Abstract
Objectives
This study aimed to evaluate the efficacy of hyaluronic acid in the viability and proliferation profile of human femoral-tibial joint cartilage affected by osteoarthritis using in vitro models of chondrocytes in a 2-dimensional (2D)- and 3-dimensional (3D)-based culture model by spheroids.
Design
In vitro study of knee cartilage affected by osteoarthritis that required surgical treatment. Samples were cultured and exposed to hyaluronic acid (100 and 500 μM; intervention group) or vehicle solution. In monolayer or 2D culture, proliferation and cell viability were measured, and nuclear morphometry was analyzed by 4′,6′-diamino-2-fenil-indol (DAPI) staining. The 3D-based culture established from the culture of articular cartilage of patients submitted to total knee arthroplasty evaluated the diameter, viability, and fusion ability of the chondrospheres created.
Results
Samples from 3 patients resulted in viable cultures, with chondrocyte cells exhibiting a potential for cell proliferation and viability to establish a culture. Hyaluronic acid (100 and 500 μM) improved chondrocyte viability and proliferation up to 72 hours in contact when compared with the control group, and no nuclear irregularities in morphology cell characteristics were observed by DAPI. In the 3D evaluation, hyaluronic acid (500 μM) improved the cellular feedback mechanisms, increasing the survival and maintenance of the chondrospheres after 7 days of analysis, showing the intrinsic capacity of chondrospheres grouped in the attempt to rearrange and reestablish new articular tissue.
Conclusions
The 2D- and 3D-based culture models with hyaluronic acid improved chondrocyte viability and proliferation and demonstrated the ability of freshly formed chondrospheres to undergo fusion when placed together in the presence of hyaluronic acid.
Introduction
Osteoarthritis (OA) is a common multifactorial chronic disorder, affecting joints such as those of the hand, knee, and hip. It promotes cartilage destruction, subchondral bone exposure, and joint deformity with progressive cartilage degeneration. 1 The incidence of OA is 35% in patients above 50 years and 80% in the age group above 65 years.2,3 It is estimated that more than 67 million people will be affected in the United States by 2030. 4
The articular cartilage (AC) is an avascular tissue, responsible for the absorption of impact and friction, and composed mainly of hyaline cartilage and chondrocytes. 5 Chondrocytes are mesenchymal cells responsible for the synthesis of extracellular matrix (ECM) in addition to growth and maintenance of AC homeostasis. 6 In the pathogenesis of OA, there is often a disturbance in the balance of chondrocyte physiological function and the activation of pro-inflammatory and catabolic factors. 7
OA of the knee joint, in particular, is widely studied as it is associated with severe physical and lifestyle disability. 8 Although as yet most drugs used to treat degenerative joint diseases are administered systemically, these diseases are ideally suited for local treatment, as commonly 1 or more well-defined sites accessible by injection are affected. While the majority of nonsurgical treatments of knee OA are ineffective, the intra-articular administration of hyaluronic acid (HA) is among the most used alternatives to handle it. 9 A major advantage of locally administered drugs is the possibility to achieve high concentrations at the site of administration while avoiding the often-serious side effects associated with systemic treatment. 10
HA is a glycosaminoglycan, is a component of cartilage matrix and synovial fluid, and acts as a lubricant and elastic shock absorber of the joint. 11 During OA, there is a decrease in the HA content of both the cartilage and synovium, and an injection of HA is used in patients who cannot tolerate the adverse side effects of pharmacological therapies or no response in nonpharmacological treatments. 12 Despite the encouraging results from local injection of drug formulations as treatment for degenerative joint disease, it is still likely that the efficacy of any drug, old or new, locally administered as a bolus, will be limited, in the articular joint characterized by full clearance within several days. 13
To clarify the behavior of chondrocytes in the global response to HA use, this study aimed at analyzing the in vitro turnover of cultured cells from AC of patients with knee OA exposed to HA.
Methods
This is an in vitro experimental study that was approved by the Ethics Committee of the University of Caxias do Sul (No. 2.503.102).
Patient Eligibility Criteria
Patients with knee OA and refractory to conservative treatment are included, according to the following criteria:
Radiological evidence of joint impairment according to Kellgren-Lawrence grade 3 or 4 osteophytes (
Signing a free and informed consent document
Kellgren and Lawrence Classification of OA of Knee. 14

OA = osteoarthritis; JSN = joint space narrowing.
Exclusion criteria are the following:
prior surgery, trauma, tumor, or infection in the joint
rheumatological, metabolic, or autoimmune diseases
continuous use of anti-arthrosis medication or corticosteroids
genu varum or genu valgum
Collection of Biological Material and Preparation for Transport
AC were collected from 3 patients submitted to total knee arthroplasty
Demographic Data—Data at the Time of Collection of AC.

AC = articular cartilage.
Isolation and Culture Cells
Human chondrocytes were isolated according to the methodologies in the literature with modifications.15-20 The material was mechanically dissociated into Petri dishes and reconditioned in its container, maintaining a maximum capacity of 10 mL by adding the biological material and 1x dPBS. The material was maintained for 30 minutes in a humidified oven at 37°C with 5% CO2. The tissue was enzymatically dissociated with the aid of 0.2 mg/mL Pronase (Sigma-Aldrich, MO) for 1 hour, followed by 1 mg/mL collagenase type I (Sigma-Aldrich, MO) for 2 hours. The cell suspension was centrifuged and the pellet was resuspended in 1 mL DMEM (High Glucose Plus—Dulbecco’s Modified Eagle’s Medium with
The resuspended cells were divided into two 25 cm2 bottles containing the same medium and supplementation conditions mentioned above. The cells were packed in a humidified oven at 37°C with 5% CO2. To establish the culture of chondrocytes, the cells received 2 mL of the same medium and supplement conditions every 3 days until completing 14 days, when the first complete exchange of media was performed, as there was cell adhesion verification using an inverted microscope (Axio Vert A1, Carl Zeiss, Germany). The culture was established around 26 days in 25 cm2 bottles containing 80% of cellular confluence. Approximately 1 × 106 cells were used for flow cytometry analyses.
Analysis and Characterization of Isolated Chondrocytes by Flow Cytometry
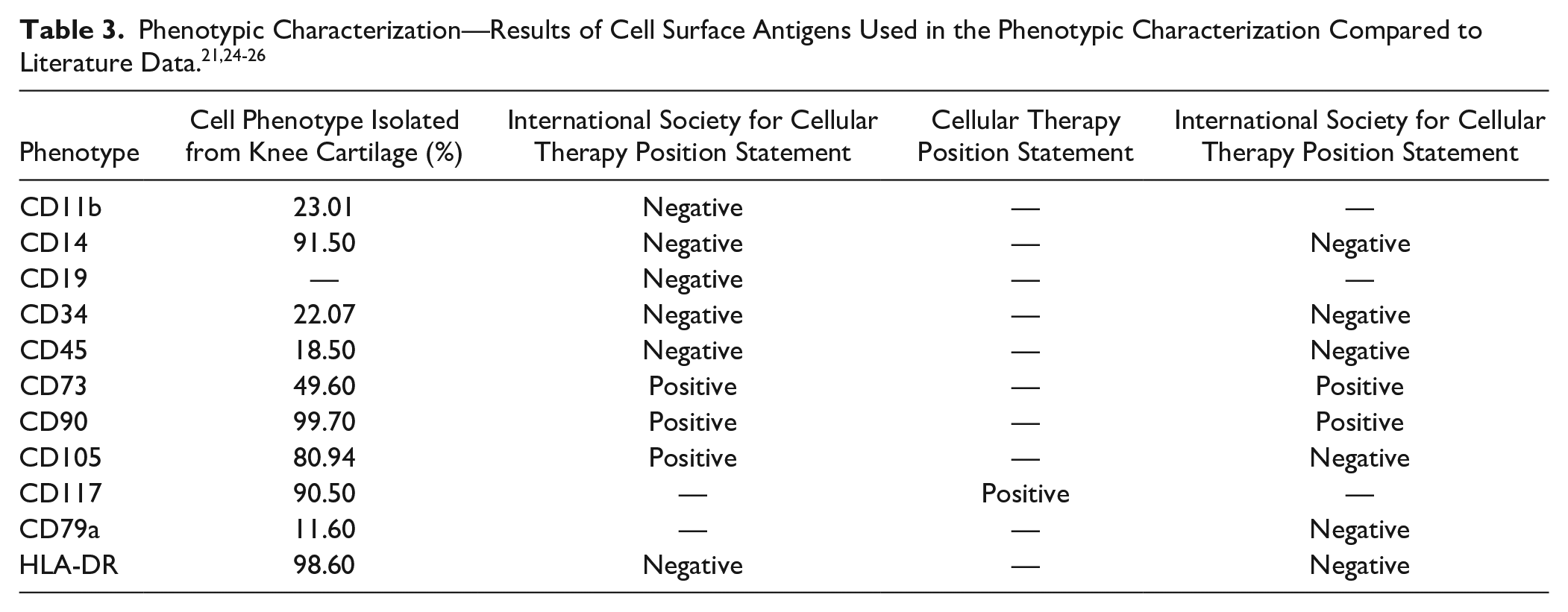
Approximately 1 × 106 cells were used for flow cytometry analyses. The cell population established in the culture was determined by flow cytometry (FACScalibur; Becton Dickison Immunocytometry Systems, San Jose, CA) with the following antibodies: anti-CD105, anti-CD73, anti-CD90, anti-CD45, anti-CD34, anti-CD11b, anti-CD79a, anti-CD14, anti-CD117 and anti-HLA-DR in accordance with the International Society for Cellular Therapy statement.21,22
General Cell Culture Protocols
The cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) with 10% FBS, 100 U/mL penicillin and 100 µg/mL streptomycin at a temperature of 37°C, a minimum relative humidity of 95%, and an atmosphere of 5% CO2 in air. For in vitro assays, at least 3 independent experiments were performed in triplicate for each patient sample.
Cell Viability Assay
The number of viable cells with metabolically active mitochondria was determined based on the mitochondrial reduction of a tetrazolium bromide salt (MTT [3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide] assay). 23 The cells were treated for 24 and 48 hours at different concentrations of Synvisc-One (SANOFI, FR) ranging from 10 to 500 µM. Cell viability was calculated using the following equation: cell viability (%) = (Abss / Abscontrol)100, where Abss is the absorbance of cells treated with different Synvisc-One concentrations and Abscontrol is the absorbance of control cells (incubated with Synvisc-One vehicle only). The experiments were analyzed by 1-way analysis of variance (ANOVA) followed by Bonferroni’s post hoc test, using GraphPad Software (San Diego, CA). Results are reported as the mean ± standard deviation. P value of 0.05 was indicative of statistical significance versus control group.
Cell Counting
To assess the proliferation grade, the human chondrocyte cells were seeded at 15-20 × 103 cells per well in 24-well plates for 24 hours. Then, the cells were treated for 24 and 48 hours with Synvisc-One (10-500 µM). After this period of incubation, the medium was collected and 200 μL of trypsin/EDTA (ethylenediaminetetraacetic acid) solution were added to detach the cells, which were counted in a hemocytometer. The cell number of the control group (nontreated cells) was considered 100%. The experiments were analyzed by ANOVA followed by Bonferroni’s post hoc test, using GraphPad Software. Results are reported as the mean ± standard deviation. P value of 0.05 was indicative of statistical significance versus control group.
Nuclear Morphology and Morphometric Analysis
The 4′,6′-diamino-2-fenil-indol (DAPI) staining provides nuclear morphological features (area, roundness, and solidity) and might be related to several mechanisms that affect cell survival processes. DAPI staining was carried out to establish the nuclear morphology of the human chondrocyte cells. Briefly, the cells were seeded in 24-well plates and incubated with different Synvisc-One concentrations for 24 to 72 hours. After incubation, the cells were washed 3 times in 1% PBS and fixed with 4% formaldehyde at room temperature for 15 minutes. The fixed cells were then washed with 1% PBS, permeabilized with 0.1% Triton X-100 in 1% PBS, and stained with a 300 nM DAPI solution (Santa Cruz, CA) at room temperature for 10 minutes. The nuclear morphology of the cells was examined under a fluorescent microscope (Carl Zeiss MicroImaging GmbH, Jena, Germany). DAPI staining clearly delineates the nuclear morphology that allows the quantification of the nuclear roundness and solidity measurements by Image J Software. Data from control cells (untreated) are used to set the parameters of the normal population. The morphometric parameters were calculated considering 100 events for each Synvisc-One concentration, 3 times in triplicate. The experiments were analyzed by ANOVA followed by Bonferroni’s post hoc test, using GraphPad Software. Results are reported as the mean ± standard deviation. P value of <.05 was indicative of statistical significance.
Preparation of Spheroids for 3D In Vitro Culture
Human chondrocytes were detached from plastic and resuspended in complete growth medium. For cell seeding to form spheroids, a suspension of 2 × 106 cells was prepared in 190 μL of DMEM supplemented with 50 μg/mL ascorbic acid (Sigma-Aldrich), 1.25 μg/mL human albumin, 100 U/mL penicillin, and 100 μg/mL streptomycin (Sigma-Aldrich) and Insulin-Transferrin-Selenium, ITS 1x (Lonza, Switzerland). Cell density was chosen according to recommendations of the silicone mold manufacturer MicroTissues 3D (3-dimensional) Petri Dish micromolds (Sigma-Aldrich). The cell suspension was carefully placed into each 81-well nonadhesive agarose micromold, and the seeding chambers were placed in 6-well culture plates and covered with complete growth media for 1 hour. Spheroid microplates were then incubated at 37°C in a humidified atmosphere with 5% CO2 for 3 days with change of medium post-24 hours.
Estimation of Spheroid Diameter and Fusion Ability
After 3 days, the spheroids formed were exposed to different concentrations of Synvisc-One (100 and 500 µM) and the average diameters (µM) were evaluated for 1, 3, and 7 days in culture using an inverted microscope (Axio Vert A1). The diameters were collected from 10 spheroids per sample and analyzed by Carl Zeiss software (MicroImaging GmbH, Jena, Germany). In parallel, the spheroids’ ability to become fused was examined. Bright-field image spheroids doublets were obtained at points 12 h, 24 h and 72 h using light inverted microscopy (Axio Vert A1).
Results
All samples from 3 patients resulted in viable cultures for experimentation, with chondrocytic human cells exhibiting potential for proliferation
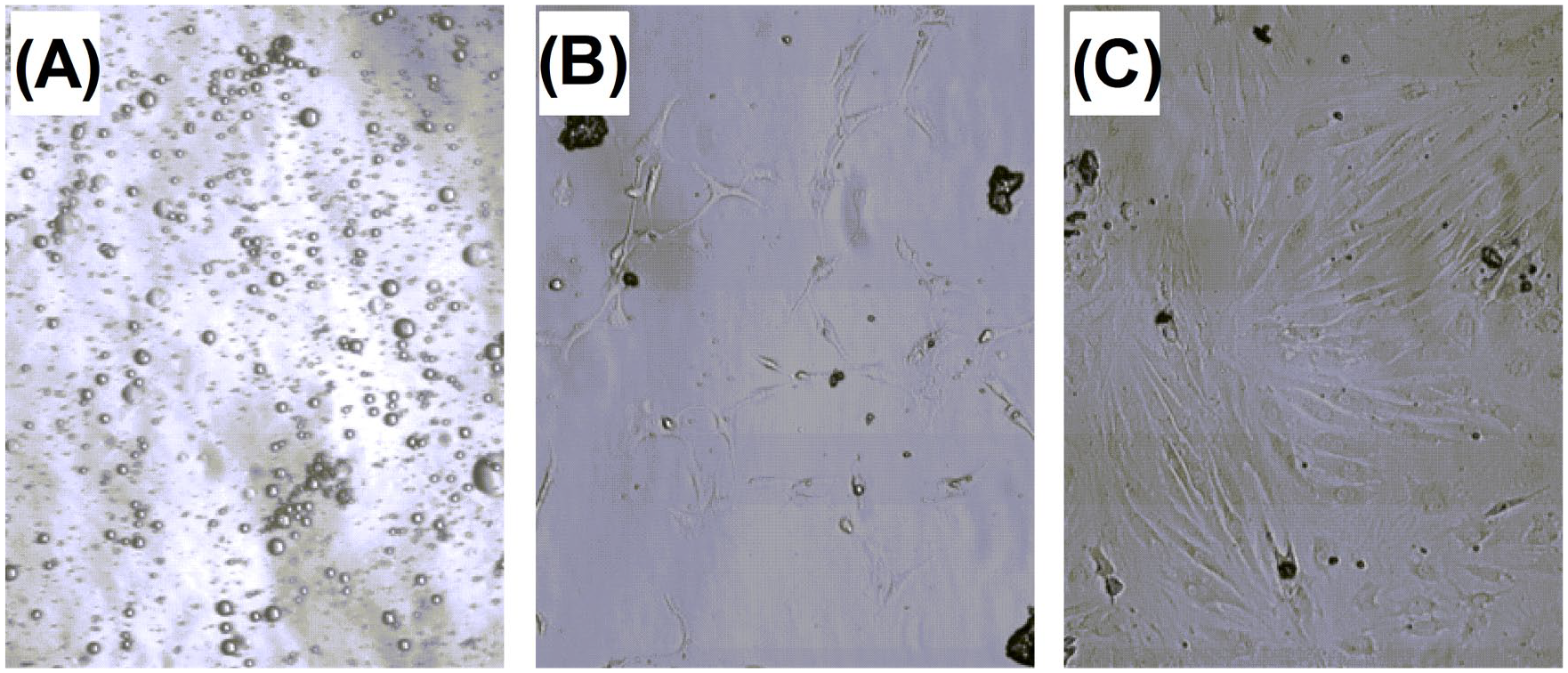
Isolation and culture.
Isolation and Culture of the Chondrocyte
Isolated chondrocyte samples were viable in 2-dimensional (2D) cultures in both groups. There was a higher chondrocyte proliferation in cultures that received 100 and 500 μM HA concentrations. This increase remained in cultures evaluated for 48 hours when compared with the control
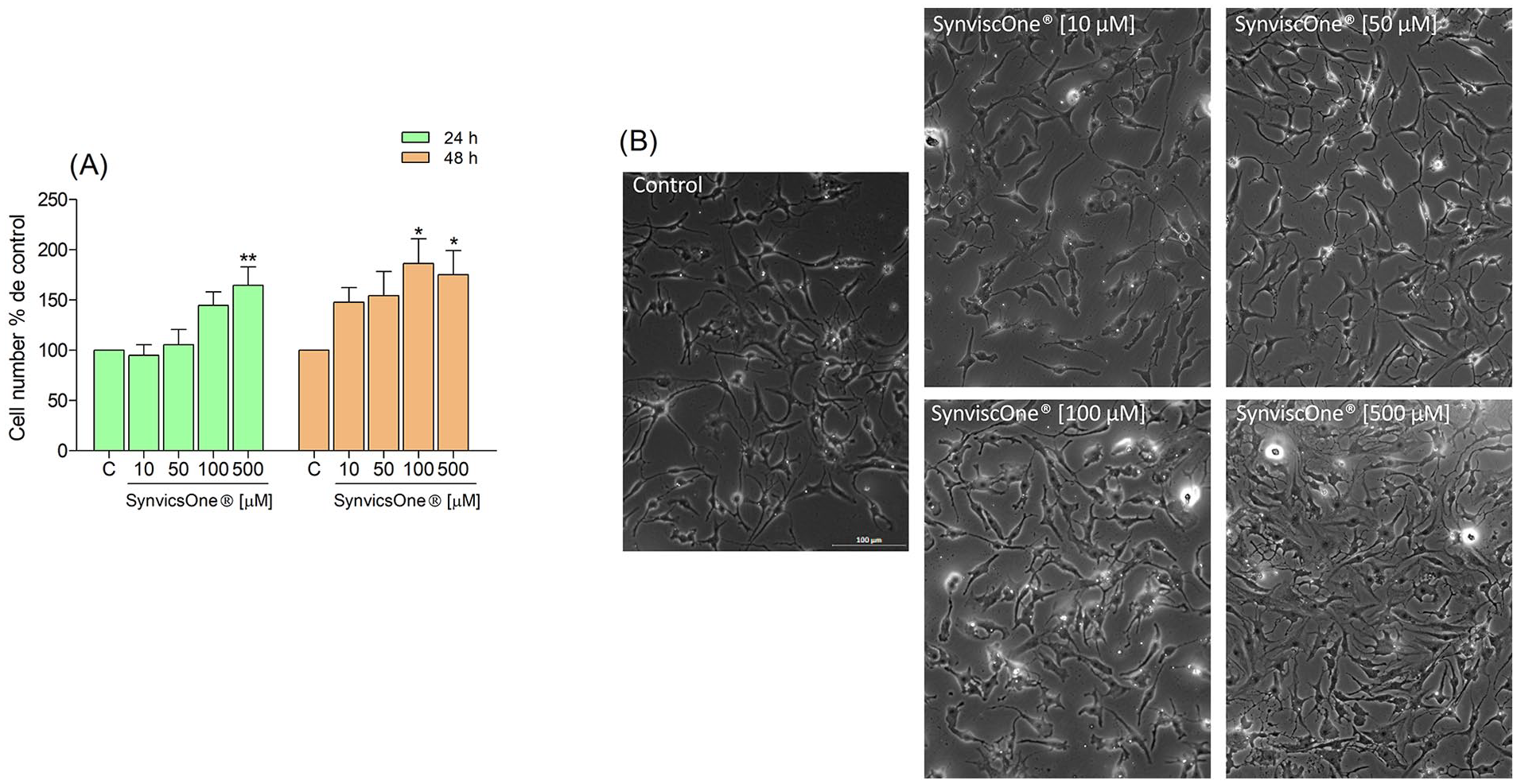
Qualitative and quantitative cell proliferation.
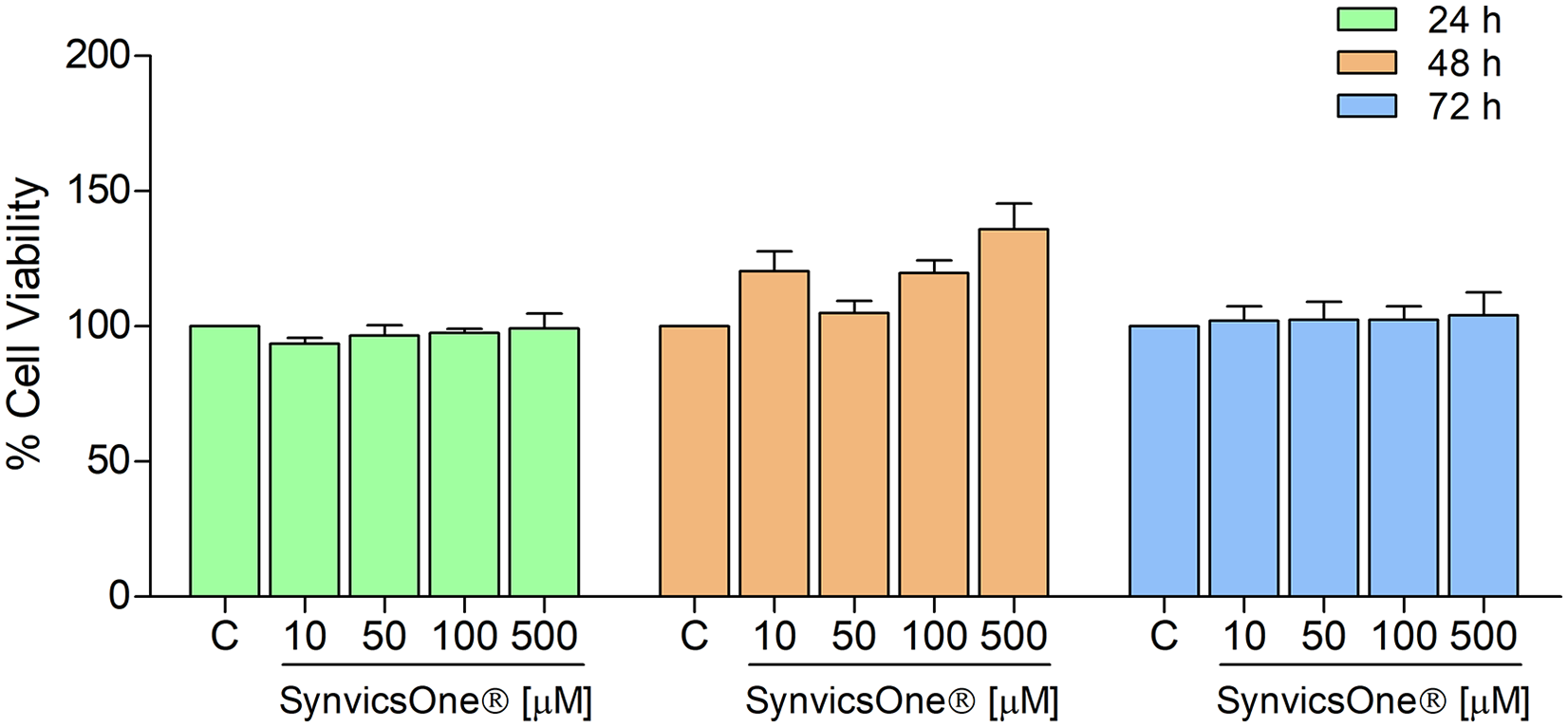
Quantitative MTT Assay. Incubation with Synvisc-One (10-500 µM) cell viability after 24, 48 and 72 hours (n = 3). Each column represents the mean ± SD. *P = .05 versus control.
Morphometric Analysis of the Chondrocytes
Human chondrocytes exposed to different concentrations of Synvisc-One up to 72 hours did not present morphological signs of nuclear irregularities by morphometric analysis. All cells analyzed presented a regular round shape and a well-defined nuclear surface
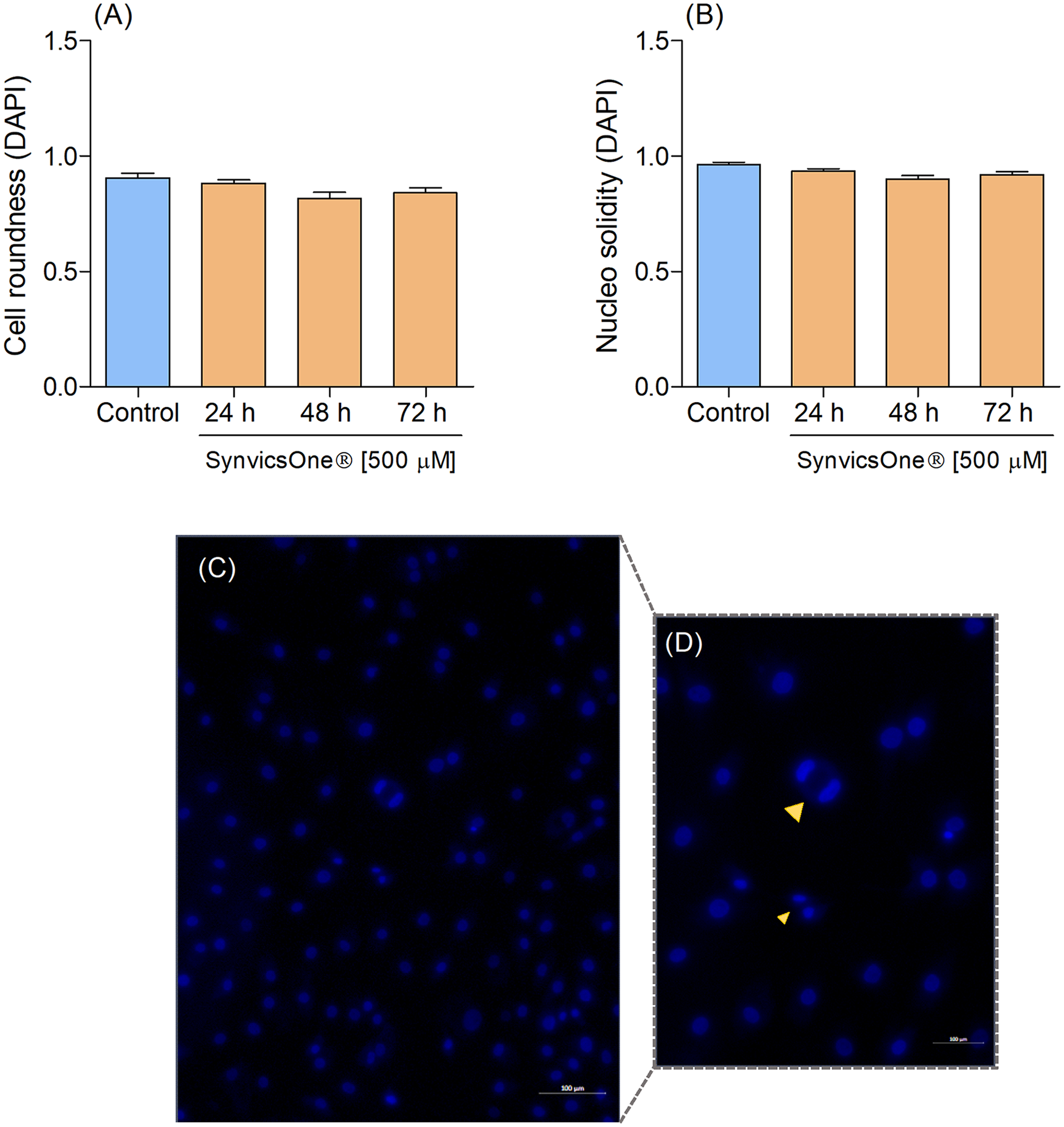
Qualitative and quantitative DAPI staining.
3D Viability and Proliferation
Cellular viability was maintained in all the analyzed groups, with results superior to the spheroid diameter of the group submitted to HA when compared with the control group
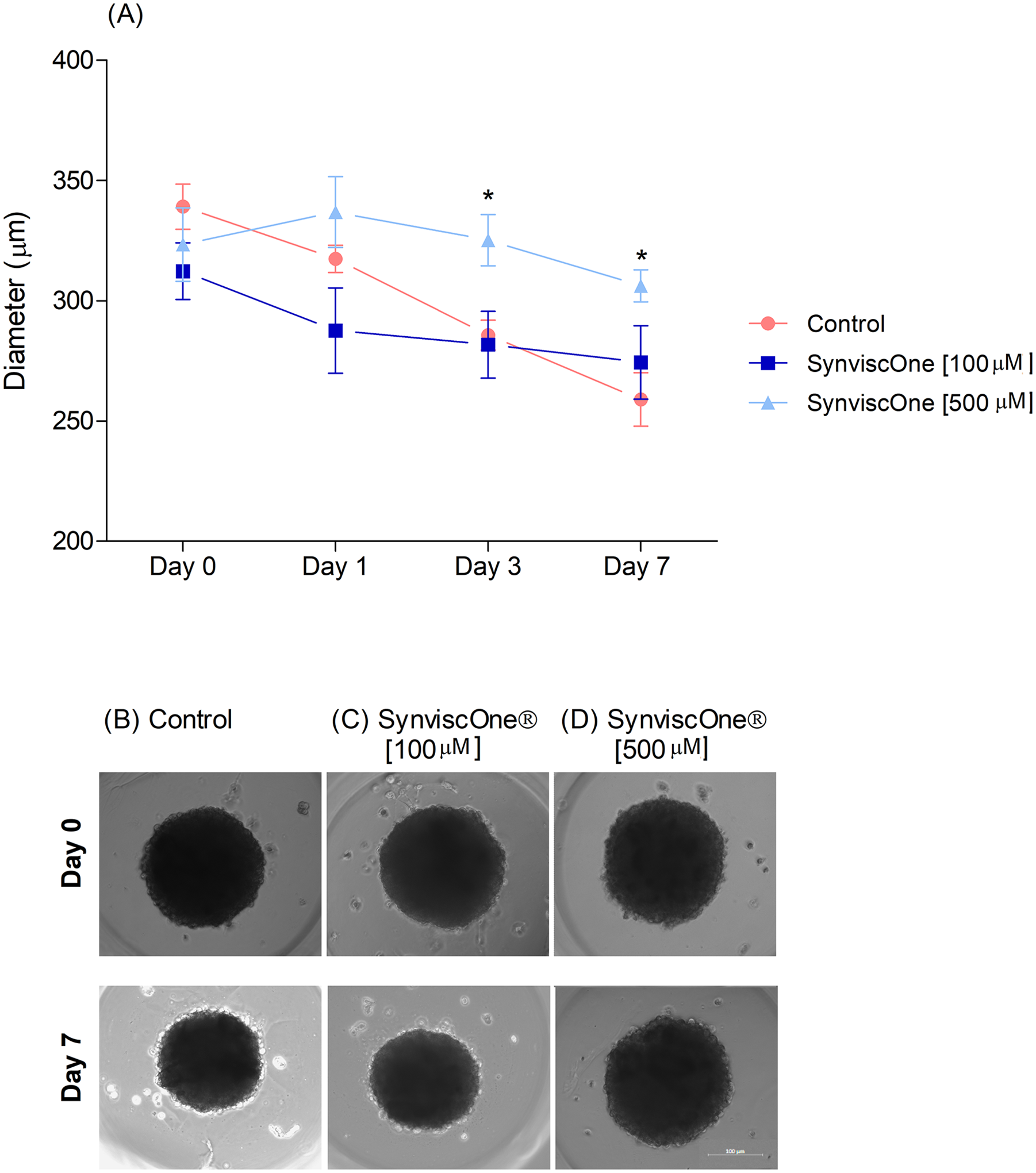
Diameter of the spheroid.
Influence of Viscosity on 3D Culture
The evaluation of the free spheroids in agarose covered wells showed the intrinsic capacity of chondrosphere grouping in the attempt to rearrange and reestablish the new articular tissue. The increased viscosity did not alter or delay clustering of the spheres

Spheroids fusion. Spheroid retained their fusion capability when exposed to Synvisc-One. Representative images of control spheroids and spheroids exposed to Synvisc-One (500 µM) in close contact after
Discussion
The study successfully established 3D-based culture from human AC of patients submitted to total knee arthroplasty and demonstrated the ability of freshly created chondrospheres to undergo fusion when placed together in the presence of HA. Assuming that the treatment of AC affected by OA in several stages remains a major challenge in the engineering of tissues and biomaterials, 27 this finding encourages us to think about the possibilities of putting together the cell culture approaches and autologous spheroid pretreated HA in OA intervention for pain and advanced defect filling.
The identification of new techniques and protocols with better chondrocyte culture conditions has demonstrated greater phenotype maintenance and cellular restoration in AC. 28 We performed the culture protocol of 3D chondrocytes isolated from human AC with an advanced degree of OA. We observed a physiological response of the chondrosphere, maintaining the feedback and proliferation mechanism of in vitro cartilage. The initial chondrosphere dimension between 350 and 400 μm corresponds to the physiological limits required for nutrition and diffusion of oxygen in the 29 chondrosphere. Maintenance of the chondrosphere diameter (~ 400 μm) after the seventh day of culture in the HA group contrasts with the control group, whose diameter presented a physiological decrease. The decrease in the physiological diameter of the chondrosphere in 7 days suggests loss of feedback with cell compaction and alteration of its phenotype. 30
The HA and other biomaterials can potentiate processes that promote the formation of AC, providing an effective means of proliferation and spheroid organization for OA. 30 Even with increasing viscosity in the HA culture, there was no impact in the chondrosphere fusion, suggesting that this characteristic does not change the physiological organization of AC. In addition, the 3D-based culture showed greater chondrocyte viability in the groups that received higher concentrations of HA (500 μM) when compared with the usual in vivo dose (between 50 and 100 μM) and with the control group and could be used to improve the quality in this cell construct and interfered directly with the effectiveness of the culture.
Apart from local effects at the injection site, including local pain and swelling, no important side effects were reported in any of trials using HA, indicating that the intra-articular therapies in clinical practice appear to be relatively safe. 31 Although the safety of HA intra-articular use in OA management is well established, single studies reported transitory beneficial effects in favor of HA injections alone in the treatment outcomes of OA joint, suggesting temporary improvement of motion and joint warmth after 12 and 24 weeks.32,33 No statistically significant difference was found between HA injection treatment and placebo for all other types of disorders, including gastrointestinal, cardiac, vascular, respiratory, nervous system, skin and subcutaneous tissue, musculoskeletal and connective tissue, renal and urinary system disorders, and hypersensitivity reaction. 31
Taken together, intra-articular HA injection presents an alternative local treatment option providing symptomatic benefit, although those associated with analgesic and corticosteroid interventions were often used to achieve great results. 31 Intra-articular injections of corticosteroids, such as triamcinolone hexacetonide and methylprednisolone acetate, are also often prescribe; however, analgesics and nonsteroidal anti-inflammatory drugs are particularly poorly tolerated by OA patients,34,35 who are frequently elderly, with comorbidities, and are receiving polypharmacy. Still, systemic absorption occurs following intra-articular corticosteroid injection, which can lead to systemic adverse events, and precautions should be taken in patients with concomitant diseases, such as hypertension and diabetes mellitus.36,37 Based upon trials of variable quality, both intra-articular corticosteroid injections and intra-articular HA injections appear not to be more efficacious than placebo in participants with OA. 38
The positive chondrocyte response in culture makes AC with OA a possible cell donor, especially in culture protocols associated with HA. This suggests that chondrocytes previously damaged by OA grown in HA can be used for implants in areas with a greater need of functional correction. In our study, HA proved to be a viable medium for the chondrocyte, potentiating the culture in relation to the control group. HA directly influences chondrosphere quality and maintenance and may increase the clinical response of implants in patients with OA or isolated joint lesions. In this trend, clinical approaches using spheroids from autologous chondrocytes for the treatment of full-size AC lesions at the knee started to be studied. Siebold et al. 39 presented 13 patients with defect sizes >5 cm2 up to 16 cm2, who were successfully treated with the implanted cartilaginous spheroids. No significant difference in the subjective assessment was observed when they were compared with the outcome reported by patients in the subgroups with smaller cartilage lesions. 39 Despite the limited cohort, the translational application possibly led to convincing clinical short-to-mid-term results with a significant increase in patients’ quality of life, satisfaction, reduction of pain, and improvement in knee function.
Primary collagen are the most abundant ECM proteins, for about two-thirds of the dry weight of mammalian adult AC, mainly collagen II and also minor collagens type IX and XI. Collagen breakdown is considered to be a key step in the OA progression. 40 Type IX collagen is extensively cross-linked with type II collagen that is crucial for the maintenance of cartilage matrix and formation of collagen net. 41 After skeletal growth ended, the synthesis rate of type II collagen by articular chondrocytes drops drastically in the adult tissue; however, some synthesis continues, and this can be accelerated up to 10-fold within 2 weeks after joint injury. 42 In parallel, the reduced level of type IX collagen may contribute to the pathogenesis of OA. 41
Better insight into molecular mechanisms by which chondrocytes act in the integrity of the collagenous component of adult AC is needed. This work did not evaluate the ability of chondrocytes to remodel the collagen at ultrastructural and molecular levels, although it is poorly understood and should be considered when translational insight is proposed. Understanding the turnover of minor collagens in 3D-based culture from human AC may provide novel and translational tool for investigating the effect of known drug targets on cartilage in preclinical or clinical settings, thereby providing proof of principle for test of those drugs in OA clinical trials.
The differentiation of 3D-based culture into chondrogenic and osteogenic lineages is improved in spheroid model when compared with 2D cell monolayers. 43 As spheroids manifest faster cell differentiation in vitro when compared with cell monolayers, the assembly of cells into spheroids upregulates several related proteins, such as type collagen and related proteins, in a period of 3 to 7 days. Compared with 2D cell culture, spheroid 3D cultures are more close to physiological tissue microenvironment, connected with ECM proteins, which ensures effective communication and facilitates intercellular signaling. The ECM from spheroids acts as a scaffold and modulates cell growth and spread. 44 Therefore, the morphogenesis of 3D spheroids is a dynamic process regulated by cell adhesion and differentiation, ECM synthesis, and constant remodeling, resembling in vivo aspects. 45
While the present work showed positive results, additional studies to improve the techniques employed, and prolonged incubation analysis (after 1, 7, 14, and 21 days in culture, at least) should be performed to estimate chondrospheres viability. Furthermore, the translation of laboratory research into clinical trials demands adaptation and standardization of methodologies for spheroid manufacture, as well as for safe application of new regenerative medicine strategies in this field.
A future prospect for the group would be to standardize a methodology and evaluate physiological variables, such as load and supernatant of the media. Controlling in vitro culture settings would expand the cell viability, the intensive accumulation of extra cellular matrix components, and architectural tissue organization. In vitro trials of different types of cartilage with various loads, such as human hand cartilage and intervertebral disk, are being started and ultimately the biomechanical properties control of chondrospheres is intended to replace AC defects on developing cartilages tissues to mimic the rolling and squeezing action of articular joints and could be safely used to improve the quality and application of the 3D chondrosphere construct.
Footnotes
Acknowledgments and Funding
The authors thank Dr. André De Martini for his support in Orthopedics. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We recognize and thank Mr. Leonel I.S. Nascimento/Rotary International-D4700 for technical contributions to LATEC/UCS.
Author Contributions
A.F., L.d.N., N.F.N., and M.P.-F. designed the experiments and analyzed the data and wrote the manuscript; A.F. and N.F.N. wrote the revised version of the manuscript; M.P.-F. and N.F.N. contributed in in vitro experiments and technical analysis; D.M. contributed with cell characterization by flow cytometry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.