Abstract
Objective
In the present in vitro study, we analyzed the chondrogenic differentiation capacity of human chondrocytes postmortally isolated from unaffected knee cartilage by the addition of transforming growth factor–β1 (TGF-β1) and/or insulin-like growth factor–1 (IGF-1) and different oxygen levels.
Design
After 14 and 35 days, DNA concentrations and protein contents of Col1, Col2, aggrecan as well as glycosaminoglycans (GAGs) of chondrocytes cultivated as pellet cultures were analyzed. Additionally, expression rates of mesenchymal stem cell (MSC)–associated differentiation markers were assessed in monolayer cultures.
Results
All cultivated chondrocytes were found to be CD29+/CD44+/CD105+/CD166+. Chondrocytic pellets stimulated with TGF-β1 showed enhanced synthesis rates of hyaline cartilage markers and reduced expression of the non-hyaline cartilage marker Col1 under hypoxic culture conditions.
Conclusions
Our results underline the substantial chondrogenic potential of human chondrocytes postmortally isolated from unaffected articular knee cartilage especially in case of TGF-β1 administration.
Introduction
For the treatment of cartilage defects, several therapeutic techniques were established. Besides Pridie drilling, microfracturing or osteochondral autograft transplantation system (OATS), autologous chondrocyte transplantation (ACT), introduced by Brittberg et al. 1 in 1994, are promising approaches for cartilage repair. A prerequisite for ACT is a high number of chondrocytic cells isolated from the patient in a first operation. In a second operation, these cells are transplanted to fill up the cartilage defect. 1 Apart from the need of a second operation steps, a further disadvantage is the de-differentiation of chondrocytic cells during in vitro expansion. 2 To support the re-differentiation process of cells into the chondrocytic phenotype, a 3-dimensional (3D) environment is favourable.3,4 Therefore, novel techniques were established, applying 3D spheroid cell cultures (also named as pellet cultures) into the cartilage defect.5-7 The main advantages of these spheroid cultures are cell-to-cell interactions that are similar to those present during the precartilage condensation step in embryonic development 8 as well as the production of natural extracellular matrix (ECM) by autologous cells.
In order to overcome the named disadvantages associated with ACT and to find an alternative cell source for cartilage repair, several in vitro studies about the chondrogenic potential of mesenchymal stem cells (MSCs) from adipose tissue or bone marrow have been carried out in pellet cultures in recent years.9-12 Although these cells are able to differentiate into the chondrogenic (osteogenic and adipogenic) lineage, the potential for forming hyaline-like tissue is limited by hypertrophy.13,14 In contrast, MSCs from synovial tissue showed an increased chondrogenic potential and a reduced hypertrophic differentiation than bone marrow–derived MSCs.15,16 These findings suggest that the production and differentiation of chondrocytic cells is more efficient when cells with a phenotype close to that of hyaline cartilage are used. 17 In addition, it is beneficial to analyze the impact of chondrogenic growth factors that play an important role for cartilage homeostasis by stimulating significant anabolic activities. 18
In search of an appropriate alternative allogenic cell source for cartilage repair, we evaluated the proliferation and differentiation potential of human chondrocytes postmortally isolated from unaffected hyaline knee cartilage. Although this cell source could have several advantages for cartilage repair techniques like ACT (similar phenotype of cells, one operation procedure, “healthy” cells), there is only a limited number of in vitro and in vivo studies dealing with this cell type.19-22
In our present study, we therefore analyzed the effect of transforming growth factor–β1 (TGF-β1), insulin-like growth factor–1 (IGF-1), and the combination of both on autonomous cell condensation to pellet cultures and the composition of the extracellular matrix produced by these cells. In previous studies, we could show that the addition of these growth factors was able to influence chondrogenic re-differentiation in 3D cultures of alginate, spheroid and matrix-associated cultures.23-25 In order to simulate physiological conditions, we examined the effect of reduced oxygen levels on the chondrogenic differentiation capacity.
Materials and Methods
Chondrocyte Isolation and Cultivation
Hyaline cartilage from knee joint was obtained postmortally from a total of 5 donors (2 males, mean age 31 ± 14 years; 3 females, mean age 32 ± 16 years) within the first 72 hours of donor’s death. The patient anamnesis and cause of death were unknown. All tissues did not show any signs of osteoarthritis or degeneration, which was examined macroscopically. The study was approved by the Local Ethical Committee (registration no. A2011-138).
For isolation of primary chondrocytes, the protocol of Jonitz et al. 23 was used. The primary chondrocytes were devided into 2 cell populations, one was cultivated at 21% oxygen level (normoxia) and the other at 5% oxygen level (hypoxia) for 2 passages. Then, cells were frozen and stored in liquid nitrogen. For all experiments, cells were thawed, centrifuged at 118 × g for 10 minutes, and incubated in a humidified atmosphere at 37°C, 5% CO2. The 2 populations that were created initially were maintained for the entire experiment under the respective oxygen partial pressure at 21% or 5%.
Flow Cytometry
Flow cytometry was performed on human chondrocytes cultured as monolayers. After cell harvesting and preparing a single cell suspension of 105 cells per 500 µL phosphate buffered saline (PBS; PAA, Coelbe, Germany) containing 1% bovine serum albumin (BSA; Sigma, Selze, Germany), cells were incubated in the dark for 30 minutes with fluorescein isothiocyanate (FITC)-conjugated CD166 antibody, phycoerythricin-conjugated CD44 and CD105 antibodies and allophycocyanine-conjugated CD29 antibody. Mouse isotype controls IgG1 and IgG2 labeled with FITC served as negative controls (all antibodies purchased from Acris Antibody, Herford, Germany). The cells were washed 3 times in PBS/BSA (1%), centrifuged and afterward resuspended in PBS/BSA (1%). Immediatly after antibody staining, flow cytometry was done on a FACS Calibur flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA) and data analysis was performed using the CellQuest software (Becton Dickinson). To determine the portion of cells expressing the respective CD antigen the ratio of the median of fluorescence intensity (MFI) of stained to unstained cells of each cell population was calculated.
Production of 3-Dimensional Pellet Cultures
In passage 3, chondrocyte pellet cultures with an amount of 5 × 105 cells per pellet were prepared. Therefore, cells (incubated at the respective oxygen level) were trypsinized and centrifuged at 118 × g for 10 minutes. Appropriate aliquots of cells were divided for incubation in 3 different medium compositions (medium 1, TGF-β1 and IGF-1; medium 2, TGF-β1; medium 3, IGF-1). Afterwards, the cell suspension was transferred into a well of a 96-well suspension cell culture plate. The different media consisted of serum-free Dulbecco’s modified Eagle medium (DMEM; Invitrogen, Darmstadt, Germany), ascorbic acid (final concentration 50 µg/mL), dexamethasone (final concentration 100 nM; Sigma-Aldrich, Munich, Germany), and ITS (complete medium to ITS in a 100:1 ratio; Becton Dickinson, Heidelberg, Germany). The concentrations of the chondrogenic growth factors were 50 ng/mL for IGF-1 (RD Systems, Wiesbaden, Germany) and TGF-β1 (tebu-bio, Offenbach, Germany), respectively. Pellet cultures were incubated at 37°C in a humidified atmosphere of 5% CO2 and 21% or 5% oxygen for 5 weeks. The culture medium was changed every second day.
DNA Isolation and Quantification
For DNA isolation, one pellet culture of each stimulating experiment was transferred in 2 mL homogenization tubes containing small steel beads (Precellys-Steel Kit, 2.8 mm; Peqlab, Erlangen, Germany) and covered with 100 µL TE-buffer. Afterward, pellets were homogenized for 30 seconds at 5000 rpm using the Minilys homogenisator (Peqlab).
DNA isolation was performed using the peqGOLD Tissue DNA Mini Kit (Peqlab) according to the manufacturer’s instructions. Therefore, samples were lysed under denaturing conditions and applied on PerfectBind DNA columns to reversibly bind DNA. After washing away cellular debris and other proteins, the DNA was eluted in Aqua dest. Finally, DNA concentrations were measured with the Qubit Fluorometer according to the manufacturer’s recommandations (Invitrogen).
Quantification of ECM Components
Protein amounts of collagen type 1 (C1CP, Quidel, Marburg, Germany) and type 2 (CP2, IBEX Pharmaceuticals, Québec, Canada) as wells as aggrecan (Acan; PG-EASIA, Invitrogen) synthesized by chondrocytic pellet cultures were analyzed using enzyme-linked immunosorbent assays (ELISAs). Both collagen ELISAs are based on the cleavage of the carboxy propeptides of newly formed collagen, which were measured in the supernatant of pellet cultures. The Acan ELISA is based on the use of an oligoclonal system where monoclonal antibodies are directed against specific epitopes of proteoglycan.
For ELISA performing, supernatants of the respective pellet cultures were collected after 14 and 35 days of incubation and stored at −20°C. All assays were conducted according to manufacturer’s instructions. Absorbances were measured at 450 nm for CPII and Acan and at 405 nm for C1CP using the Opsys MR microplate reader (Dynex Technologies, Denkendorf, Germany). Afterward, protein contents were normalized to DNA content (µg protein/µg DNA).
Blyscan Glycosaminoglycans Assay
The Blyscan assay (Biocolor Pharmaceuticals Inc., Carrickfergus, UK) is a quantitative dye-binding method to measure sulfated proteoglycans and glycosaminoglycans. After 14 and 35 days, supernatants from pellet cultures of each medium combination were collected and stored at −20°C. Subsequently, a papain digestion was done overnight at 60°C. Therefore, 150 µL of supernatant was filled up to 1 mL with 50 mM phosphate buffer (1 M NaCl, 5 mM cystein-HCl, 1 mM EDTA (all Merck, Darmstadt, Germany); pH 6.8) and treated with 20 units/mg papain (from papaya latex, Sigma-Aldrich). The Blyscan assay was performed according to the manufacturer’s instructions. Absorbance was measured at 656 nm using the Opsys MR microplate reader (Dynex Technologies). The contents of glycosaminoglycan (GAG) were normalized to the DNA content (µg GAG/µg DNA).
Data Illustration and Statistical Analysis
All metrical data were subjected to normalization. This means, for each donor and culture condition (growth factor treatment under the respective oxygen level), all data were collected to determine the minimum and maximum value. Afterward, the position of a distinct culture condition was calculated within these extremes (minimum, maximum) in order to scale all values to a range from zero (lowest value measured) to one (highest value measured). Therefore, donor-specific differences in absolute values are left out of consideration. Data in figures are presented as box plots. Boxes denote interquartile ranges, horizontal lines within the boxes denote medians, and whiskers denote minimum and maximum values. For all analyses, cultures of human chondrocytes of a minimum of three independent donors were used. Since the data obtained were not normally distributed, the statistical significance between 2 datasets was calculated with the Mann-Whitney U test using SPSS 20 (IBM Deutschland, Ehningen, Germany). The level of significance was set to a P value less than or equal to 0.05.
Results
Expression of MSC-Associated Markers
MSC-associated marker expression was highly uniform between the different donors. All postmortally isolated chondrocytes were found to be CD29+/CD44+/CD105+/CD166+ and there were no significant differences in marker expression between hypoxic and normoxic condition ( Fig. 1 ). Lower expression levels were found for CD29, CD105, and CD166 whereas CD44 was highly expressed in all monolayer cultures ( Fig. 1 ).
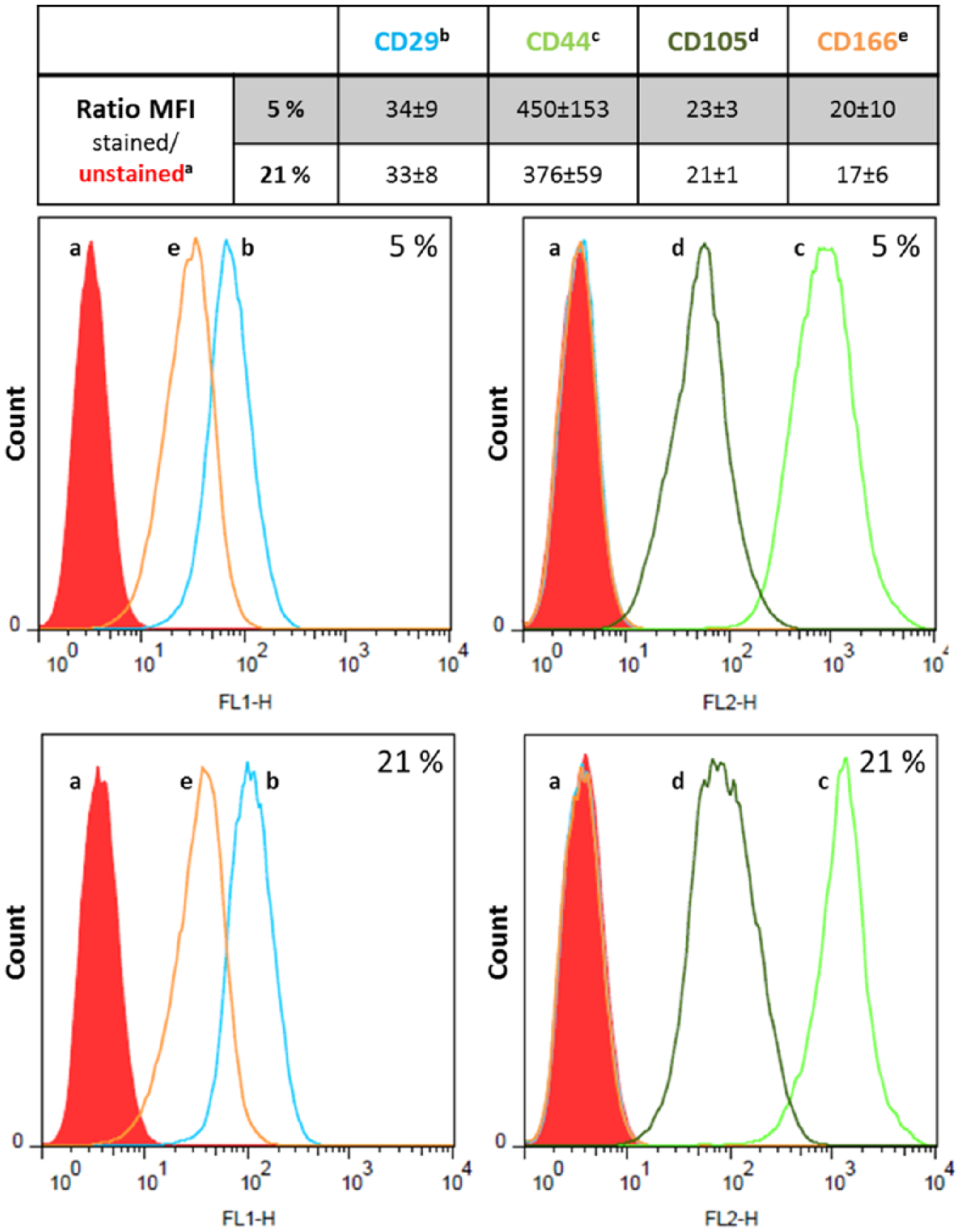
Expression rate of the mesenchymal stem cell (MSC)–associated markers CD29, CD44, CD105, and CD166 on chondrocytic cells in monolayer culture under hypoxia and normoxia. Postmortally isolated human chondrocytes were cultivated over 4 passages in monolayer cultures under the respective oxygen concentration. Afterward, aliquots of 1 × 105 cells were stained with fluorescence dye-labeled antibodies of CD29, CD44, CD105, and CD166. The respective CD expression rate was determined by calculating the ratio of the median of fluorescence intensity (MFI) of stained cells to unstained ones (n = 3, mean ± SEM). No statistical significance was provable (Mann-Whitney U test).
Pellet Formation and Proliferation of Human Chondrocytes in Suspension Plates
The tested growth factors supported the formation of 3D pellet cultures in suspension plates within 48 hours. After 5 weeks of incubation, cell pellets with a hyaline-like appearance were visible ( Fig. 2 ). In order to analyze proliferation of cells in pellet cultures, we determined the DNA content after 2 and 5 weeks of incubation ( Fig. 3 ). Under hypoxia, we observed higher DNA contents in IGF-1 treated pellets after 14 days compared with the single (significant difference, P = 0.029) and combined stimulation with TGF-β1 ( Fig. 3a ). This was in contrast to normoxic-cultivated cultures where similar DNA contents were determined after all 3 different growth factor treatments ( Fig. 3b ). Moreover, DNA contents of TGF-β1 and IGF-1-stimulated pellets after 14 days of hypoxia were significantly (P = 0.032) lower compared with normoxia. After 35 days of incubation, an increase of DNA amount was found for the combined growth factor treatment under hypoxia whereas a slight decrease was shown for both single growth factor treatments. Under normoxia, a noticeable decrease between 14 and 35 days in all stimulation groups were measured, which was significant for TGF-β1 and IGF-1-stimulated pellets (P = 0.032). Additionally, IGF-1 treatment led to significantly reduced DNA amounts (P = 0.029) under normoxia compared with hypoxic culture conditions. Apart from these findings, similar proliferation rates were observed for all growth factor supplementations after 35 days under the respective oxygen environement.
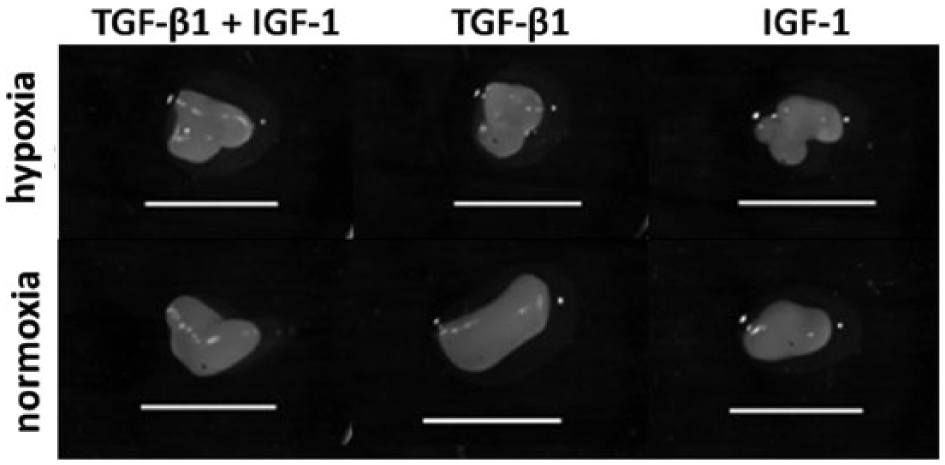
Morphology of chondrocytic pellet cultures after five weeks of incubation under hypoxia and normoxia. Chondrocytes were cultivated under different medium conditions in 96-well suspension plates where they organised themselves into pellets. Bar: 2.5 mm.
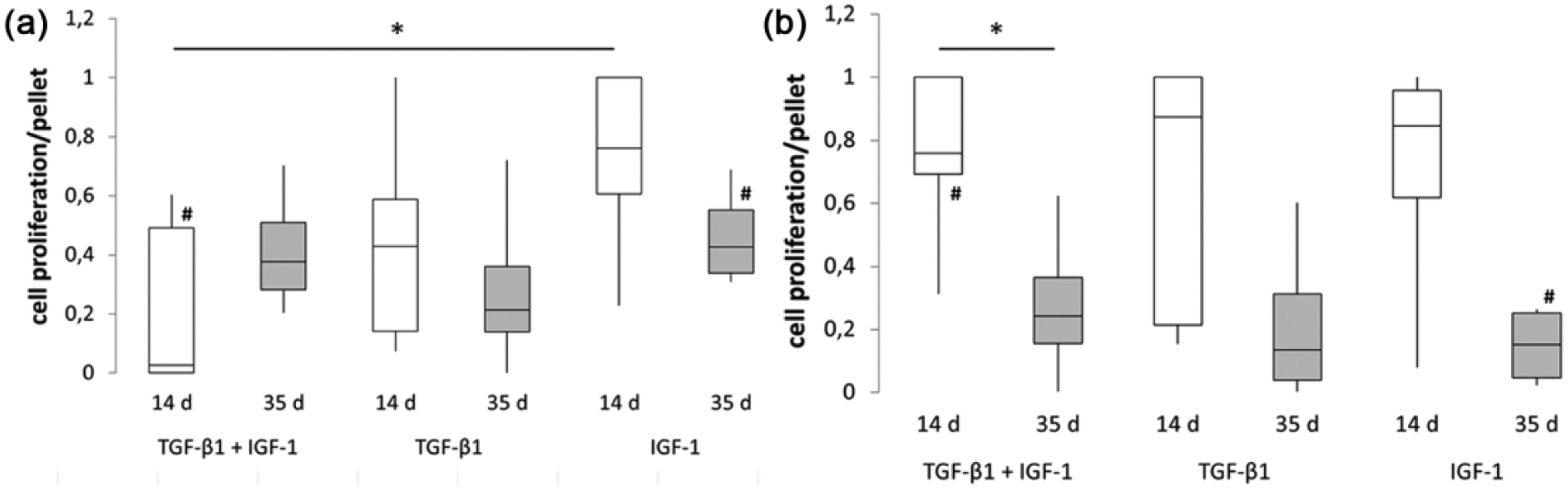
Proliferation of chondrocytic cells in pellet cultures after 2 and 5 weeks of incubation under hypoxia (
Expression of ECM Components
To determine the composition of the ECM of chondrocytic pellet cultures, the contents of collagen type 1 (C1CP) and type 2 (CP2) as well as aggrecan (Acan) and GAGs after 2 and 5 weeks of incubation were analyzed ( Table 1 ).
Synthesis Rates of Extracellular Matrix ECM Components by Chondrocytic Cells in Pellet Cultures After 14 and 35 Days of Incubation under Hypoxia (5%) and Normoxia (21%).
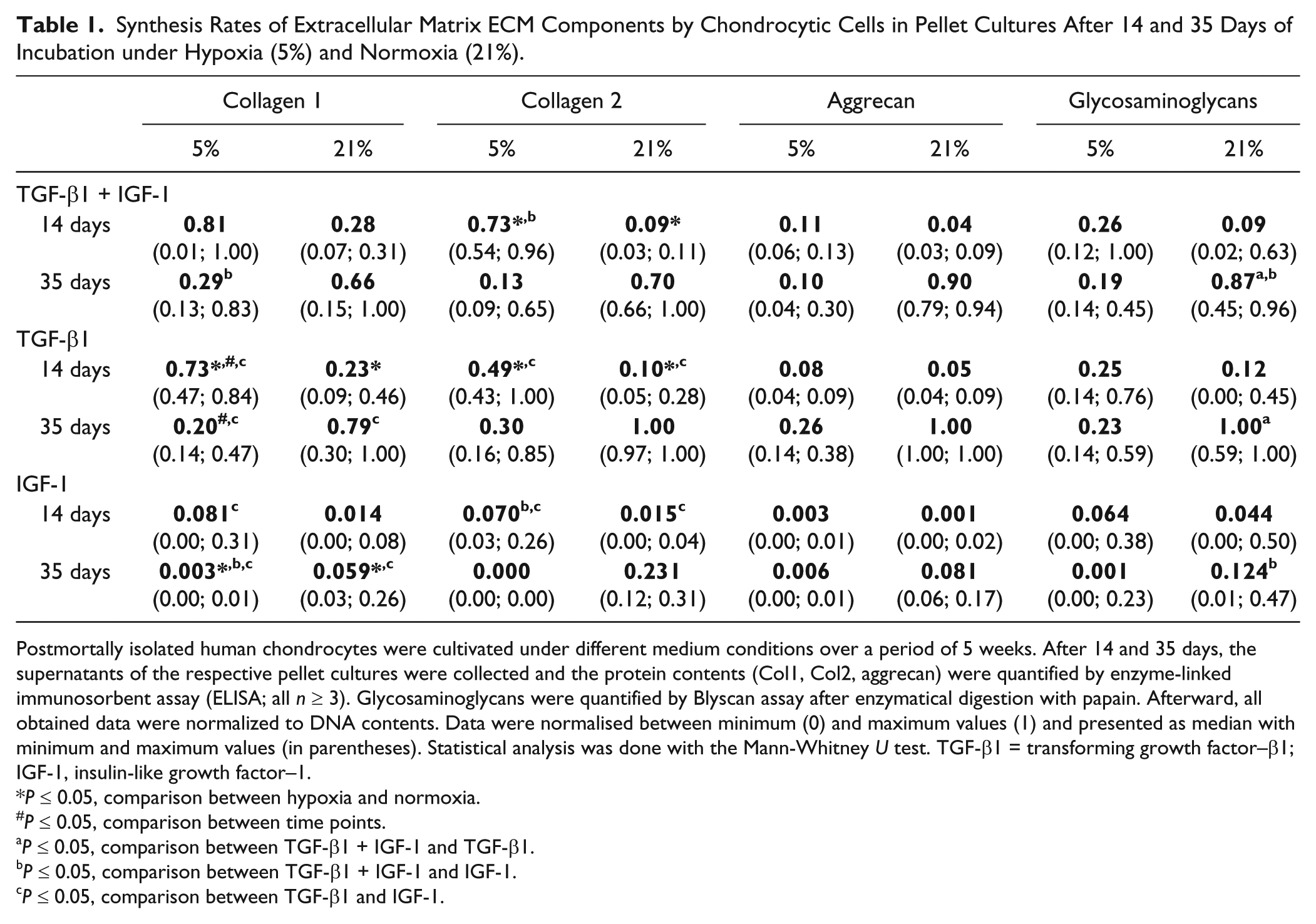
Postmortally isolated human chondrocytes were cultivated under different medium conditions over a period of 5 weeks. After 14 and 35 days, the supernatants of the respective pellet cultures were collected and the protein contents (Col1, Col2, aggrecan) were quantified by enzyme-linked immunosorbent assay (ELISA; all n ≥ 3). Glycosaminoglycans were quantified by Blyscan assay after enzymatical digestion with papain. Afterward, all obtained data were normalized to DNA contents. Data were normalised between minimum (0) and maximum values (1) and presented as median with minimum and maximum values (in parentheses). Statistical analysis was done with the Mann-Whitney U test. TGF-β1 = transforming growth factor–β1; IGF-1, insulin-like growth factor–1.
P ≤ 0.05, comparison between hypoxia and normoxia.
P ≤ 0.05, comparison between time points.
P ≤ 0.05, comparison between TGF-β1 + IGF-1 and TGF-β1.
P ≤ 0.05, comparison between TGF-β1 + IGF-1 and IGF-1.
P ≤ 0.05, comparison between TGF-β1 and IGF-1.
Under hypoxia, synthesis of C1CP was initially high after stimulation with TGF-β1 and IGF-1 as well as TGF-β1. After 35 days of stimulation, a reduction in protein content was found, which was significant for treatment with TGF-β1 (P = 0.029). Additionally, significant differences were determined between the combined IGF-1 and TGF-β1 stimulation after 35 days (p = 0.029) as well the single treatments with TGF-β1 or IGF-1 after 14 (P = 0.029) and 35 days (P = 0.029). Under normoxic culture conditions, lower C1CP contents were determined after stimulation with the 3 growth factor combinations compared with hypoxia after 14 days. In contrast, a time-dependent increase of C1CP was found for all stimulation groups under normoxia. A significant difference between the single treatment with TGF-β1 as well as IGF-1 was measured. Under both oxygen partial pressures, IGF-1-treated pellet cultures showed the lowest C1CP contents, with significantly lower values (P = 0.029) under hypoxia compared with normoxia after 35 days of culture.
Under hypoxic culture conditions, significant higher amounts of CP2 were found after combined stimulation with TGF-β1 and IGF-1 (P = 0.029) and TGF-β1 only (P = 0.029) compared with IGF-1 treatment after 14 days. We found comparable results under normoxia. After 35 days of incubation, a reduction in protein content was found under hypoxic culture conditions, while it was increased under normoxia. The single treatment with IGF-1 resulted in the lowest protein amount. Comparable to hypoxia, a significant difference between the single TGF-β1and IGF-1-treatments was found under normoxia (P = 0.029).
After 14 days of stimulation, aggrecan protein expression was low for all growth factor combinations and under both oxygen levels. Under hypoxia, a time-dependent increase of aggrecan content was determined after stimulation with TGF-β1 only, whereas under normoxia, a distinct rise was visible under any growth factor stimulation.
As was true for aggrecan synthesis rates under hypoxia, sulfated proteoglycans, and GAGs were also synthesized at rather constant rates during the stimulation period. Under normoxia, a time-dependent increase of synthesis rates for GAGs were found under the 3 growth factor stimulation conditions. As found under hypoxia, IGF-1 treatment led to the lowest protein contents, while the highest content was found for TGF-β1-treated cells after 35 days of stimulation.
Discussion
The aim of the present study was to evaluate the ability for pellet formation and the chondrogenic differentiation potential of chondrocytic cells, which were postmortally isolated from healthy cartilage, and represent a potential allogenic cell source for cartilage regeneration. The isolation of viable chondrocytic cells was possible within 72 hours after the donor’s death and the expression of the MSC-associated markers CD29+/CD44+/CD105+/CD166+ indicates the de-differentiation of cells to a chondroprogenitor status. Moreover, cells expressing CD29, CD105, and CD166 are known to have the capacity for chondrogenic differentiation. 23 The re-differentiation potential of de-differentiated cells derived from hyaline cartilage was comparable to that of MSCs from adipose tissue, 26 thus substantiating the progenitor status. The high synthesis rate of CD44 (hyaluronan receptor) may also predict the production of ECM by these cells since expression of CD44 was described to occur at the same time as matrix deposition within intercellular regions and CD44 plays an important role for cell to matrix interactions during chondrogenesis. 27
During chondrogenesis in vivo, MSCs undergo different steps: from proliferation over condensation to lastly differentiation. 28 For cartilage formation in vitro, cells have to undergo the same process. In order to achieve good differentiation results, Dexheimer et al. 28 suggested that MSCs with higher metabolism are able to proceed through the steps from monolayer via determination into the chondrocytic lineage. In our study, we found that de-differentiated chondrocytes, postmortally isolated from unaffected knee cartilage, were able to undergo condensation from cell suspension to pellets when supplemented with chondrogenic growth factors. This process was associated with an acidification of the cell culture medium—as observed by the color change in phenol red—indicating metabolically active cells. Indeed, these cells were then able to synthesize hyaline-like cartilage components. The extent of the ECM synthesis depended on oxygen tension and growth factor treatment. While apparent differences of ECM production between hypoxic (5%) and normoxic (21%) culture conditions were observed, the quantities of the single ECM components were mainly affected by the choice of growth factor.
TGF-β1 is known for its chondroinductive properties 29 and to inhibit hypertrophy of chondrocytes,30,31 suggesting the impact of a targeted growth factor treatment on ECM deposition. It was further reported that the absence of TGF-β1 led to the formation of fibrocartilaginous tissue with hypertrophic characteristics.32,33
Although IGF-1 is known to be an anabolic factor for stimulating matrix synthesis, 34 in vitro treatment with exogenous IGF-1 resulted in a reduced production of hyaline cartilage matrix, 18 which was also confirmed by the results of our present study. In a previous work, we analyzed the ECM composition of chondrocytic cells from osteoarthritic (OA) articular cartilage and found that IGF-1 treatment led to similar results on matrix deposition compared with cells from healthy cartilage. 24 Nonetheless, cells from OA cartilage showed better results for hyaline matrix formation after the single treatment with IGF-1 under hypoxia. Additionally, an additive effect of the combined stimulation was visible. 24 Here, we suggested that the enrichment of reactive oxygen species in OA cartilage resulted in a reduced activity of IGF-1. 35 Hypoxia, on the other hand, could restore IGF-1 effects by decreasing oxidative stress. In contrast to chondrocytic pellet cultures derived from OA cartilage, enhancement of hyaline cartilage–like protein synthesis rate was not detectable in nonaffected chondrocytes. These results suggest a varied availability and a different effect mechanism of exogenous IGF-1 in OA and healthy cartilage; however, a donor-specific response to growth factor stimulation has to be taken into consideration. We also previously observed clear differences in hyaline matrix components between cells from healthy and OA cartilage. 36 Under identical culture conditions, chondrocytic pellet cultures of healthy cartilage sustained a better Col2/Col1 protein ratio than those from OA cartilage. 36
In cell-based models for cartilage repair, a detrimental factor is the development of fibrocartilage by enhanced collagen type 1 expression rates as well as hypertrophy of cells. The results of our study indicated decreasing collagen type 1 contents within pellet cultures under hypoxic conditions suggesting a positive impact of the approximation of physiological conditions. Although we did not investigate hypertrophic markers such as collagen type X, alkaline phosphatase (ALP), or matrix metalloproteinase 13 (MMP13) in this study, we have got gene expression results of alkaline phosphatase and collagen type X rates from postmortally isolated chondrocytes in pellet cultures with reduced mRNA levels for both hypertrophic markers. 37 Moreover, we analyzed the ALP activity of alginate-encapsulated OA chondrocytes under normoxia in a previous work using the same dosage of TGF-β1 and IGF-1. We did not detect an increased ALP activity of stimulated cells 23 for the combination of TGF-β1 and IGF-1, which supports the assumption that the used growth factor concentrations did not induce a hypertrophic phenotype. However, further studies are necessary to analyze the effects of sole and combined growth factor treatment on hypertrophy in unaffected chondrocytic pellet cultures on protein level.
The results indicate that postmortally isolated chondrocytes showed good viability and synthesis rates of hyaline-like ECM proteins suggesting these cells as a suitable source for cartilage repair. For therapeutical approaches like ACT, postmortally isolated chondrocytes instead of autologous cells could be used for defect regeneration. Dhollander et al. obtained satisfactory midterm clinical outcomes of ACT using postmortally isolated chondrocytes embedded in alginate matrix in a human study.20-22 However, defect repair with hyaline-like ECM was only shown for 15% out of 13 patients whereas mixed tissue was found in 46% and fibrocartilage in 31% of cases. 22 The insufficient hyaline matrix protein synthesis rate, at least partially, may result from the lack of medium supplementation with cartilaginous growth factors during in vitro cultivation and the use of non-dedifferentiated, alginate-embedded chondrocytes. Finally, immunological aspects have to be taken into account in allogenic approaches. Therefore, further in vitro and in vivo studies are necessary to assess the suitability of this cell source for clinical use. By analyzing the immunological response of allogenic cartilage constructs in rabbits by Arzi et al. 38 a negligible to mild immune response was shown for tissue engineered allogenic cartilage. The authors concluded that the immune tolerance to allogenic implants depends on the proximity of the transplantation site to the synovium mediating the immune response within the joint. 38 Since the data from small animal models are not transferable to the human situation, further animal tests, for example, in sheep, have to be taken into consideration before testing allogenic cells in humans. However, there are also some preliminary results from the mid-term follow-ups of the human study by Dhollander et al. 20 in which the patients did not report detrimental effects after allogenic cell transplantation.
In summary, we could demonstrate that postmortally isolated chondrocytes are able to maintain the chondroprogenitor status. Moreover, these cells could re-differentiate in 3D pellet cultures whereby the addition of TGF-β1 as well as reduced oxygen partial pressure supports the accumulation of hyaline-like ECM components. These results indicate that postmortally isolated chondrocytic cells may be an alternative allogenic cell source for approaches aiming at cartilage repair, especially when TGF-β1 is used during in vitro expansion of cells. In future studies, it has to be clarified whether these promising in vitro results can be translated to the in vivo situation.
Footnotes
Acknowledgment and Funding
We gratefully thank Mr. B. Heskamp for his technical support. This work was supported by the FORUN project 889004 of the University Medical Center Rostock. The funding source had no role in study design, data collection, analysis, and interpretation as well as manuscript writing and submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Local Ethical Committee (Registration No. A2011-138).
