Abstract
Intra-articular injections of hyaluronic acid (HA) are widely used in the treatment of osteoarthritis. HA half-life varies between products which might explain differences in effectiveness between viscosupplements.
Aim
To compare the resistance to degradation of linear and cross-linked viscosupplements using a rheological model combining mechanical and oxidative stresses, mimicking what happens inside the joint following HA injection.
Methods
The rheological properties of 8 HAs were measured using a stress-imposed Rheometer DHR3. Strain sweeps were carried out to evaluate the rheological properties at rest from 0.001 to 3000% at a frequency of 1 Hz. The complex modulus G*, in Pa, and the phase tangent tan δ, dimensionless, in the linear viscoelastic domain (LVED) were extracted. The oxidation tests were conducted by exposing the product to H2O2 for 30 minutes. The effect of oxidation was evaluated by measuring variations of G* and tan δ, using an oscillation time sweep. Those tests were carried out at a frequency of 1 Hz and at 1% strain in the LVED.
Results
At rest, the different samples exhibited various viscous behaviors. During mixing process, G* decreased from −6.4% to −31.3%. G* of low-molecular-weight HAs decreased more than that of medium molecular weight (MW) and cross-linked products. After oxidative stress, G* variation ranged from −10.1% to −46.3%. Cross-linked HAs and those containing mannitol resisted the best to degradation.
Conclusions
We showed large variations in resistance to degradation between viscosupplements. The duration of effectiveness of these products deserves to be compared in randomized clinical studies.
Introduction
For more than 30 years, intra-articular injections of hyaluronic acid (also called viscosupplementation) have been used to relieve pain and improve mobility in patients suffering from knee osteoarthritis (OA). Hyaluronic acid (HA) is a high-molecular-weight (HMW) non-sulfated glycosaminoglycan consisting of alternating units of N-acetylglucosamine and
In OA, SF elastoviscosity is dramatically lowered when compared with healthy SF because of a decrease in both HA MW and concentration. 3 Consequently, the ability of SF to protect cartilage decreases, favoring cartilage degradation. Beyond its mechanical properties, HA exhibits many biologic functions that depend on interactions with specific binding proteins (hyaladherins) such as the cell surface hyaluronan receptor (CD44), HA-mediated motility (RHAMM), and hyaluronan-binding proteins.5,6
Viscosupplementation is a treatment used worldwide for limb OA (mainly knee OA), which aimed to restore the rheological homeostasis of the SF by injecting a solution of HMW HA intra-articularly. 2 The aim of the treatment is to protect articular cartilage from degradation and consequently to decrease pain and improve joint motion. Viscosupplementation effect is not fully clarified and is probably due to several mechanisms such as promotion of endogenous HMW HA production, interaction with pain receptors, and various anti-inflammatory effects. 7 Although this therapy has been used for more than 3 decades and is recommended in several guidelines for the management of OA,8-13 the use of viscosupplementation in OA remains a topic for debate regarding treatment efficacy and safety and the best dosing regimen. These discrepancies may originate from differences between the marketed HA products 14 that widely vary in MW (from 0.7 to more than 90 MDa), concentration (ranging from 0.8 to 32 mg/mL), origin (cockscomb extraction or bacterial biofermentation), and molecular structure (linear or cross-linked). HA is a very unstable and heat-sensitive molecule. The molecular mass and the viscosity may be decreased due to the damage of bonds in the polymeric chains. HA is susceptible to degradation under a variety of conditions such as heat, acid hydrolysis, and oxidative depolymerization reactions. Conversely, HA remains relatively stable during the freeze-drying process. 15 The major problem faced by manufacturers of HA is that the latter, once injected, is rapidly degraded, limiting the time of intra-articular residence to only a few days. 16 HA degradation is the result of 2 different mechanisms. The first one is mediated by specific enzymes (hyaluronidases) while the second one is nonspecific, due to reactive oxygen species (ROS).5,17 OA is a degenerative joint disease of multifactorial origin in the pathogenesis of which ROS play a deleterious effect.18,19 Interleukin-1b (IL-1b) activates the production of large amounts of ROS which are themselves involved in the production and/or the activation of collagenase matrix metalloproteinase-1 and in chondrocyte apoptosis. 18 There is substantial evidence that ROS are involved in HA degradation in inflammatory joint diseases, such as rheumatoid arthritis and OA. 17 Optimizing clinical effectiveness of viscosupplementation by reducing HA degradation in situ, and therefore increasing the time of contact with the diseased tissue, 20 can be obtained by the addition of ROS scavenging molecules (mannitol or sorbitol) 21 and/or by cross-linking.
Our hypothesis is that viscosupplements, whether linear or cross-linked, may differ greatly from each other and their resistance to degradation varies accordingly. The aim of the present study is to compare in vitro the resistance to ROS-mediated degradation of 8 marketed viscosupplements that differ in molecular structure, viscoelastic behavior, and HA concentration, by studying their rheological properties before and after mixing them with hydrogen peroxide (H2O2), a well-known method to degrade polysaccharides through a radical mechanism.1,22
Methods
Viscosupplements to be Assessed
In this study, 8 HA viscosupplements, 4 linear and 4 cross-linked, were compared. The characteristics of the investigated viscosupplements are given in Table 1 . Both HAPPYCROSS® and OSTENIL PLUS® contain mannitol.
Characteristics of the Studied Hyaluronic Acid Viscosupplements.
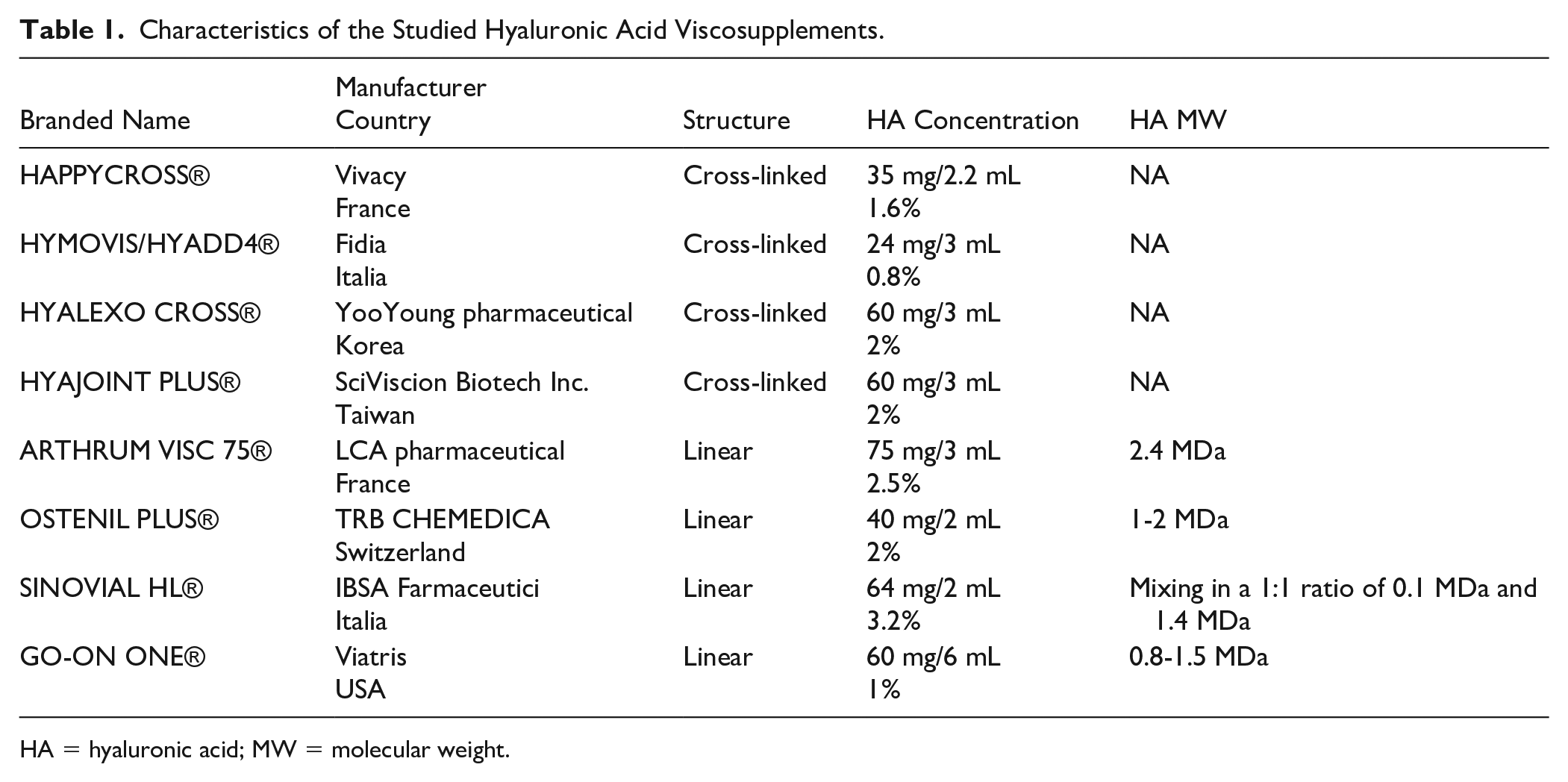
HA = hyaluronic acid; MW = molecular weight.
Rheological Assessment
The rheological properties were measured using a stress-imposed Rheometer DHR3 (TA Instruments; New Castle, DE, USA) with a rough 25-mm-diameter plate–plate geometry according to ISO 3219-1 and ISO 3219-2 standards for good rheometric practice. During the experiments, the temperature was regulated by a Peltier effect module. The lower plane was conditioned to the measurement temperature of 25°C, with an accuracy of 0.5°C.
The samples were put on the lower plate directly from their syringes without any needle. The upper plate was lowered to a trim gap of 1,050 µm. Any excess product was removed with a spatula. The gap was set to 1,000 µm. Each experiment was repeated 3 times, to obtain the mean and standard deviation.
Determination of Viscoelastic Properties
Strain sweeps were carried out to evaluate the rheological properties at rest and in particular if the samples were gelled or not. They were performed from 0.001 to 3000% at a frequency, f, of 1 Hz. The complex modulus G*, in Pa, and the phase tangent tan δ, dimensionless, in the linear viscoelastic domain (LVED) were extracted.
G* is the complex resultant of the elastic and viscous moduli G′ and G″. It reflects the overall consistency of the product at rest. Tan δ is the ratio between G″ and G′. It is related to the molecular network morphology. Any change in its value indicates a change in the microstructure. If tan δ in the LVED is inferior to 1, the product is considered gelled.
In a previous rather similar study, 20 the assessed rheological parameters were G′, G″, and the complex viscosity η* which is equal to G* divided by the constant 2πf where f is the frequency. The viscosity is a dynamic parameter normally obtained in flow conditions. As strain sweeps tests are mainly quasi-static, it therefore seems more relevant to work with modules.
Induction of Oxidative Stress
The 8 HA viscosupplements mentioned above were subjected to oxidative stress generated by the addition of H2O2 (30%, Sigma Aldrich) at a final concentration of 5.4% (v/v).
A volume of 1 mL of each HA commercial sample was put on the lower plate. 0.1 mL of H2O2 was added giving a ratio of 10:1. The mix was stirred for 10 seconds with a spatula. The upper geometry was then lowered in the same way as described above. Delay time between addition of H2O2 and first measurement was around 1 minute.
The oxidation tests were conducted by exposing the product to H2O2 for 30 minutes. The effect of oxidation was evaluated by measuring the evolution of G* and tan δ in real time, using an oscillation time sweep. Those tests were carried out at a frequency of 1 Hz and at 1% strain in the LVED. The initial and final values of the complex modulus G* and of the phase tangent tan δ were extracted as well as the G* maximum and tan δ minimum.
Results
Strain Sweeps
At rest, without any oxidative stress, the different samples of this study exhibited different behaviors. As shown in Table 2 , the consistency G* presented a variation over 3 decades (from 3.73 to 378 Pa). A high G* was linked to either a cross-linked or HMW structure. Nonetheless, HYMOVIS® has a low constancy equivalent to that of SINOVIAL HL® despite being cross-linked. This lies probably in the specificity of its cross-linking, which is not obtained with 1.4-butanediol diglycidyl ether (BDDE) but using hexadecylamide, giving formation of reversible interactions yielding a gel-like structure.
Average Values of Complex Modulus and Phase Tangent Obtained with the Strain Sweeps in the LVED.
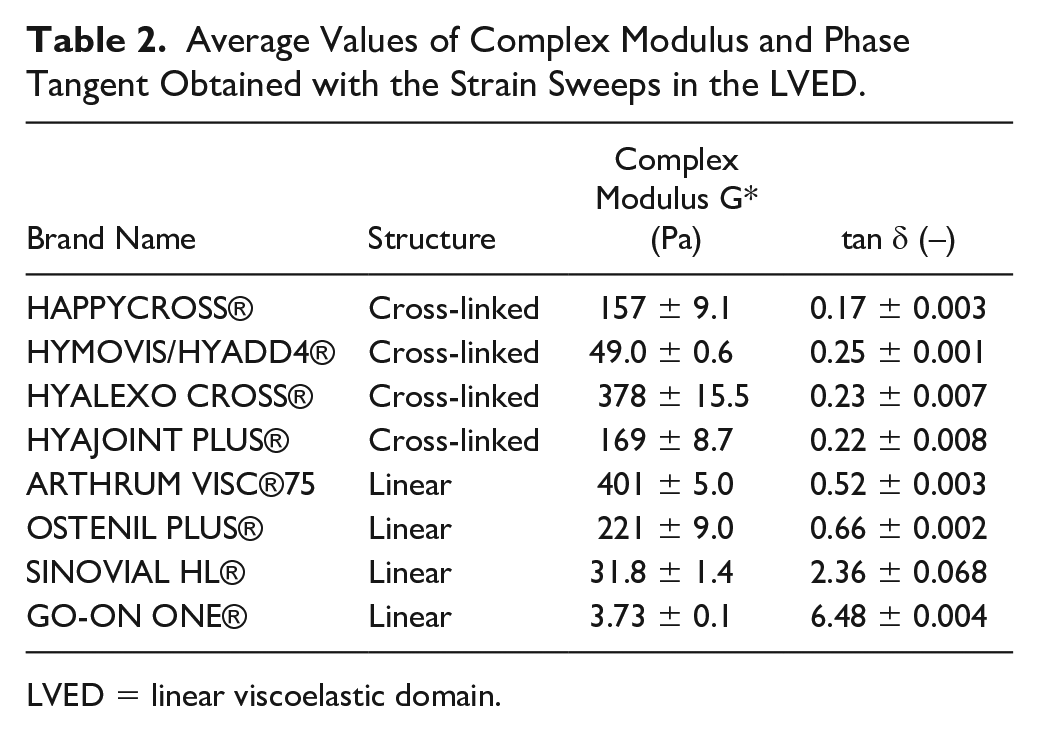
LVED = linear viscoelastic domain.
In terms of tan δ, as shown in Figure 1 , all the cross-linked samples, including HYMOVIS®, exhibited a gel-like behavior with values around 0.2 which means a strong contribution of G′, the storage modulus, the solid part. For linear products, a gel is obtained with the HMW HAs (OSTENIL PLUS® and ARTHRUM VISC® 75). However, those gels were less solid-like than cross-linked HAs. The linear samples with lower MW (SINOVIAL HL® and GO ON ONE®), that are not gelled, had tan δ superior to 1. They can be called structured liquids. With all these samples we had a fairly complete panel of what may exist in terms of rheological behavior of viscosupplements.
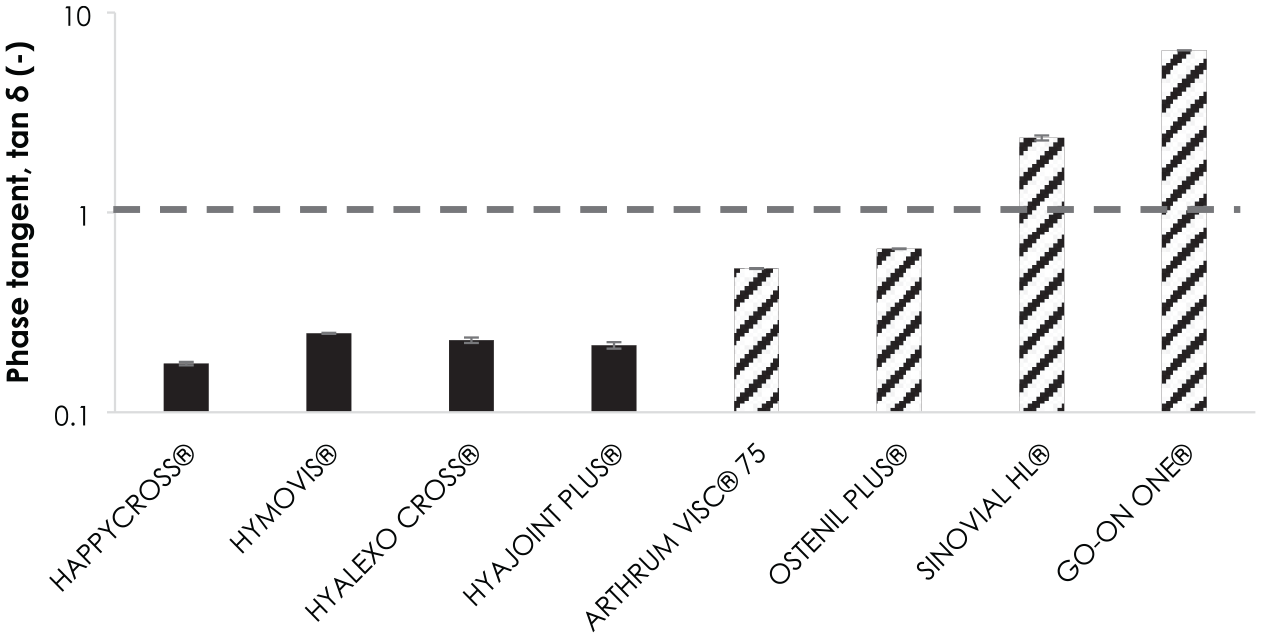
Comparison of tan δ values for the different viscosupplements in the viscoelastic linear domain without oxidative stress. The horizontal dotted line, for tan δ equal to 1, marks the boundary between soft solid (tan δ < 1) and structured liquid (tan δ > 1). The full histograms correspond to partially or totally cross-linked products and the striped ones to linear products.
Study of Oxidation Kinetics
The study of tests under oxidative stress showed, for most samples, an evolution in 2 phases with first an increase of complex modulus G* for a few minutes, followed by a decrease of this modulus over time (
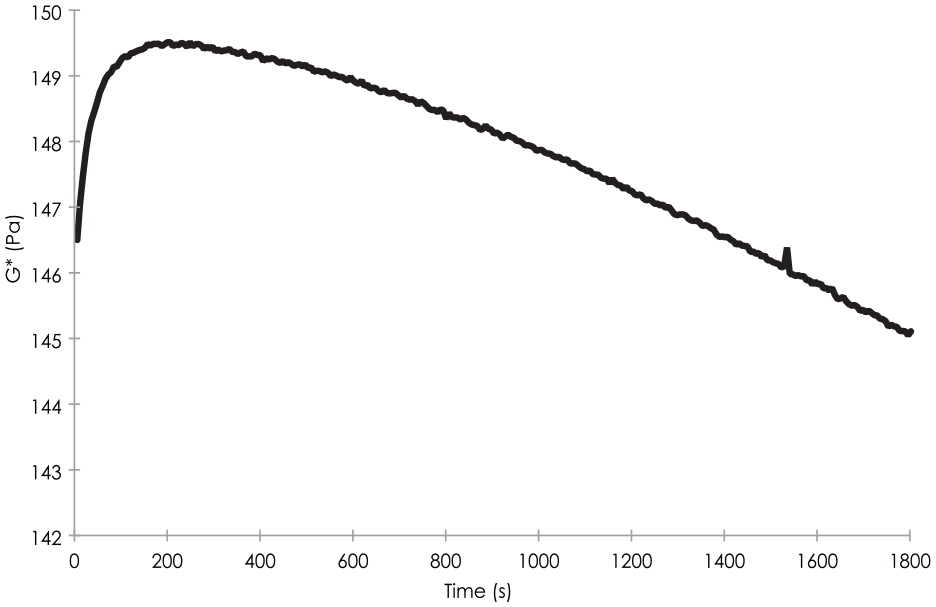
Evolution of the complex modulus G* over time after contact with the oxidizing agent. The curve presented here is that obtained for the HAppycross© sample.
Mechanical stress
We first evaluated the mechanical impact of the mixing of HA with hydrogen peroxide on the rheological properties. During oxidation tests, the more consistent the sample, the more complicated the mixture is and the more it requires mechanical action on the product. It is possible to evaluate the impact of this mixing while considering the thixotropy (i.e., the temporal recovery of rheological properties following the mechanical stress of the mixture) by calculating the G* and tan δ variations between the viscoelastic linear domain without oxidative stress and the maximum—or minimum for tan δ—values during the oxidation tests.
On fragile samples, mixing induced a decrease in modules and a change in the nature of the microstructure.
Figure 3A
shows that the greatest variations were recorded for linear samples especially SYNOVIAL HL® and GO-ON-ONE®. The implementation had a small influence on the HAPPYCROSS® sample. For the other cross-linked samples, the influence was more pronounced probably because the mixing with the oxidizing agent induced fragmentation of the samples. Concerning the nature of the microstructure, it is considered that it is not altered below a 10% variation. In this respect, all samples apart from GO-ON-ONE® did not have their microstructure impacted (

Complex viscosity G* (
Oxidative stress
To evaluate the impact of the oxidative stress, by taking into account the mixing process, the percentage of variation of G* or tan δ was evaluated. It was calculated as the relative difference between the values in the LVED and the final values of the oxidizing tests (ΔG*tot and tan δtot). Figure 4A shows the variations of ΔG*tot over the 20 minutes of the test. Although a decrease was always observed, the moduli of linear products decreased more than those of cross-linked products.
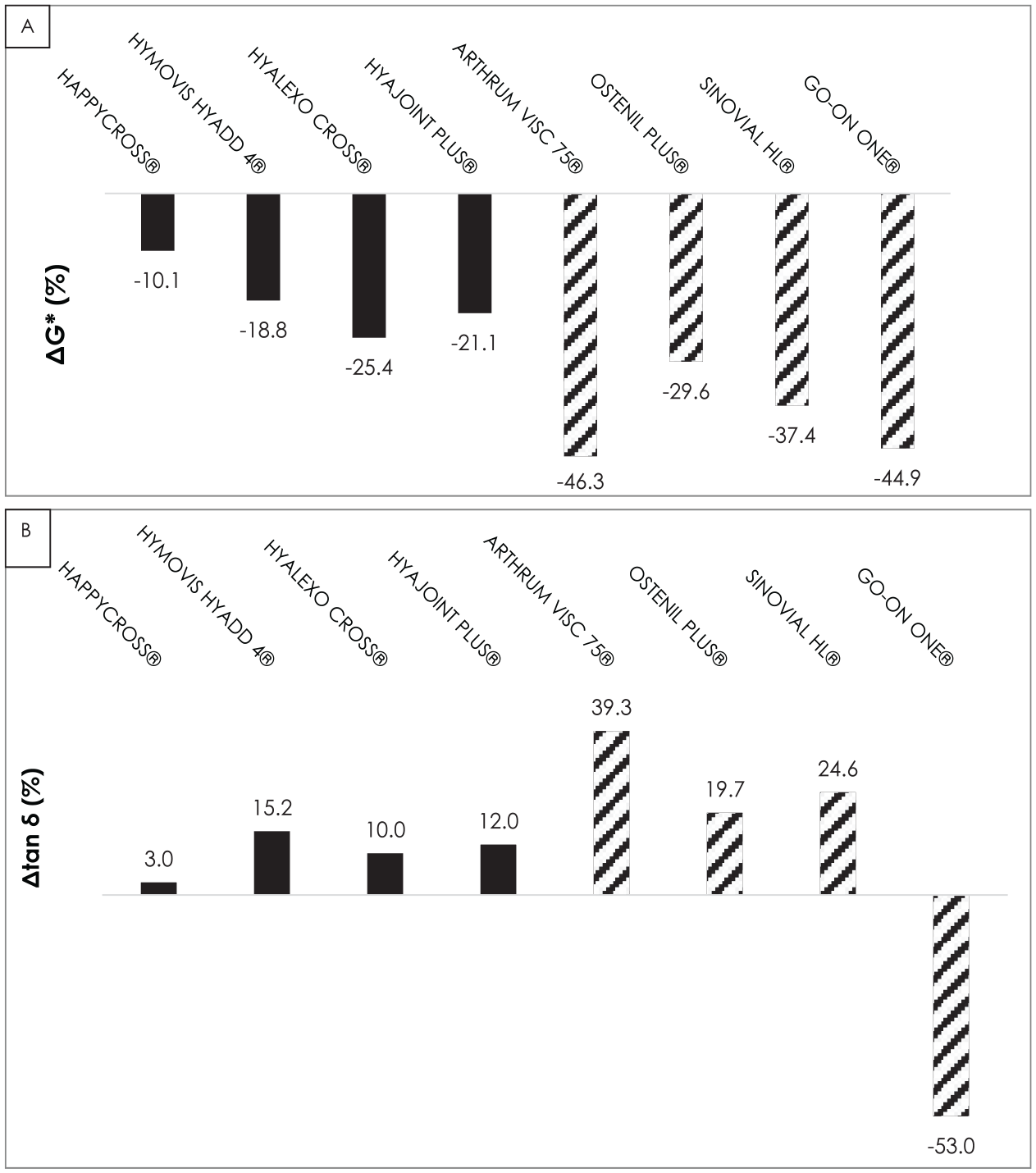
Complex modulus ΔG*tot (
The signature of the microstructure was evaluated through the tan δtot variation. Figure 4B shows that all the cross-linked products exhibited either no microstructure modification (i.e., HAPPYCROSS®) or a more pronounced viscous liquid character inducing an increase in tan δ. For the linear viscosupplements, the changes also go toward a more pronounced viscous liquid character with the exception of GO-ON-ONE® which exhibited a decrease in tan δ.
Discussion
The decrease in G* modulus and the variations in tan δ demonstrate that oxidative stress deteriorates the viscosupplements through microstructure changes. The results, showed in Figure 4 , suggest that hydrogen peroxide alters the chains of HA by fragmenting them, making them more liquid-like. This change is more pronounced for linear product. For GO-ON-ONE®, there is an exception where the strong degradation is linked to a drop in G* that goes with a decrease in tan δ. Unsurprisingly, our study shows large differences in resistance to oxidative stress-related degradation between linear and cross-linked HA. More surprisingly, it also highlights significant differences between linear HA products. However, we did not find large differences between cross-linked HAs, whatever the HA concentration was (ranging from 0.8% to 2%), apart from the HAPPYCROSS® which only shows a 10% decrease in consistency and almost no change in microstructure.
Our study reports, imperfectly of course, what can happen in OA joints when HA is injected. HA is subjected to both mechanical (joint movement) and oxidative (presence of ROS) stress. We clearly showed that cross-linking, and thus a gelled character, gives the advantage to limit the deleterious influence of both oxidative and mechanical stress on the macromolecule structure. Conversely, the impact of HA-MW on the resistance to degradation (that was not directly measured, but was assessed using the complex viscosity values, which indirectly reflect the MW) does not seem of major importance. This was suggested by the nearly same resistance to H2O2-related degradation of Go-On One® (ΔG* = −46.3%) and Arthrum Visc® 75 whose MW is much higher (ΔG* = −44.9%). Similarly, its concentration does not seem to protect, either, HA from ROS degradation, since the most concentrated viscosupplement, Sinovial HL® (HA concentration 3.2%), was the one that resisted the least oxidative degradation.
In contrast, less differences were found between cross-linked HAs both in G* (−10.1% to −25.4%) and tan δ (+3% to +15.2%). Cross-linking consists in linking together different chains of linear HA by covalent bonds, either chemically or physically. Hence, the rheological properties, stability, and half-life of the native HA can be enhanced by creating a 3-dimensional (3D) structure and forming a hydrogel. 23 Several studies showed that the half-life of the injected HA was greatly prolonged by the cross-linking process, in addition to the fact that the latter improves the viscoelasticity. Thanks to cross-linking, the HA residence time can reach up to 28 days.24,25 This finding does not prove that cross-linked HAs have a long-lasting effect than linear ones. However, the only 3 viscosupplements that have shown clinical efficacy for up to a year are all cross-linked.26-28 Several single-dose linear viscosupplements have been shown to be non-inferior to a cross-linked comparator.29-31 However, these studies only lasted 6 months and did not make it possible to judge the respective efficacy of the long-term treatments.
It is also interesting to underline that only HAPPYCROSS®, which combines cross-linking and mannitol,32,33 presented a decrease in the complex modulus ΔG*tot < 10%. The HAPPYCROSS® and OSTENIL® samples contain Mannitol as an antioxidant. These 2 products, in their respective category (cross-linked and linear), were the least affected by oxidative stress (
Unfortunately, in the present study, we were not able to investigate whether the type of sterilization predisposes the hydrogels to a greater or lesser degradation. Steam heat was used for the sterilization of all the studied viscosupplements. This process generally needs temperatures between 121°C and 130°C for short periods, 15 to 20 minutes, but we could not have access to the precise conditions of sterilization of each product studied. However, it has been clearly demonstrated that heat sterilization modifies both the micro and macrostructures of gels.34,35
Conclusion
In summary, the present study shows very large differences of resistance to degradation between viscosupplementation products. We have been able to show that under the effect of a combined mechanical and oxidative stress, the reduction in viscosity could vary according to a ratio of 1 to 5 depending on the products. Both cross-linking and mannitol make it possible to decrease HA depolymerization when subjected to an oxidative stress. These differences might explain the wide variations of half-life between HA products. Although it can be assumed, it is not known whether such differences have a significant impact on the clinical effectiveness or the duration of the products’ effectiveness. Face-to-face clinical trials are needed to assess the impact of the half-life on the magnitude of the effect and the duration of the efficacy of the different viscosupplements.
Footnotes
Author Contributions
TC, JP, and GD designed the protocol. GD carried out the measurements. GD and TC analyzed the data and wrote the manuscript. JP reviewed and approved the manuscript.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by LABRHA SAS, laboratoire de rhumatologie appliquée, Lyon, France.
Data Availability
The datasets for this study can be found in Rheonova, Université Grenoble Alpes, 1 Allée de Certèze, 38610 Gières, France.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TC received fees from LABRHA for expert and consultant services.
Ethical Approval
Not applicable, this work does not involve the human person.
