Abstract

Introduction and Summary
Osteoarthritis (OA) is a common degenerative disorder affecting whole synovial joints. Characteristic features include progressive loss of mechanically weakened cartilage, which eventually exposes subchondral bone, and movement results in extreme pain and ultimately joint failure. 1 Understanding early extracellular matrix (ECM) damage is a key research objective, with aggrecan degradation being a hallmark of OA. 2 Early OA shows strong immunostaining for the major proinflammatory cytokines interleukin-1 beta (IL-1β) and tumor necrosis factor alpha (TNF-α), suggesting an important role in OA development. 3 These stimulate production of two groups of proteolytic enzymes, the metalloproteinases and the A Disintegrin and Metalloproteinase with Thrombospondin Motifs (ADAMTS) family, both causing aggrecan degradation. ADAMTS-4 (aggrecanase-1) 4 and ADAMTS-5 have been implicated as aggrecan fragments in OA match those produced by these enzymes. 5 ADAMTS-4 is induced by IL-1β and TNF-α, whereas ADAMTS-5 is constitutively expressed in normal cartilage. 6 Relative mRNA levels of ADAMTS-4 are ~4-fold higher in knee OA than in normal cartilage and correlate with degenerative progression. 7 However, inhibitory elements may be upregulated and posttranscriptional modifications occur, so this does not necessarily constitute higher ADAMTS-4 protein levels. Although ADAMTS-4 knockouts showed no effect against cartilage degeneration in an animal instability model, 8 the pathway(s) for cartilage loss in idiopathic human OA may be different.
Early microscopic changes to chondrocytes in otherwise normal cartilage might herald the start of the vicious cycle of OA. Imaging fluorescently-labeled in situ human chondrocytes by confocal scanning laser microscopy (CLSM) identified a small, but potentially important, population of abnormally-shaped chondrocytes.9,10 They exhibit cytoplasmic processes, with the cells resembling a fibroblastic rather than a chondrocytic phenotype, 11 which might reflect or lead to deleterious matrix metabolism. 12 Abnormal chondrocyte morphology correlated with increased cell-associated IL-1β, decreased chondron-localized collagen type VI, 13 and increased collagen type I. 14 It is possible that IL-1β stimulation results in localized aggrecan depletion, the development/acceleration of abnormal morphology, and a fibro-cartilageous phenotype. Thus in nondegenerate human cartilage, these peculiar chondrocytes may possess increased ADAMTS-4 levels in contrast to normal (elliptical/rounded) chondrocytes. Alterations to ADAMTS-4 levels associated with chondrocyte clusters are also of interest as proliferation is a characteristic of later OA and could cause additional ECM weakness.
Using CLSM imaging, fluorescently-labeled in situ chondrocytes were classified cell-by-cell into those with (1) normal morphology, (2) abnormal morphology (≥1 process/cell), and (3) cells (≥4) in a cluster, within grade 0 (G0; normal nondegenerate) and grade 1 (G1; mild OA) human cartilage. On the same cells, we semiquantitatively assessed ADAMTS-4 immunofluorescence (IF) and determined its relationship with chondrocyte morphology. The results demonstrated marked heterogeneity in the morphology and ADAMTS-4 levels of human chondrocytes. In macroscopically normal cartilage, a small but significant population of abnormal chondrocytes was present possessing higher levels of ADAMTS-4 than normal or clustered cells. These early changes to ADAMTS-4 levels might indicate that ECM regions are more sensitive to mechanical loads and focal points for OA cartilage degeneration.
Methods, Results, and Discussion
Femoral heads (FHs) were obtained from N = 14 patients (8 female and 6 male patients [range 44-89 y; mean 71 y]) undergoing surgery for a femoral neck fracture. Ethical permission (Tissue Governance [NHS], Lothian) and consent were obtained. Cartilage areas were visually graded G0 or G1 using Osteoarthritis Research Society International (OARSI) criteria, and explants were taken. 11 Chondrocyte morphology was identified using 5-chloromethylfluorescein-diacetate (CMFDA). Spheroidal/elliptical cells without cytoplasmic processes were classified as normal cells. A cytoplasmic process was defined as a CMFDA-labeled protrusion of ≥2 μm. Chondrocyte clusters possessed ≥4 cells within the lacunar space. Sections (30 μm) were incubated with ADAMTS-4 antibody (PA1-1749A, 1:100; Invitrogen, Waltham, MA), then incubated with secondary antibody (AlexaFluor ab175471, 1:200; Abcam, Cambridge, United Kingdom). Sections were mounted (propyl-gallate; Sigma-Aldrich, Dorset, United Kingdom), imaged (Zeiss LSM800; Carl Zeiss Microscopy Ltd., Cambridge, United Kingdom), and analyzed (Imaris; Oxford Instruments, United Kingdom). A section from each explant was used as a negative control with background secondary antibody fluorescence subtracted to identify specific labeling. A cell-by-cell mask selection allowed analysis of cell-specific (punctate) IF which localized with CMFDA labeling. For cell clusters, the selection encompassed the whole cluster, and fluorescence was assessed on a “per-cell” basis. Fluorescence intensity was presented in relative fluorescence units. Asterisks represent P ≤ 0.05*, P ≤ 0.01**, and P ≤ 0.001***. FHs (N) and cells (n) were presented as N (n).
There were clear differences between cells in G0 and G1 cartilage ( Fig. 1A(a,b) ). At low magnification, most cells in G0 were normal; however, at high magnification, abnormal chondrocytes were observed ( Fig. 1A(a)(iii) ). In G1, clustering and cytoplasmic processes were detected at low and high magnification ( Fig. 1A(b)(i-iv) ). Chondrocyte morphology classified into normal, abnormal, or clusters was performed by eye. There was a decrease (90%-50%) in normal chondrocytes between G0 and G1 (P < 0.01; Fig. 1B(a) ). However, there was an ~2-fold increase in abnormal cells (P < 0.05; Fig. 1B(b) ) and a >6-fold increase in cells within a cluster (P < 0.001; Fig. 1B(c) ). Occasionally, these cells exhibited processes ( Fig. 1A(b)(ii) ) and were classified as a cell in a cluster. The average length of processes increased from 3.81 ± 0.67 μm to 6.44 ± 1.16 μm (G0 vs. G1 cartilage [4 (77); P = 0.0125]).
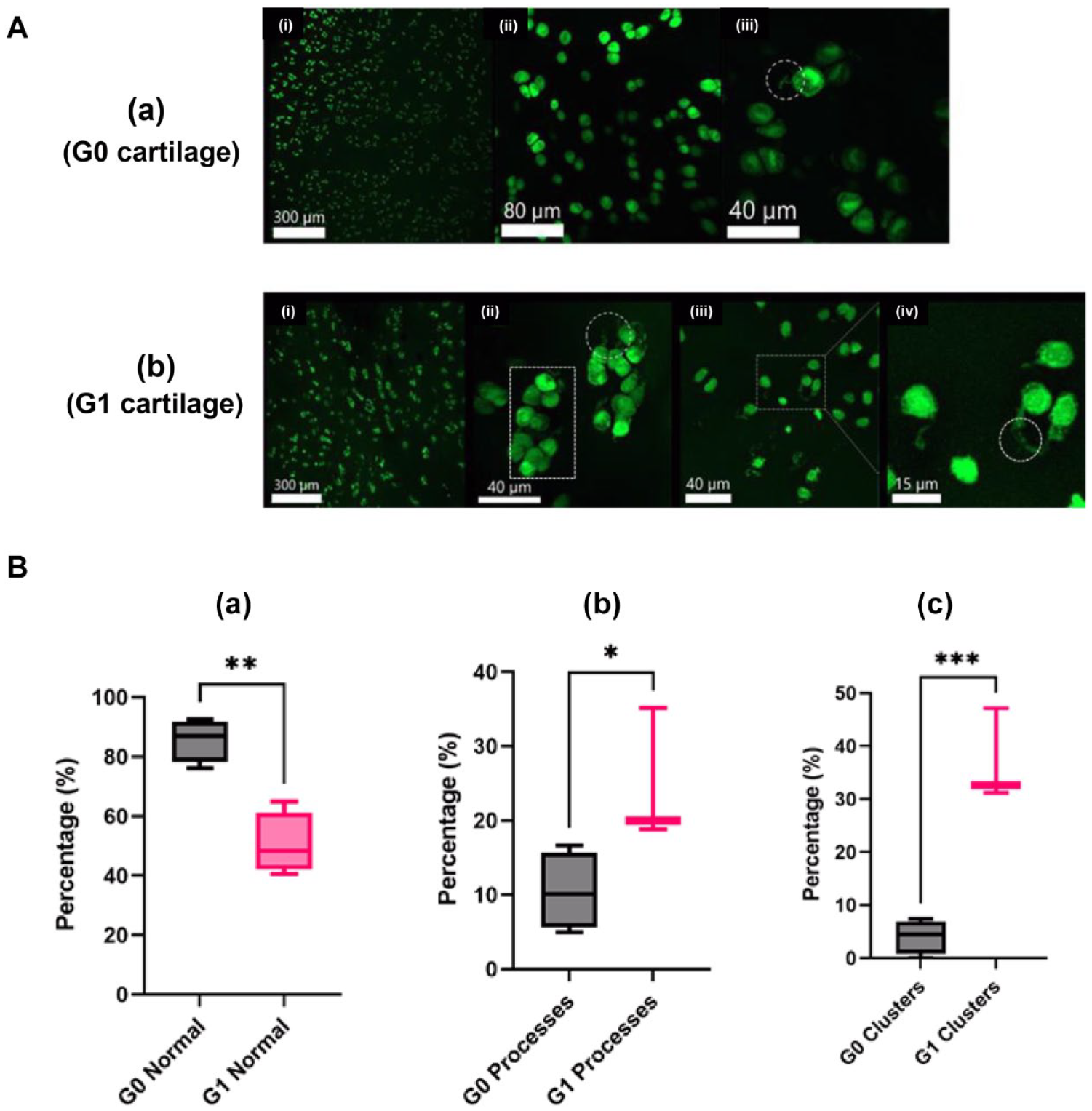
Variations in the morphology of in situ human chondrocytes in non-degenerate and mildly-degenerate articular cartilage. (
Having identified the chondrocyte morphologies, their association with ADAMTS-4 levels was assessed. There was clear ADAMTS-4 immunofluorescence (IF) ( Fig. 2A(ii&iv) ) which matched the CMFDA-labeled cells, appearing as punctate labeling closely delineating the same size/morphology suggesting cell-associated labeling ( Fig. 2A(i&iii) ). CLSM visualization highlighted the heterogeneous nature of ADAMTS-4 labeling. Both diffuse and punctate labeling was evident with strong labeling in some cells close to weakly labeled cells ( Fig. 2A(ii&iv) ). For all cell types assessed in either G0 or G1 cartilage, there was no difference between mean and punctate fluorescence levels (P = 0.5245 and P = 0.4080, respectively). This indicated mean levels over a relatively large area or when measured as cell-associated protein did not change with early cartilage degeneration. However cell-associated (punctate) labeling was higher (P < 0.001) in both G0 and G1 than mean levels which was expected, as it related to cell-specific labeling which covered smaller and more intense areas of labeling. This generalized chondrocyte labeling could mask the heterogeneous distribution of ADAMTS-4 between the various morphologies (Fig. 1A). Accordingly, >4,000 cells within G0 and G1 cartilage were imaged, and morphology and ADAMTS-4 IF analyzed cell by cell. Despite the data spread, abnormal cells demonstrated higher intensity than normal (P = 0.0440) or clustered chondrocytes (P < 0.0001; Fig. 2B(i) ).
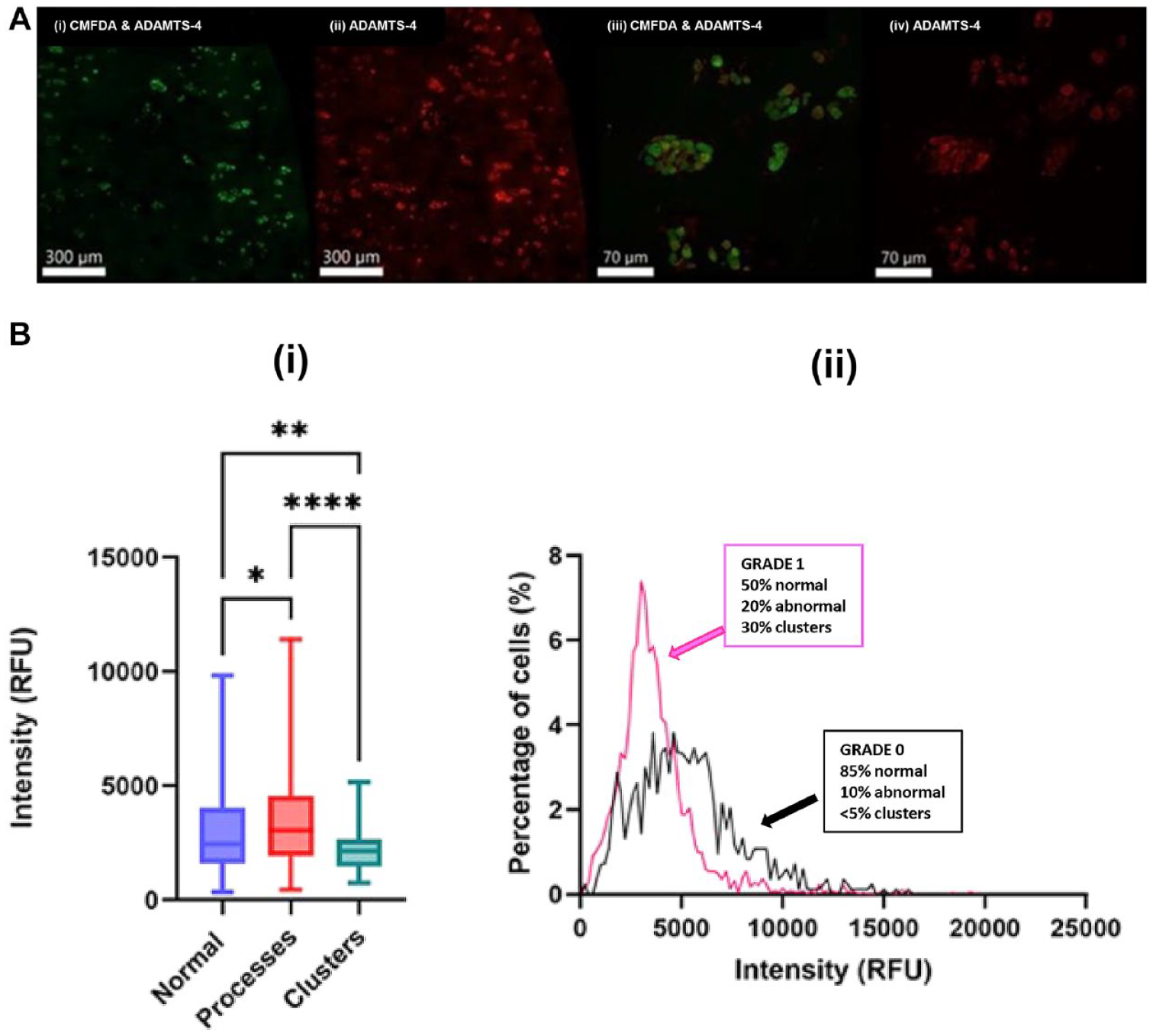
Cell-specific labelling and distribution of ADAMTS-4 for in situ human chondrocytes observed by immunofluorescence and CLSM. Panel (
With cartilage degeneration, there was a marked increase in the proportion of abnormal cells and clustered cells ( Fig. 1B(a-c) ). Thus, in G1 cartilage, it was possible increased numbers of abnormal cells with greater levels of ADAMTS-4 would be offset by the increased number of cell clusters that had lower levels of the enzyme. This could account for the lack of a difference between overall levels of the enzyme between G0 and G1 noted above. Accordingly, the percentages of chondrocytes in G0 and G1 cartilage with a given IF intensity were plotted ( Fig. 2B(ii) ). Although there was a broad distribution in both grades, the levels in G0 cartilage (with normal and abnormal cells, but negligible clusters) was higher and broader than those in G1 cartilage. For G1 cartilage, all three cell categories were present ( Fig. 1B(a-c) ), but there was a higher proportion of cells in clusters possessing lower levels of ADAMTS-4 as reflected in the sharper, lower IF intensity peak. There was no difference between ADAMTS-4 labeling for normal cells (P = 0.9980), cells with processes (P > 0.9999), or cells in a cluster between G0 and G1 (P = 0.9744; [3 (436)]) suggesting morphological/clustering properties of the cells determined ADAMTS-4 levels and not cartilage grade. Although we have graded cartilage, our description of a small population of morphologically abnormal chondrocytes in apparently G0 cartilage ( Fig. 1B(a). ) raises an interesting question about cartilage grading. Chondrocyte death and clustering in histological sections are important criteria for cartilage grading. However, observing subtle changes in chondrocyte morphology requires different approaches such as those described here. We accept the limitation that the G0 cartilage studied here might have microscopic characteristics of very early cartilage degeneration and, as such, might not be “truly” nondegenerate. Nevertheless, we feel that the identification of aberrant chondrocytes provides an important insight into initial changes associated with cartilage degeneration.
CLSM and imaging identified marked heterogeneity in ADAMTS-4 levels which depended on chondrocyte shape and clustering. This approach assesses the responses of single in situ cells and revealed subtle and local properties of chondrocytes obscured with whole tissue samples. Morphologically-abnormal chondrocytes labeled more strongly for ADAMTS-4 than for normal or clustered chondrocytes ( Fig. 2B(i) ). As the chondrocyte population changed with cartilage degeneration (i.e., with more abnormal cells and clusters), this was reflected in a shift to the left of the distribution curve as there was a large number of cells in clusters with lower levels of ADAMTS-4 ( Fig. 2B(ii) ). There was also heterogeneous labeling in each of the chondrocyte categories, but there was no a priori reason that cells in each category should behave identically. Examples showed chondrocytes with no clear ADAMTS-4 labeling beside strongly-labeled cells and even cells within the same lacuna exhibiting varying levels of labeling ( Fig. 2A(iv) ). This could be due to variations in primary and/or secondary antibody penetration. However, heterogeneity was observed in single sections and images where all cells would be exposed to identical antibody levels. A more likely explanation could be that access of factors controlling ADAMTS-4 production are variable and dependent, e.g., on local ECM permeability. Although IL-1β upregulates ADAMTS-4 gene expression in human chondrocytes and cartilage explants, 3 cell-associated levels of IL-1β are very different between in situ human chondrocytes of varying morphology. 13 Thus, IL-1β availability either from intracellular metabolism or an inflammatory response could be variable accounting for the heterogeneity in ADAMTS-4 levels. The estimated t½ of ADAMTS-4 exocytosis is ~220 minutes, 15 and it is possible that there are varying levels of ADAMTS-4 depending on local IL-1β concentrations. We also do not know the aggrecan levels around chondrocytes. However, as they may be reduced in the vicinity of morphologically abnormal cells, this could lead to focal regions of mechanical weakness.
Footnotes
Acknowledgments and Funding
The authors are very grateful to Ms. Fiona Gibson from Edinburgh Orthopaedics for coordinating consent, collection, and delivery of femoral heads. They also thank Dr. Anisha Kubasik-Thayil for assistance with the CLSM. Arthur Herren was funded through the ERASMUS exchange scheme between the University of Edinburgh and the Karolinska Institutet.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was obtained from Tissue Governance [Ref: SR1295-AM02, NHS Lothian] and consent acquired from all patients involved in this study.
