Abstract
Objective
The objective of this study was to assess a novel 3D microstructured scaffold seeded with allogeneic chondrocytes (cells) in a rabbit osteochondral defect model.
Design
Direct laser writing lithography in pre-polymers was employed to fabricate custom silicon-zirconium containing hybrid organic-inorganic (HOI) polymer SZ2080 scaffolds of a predefined morphology. Hexagon-pored HOI scaffolds were seeded with chondrocytes (cells), and tissue-engineered cartilage biocompatibility, potency, efficacy, and shelf-life in vitro was assessed by morphological, ELISA (enzyme-linked immunosorbent assay) and PCR (polymerase chain reaction) analysis. Osteochondral defect was created in the weight-bearing area of medial femoral condyle for in vivo study. Polymerized fibrin was added to every defect of 5 experimental groups. Cartilage repair was analyzed after 6 months using macroscopical (Oswestry Arthroscopy Score [OAS]), histological, and electromechanical quantitative potential (QP) scores. Collagen scaffold (CS) was used as a positive comparator for in vitro and in vivo studies.
Results
Type II collagen gene upregulation and protein secretion was maintained up to 8 days in seeded HOI. In vivo analysis revealed improvement in all scaffold treatment groups. For the first time, electromechanical properties of a cellular-based scaffold were analyzed in a preclinical study. Cell addition did not enhance OAS but improved histological and QP scores in HOI groups.
Conclusions
HOI material is biocompatible for up to 8 days in vitro and is supportive of cartilage formation at 6 months in vivo. Electromechanical measurement offers a reliable quality assessment of repaired cartilage.
Keywords
Introduction
Advancements in biomaterial fabrication and tissue engineering procedures have enabled a wide spread of technologies for articular cartilage regeneration. 1 Scaffold-based tissue engineering approach has long been a reasonable approach to restore or improve damaged cartilage tissue in the field of regenerative orthopedics. 2 To improve scaffold biological activity, a selection of biomaterials and manufacturing methods have been analyzed, both of which are essential initial elements for a successful subsequent in vivo application. 3
A cellular component is often included in the fabricated scaffold, thus resulting in a tissue-engineered product (TEP). A positive feedback from scaffolding materials results in a cellular activation and improved potency of a TEP in vitro. This is mainly influenced by a number of factors, such as fabrication method, material, and morphological parameters of a scaffold. 4 Biocompatibility of the scaffold can then be determined by efficacy and potency studies in vitro, which are mostly dependent on adequate amount and type of cells seeded and culture methods. 5 Despite the emergence of stem cells in orthopedic regenerative medicine, somatic cells have convincingly supported a long-term positive preclinical and clinical data for articular cartilage defect treatment. 6 In addition, different approaches to reduce the use of a 2-stage matrix-applied chondrogenesis procedure by combining somatic and stem cells and excluding the need for cultivation has still yet to prove for the long-term clinical outcome.7,8
Direct laser writing in pre-polymers (DLW-PP) technique (also known as 2-photon polymerization [2PP] or multi-photon processing [MPP]) is especially attractive for the fabrication of scaffolds in tissue engineering applications as it enables the predefinition of precise geometry and dimensions.9-12 Versatility of the fabrication method enables the creation of scaffolds, which can be tailored to an individual patient. In addition, recent research on the throughput-augmenting techniques of the DLW-PP has resulted in speeding-up the fabrication of microstructured objects, successfully mitigating the main disadvantage of point-by-point fabrication that was once a bottleneck of this technique.11,13,14 In vitro biocompatibility of the HOI material used for the scaffold fabrication has been shown previously, while effectiveness and safety evaluation relied on preliminary in vivo studies.15,16 TEPs prepared for clinical use must be cultivated for a certain period of time before implantation to ensure optimal potency. Thus, interruptions and delays of transplantation procedure might require prolonged cultivation and adequate evaluation of shelf-life as represented by sustained efficacy and potency in vitro. 17
A quick and reliable method to objectively evaluate several key parameters of cartilage quality, including cellular viability and histological and biomechanical parameters, has been proposed by analyzing the electromechanical properties of cartilage. 18 Cartilage repair quality, by means of electromechanical properties, after treatment with a cellular-based TEP, has not yet been evaluated in any of the preclinical studies reported in the literature.
The primary objective of this work was to compare the biocompatibility of 3D customized DLW-PP scaffold to a routinely used commercially available CS in vitro. The secondary objective was to evaluate repaired cartilage quality in a long-term in vivo preclinical model.
Materials and Methods
Scaffold Manufacturing
The 3D microstructured polymeric scaffolds were fabricated by employing the DLW-PP technique. Computer models were designed using conventional CAD software or specially designed 3DPoli package (Femtika, Vilnius, Lithuania) and created by focusing a femtosecond laser beam into the volume of a photosensitive pre-polymer. Polymerization reaction is initiated due to multi-photon absorption localized within the vicinity of the focal volume, which turns a gel-like material into a solid state. Subsequently, different parts of the material are exposed to the laser light point-by-point until a scaffold of specific microarchitecture is materialized. The DLW-PP setup employed an ultrafast laser, which provided 300 fs, 200 kHz, and 515 nm pulsed light radiation (Pharos, Light Conversion, Vilnius, Lithuania). More specific details about the setup and fabrication process can be found elsewhere. 11 The material chosen for the scaffold fabrication was a hybrid organic-inorganic silicon-zirconium containing photopolymer SZ2080 (FORTH-IESL, Heraklion, Greece), which consists of 20% inorganic and 80% organic parts. 19 To make the material more sensitive to the laser light, it was photo-sensitized with 1% of 2-benzyl-2-(dimethylamino)-4′-morpholinobutyrophenone (IRG, Sigma Aldrich, St. Louis, MO) photo-initiator.
HOI scaffolds were fabricated as hexagonal structures and consisted of 3 identical layers. Each layer had an offset of half a period in relation to the lower layer. The overall scaffolds dimensions were 2.1 × 2.1 × 0.21 mm3 and single hexagon diameter was predefined at 100 µm wide. Height and width of the rod constituting a scaffold structure was set at 15 µm. Predetermined scaffold top pore characteristics were described as a distance between 2 parallel rods and were set at 42 × 49 µm2, which reflected the actual narrowest passage from top to bottom of the scaffold. The typical side pore was set at 51 × 54 µm2 in width and length, respectively ( Fig. 1a and b ).

SEM images of manufactured HOI top (
A commercially available collagen sponge (CS, Septodont, Maidstone, UK) was used as a direct comparator. CS is composed of native, non-denaturated, freeze-dried collagen of bovine origin type I collagen ( Fig. 1c ). CS was cut to the same dimensions to be compared with each other.
CS scaffolds were individually received under sterile conditions, while HOI scaffolds were chemically disinfected in 70% ethanol solution overnight and UV irradiated for 2 hours. The next day scaffolds were washed in phosphate-buffered saline (PBS) solution and left to dry for subsequent cell seeding.
Scaffold Morphology Analysis
Scanning electron microscopy (SEM) on HOI and CS was performed by Hitachi TM-1000 (Hitachi High-Technologies Co., Tokyo, Japan), as previously described. 16
A cell filling of HOI pores was calculated as a percentage of empty and filled pore areas from SEM photographs by 2 researchers at days 4, 8, and 12. CS cell distribution was evaluated at day 12 prior to implantation by histological staining with hematoxylin and eosin dye (H&E).
Cell Isolation, Culture, and Posology
All experimental procedures were approved and conducted according to the standard guidelines and protocols by the Animal Health and Welfare Department, State Food and Veterinary Service of the republic of Lithuania. New-Zealand rabbits (male and female; 4-5 months old; 3-4 kg body weight) were housed separately in cages under ordinary conditions (21 ± 1°C, 12/12 light/dark and a 45% relative humidity) with free access to food and water.
Rabbit chondrocytes were isolated as previously described. 16 Briefly, allogeneic rabbit articular cartilage biopsy from the non–weight-bearing area was minced and digested by 2.5% trypsin (Gibco, Invitrogen, Carlsbad, CA), followed by overnight collagenase XI (Sigma Aldrich) digestion. Isolated cells were plated, cultured, and harvested when 80% confluence was reached. Cells were grown in DMEM/F12 (Hyclone, Logan, UT) supplemented with 10% fetal calf serum (Lonza, Basel, Switzerland), 100 U/mL−1 penicillin and 100 µg/mL−1 streptomycin (Sigma) at 37°C in a humid atmosphere with 5% CO2.
Scaffolds were soaked in proliferation medium 1 day before seeding. Posology was carried by calculating a cell dose required for seeding and was based on the volume of the defect and the corresponding scaffold used. After tripsinization with 0.25% trypsin, third passage cells were counted and resuspended in vials with culture medium at 107 in 1 mL concentration. Cells were seeded in 10 µL doses on presterilized HOI and CS in a dropwise fashion, resulting in 105 cells per scaffold. Seeded scaffolds were placed for 2 hours in the CO2 incubator to allow cellular adhesion before the remaining medium was added.
Seeded scaffolds were cultured up to 12 days. The medium was changed every 3 to 4 days. Scaffolds with cells were harvested after 4, 8, and 12 days of culture and sent for in vitro analysis. Also, on the day 12 of culturing, scaffolds were prepared for in vivo implantation.
Protein Secretion Analysis
Type II collagen production was analyzed at 4, 8, and 12 days after seeding using enzyme-linked immunosorbent assay kit (rabbit collagen type II ELISA kit, BioSite). The test was performed on 7 samples per condition, according to manufacturer’s protocol. The optical density absorbance was read at 450 nm in a microplate reader (Multiskan GO, Thermo Scientific). A standard curve was plotted as the relative optical density of each standard solution versus the respective concentration of the standard solution. Type II collagen concentration of the samples was interpolated from the standard curve.
Gene Expression Analysis
For the chondrogenic gene mRNA analysis, we examined COL2A1 (type II collagen) and COL10A1 (type X collagen) mRNAs’ expression dynamics of cells at day 0 before the seeding on scaffolds and on days 4, 8, and 12 in vitro. The total RNA was extracted from the samples using an ISOLATE II RNA Micro Kit (Bioline, London, UK) according to the manufacturer’s instructions. Designed primer pairs, probes, and the condition of amplification are represented in the supplementary data (Suppl. Table S1). To analyze the data obtained, the 2−ΔΔCT method was applied for the relative gene expression data evaluation. European rabbit GAPDH gene expression was used for data normalization.
Long-Term In Vivo Biocompatibility
Nine rabbits containing 18 bilateral osteochondral defects were used in the study. Surgical procedures were performed aseptically in an operating theatre. Anesthesia was induced intramuscularly and maintained intravenously. Knee joints were approached via the lateral parapatellar approach, followed by medial patellar dislocation. Critical size osteochondral defect (diameter: 3 mm; depth: 2 mm) was created through the articular cartilage and subchondral bone at the weight-bearing area of the medial femoral condyle using an electric drill.
Every defect was extensively washed with saline before implanting a polymerized fibrin clot. Briefly, a mixture 1 mL of autologous blood plasma, 250 µL of thrombin, and 250 µL of CaCl2 were mixed to prepare the fibrin clot. It was incubated for 5 minutes at room temperature just before adding it to the defect to secure the scaffold or fill the scaffold-free defect.
Osteochondral defects were divided into experimental groups based on the randomly received treatment: defects treated using HOI hexagon-pored scaffolds with cultured cells (HOI-cells, n = 4) and without cells (HOI-only, n = 4). Similarly, positive control groups included collagen scaffolds with cultured cells (CS-cells, n = 3) and without cells (CS-only, n = 3). Negative control group comprised defects without scaffolds (scaffold-free, n = 4). After the implantation patella was relocated, knee capsule was closed with interrupted 3-0 resorbable Monocryl suture (Ethicon, Johnson & Johnson Medical, Somerville, NJ). Overlying skin was approximated with 4-0 subcutaneous continuous suture (Ethicon) and disinfected.
After intervention, the rabbits were housed under regular conditions and were allowed to move freely in individual cages. Rabbits were euthanized after 6 months and samples for subsequent examination were collected.
Macroscopic Evaluation of Repair Cartilage
Two independent researchers performed macroscopic grading according to a modified Oswestry Arthroscopy Score (OAS) at 6 months after implantation. The OAS system comprises a distinct evaluation of several cartilage repair parameters: graft level, integration with surrounding cartilage, appearance of the surface, and color of the repair tissue. Stiffness on probing was excluded from the original scoring system in our study, because a more objective electromechanical parameter of the repair tissue was evaluated. The maximum OAS score of 8 represents normal cartilage (supplementary data, Suppl. Table S2).
Electromechanical Analysis of Repair Cartilage
Electromechanical properties of the repair tissue were evaluated with Arthro-BST device (Biomomentum Inc., Laval, Quebec, Canada) 6 months after implantation, as previously described. 20 Briefly, negatively charged proteoglycan molecules in the collagen network are balanced by mobile positive ions in an interstitial fluid. Cartilage compression results in interstitial fluid movement; thus, mobile positive ions are displaced relative to the fixed negative charges. This flow generates streaming potentials which reflect cartilage composition and function.21-23
A higher electromechanical QP mainly reflects increased extracellular matrix disintegration and inferior load-bearing capacity of the cartilage, while low QP indicates strong electromechanical properties and superior load-bearing capacity. However, low QP can similarly be at the lower end of the scale, due to a cartilage thinning until reaching no electromechanical response with complete cartilage loss and bone exposure. 24
Measurements of the weight-bearing area on lateral femoral condyle were made during the surgery at 6 months after implantation, following femoral joint harvest and were used as a healthy cartilage control (n = 9). QP measurements were recorded 3 times on each control and treated defect to obtain median values.
Histological Analysis of Repair Cartilage
For histological analysis, distal ends of femurs were cut above the condyles, fixed in a 10% neutral buffered formalin solution, and embedded in paraffin blocks. Five-micrometer-thick serial sections were deparaffinized and stained with toluidine blue and Safranin-O stain (both Fisher Scientific, Pittsburgh, PA). 25 Sections were analyzed using a digital image microscope (Olympus BX61, Center Valley, PA) equipped with a camera (Olympus DP72, Olympus, Japan) to assess glycosaminoglycans, proteoglycans, and collagen production. Images were scored blindly using a modified O’Driscoll histological scoring system. 26 Higher score indicated superior cartilage repair, with a total maximal score of 24 (supplementary data, Suppl. Table S3). Immunological reaction of host tissue to the scaffold at the implantation site was evaluated from histological sections by infiltration of inflammatory cells.
Statistical Analysis
The quantitative data are expressed as a mean (standard deviation). Statistics were performed using GraphPad Prism 7.04. Statistical results were obtained using Kruskal-Wallis multiple comparison test and presented as the mean and standard deviation (SD). Statistical significance between experimental groups is indicated with (*), which represents a P < 0.05 and (**) representing P < 0.01.
Results
Cell Seeding and 3D Growth Analysis
Cells seeded on hexagonal-pored HOI exhibited a continuous adherence to the horizontal and vertical rods of the scaffold. Proliferation of cells and extracellular matrix (ECM) production within the scaffold have maintained up to 12 days in vitro. The cells could be seen positioned on horizontal and vertical rods, elongated or oval-shape. Connections between nearby rods were mainly made by wrapping and bridging the interconnected nearby perpendicular rods. Cell number and interconnecting ECM content have outgrown all the layers of HOI and kept increasing up to day 12, filling the void space and covering most of the pore diameter at day 12 ( Fig. 2 ).

Cells and interconnecting ECM distribution throughout 3-dimensional hexagonal-pored HOI as a percentage of empty and filled pore areas from SEM photographs at days 4 (
Incremental pore coverage was evident throughout the cultures up to day 12 prior to the implantation. It improved significantly at day 12 compared to day 4 (P = 0.0114) and covered 71 ± 6.5% of a single pore ( Fig. 3 ).
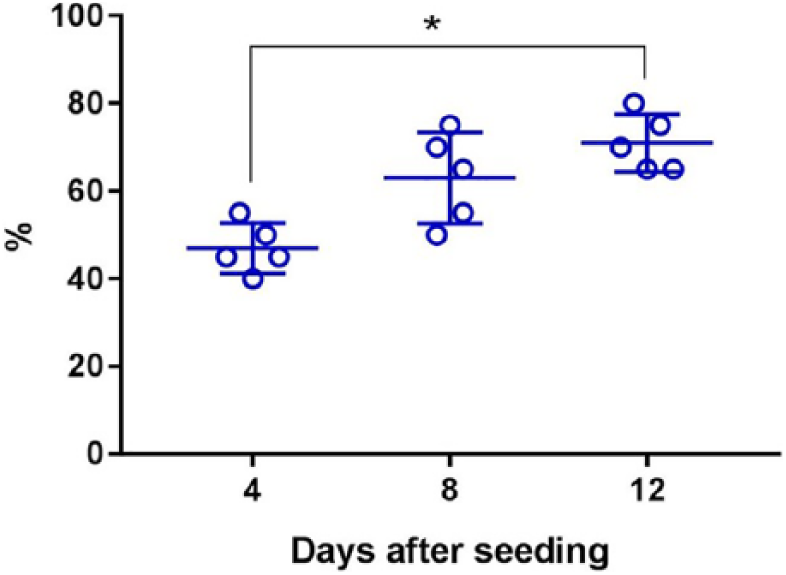
Dynamics of HOI pore coverage by seeded cells. Percentage of filled pore has increased significantly from days 4 to 12, revealing biocompatible morphological conditions for sustained cell proliferation. *P < 0.05.
A positive control of cell-seeded CS revealed random cell distribution from top to bottom and interconnection of nearby collagen folds and creases. Despite all the voids clearly occupied by cells, the bottom layer of CS had fewer cells compared to the top and middle layers (supplementary data, Suppl. Fig. S1). Significantly more iterative pore morphology in HOI allowed superior and more even cell distribution throughout the scaffold compared to CS. Both scaffolds kept physical integrity and did not lose material during in vitro culture.
Cell Seeded Scaffold Potency Analysis
HOI with cells sustained chondrogenesis under regular culture conditions, by measuring type II collagen secretion to the media when compared to the secretion level of cells that were used for scaffold seeding (monolayer cells) ( Fig. 4 ).
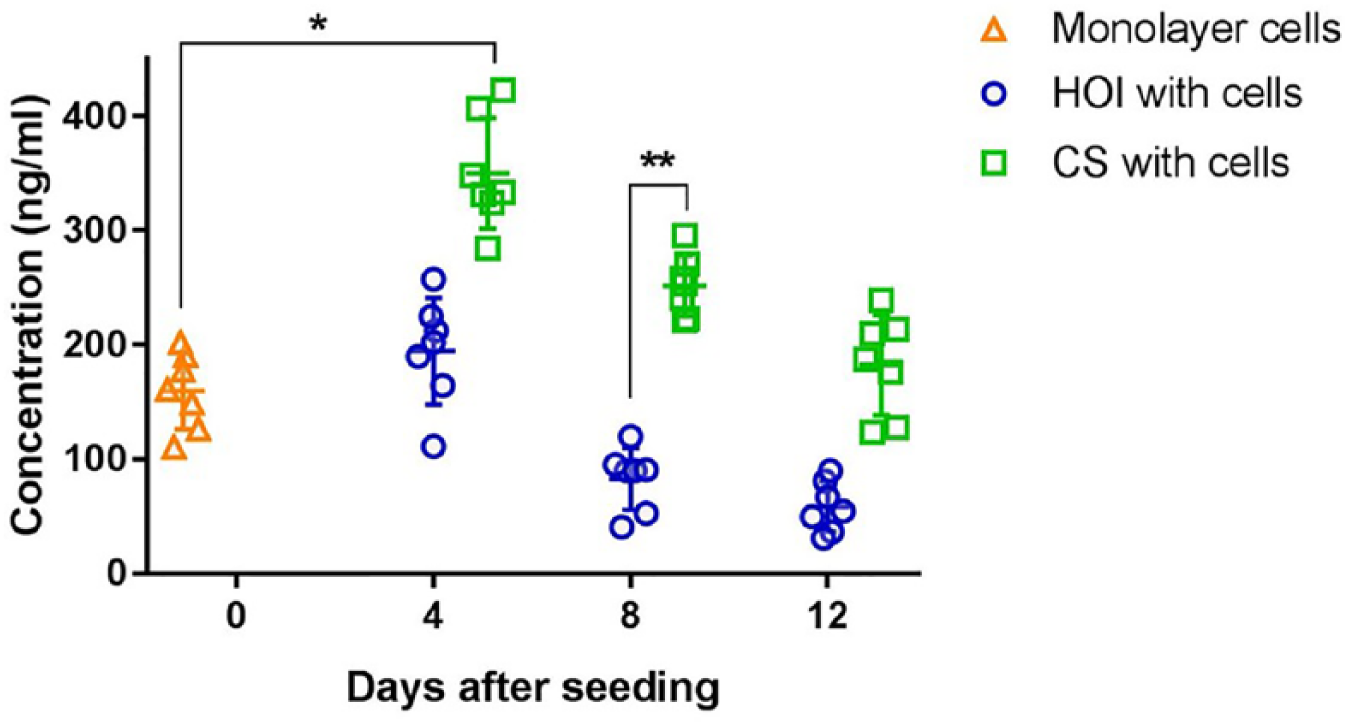
Type II collagen protein secretion, as measured by ELISA in monolayer, HOI, and CS groups at days 0, 4, 8, and 12. Both groups retained the level of secreted protein from days 4 to 12, with a numerical decrease throughout the period. CS had the superior initial type II collagen secretion capacity at day 4 and at day 8, when compared to monolayer cells and HOI, respectively. *P < 0.05, **P < 0.01.
Protein excretion in HOI-cells group retained its level up to day 12; however, a slight decrease was evident throughout the period. Protein secretion improved at day 4 (P = 0.03) in CS-cells, then retained its previous level up to day 12. Numerical protein secretion decrease was also noted in CS-cells up to day 12. When scaffolds with cells were compared to each other, a greater amount of type II collagen secretion was noted in CS-cells group at day 8 (P = 0.0045), highlighting a superior initial phase of cell-scaffold biocompatibility in this group. A numerical superiority was noted in CS-cells at day 12, when compared to HOI-cells at the respective endpoint of the study.
Chondrogenic COL2A1 expression was upregulated in both groups up to day 8 with a numerical decrease at day 12 in both groups ( Fig. 5a ).
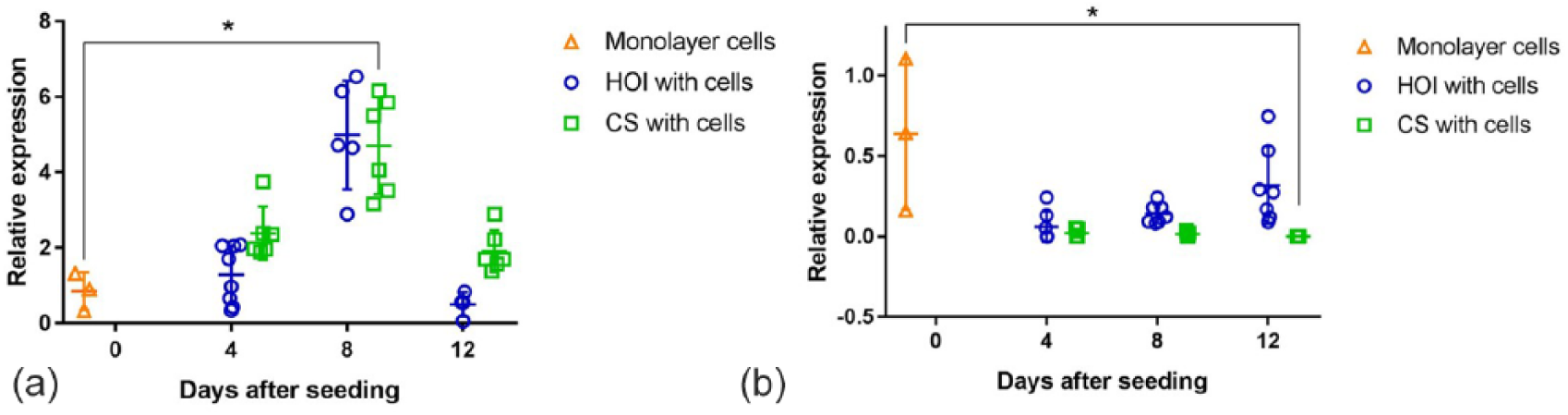
COL2A1 (
Even though the mean level of COL2A1 expression increased in HOI-cells at days 4 and 8, it was comparable to day 0 (P = 0.11). CS-cells retained superior mean level of expression up to day 12, when compared to the gene expression of monolayer cells; however, it improved significantly in the middle of culture period at day 8 (P = 0.0306). No significant differences among the 2 scaffolds were observed; however, mean values were lower in HOI-cells group, compared to CS-cells.
Mean expression of fibroblastic COL10A1 was downregulated in HOI-cells and CS-cells groups, thus supporting cell redifferentiation on a genotypic level ( Fig. 5b ). However, COL10A1 expression remained numerically lower throughout the culture period only in HOI-cells group, thus supporting the improved chondrogenic characteristic. CS-cells group was superior compared to monolayer cells by numerically lower fibroblastic expression values. In addition, it improved significantly, by expressing less COL10A1 at day 12 (P = 0.0355). CS-cells scaffold expressed less fibroblastic gene than HOI-cells group at day 12 (P = 0.0077). In vitro potency analysis values are set in supplementary data (Suppl. Table S4).
Macroscopic Evaluation
No swelling, signs of inflammatory, or immune responses to implanted materials on operated knees were observed at 6 months after implantation. CS-cells group had the highest mean OAS score among all experimental groups ( Fig. 6 ).
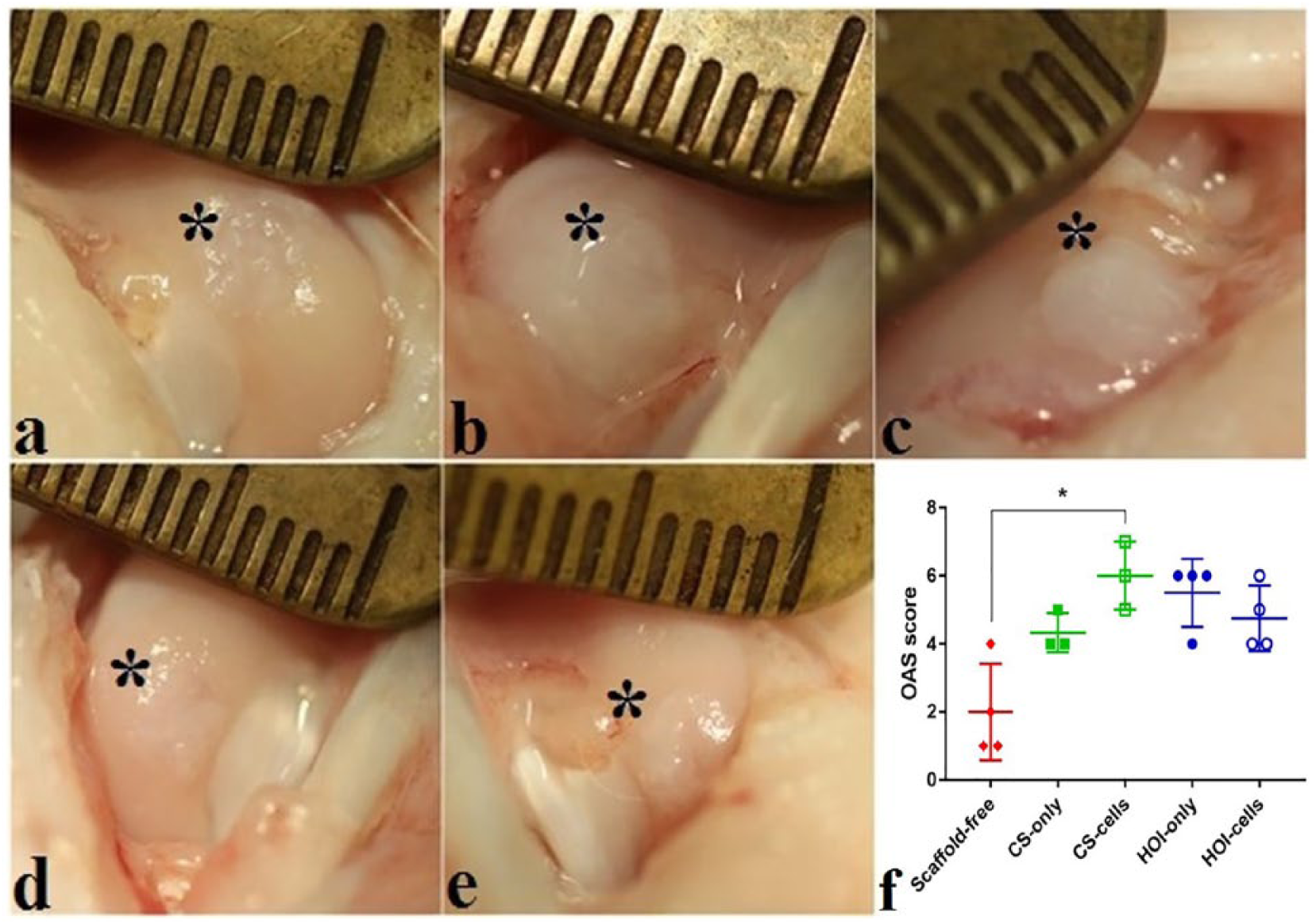
Macroscopic evaluation of experimental groups at 6 months after treatment in (
CS-cells group OAS evaluation revealed the most hyaline-like cartilage, when compared to scaffold-free group (P = 0.0347). Other experimental groups did not differ among each other significantly; however, a tendency for superior outcome in HOI-only (P = 0.076) and HOI-cells (P = 0.69) compared to scaffold-free group was also noted. A numerically inferior outcome in HOI groups was mainly influenced by more fine fronds on the cartilage surface when compared to a smoother cartilage surface in CS groups. Macroscopic values are set in supplementary data (Suppl. Table S5).
Histological Analysis
Mean cartilage repair scores were superior in all treatment groups compared to the scaffold-free group at 6 months; however, a significantly superior histological outcome was scored in CS-cell (P = 0.035) group only ( Fig. 7 ).
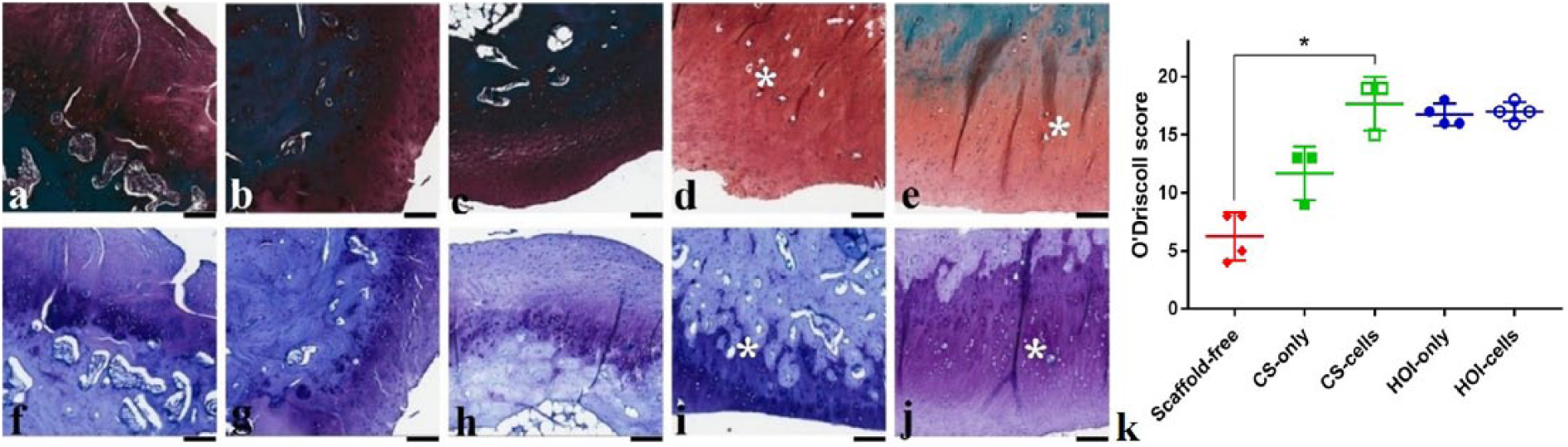
Histological evaluation of experimental groups at 6 months after treatment in (
A clear numerical advantage of HOI-cell group over scaffold-free group was also noted (P = 0.057). Other experimental groups were comparable among each other; however, HOI-only group had a tendency for superior histological restoration compared to the control group, as well (P = 0.103). Restoration of subchondral bone was not achieved in any of the samples, with slight contour changes throughout experimental groups. HOI structural elements were dispersed in cartilage and subchondral bone layers enfolded in the host tissue. No evidence of infection, donor tissue rejection, or immune response was noted in any histological samples of experimental groups. Histological values are set in supplementary data (Suppl. Table S5).
Electromechanical Testing
Electromechanical parameter measured at the cartilage repair sites treated with HOI-only, HOI-cells, and CS-cells groups were lower than untreated scaffold-free group, resulting in superior repair for scaffold-based groups, especially cell-seeded scaffolds ( Fig. 8 ).
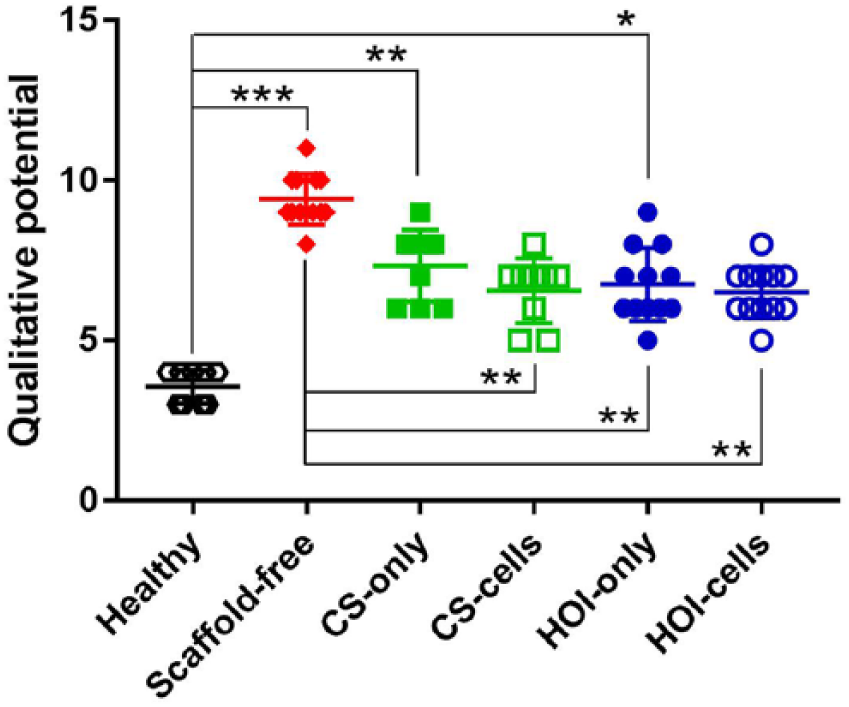
Electromechanical measurement in cartilage-treated areas with Arthro-BST. Electromechanical parameter measured at HOI-only, HOI-cells, and CS-cells was lower than scaffold-free group, thus resulting in superior repair in these groups. HOI-cells and CS-cells exhibited improved values of electromechanical parameter and were comparable to healthy cartilage. *P < 0.05, **P < 0.01, ***P < 0.001.
Significantly inferior electromechanical properties were noted in CS-only (P = 0.002), HOI-only (P = 0.022), and in scaffold-free (P < 0.0001) groups compared to healthy cartilage. However, HOI scaffolds with cells (P = 0.0014) or without (P = 0.0059) exhibited improved values of electromechanical parameter compared to scaffold-free group. Despite the noninferiority between healthy cartilage values compared to CS-cell (P = 0.069) and HOI-cell (P = 0.066) groups, the best repair potential has been shown for cell seeded groups, as expressed by improved intrinsic electromechanical properties of cartilage. Electromechanical values are set in supplementary data (Suppl. Table S5).
Discussion
This study demonstrated a sustained cartilage repair at 6 months after 3D HOI scaffold implantation. This was supported by macroscopical, histological, and electromechanical analysis. To our knowledge, this is the first in vivo study to analyze electromechanical properties of a TEP in a long-term preclinical study.
Chondrocytes have long been a first choice of cells for articular cartilage repair, due to their genotypic and phenotypic compliance to the cartilage treatment area. Morphological, genotypic, and phenotypic profiles of chondrocytes have been well established and in vivo efficacy of cellular TEPs has been proven in long-term in vivo human studies.27-29
Physical and chemical properties of TEP influence cell attachment and the resultant potency in vitro and efficacy in vivo. DLW-PP and SZ2080 material enable predetermining parameters such as pore size, shape, and porosity, which would support the ingrowth of cells and extracellular matrix ECM deposition. A described discrepancy among gene expression and protein secretion prompted PCR (polymerase chain reaction) and ELISA (enzyme-linked immunosorbent assay) analysis in vitro. 30 Despite the decline of protein secretion, HOI retained the same protein translation level as that of monolayer chondrocytes used for seeding. A period up to day 4 should be analyzed in detail, to determine if the spike of gene expression has occurred earlier. From the clinical translation perspective, most scaffolds tend to be cultured for a very brief period after seeding, with the focus on accelerated implantation. This enables faster and more qualitative in vivo redifferentiation, even compared to recently introduced bioreactors, which are still ways of addressing functional and structural mimics of in vivo conditions, after prolonged in vitro culture. 31
Cell-based cartilage regeneration has been best established for chondral defects, requiring additional bone grafting for subchondral lesions. 32 Despite a complicated osteochondral defect used in our study, improvement at 6-month follow-up was evident in most of the experimental groups, especially HOI and CS scaffold groups with cells. Both HOI groups had a tendency for better OAS compared to a scaffold-free group, mainly because of superior tissue integration and smoother cartilage surface in the defect area. Interestingly, OAS score was numerically inferior in a HOI group with cells, when compared to HOI without cells, representing superior macroscopical outcome in HOI without cells group. This might be the result of cells’ interference in a scaffold full integration by suppressing the attractiveness of host tissue cells. A recent study showed an increased fibrosis in a cell-based scaffold, compared to cell-free scaffold, suggesting an inhibitory cell effect on host tissue integration. 33 The property of cell migration toward scaffold should be analyzed in the future studies.
Histologically cell groups had a tendency for superiority over cell-free groups, mainly influenced by the hyaline-like cellular morphology and over 75% of area filled with chondrocytes, both of which are influenced by the addition of cells. HOI structural elements were enfolded both in cartilaginous and subchondral layers, with no signs of inflammation. Nevertheless, subchondral bone was not fully restored and slight bone plate changes were evident throughout all of the samples. Biphasic scaffolds with different mechanical and spatial parameters have been proposed to provide a superior osteochondral repair. 34 Strategies for choosing the right morphological scaffold parameters vary among groups.35-37 In addition, the subchondral interface between bone marrow and calcified cartilage layer containing vessels and innervation must be addressed as well. 38
An increasing number of in vivo experimental TEP studies have prompted a need for a quick and reliable assessment of treatment outcome, other than invasive diagnostic methods, especially in long-term preclinical studies. Electromechanical properties have been shown to reflect cartilage quality and correlate to histological and biomechanical parameters, apoptosis, and inversely correlate to chondrocyte viability.18,20,39 In addition, electromechanical analysis has been shown to be more sensitive than invasive biomechanical testing. 40 In our study, we have observed higher electromechanical potential in the repair cartilage compared to healthy cartilage, revealing incomplete cartilage regeneration. Nevertheless, scaffold groups with cells had a significantly higher score compared to scaffold-free groups, translating to better cartilage quality. In addition, inferiority to a healthy cartilage was only marginal in both groups with cells, revealing satisfactory in vivo cartilage regeneration. Results from Arthro-BST were comparable to histological results of cartilage repair quality in our study. Inclusion of QP measurement has already been applied in explanted human osteochondral cores, and correlation to other cartilage quality parameters have suggested a possible use of this technique to detect underlying cartilage defects, otherwise not visible with a macroscopic analysis. 18 Electromechanical measurement can serve as a diagnostic in vivo method to quantify damaged and regenerated cartilage, by reflecting essential cartilage quality parameters.
Supplemental Material
Supplementary_data – Supplemental material for Osteochondral Repair and Electromechanical Evaluation of Custom 3D Scaffold Microstructured by Direct Laser Writing Lithography
Supplemental material, Supplementary_data for Osteochondral Repair and Electromechanical Evaluation of Custom 3D Scaffold Microstructured by Direct Laser Writing Lithography by Justinas Maciulaitis, Milda Miskiniene, Sima Rekštytė, Maksim Bratchikov, Adas Darinskas, Agne Simbelyte, Gintaras Daunoras, Aida Laurinaviciene, Arvydas Laurinavicius, Rimtautas Gudas, Mangirdas Malinauskas and Romaldas Maciulaitis in CARTILAGE
Footnotes
Supplemental Material
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant (No. SEN-20/2015) from the Research Council of Lithuania.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from Animal Health and Welfare Department, State Food and Veterinary Service of the republic of Lithuania (G2-36).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
