Abstract
Objective
The objective was to evaluate clinical outcome and safety of arthroscopic, autologous minced cartilage implantation for acetabular cartilage lesions observed during hip arthroscopy to treat femoroacetabular impingement syndrome (FAIS).
Design
Eleven male patients, average age: 29.4 ± 5.4 years, average body mass index (BMI): 24.2 ± 2.2 kg/m2, scheduled for hip arthroscopy due to FAIS accompanied by an acetabular cartilage lesion were included in the case series. Cartilage tissue was harvested and minced from the loose cartilage flap at the chondrolabral lesion by arthroscopic shaver, augmented with autologous conditioned plasma, implanted into the defect, and fixated by autologous thrombin. Concomitant interventions were performed as indicated. The patients were evaluated preoperatively and at 24-month follow-up, using the International Hip Outcome Tool-12 (iHOT-12) and Visual Analog Scale (VAS) pain score and by magnetic resonance imaging (MRI) using the Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) grading scale at the 2-year follow-up.
Results
The defect size was on average 3.5 cm2 (1.5-4.5 cm2). From preoperatively to 2 years postoperatively, the iHOT-12 significantly improved from 50.2 ± 18 to 86.5 ± 19 (P < 0.0001), and pain score decreased from 5.6 ± 1.8 to 1.0 ± 1.5 (P < 0.0001) on the Visual Analog Scale pain score. Regarding functional outcome and pain, 10 of the 11 patients and all patients reached the minimal clinically important difference (MCID), respectively. The postoperative average MOCART score was 87.2 (± 9.2). No adverse events or reoperations were observed.
Conclusions
Arthroscopic, autologous minced cartilage implantation for treating full-thickness acetabular cartilage lesions in FAIS shows statistically and clinically significant improvement at short-term follow-up.
Introduction
Femoroacetabular impingement syndrome (FAIS) is a common cause of hip and groin pain and is recognized as a prearthritic deformity.1,2 Hip arthroscopy is considered the gold standard for surgical treatment of FAIS, and recent studies have reported promising long-term outcomes with high rates of hip joint survival.3,4 However, acetabular cartilage delamination caused by shear forces at the chondrolabral junction is frequently observed during hip arthroscopy,5 -7 a finding linked to poor outcomes and significantly negatively affects long-term prognosis following arthroscopic hip surgeries. 8 In the past, microfracture has been predominantly used to treat focal cartilage lesions at the acetabulum. 9 However, due to heterogeneous study results and the fact that subchondral acetabular cyst formation was frequently observed, some authors no longer recommend this cartilage repair technique. 10 As an alternative method, autologous chondrocyte implantation (ACI) has shown good to excellent results in a case series, including treatment of large cartilage lesions. 11 While from a scientific point of view, ACI can be recommended to treat focal cartilage lesions of the acetabulum, the technique has distinct disadvantages in practical application, as it requires two operations for arthroscopic harvesting of cartilage tissue, subsequent in vitro isolation and expansion of chondrocytes and finally reimplantation of processed chondrocytes in a second operation. These circumstances, regulatory obstacles, and high costs restrict the general availability of ACI. 12 Minced cartilage implantation (MCI) has recently attracted attention as a promising one-stage alternative. MCI was first described as open surgery at the knee joint in 1983. 13 The currently evolved MCI technique allows for a minimally invasive, arthroscopic, and completely autologous procedure by utilizing an arthroscopic shaver for harvesting and mincing of the cartilage tissue, patient-specific autologous conditioned plasma (ACP) for augmentation of cartilage tissue, and autologous thrombin to fixate the implanted cartilage tissue into the lesion. 14 Recently, the practicability of this evolved MCI technique to treat cartilage defects at the acetabulum during hip arthroscopy has been described. 15 In the context of FAIS, cartilage lesions are typically observed as an acetabular cartilage lamella, which is well suited to be harvested by an arthroscopic shaver and serves as an ideal source for subsequent MCI as high viability of chondrocyte acetabular lamellae has been documented in the past. 16 In addition, good viability has also been demonstrated for the cartilage overlying the femoral cam morphology,16,17 which can serve as a secondary source of cartilage tissue during arthroscopic femoroplasty and concomitant MCI. Although MCI has been known for 40 years, clinical evidence on the efficacy of this method is low. While several case series for the knee joint yielded promising short-term results,18,19 proof of the efficacy of MCI at the hip joint has yet to be demonstrated. Therefore, this study aimed to assess clinical results, reoperation rate, and possible adverse events in the context of arthroscopic treatment of focal cartilage lesions at the acetabulum by autologous MCI. The hypothesis was that MCI for treating focal acetabular cartilage lesions would reduce pain and significantly improve patient-reported outcomes without facing adverse events.
Materials and Methods
Patient Recruitment
Consecutive patients with clinical and radiological diagnosis of symptomatic FAIS 20 failure of conservative treatment, age >18 years, intraoperatively diagnosed acetabular cartilage damage ≥Haddad 3, 21 Tönnis grade <2, and follow-up >2 years were included in the presented case series. Hip dysplasia (lateral center-edge angle [LCEA] of Wiberg <25°), osteoarthritis (Tönnis grade >1), acetabular cartilage damage < Haddad 3, history of pediatric hip disorders, chronic pain syndrome, and refusal to participate in this study were exclusion criteria for participation in this case series. Prospective data collection was enabled by the clinic’s implemented hip registry, which includes a sampling of demographic data, operative procedures, and patient-reported outcomes on all patients undergoing hip surgery in the executing clinic. The hip registry was approved by the local ethics committee (F-2019-006). Informed consent of all patients was obtained before surgery.
The present case series included 11 consecutive patients who met the above-described inclusion criteria. All patients were male. The mean age of included patients was 29.4 ± 5.4 years, the average body mass index (BMI) was 24.2 ± 2.2 kg/m2, 75% showed osteoarthritis grade 0, and 25% showed osteoarthritis grade 1 according to the Tönnis classification. The mean size of treated cartilage lesions was 3.5 cm2. The intraoperative assessment showed cam-type FAIS in seven and combined FAIS in four patients. Femoroplasty was performed in all patients, and additional acetabuloplasty was necessary in four cases. Furthermore, seven patients underwent labral repair, and one underwent labral reconstruction (Table 1).
Patient Demographic Data and Baselines.

FAIS = femoroacetabular impingement syndrome.
Operational Technique
Two fellowship-trained hip surgeons (A.Z., C.S.) performed all hip arthroscopies in the supine decubitus position using two to three standardized portals. A post-traction system was used. Both the central and the periphery compartments were accessible. Acetabuloplasty, femoroplasty, and a combination procedure were used to treat pincer, cam, and mixed-type FAIS. If preservation of the labrum was feasible, a labrum repair was performed; otherwise, the labrum was reconstructed. Periportal capsulotomies were performed on each patient. An interportal capsulotomy was performed and later restored in the case of a significant cam morphology. If during the diagnostic part of the arthroscopy, an acetabular cartilage lesion characterized as Haddad 3 or 4, measuring 1-6 cm2, and accompanied by an intact or reconstructable labrum was present, the indication for concomitant cartilage repair surgery by MCI was set. For time management reasons at this stage, the anesthetist was asked to draw venous blood from one antecubital region to produce ACP and autologous thrombin. To this end, 45 ml of patient blood was drawn into three 15-ml ACP double syringes (Arthrex) under sterile conditions. The blood was centrifuged according to the manufacturer’s specifications to obtain the supernatant of ACP in the double syringes. The supernatant, which according to the manufacturer, including platelet-rich and leukocyte-poor plasma, was then deducted into the smaller syringe of the closed and sterile double-syringe system. The ACP gained this way was partly further processed to autologous thrombin with the help of the Thrombinator (Arthrex), a device that activates the coagulation cascade. The rest of the ACP was saved for later use. To achieve optimal accessibility of the cartilage lesion, a distal ventrolateral (DVL) portal was established. The loose cartilage flap and cartilage from the border of the treated lesions were debrided with a provided arthroscopic shaver (Sabre 4.0 mm, Arthrex). At this stage, the GraftNet (Arthrex) was interposed into the standard suction of the shaver to collect the cartilage tissue that was intra-articularly harvested and minced by the arthroscopic shaver. The defect size was determined with an arthroscopic measurement probe (Arthrex). The minced cartilage tissue was after that augmented with ACP. After thorough debridement of the cartilage lesion, the ACP-augmented minced cartilage tissue was arthroscopically implanted into the defect with the help of a tuohy application cannula and fixated by autologous thrombin. Two minutes was allowed to achieve primary cartilage and thrombin clot stability before an ACP-thrombin mixture was applied on top of the implanted tissue as described elsewhere. 15
Postoperative Regime
Postoperatively, patients were required to partially bear 20 kg of weight for 6 weeks. Hip flexion was limited to a maximum of 90° for 6 weeks and controlled by a brace. Continuous passive motion was recommended for 4 hours daily to reduce intra-articular adhesion risk. 22 A 3-week course of oral nonsteroidal anti-inflammatory drugs achieved pharmaceutical prophylaxis of ossification.
Outcome Measures
The patient-reported outcome was obtained preoperatively and 2 years postoperatively with the International Hip Outcome Tool-12 (iHOT-12) patient-reported outcome scale and the Visual Analog Scale pain score (VAS, 0-10). The minimal clinically important difference (MCID) was calculated using the half-standard deviation (SD) method as previously described.22,23
In addition, magnetic resonance imaging (MRI) of the hip joints was performed on a 1.5-Tesla MRI scanner at the 2-year follow-up. The Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) grading scale, a well-known instrument for evaluating cartilage healing with strong interobserver reliability and clinical result correlation, was utilized to assess the implanted minced cartilage.24,25
Statistical Analysis
The data were assessed for normality and are presented as the mean ± SD and range. Paired two-tailed t tests assessed differences between the preoperative and postoperative data. All analyses were performed using GraphPad Prism (version 9.3.1; GraphPad Software, San Diego, CA). The level of significance was set to 0.05.
Results
The iHOT-12 significantly improved from 50.2 ± 18 preoperatively to 86.5 ± 19 at 2 years follow-up (P < 0.0001). Ten of the 11 patients (91%) reached the MCID, calculated as 11-point difference in the iHOT-12 scale. According to VAS, the pain was significantly reduced from 5.6 ± 1.8 to 1.0 ± 1.5 at 2-year follow-up. Considering pain, all patients reached MCID, which was a 1.4-point difference on the VAS (Table 2). No intraoperative or postoperative complications were observed. To date, none of the included patients has undergone revision surgery.
Outcome Parameter.
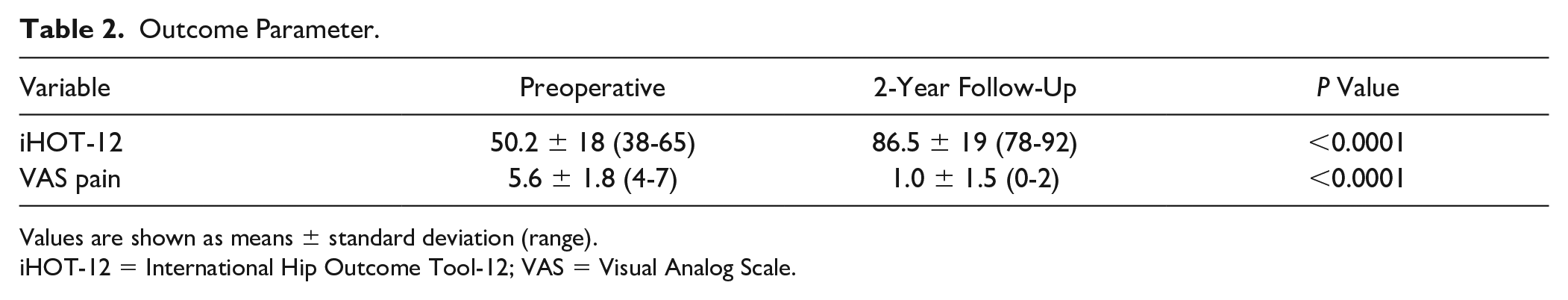
Values are shown as means ± standard deviation (range).
iHOT-12 = International Hip Outcome Tool-12; VAS = Visual Analog Scale.
In the assessment of the MOCART score, an average value of 87.2 (± 9.2) points was obtained at the 2-year follow-up. The filling was complete in 10 of the 11 patients (91%) (Fig. 1). Ten patients showed full integration at the border zone. There were no signs of osteophytes in the repaired cartilage or bone edema.
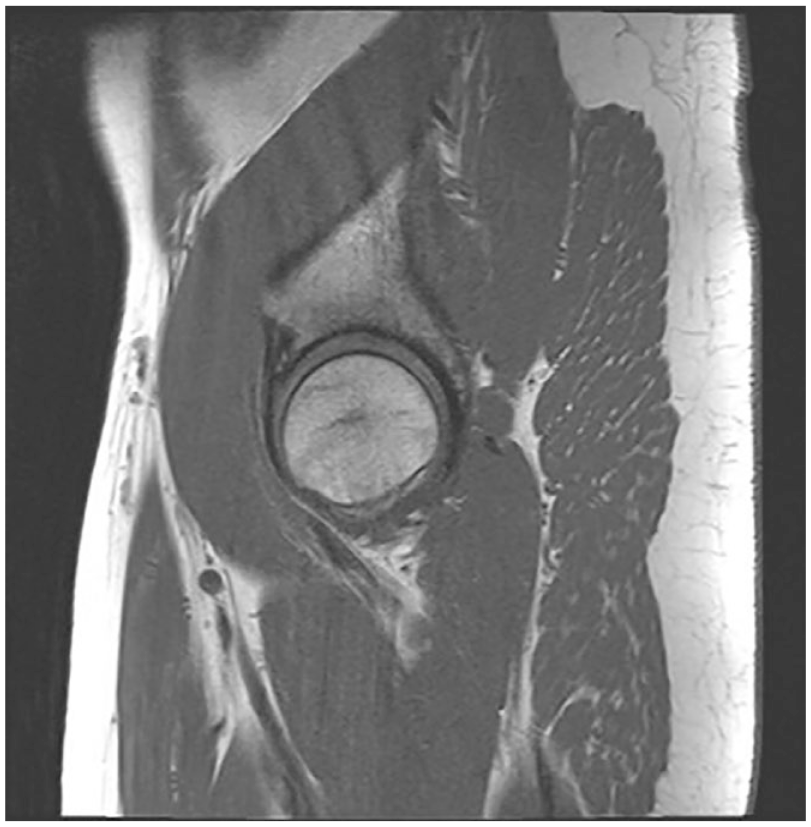
Representative sagittal MRI of a hip joint at a 2-year follow-up after MCI for an acetabular cartilage lesion, showing complete defect filling and a small cystic formation in the adjoining labrum. MRI = magnetic resonance imaging; MCI = minced cartilage implantation.
Discussion
The main findings of this study are that arthroscopic, autologous MCI for the treatment of focal cartilage defects at the acetabulum in combination with arthroscopic treatment of FAIS led to a statistically and clinically significant reduction of pain and improvement of functional outcome of the operated hip joint at 2-year follow-up. No intraoperative or postoperative complications were observed in the 11 patients investigated, and no reoperation was necessary within 24 months.
Most of our MCI knowledge is derived from in vitro investigations. To this end, it was shown that extensively minced cartilage tissue with a paste-like appearance can activate chondrocytes and induce extracellular matrix production from these cells. 26 Another essential factor in MCI is that chondrons contained in the minced cartilage fragments are transplanted. Chondrons are chondrocytes with surrounding pericellular matrix (PCM).27,28 It seems that minced cartilage with most of the PCM intact may induce a better chondrogenic response compared with implantation of isolated chondrocytes such as in ACI Gen 1. If such a difference exists between minced cartilage and the different available third-generation ACI implants remains to be shown.28 -32
Sustained chondrocyte viability has been demonstrated for innovative mincing techniques with a manual hand device 33 and, lately, with an arthroscopic shaver. 34 Possible benefits of the augmentation with autologous platelet-rich plasma (PRP) are uncertain as PRP showed a dose- and time-dependent positive effect on the proliferation and viability of isolated chondrocytes in vitro 35 but did not have an impact during MCI in an animal model. 36 Autologously produced fibrin is particularly suitable for sealing the transplanted cartilage fragments, as this avoids using exogenous protein. 37 Unlike most cartilage defects of the knee joint, the typical cartilage damage observed during hip arthroscopy is a loose cartilage flap at the chondrolabral junction of the acetabulum. Because in vitro investigation has shown sustained proliferation potential and viability of chondrocytes isolated from such cartilage flaps, they can serve as an ideal source of cartilage tissue during MCI, thus avoiding the issue of donor-side morbidity. 16 In addition, good viability has also been demonstrated for the cartilage overlying the femoral cam morphology;16,17 therefore, if femoroplasty is performed, the attached cartilage can serve as a reserve reservoir for cartilage tissue. These findings make acetabular chondrolabral lesion especially suitable for autologous, arthroscopic MCI treatment.
Clinical evidence on the efficacy of MCI is limited. The few available studies focus on the knee joint and report significant improvement of functional outcome at short-term follow-up, comparable to results reported for established cartilage repair techniques like ACI. 38 However, because controlled studies comparing MCI with an established technique are absent, MCI is regarded as a method with potential but needs more supporting evidence to be recommended for specific indications. 39
Unlike for the knee joint, clinical evidence and treatment recommendation on MCI are absent for the hip joint; in the past, microfracture was the standard of care for the arthroscopic treatment of cartilage lesions at the acetabulum. In a comparative case series including 109 patients with acetabular cartilage lesions and FAIS, the results of the microfracture and autologous matrix-induced chondrogenesis (AMIC) were compared. A significant improvement of the modified Harris Hip Score (mHHS) from preoperative 47.6 and 44.5 to 77.0 and 78.8 at 6-month follow-up was observed for both groups. In the microfracture group, the mHHS showed significantly worse values from 2- to 7-year follow-up than the corresponding AMIC group. The conversion rate to total hip arthroplasty was 22% in the microfracture group, while no conversions to hip arthroplasty were documented in the AMIC group. 40 Several clinical studies have shown no advantages of concomitant microfracture over arthroscopic treatment of FAIS without microfracture.41 -45 Microfracture causes significant changes in the subchondral bone and is associated with subchondral cyst formation, probably explaining the observed deterioration of long-term outcome. In conclusion, some authors do not recommend any cartilage repair technique that disrupts the subchondral plate of the acetabulum. 10 It remains to be seen to which extent these findings also account for AMIC, as microfracturing or nanodrilling is a major part of this procedure.
In recent years, ACI efficacy was also investigated for treating larger acetabular cartilage defects measuring 2-6 cm2 with a mean of 4.9 cm2. Good to excellent results were reported at 3-year follow-up, with a statistically significant increase in the mHHS from 64 to 91 points, the iHOT-33 scale from 44% to 86%, and the subjective hip value (SHV) from 54% to 87%. The minimal clinically significant difference was met by 93% of patients according to the mHHS and 100% of patients according to the iHOT-33. 11 The clinical outcome was confirmed by a recent study, which showed complete defect filling in 87.5% of patients and a mean MOCART score of 82.2 in postoperatively performed MRIs at 2-year follow-up. 46 Putting the findings of the executed case series into perspective, it has to be mentioned that the MCI technique does not disrupt the subchondral plate, and thus, adverse effects of subchondral cyst formation that were observed for microfracture should not account for MCI. The clinical outcome demonstrated by the iHOT-12 was improved to a mean of 86.5%, thus comparable to the results of ACI and better than those reported for microfracture or AMIC. Likewise, a similar result was obtained in the MOCART score. Unlike ACI, only one operation per patient was necessary to complete the treatment of acetabular cartilage lesions.
An important point to discuss is that cartilage therapy alone is very rarely performed as a stand-alone procedure in FAIS surgery. Thus, concomitant procedures such as resection and correction of bony morphologies or therapy of the labrum are often necessary, so that usually a combination of several procedures is present. It is thus difficult to determine what influence the individual procedure has on the improvement of the clinical scores. This point applies to all publications mentioned and discussed above. Nevertheless, we observe better clinical scores with the described technique than microfracturing or AMIC and comparable results to ACI. A recent review demonstrated significantly better patient-related outcomes in patients who underwent treatment with bone marrow aspirate concentrate (BMAC), microfragmented adipose tissue concentrate, AMIC, and a combination of AMIC and BMAC compared with microfracture. 47 Therefore, we believe that the cartilage therapy performed may have a significant impact on the outcome, even in the absence of a control group.
Limitations
Although this study features the largest group of patients treated with MCI in the hip, it does have some limitations. One of the limitations is the relatively small sample size. Furthermore, a major weakness of the study is the absence of a randomized control group that compares the effectiveness of MCI with other cartilage therapies. Another disadvantage is the relatively short follow-up period, because for some cartilage repair techniques, initially good outcome and deterioration have been observed. In addition, it is impossible to precisely quantify how concomitant interventions such as labrum repair or femoroplasty influenced the results.
Conclusion
The MCI shows clinically and statistically significantly improved results at 2 years of follow-up for treating acetabular cartilage defects in combination with FAIS surgery. The MCI appears to be especially suitable in the hip joint, as in many cases, a chondrolabral flap is present that can serve as a cartilage source. Furthermore, MCI avoids alterations to the subchondral plate which was shown to be associated with subchondral bone changes and might explain inferior outcome following microfracturing of the acetabulum.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.Z. received personal fees from Arthrex. The other authors report no conflict of interest.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Clinician Scientist Program at University Medicine Greifswald (S.G.).
Ethical Approval
This study was approved by institutional review board (IRB)/Ethik-Kommission Landesärztekammer Baden-Württemberg, Germany.
