/lane) was separated by 10% or 12% SDS-PAGE (FUDE, Hangzhou, China) at 200V for 45 minutes, followed by wet transfer to polyvinylidene fluoride (PVDF) membranes (Merck MiLipore, Darmstadt, Germany) at 300 mA for 60 minutes. Membranes were blocked in Tris-buffered saline 0.01% Tween 20 (TBST) containing 5% nonfat milk for 2 hours. After the incubation with primary antibodies MMP3 (Abcam, 1:1000 dilution, Cambridge, UK), MMP13 (proteintech, 1:2000, Wuhan, China), and β-actin (Fude, 1:10000, Hangzhou, China) at 4°C for 12 hours, eluted them with TBST and incubated with a secondary antibody (Abcam, 1:10000 dilution, Cambridge, UK) for 1 hours. After washing, spots were imaged with Tanon 5200 Multi Chemiluminescent Imaging System and analyzed with AlphaView (NatureGene Corp, New Jersey, United States).
Dual-Luciferase Reporter Assay
BMSCs (2 × 104 cells/well) were seeded in 24-well plates. After culture for 24 hours, novel-miR-81mimic (100 nM) with plasmids of IL16 WT 3’UTR (100 ng) or MUT 3’UTR (100 ng), novel-miR-81inhibtor (100 nM) with plasmids of IL16 WT 3’UTR (100 ng) or MUT 3’UTR (100 ng) were co-transfected with Lipofectamine 3000 into cells (Thermo Fisher Scientific, Guangzhou, China) according to the manufacturer’s instructions. After 48 hours of transfection, firefly and Renilla luciferase activities were calculated by Dual-Luciferase Reporter Assay System Kit (Promega Corporation, Madison, United States).
Rats OA Model
All animal experiments were performed according to the guiding principles of the Laboratory Animal Centre of Guangzhou University of Chinese Medicine. SD rats (male; 200-220 g) were supplied by the Laboratory Animal Center of Guangzhou University of Chinese Medicine (license number: SCXK 2018-0034, quality certificate number: 44005800013457). All rats lived in the Animal Centre of Guangzhou University of Chinese Medicine (Guangzhou, China), with artificial lighting from 7 a.m. tantamount to 7 p.m., room temperature 22 °C and controlled humidity. All rats were healthy and had never received any medication during the experiment. Rats can continue to get a reproductive diet and water and were Specific Pathogen Free (SPF). All surgical procedures were performed under sterile conditions and general anesthesia, and all efforts were made to alleviate the suffering of rats. Sodium pentobarbital (Chemmart, Beijing, China) was used to anesthetize rats through the abdominal cavity. The medial collateral ligament was cut off, and the medial meniscus was removed. 14 The skin was sutured in layers, and the rats were free to move, eat, and drink after surgery.
Intra-Articular Injection of the Novel-miR-81 Agomir
The rats were randomly divided into 3 groups (n = 6 per group): control; meniscal/ligament injury (MLI) (model); MLI plus novel-miR-81 agomir. After 1 month of surgery, rats in control and model group were injected with 60 µl saline via joint cavity, and rats in model + novel-miR-81 agomir were injected with 60 µl novel-miR-81 agomir (10 µM). Injections were given once weekly into the MLI-operated knee for four consecutive weeks. Animals were euthanized by injection of sodium pentobarbital (200 mg/kg body weight) at the 9 weeks after surgery. Eighteen Rats had a total of 36 knees. Half of the knees were used for histochemical analysis, and the other half for RT-qPCR.
Hematoxylin and Eosin (HE) Staining
The joints of rats were fixed with 4% paraformaldehyde, embedded in paraffin, and sliced into 5 μm sections. HE staining was carried out according to the routing scheme. After deparaffinization and rehydration, sections were stained with hematoxylin solution for 5 minutes, differentiated with 0.5% hydrochloric acid ethanol solution, and rinsed in distilled water. The sections were subsequently stained with eosin solution for 15 minutes, dehydrated with gradient ethanol, and cleared in xylene. The tissues were photographed by a microscope.
Safranin O and Fast Green
The joints of rats were fixed with 4% paraformaldehyde, embedded in paraffin, and sliced into 5 μm sections. After deparaffinization and rehydration, sections were stained with Fast Green (Servicebio, Wuhan, China) for 1 to 5 minutes, differentiated with 1% hydrochloric acid ethanol solution, and rinsed in distilled water. The sections were subsequently stained with Safranin O (Servicebio, Wuhan, China) for 5 to 10 seconds, dehydrated with anhydrous ethanol, and cleared in xylene. The tissues were photographed by a microscope.
Immunohistochemistry (IHC)
The joints of rats were fixed with 4% paraformaldehyde, embedded in paraffin, and sliced into 5 μm sections. Tissue sections were dewaxed, hydrated, subjected to antigen retrieval, and exposed to antigenic sites according to conventional methods. The sections were incubated for 15 minutes in 0.03% Triton X-100. Endogenous tissue peroxidase activity was blocked with 3% hydrogen peroxide for 20 minutes. After incubation with goat serum for 30 minutes to block non-specific binding, the sections were incubated with primary antibody at 4 °C overnight, including MMP13 (Abcam, 1:500 dilution, Cambridge, UK), Collagen II (Abcam, 1:250 dilution, Cambridge, UK). The sections were incubated with secondary antibody (Histostain TM-Plus Kits, Biotechnology, Beijing, China) for 30 minutes and then the horseradish-labeled streptomycin for 30 minutes. The color was interpreted with DAB (Biosynthesis, Beijing, China) for 20 minutes. The sections were added dropwise with hematoxylin for 1 minute, dehydrated with a gradient of ethanol, and clarified in xylene. The tissues were photographed by a microscope.
Statistical Analysis
Data were reported as means ± standard deviation (SD). Statistical analysis was performed by unpaired 2-tailed Student’s t test and 1-way analysis of variance (ANOVA) by GraphPad Prism 7.0 software (GraphPad Software, Inc.). Results with P < 0.05 were considered to indicate a statistically significant difference.
Results
IL16 Was Upregulated in Rats OA Model
To identify the expression level of IL16 in OA, we performed qRT-PCR analysis on cartilage tissues from rats OA model and healthy controls. Expression of IL16 has been shown to be significantly increased in cartilage tissues of rats OA model compared with normal cartilage tissue ( Fig. 1A ).
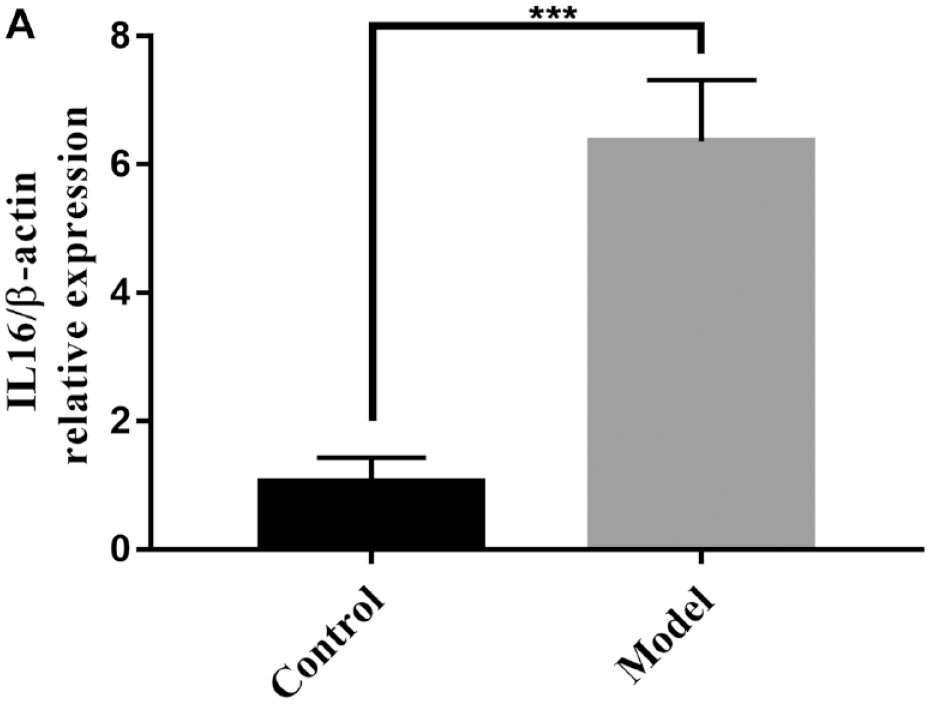
Expression of IL16 in rats osteoarthritis model. (
IL16 Was Upregulated in ADTC5 Cells OA Model
IL1β and ADTC5 cells were used to mimic OA under in vitro cell culture conditions. ADTC5 cells were treated with 10 ng/mL IL1β for 24 hours. The mRNA and protein levels of MMP3 and MMP13 were strongly upregulated by IL1β treatment ( Fig. 2A-C ). Based on these results, we confirmed the successful mimicking of the OA environment in vitro. The expression levels of IL16 were increased upon treatment with IL1β by qRT-PCR ( Fig. 2D ).
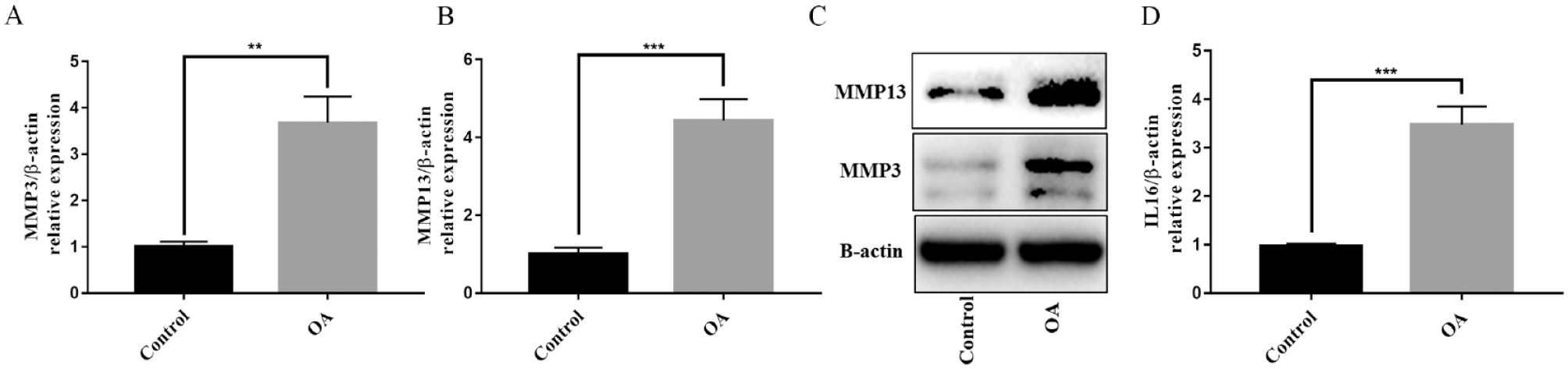
Expression of IL16 in ADTC5 cells OA model. (
SiIL16 Protected ADTC5 Cells from IL1β-Induced Injury in ADTC5 Cells OA Model
We transfected siIL16 or siNC into ADTC5 cells under IL1β stimulation to investigate the possible effects of IL16 on IL1β-induced chondrocyte injury in vitro. qRT-PCR analysis showed that the expression of IL16 mRNA was obviously downregulated after siIL16-01 transfection into IL1β-stimulated ADTC5 cells ( Fig. 3A ). Therefore, siIL16-01 was used as the follow-up research object. In IL1β-stimulated ADTC5 cells, we also found that siIL16 downregulated the mRNA and protein expression of MMP3 and MMP13 ( Fig. 3B-D ).
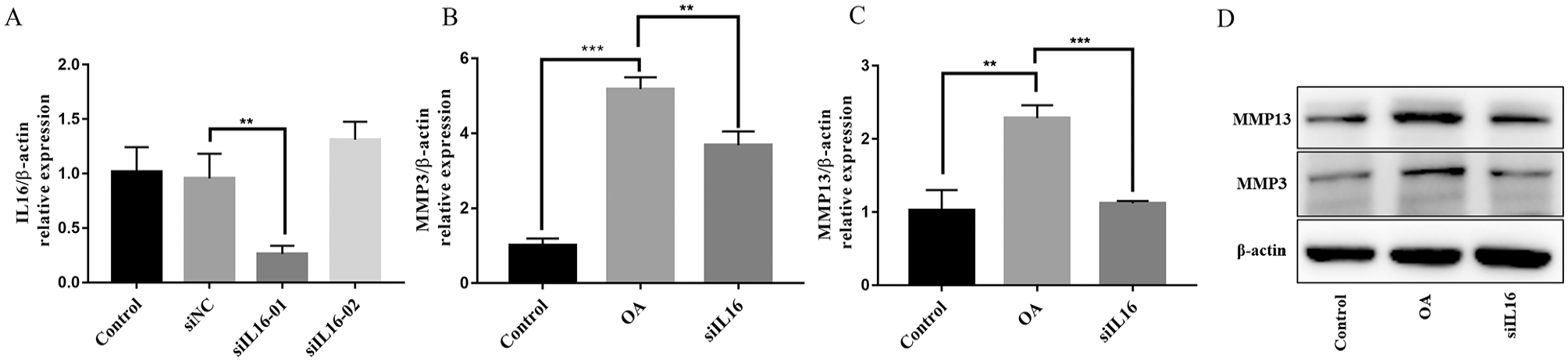
siIL16 significantly reversed IL1β-induced cartilage matrix degradation in vitro. (
IL16 Was a Target Gene of Novel-miR-81 In Vitro
IL16 was predicted to be the potential target of novel-miR-81 by bioinformatics tools.
15
We conducted gene ontology (GO) analysis of the target genes of novel-miR-81 and found that the biological process of novel-miR-81 was mainly enriched in immune response (
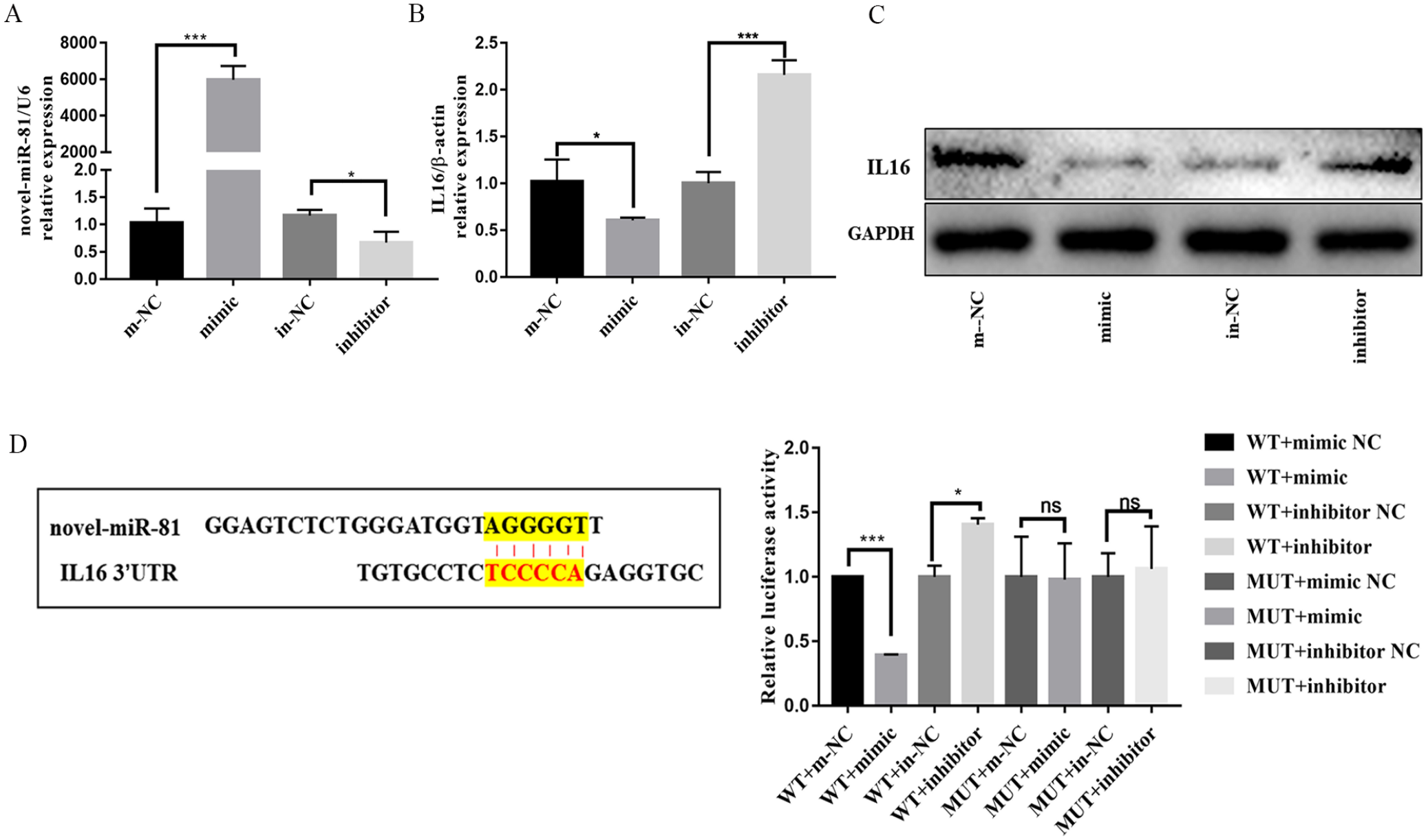
IL16 is a potential target of novel-miR-81. (
Novel-miR-81 overexpression inhibited MMP3 and MMP13 expression in ADTC5 cells OA model
The expression of novel-miR-81 was decreased after treated with IL1β ( Fig. 5A ). MMP3 and MMP13 protein expression were reduced to varying degrees in the novel-miR-81 mimic group compared with OA group. Taken together, novel-miR-81 mimic reversed IL1β-induced degradation of chondrocyte cartilage matrix ( Fig. 5B ).
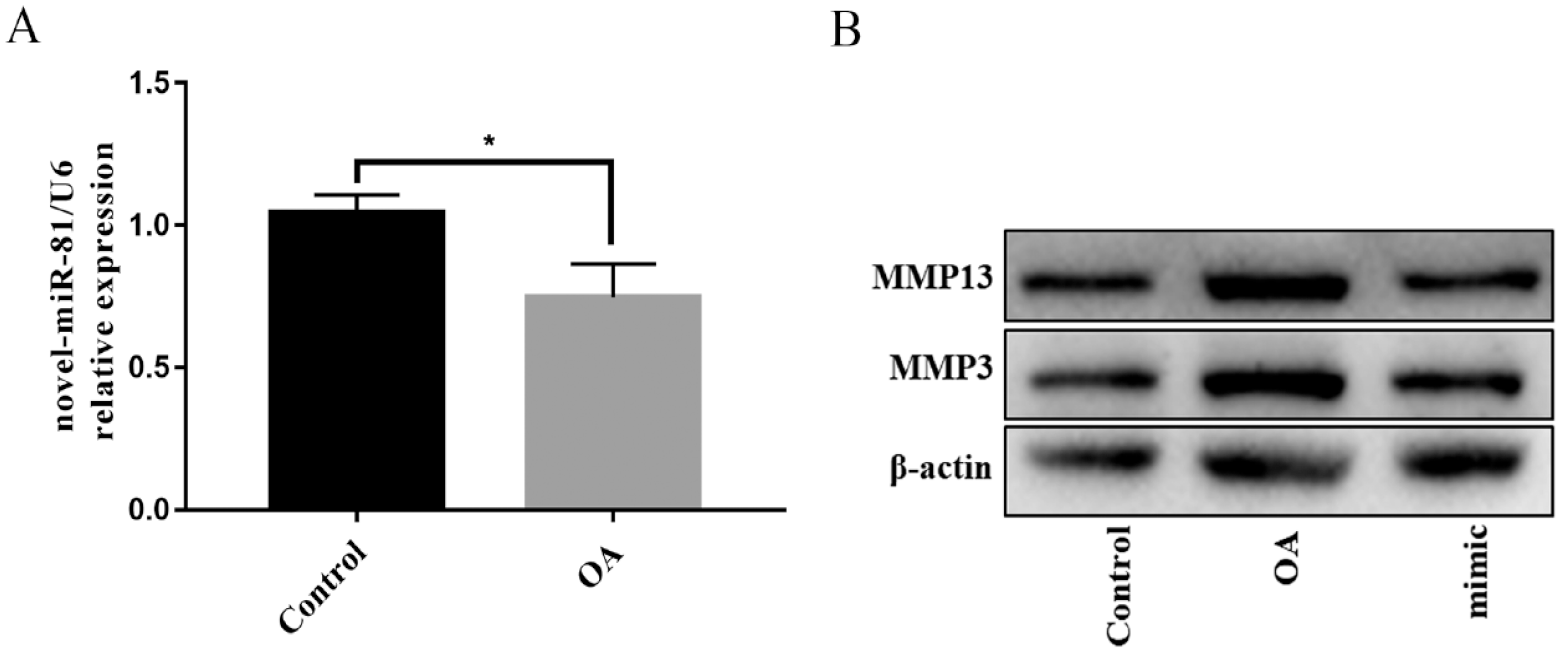
Up-regulation of novel-miR-81 significantly reversed IL1β-induced cartilage matrix degradation. (
Intra-Articular Injection of Novel-miR-81 Agomir Attenuated the Progression of Rats OA In Vivo
To confirm the effects of intra-articular injection of novel-miR-81 agomir on OA, we performed HE staining, Safranin O and Fast Green staining and IHC staining ( Fig. 6A ). HE staining showed that the model group had significant cartilage damage compared with the control group, while novel-miR-81 agomir had a repair effect on cartilage damage ( Fig. 6A ). The OA cartilage damaged area exhibited significant proteoglycan loss as shown in Safranin O and Fast Green. Safranin O and Fast Green showed that novel-miR-81 agomir had a higher level of cartilage matrix than the model group ( Fig. 6A ). IHC staining showed that MMP13 positive cells were significantly decreased in novel-miR-81 agomir group compared with model group, and Collagen II positive cells were significantly increased in novel-miR-81 agomir group compared with model group ( Fig. 6A ). Novel-miR-81 agomir markedly abolished cartilage destruction, inflammatory infiltration and articular chondrocyte cellularity loss in rats OA model. The expression of novel-miR-81 was significantly increased in the novel-miR-81 agomir group ( Fig. 6B ). The expression of MMP3 and MMP13 were significantly decreased in the novel-miR-81 agomir group compared with the model group by qRT-PCR ( Fig. 6C and D ). Novel-miR-81 agomir may significantly inhibit the expression of IL16 ( Fig. 6E ). These results indicated that intra-articular injection of novel-miR-81 could partially attenuate OA progression in rats.
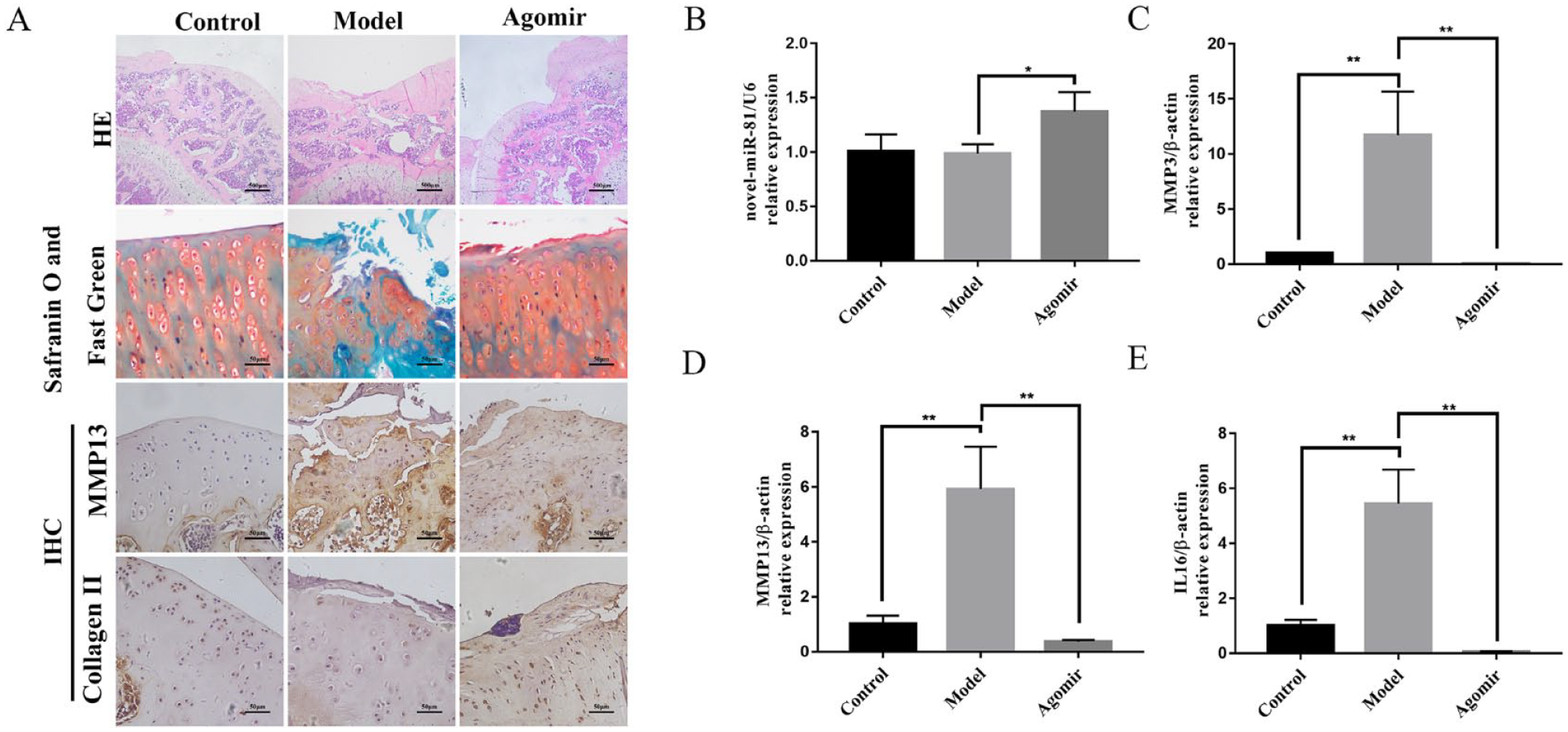
Novel-miR-81 alleviates OA in vivo. (
Discussion
IL16 plays an important biological role in the immune process of the body. 16 The synovial fluid of patients with internal rheumatoid arthritis was increased compared with the patients without internal rheumatoid arthritis, and the expression of IL16 was significantly increased. 17 MMP3 directly destroyed joints in patients with internal rheumatic joints by degrading cartilage and bone. 18 The serum proteomic analysis, suggested that IL16 is the most MMP3-related cytokine and IL16 could be used as an evaluation index for early treatment of patients. 18 Studies have also shown IL16 gene polymorphisms to be associated with the risk of knee OA.9,10 IL16 rs11556218 polymorphism, representing an Asn to Lys substitution in exon 6 of the IL16 gene, has a significant effect on the risk of knee OA; individuals carrying the rs11556218 G allele had a significantly decreased risk of developing knee OA compared with those carrying the T allele. 10 However, it is unknown whether siIL16 can attenuate the progression of OA. In this study, IL16 was upregulated in cartilage tissues of rats OA model. In ADTC5 cell transfected with siIL16, the expression of IL16, MMP3, and MMP13 was significantly reduced. siIL16 protected ADTC5 cells from IL1β-induced injury in vitro.
miRNAs regulate gene expression by guiding Argonaute (AGO) proteins to target sites in the 3’ untranslated region (UTR) of mRNAs. 19 IL16 was identified as a target of novel-miR-81 by using bioinformatics. This target association was further confirmed by constructing luciferase reporter genes containing IL16 WT 3’UTR and MUT 3’UTR. In addition, the mRNA expression of IL16 was significantly downregulated by novel-miR-81 mimic and upregulated by novel-miR-81 inhibitor, as revealed by qRT-PCR. miRNAs have biological functions of regulating chondrocyte apoptosis and proliferation, extracellular matrix metabolism, and inflammatory responses, and then play an important role in the pathogenesis of OA.20 -22 In this study, overexpression of novel-miR-81 attenuates inflammatory responses in ADTC5 cells OA model.
Intra-articular injection of drugs, such as Corticosteroids and Hyaluronic acid compounds, is a widely accepted treatment for OA, 3 and intra-articular injection of miRNAs (such as miR-140, 23 miR-214-3p, 11 miR-34a-5P, 24 miR-144-3p, 25 etc.) has been used as a treatment for OA in animal models in recent years. However, no study has reported the intra-articular injection of novel-miR-81 as a treatment for OA. Therefore, we utilized a surgically induced OA rats to test the hypothesis that novel-miR-81 could attenuate OA progression by reducing the expression of inflammatory factors. Intra-articular injection was an effective means of administering novel-miR-81 in OA rats. Novel-miR-81 expression in cartilage tissue was significantly higher in the novel-miR-81 agomir group than control group and model group. Moreover, Collagen II expression levels in cartilage tissue were significantly higher, while MMP13 expression was significantly lower in the novel-miR-81 agomir group than in the model group. The above suggests that novel-miR-81 is expected to be a new target for OA treatment.
This study has some limitations. Although the classical MLI model of posttraumatic OA was used, 14 it is different from the clinical development process of OA. OA is associated with gender, aging, obesity, immune response, and other factors. 26 Another limitation of this study is that the sample size is not estimated by power analysis in the experimental design. Although this study showed difference significantly with a small sample size, performing power analysis in the experimental design would make the whole experiment more rigorous.
Conclusion
Novel-miR-81 could reduce inflammatory response of OA by inhibiting IL16. Novel-miR-81/IL16 may be a potential therapeutic molecular target for OA.
Supplemental Material
sj-png-1-car-10.1177_19476035231168387 – Supplemental material for IL16 Regulates Osteoarthritis Progression as a Target Gene of Novel-miR-81
Supplemental material, sj-png-1-car-10.1177_19476035231168387 for IL16 Regulates Osteoarthritis Progression as a Target Gene of Novel-miR-81 by Ziwei Luo, Qianting Han, Jianghua Lu, Xiyan Ouyang, Yueying Fan, Yangping Liu, Xianxi Zhou, Jiechen Kong, Helu Liu, Aijun Liu and Dongfeng Chen in CARTILAGE
Supplemental Material
sj-png-2-car-10.1177_19476035231168387 – Supplemental material for IL16 Regulates Osteoarthritis Progression as a Target Gene of Novel-miR-81
Supplemental material, sj-png-2-car-10.1177_19476035231168387 for IL16 Regulates Osteoarthritis Progression as a Target Gene of Novel-miR-81 by Ziwei Luo, Qianting Han, Jianghua Lu, Xiyan Ouyang, Yueying Fan, Yangping Liu, Xianxi Zhou, Jiechen Kong, Helu Liu, Aijun Liu and Dongfeng Chen in CARTILAGE
Footnotes
Acknowledgments and Funding
Thanks for the Guangzhou Chinese Medical University for providing the experimental place and equipment and also thanks for all the participating researchers for their hard work. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangdong Province (2017A030312009), National Natural Science Foundation of China (82174368), Bao’an TCM Development Foundation(2020KJCX-KTYJ-130, 2020KJCX-KTYJ-110), Shenzhen Bao’an Science and Technique Project (No. 2020JD569), and Sanming Project of Medicine in Shenzhen (No. SZZYSM202106009).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal studies were performed in Laboratory Animal Centre of Guangzhou University of Chinese Medicine, according to protocols approved by the Institutional Animal Care and Use Committee of Guangzhou Chinese Medical University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
