Abstract
Objective
Chondrocytes are responsible for remodeling and maintaining the structural and functional integrity of the cartilage extracellular matrix. Because of the absence of a vascular supply, chondrocytes survive in a relatively hypoxic environment and thus have limited regenerative capacity during conditions of cellular stress associated with inflammation and matrix degradation, such as osteoarthritis (OA). Glucose is essential to sustain chondrocyte metabolism and is a precursor for key matrix components. In this study, we investigated the importance of glucose as a fuel source for matrix repair during inflammation as well as the effect of glucose on inflammatory mediators associated with osteoarthritis.
Design
To create an OA model, we used equine chondrocytes from 4 individual horses that were differentiated into cartilage pellets
Results
We found that elevated glucose levels preserve glucose uptake, hyaluronan synthesis, and matrix integrity, as well as induce anti-inflammatory actions by maintaining low expression of Toll-like receptor-4 and low secretion of glutamate.
Conclusions
Adequate supply of glucose to chondrocytes during conditions of inflammation and matrix degradation interrupts the detrimental inflammatory cycle and induces synthesis of hyaluronan, thereby promoting cartilage repair.
Introduction
In spite of its apparent simple structure, articular cartilage is a complex tissue with intricate extracellular matrix (ECM) composition and cellular organization. Cartilage ECM is composed primarily of type II collagen and an interlocking mesh of proteins and proteoglycans, hyaluronan, and chondroitin sulfate. 1 It is becoming increasingly clear that proteoglycans and hyaluronan generate intricate transduction networks comprising signaling pathways involved in immune responses and inflammatory pathways, such as Toll-like receptor (TLR)-2 and TLR-4 signaling. 1
Chondrocytes constitute the only cell type in cartilage and are quiescent cells with limited turnover of the ECM network in the resting steady state. 2 Chondrocytes respond to mechanical and biochemical changes in ECM through signaling events by various cell surface growth factor receptors and adhesion molecules, 3 thereby maintaining the matrix components under normal, low-turnover conditions. The chondrocyte has a low metabolic activity, surviving under relatively hypoxic conditions and with a nutritional supply solely through the synovial fluid. This cell, which is ultimately responsible for remodeling and maintaining the structural and functional integrity of the cartilage matrix, therefore possesses limited regenerative capacity. 4 During conditions of abnormal cartilage stress, such as osteoarthritis (OA), chondrocytes undergo a phenotypic shift, characterized by increased cell proliferation, cluster formation, and increased production of both matrix proteins and matrix-degrading enzymes, resulting in cartilage degradation. 5
OA is a low-grade inflammatory progressive disease, frequently associated with joint pain, swelling and stiffness leading to significant functional impairment and disability.
6
The correlation between the increased levels of catabolic enzymes and inflammatory mediators such as prostaglandins, nitric oxide, and increased levels of interleukin-1β (IL-1β) and tumor necrosis factor (TNF)-α in OA synovial fluids and joint tissue is well documented.
7
In addition, chondrocytes produce cytokines, chemokines, alarmins, prostanoids, and adipokines and express numerous cell surface receptors for cytokines and chemokines, as well as toll-like receptors (reviewed in Houard
Because of the absence of vascular and lymphatic supply in the adult cartilage, distribution of nutrients to the chondrocytes occurs solely by diffusion from synovial fluid. The adequate provision of glucose to articular chondrocytes is essential to sustain their predominantly anaerobic metabolism; glucose is also a precursor for the ECM macromolecules synthesized by these cells.8-10 Thus, a higher demand for glucose would be expected in OA and other inflammatory conditions but might be limited for chondrocytes due to the avascular nature of the cartilage tissue. Limited glucose supply might compromise cell function and potentially result in an imbalance of matrix synthesis and degradation, leading to osteoarthritis. 11 It is therefore plausible that a limited supply of glucose largely contributes to the limited repair capacity of the matrix by the chondrocytes during OA.
Here, we investigate the importance of glucose as a fuel source for matrix production and cartilage repair during conditions of inflammation associated with OA in a 3-dimensional (3D)
Methods
Tissue Biopsies and Chondrocyte 3-Dimensional Culture
Macroscopically normal cartilage samples were obtained from 4 horses (2-4 years old). Isolation and expansion of chondrocytes were performed as previously described. 12 The study protocol was approved by the Ethical Committee on Animal Experiments, Stockholm, Sweden (N378/12). The protocol for 3D chondrocyte pellet cultures was slightly modified from previously described procedures. 12 Expanded chondrocytes (seeded 300,000 cells/pellet) were cultured for approximately 3 weeks in a 96-well, ultra-low attachment, flat bottom plate (Corning Life Sciences, Lowell, MA, USA) at 37°C in a chondrogenic medium, consisting of Dulbecco’s modified Eagle’s medium (DMEM) with 25 mM glucose concentration (Gibco or Lonza), supplemented with 14 µg/mL ascorbic acid (Sigma-Aldrich), 10−7 M dexamethasone (Sigma-Aldrich), 1 mg/mL human serum albumin (Equitech Bio, Kerville, TX, USA), 1× insulin–transferrin–selenium (Gibco, Invitrogen), 5 µg/mL linoleic acid (Sigma-Aldrich), 1× penicillin-streptomycin (GE Healthcare Life Sciences HyClone South Logan, Utah, USA), and 10 ng/mL human transforming growth factor (TGF)β-1 (R&D Systems, Abingdon, UK) to form a 3D pellet and generate a defined matrix.
When differentiated 3D chondrocyte pellet were formed, pellets were stimulated with 5 ng/mL IL-1β (R&D Systems Inc., Minneapolis, MN) in DMEM media with 5 mM or 25 mM glucose concentration, supplemented with the additives described above, for 72 hours.
Glucose Uptake
Glucose uptake in 3D equine cartilage pellets were performed essentially as described earlier for monolayer cultured chondrocyte cells.
13
The 2-deoxy-
Isolation of RNA
Frozen cell pellets were homogenized using 3-mm stainless steel beads and TissueLyser (Qiagen, Valencia, CA, USA). One milliliter QIAzol (Qiagen, Valencia, CA, USA) was added followed by 0.2 mL chloroform and the mixture was centrifuged at 13,000 rpm for 15 minutes at 4°C. Total RNA was extracted using RNeasy Micro Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions. DNAse I was used to remove contaminating genomic DNA from the isolated RNA. RNA concentrations were measured with NanoDrop 1000 (Thermo Fisher Scientific, Waltham, MA, USA), with a 260/280 nm ratio between 1.7 and 2.1 considered a good purity.
Quantitative Real-Time Polymerase Chain Reaction Analysis
cDNA was prepared from total RNA using High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). Commercially available equine TaqMan Gene Expression Assays all from Applied Biosystems were used (see table below for details of primers and probes). Samples were analyzed in duplicates. The relative comparative CT method was used to analyze the real-time polymerase chain reaction (PCR) data.
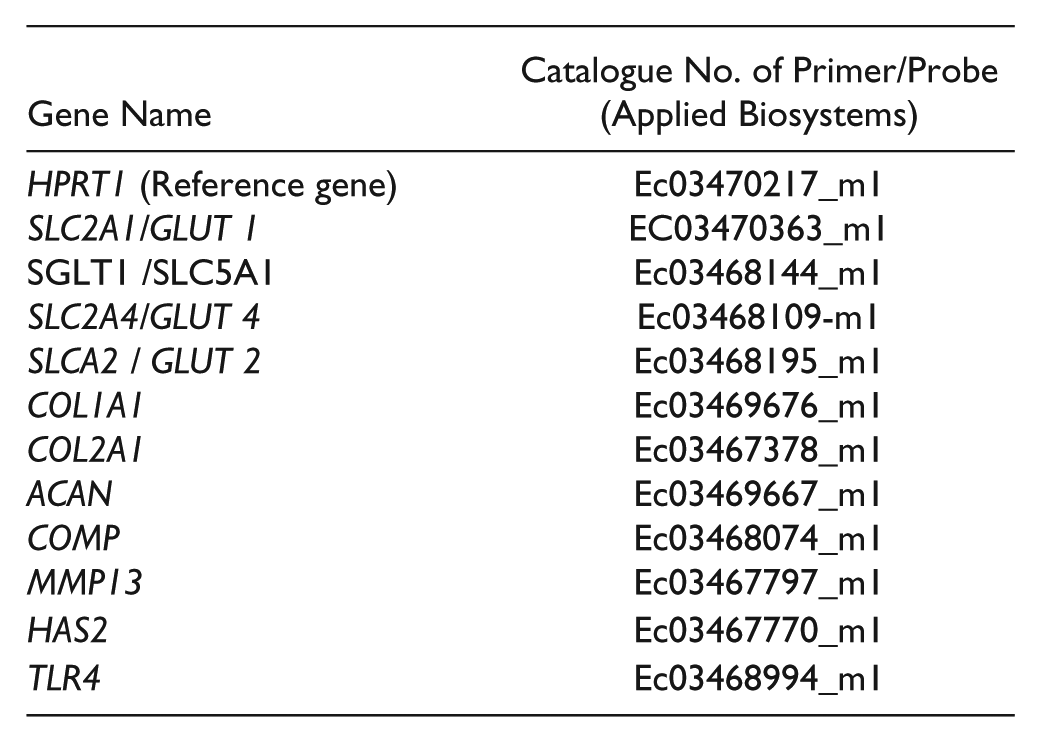
Gene Expression of Glucose Metabolism Using PCR Array
Expression of genes involved in glucose metabolism was analyzed by RT2 Profiler PCR Array (Qiagen, Valencia, CA, USA). cDNA for this analysis was prepared by RT2 First Strand Kit (Qiagen, Valencia, CA, USA). Gene expression was analyzed by the real-time-PCR ViiA7 system (Applied Biosystem, Foster City, CA, USA).
Analysis of Protein Interaction Network
To investigate plausible interactions between mediators of glucose metabolism and hyaluronan synthesis mediators, the search tool STRING was used. 14 Analysis settings were set for evidence-based interactions (indicated by lines between nodes) from textmining (yellow), gene neighborhood (green), experimentally determined (pink), gene fusions (red), curated databases (light blue), co-occurrence (dark blue) and co-expression (black). Colored nodes = query proteins and first shell interactions. Small nodes = proteins of unknown 3D structure. Large nodes = some 3D structure is known or predicted.
Glutamate Analysis
Cell culture medium was treated with sulfosalicylic acid and the free amino acids were separated with ion change chromatography in an automatic analysis system. Glutamate was detected with ninhydrin in a photometer at 570 nm at Clinical Chemistry Lab, Sahlgenska University Hospital, Gothenburg, Sweden.
Hyaluronan Analysis
The concentration of hyaluronan in cell culture medium was determined by a Quantikine ELISA according to according to the manufacturer’s instructions (R&D Systems Inc, Minneapolis, MN, USA).
Lactate Analysis
Lactate concentration in cell culture medium was determined by an enzymatic colorimetric method using lactoxidase, peroxidase, and hydrogen peroxide as enzymes and substrate. The color change was detected at 552 nm (Clinical Chemistry Lab, Sahlgenska University Hospital, Gothenburg, Sweden).
Histological Staining
Sections of chondrocyte pellets were stained with safranin O to visualize proteoglycan content and evaluated with light microscopy as described in Ley
Equine cartilage explants were incubated for 24 days in 5 mM glucose or 25 mM glucose with or without 10 ng/ml IL-1β. The explants were sectioned and stained with hematoxylin and eosin, followed by light microscopy analysis.
Statistical Analysis
All data are presented as mean ± standard error of the mean (SEM). One-way analysis of variance with multiple comparisons was used for statistical analysis. A
Results
To create an OA model, we used equine chondrocytes from four healthy individual horses that we differentiated into cartilage pellets
Elevated Levels of Glucose Rescue Glucose Uptake Following IL-1β Stimulation
We found that glucose uptake was decreased by 50% following IL-1β stimulation at 5 mM glucose, whereas glucose uptake remained sustained following IL-1β stimulation at 25 mM glucose ( Fig. 1A ).
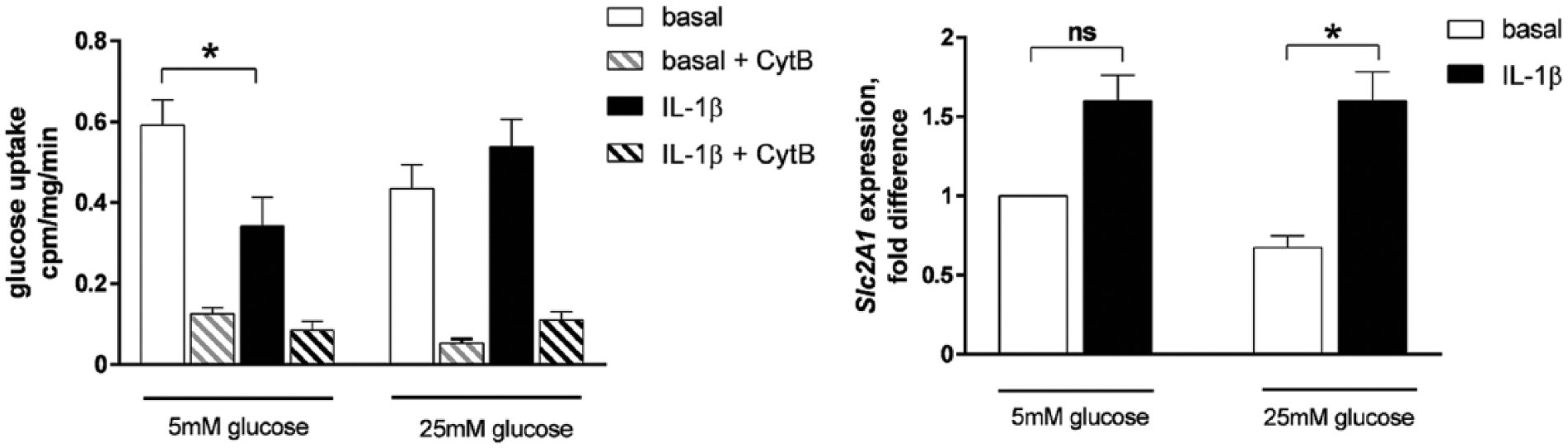
Glucose uptake in response to interleukin-1β (IL-1β) at 5 mM and 25 mM glucose. Equine chondrocytes were differentiated into cartilage pellets
Blocking glucose transporters (GLUT) with cytochalasin B resulted in a marked diminished uptake of glucose for all conditions ( Fig. 1A ), whereas inhibitors of sodium-dependent glucose cotransporters (SGLTs) had no effect (data not shown). Thus, the majority of the glucose taken up in these cells as well as the effect of IL-1β on glucose uptake occurred through GLUT in the chondrocytes. We found the GLUT-1 gene to be robustly expressed ( Fig. 1B ), whereas GLUT-2 or GLUT-4 were not expressed in the differentiated chondrocytes (data not shown). GLUT-1 gene expression was increased following IL-1β stimulation at 25 mM glucose ( Fig. 1B ).
Elevated Glucose Concentration Sustains Matrix Production Following IL-1β Stimulation
We hypothesized that the sustained glucose uptake at 25 mM glucose following IL-1β stimulation was associated with an intact expression and production of cartilage extracellular matrix molecules, which are otherwise known to be subjected to degradation during inflammatory states such as OA. Chondrocyte pellets subjected to IL-1β treatment showed increased staining intensity, larger size, as well as a more intact outer matrix cell layer when incubated at 25mM glucose concentration compared to 5 mM glucose conditions as revealed by histological analysis using safranin O staining ( Fig. 2A and B ). Hematoxylin and eosin staining of equine cartilage explants showed modest fibrillation of the superficial layer of the articular cartilage following IL-1β stimulation at 5 mM glucose, whereas the articular cartilage was intact at 25 mM glucose ( Fig. 2C ), supporting our data for differentiated chondrocytes in cell culture. These data suggest that 25 mM glucose concentration protects the extracellular matrix from degradation during inflammatory conditions.
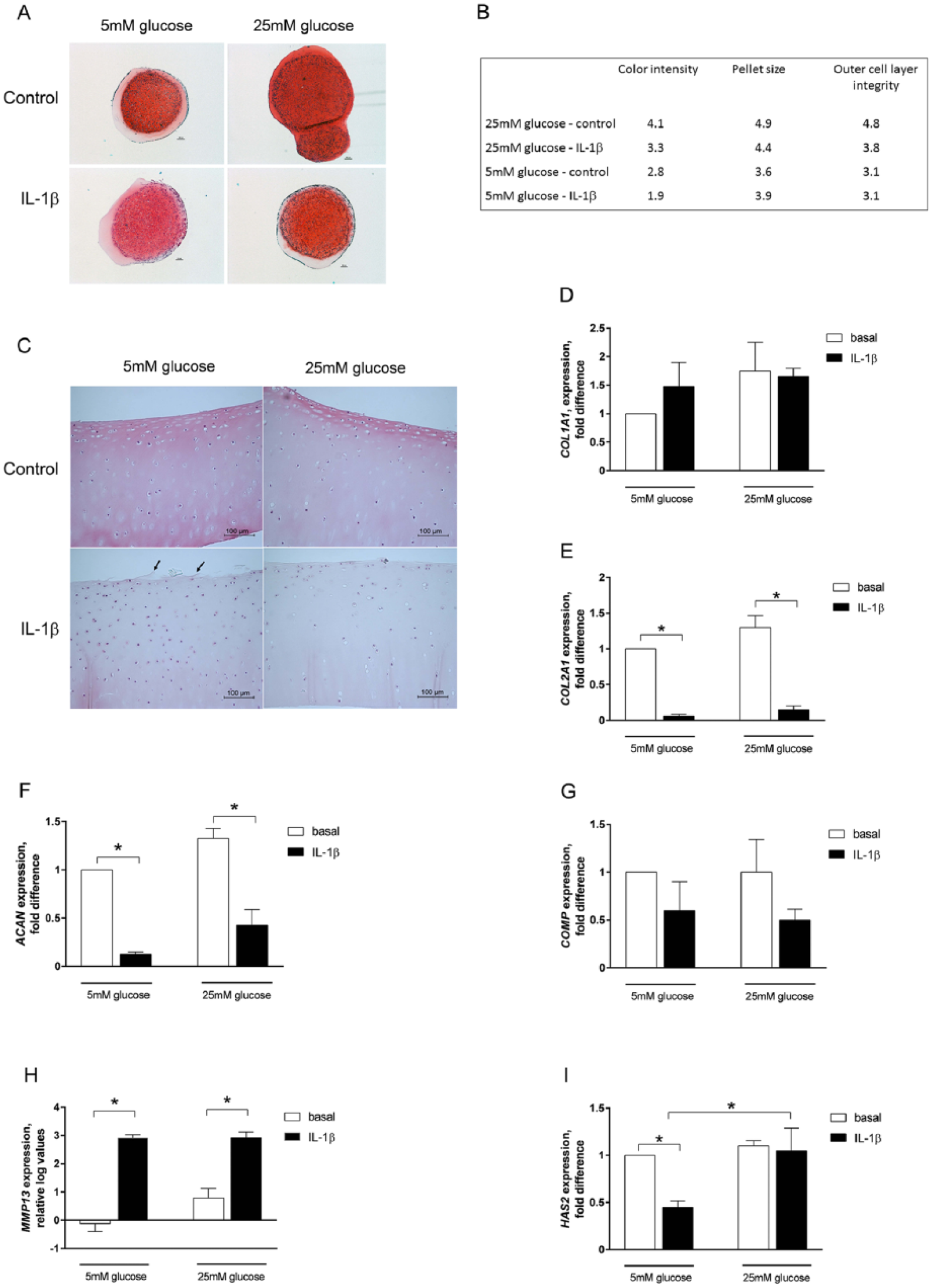
(
At 5 mM glucose, IL-1β stimulation resulted in a marked decreased expression of the key matrix genes
Sustained HAS2 Gene Expression in Response to Elevated Glucose Levels Is Accompanied by Anti-Inflammatory Signals
Based on the
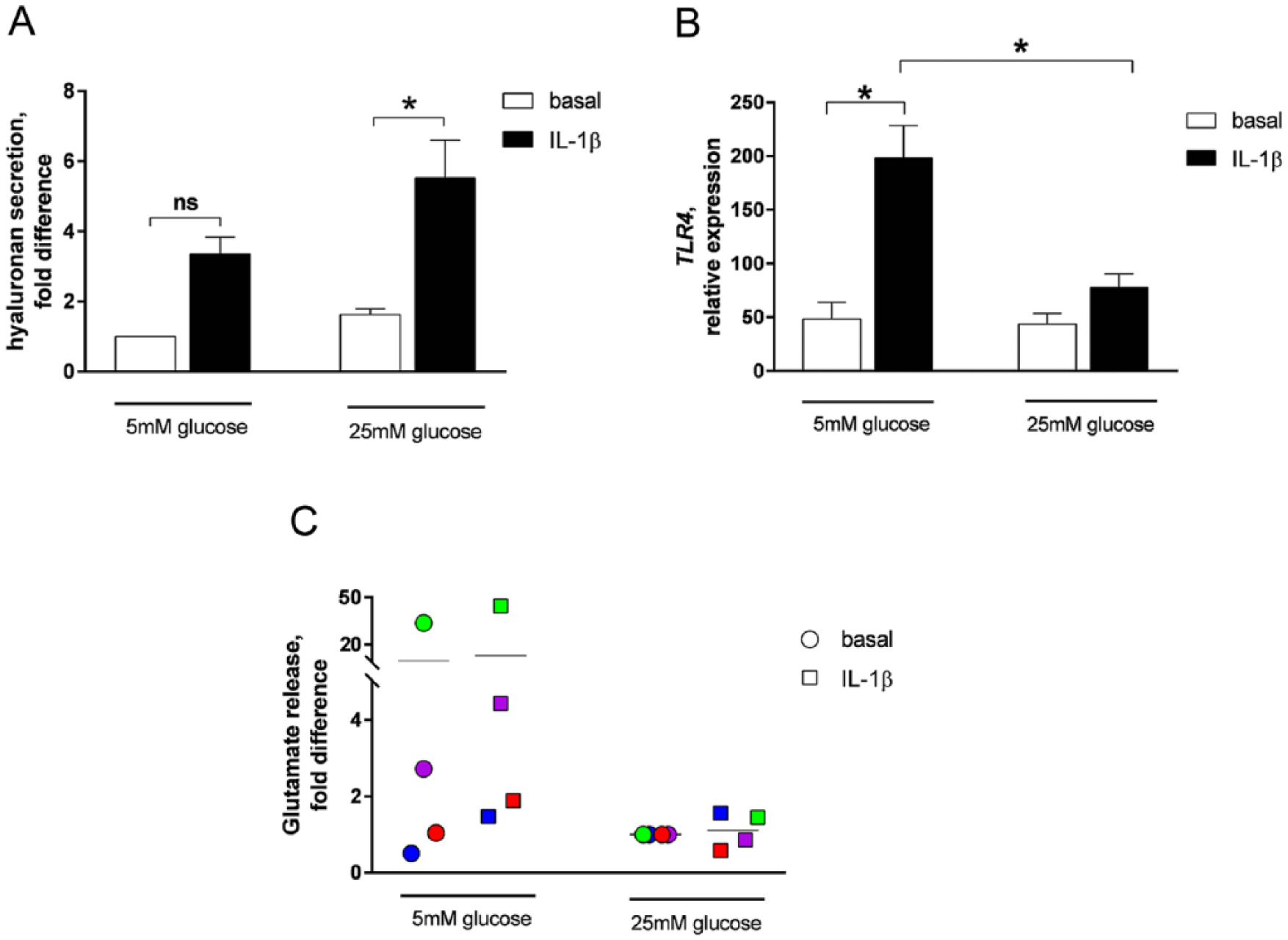
Evaluation of anti-inflammatory activities. Equine chondrocytes were differentiated into cartilage pellets
Taken together, hyaluronan secretion from differentiated chondrocytes following IL-1β stimulation is increased at 25 mM glucose compared with 5 mM glucose. The sustained
Elevated Glucose Levels Result in an Increased Glycolytic Rate in Differentiated Chondrocytes
To identify mechanistic mediators of the association between the sustained glucose uptake and
Equine Chondrocytes were Differentiated into Cartilage Pellets
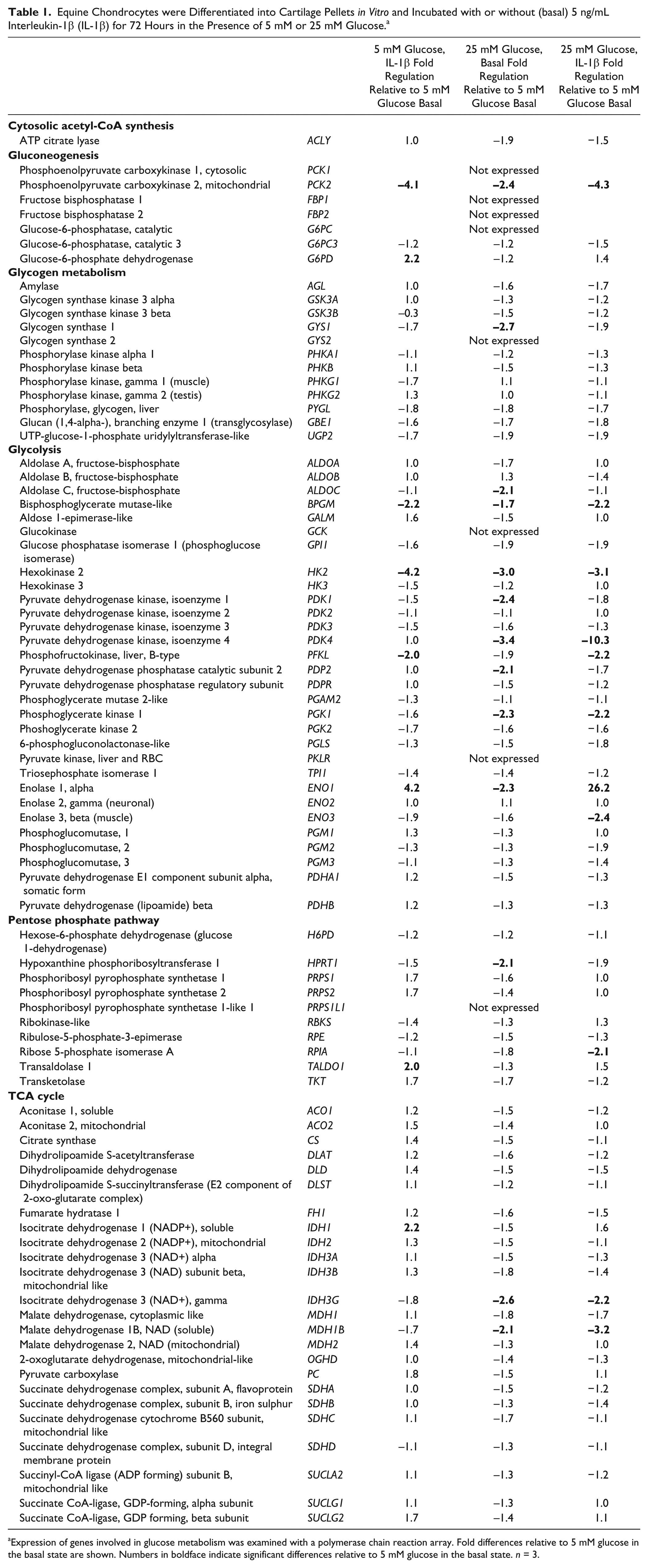
Expression of genes involved in glucose metabolism was examined with a polymerase chain reaction array. Fold differences relative to 5 mM glucose in the basal state are shown. Numbers in boldface indicate significant differences relative to 5 mM glucose in the basal state.
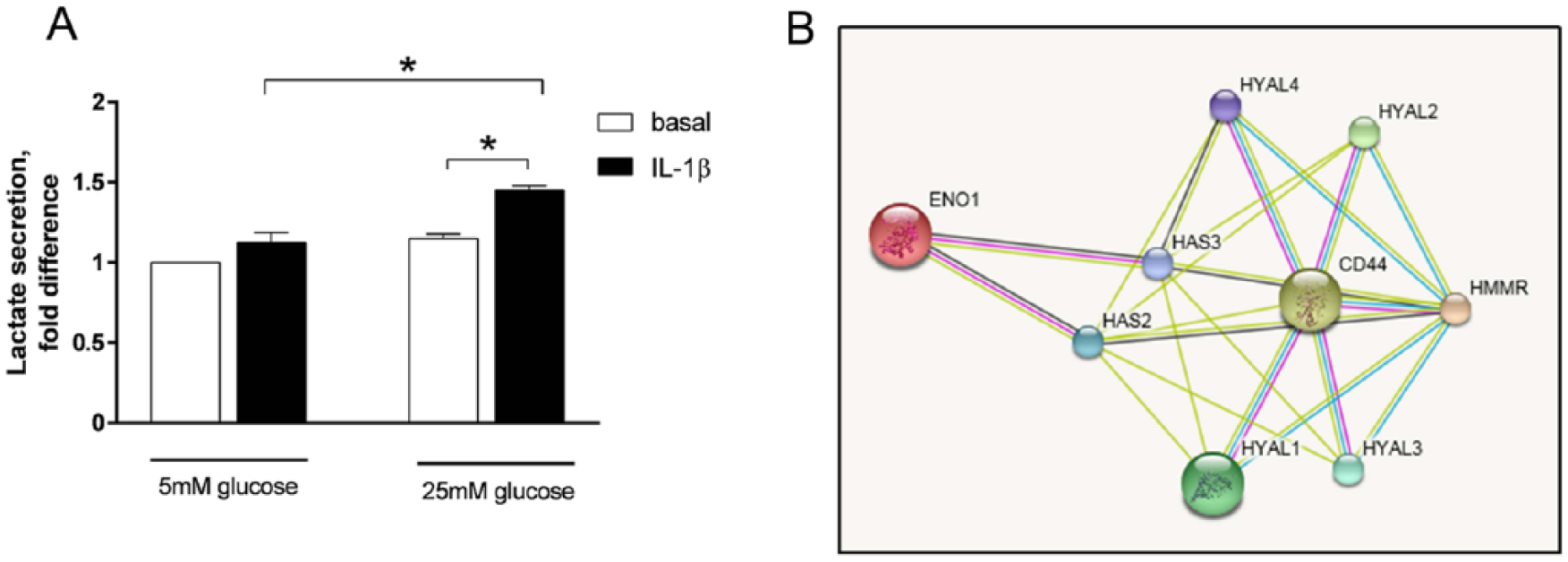
Lactate secretion and relation of ENO1 with HAS2. Equine chondrocytes were differentiated into cartilage pellets
Taken together, when investigating glucometabolic pathways, the most pronounced effect of elevated glucose levels in response to IL-1β stimulation was observed for glycolytic mediators, of which
Discussion
In this study, we investigated the effect of physiological and supraphysiological levels of glucose on glucose uptake, matrix production and glutamate release in an OA model of differentiated 3D chondrocyte pellets. In contrast to physiological levels of glucose (5 mM), elevated levels (25 mM) resulted in sustained glucose uptake and
Previously reported studies on the effect of IL-1β on glucose uptake in isolated primary chondrocytes support our findings in differentiated chondrocyte pellets, showing that, at high glucose concentrations, IL-1β stimulation sustained or increased glucose uptake in chondrocytes,
15
whereas glucose uptake was reduced in response to IL-1β at low glucose concentrations (2.5 mM or lower).
11
One report by Shikhman
The effect of IL-1β on degradation of matrix molecules is well known and was confirmed in our study with marked effects on
The
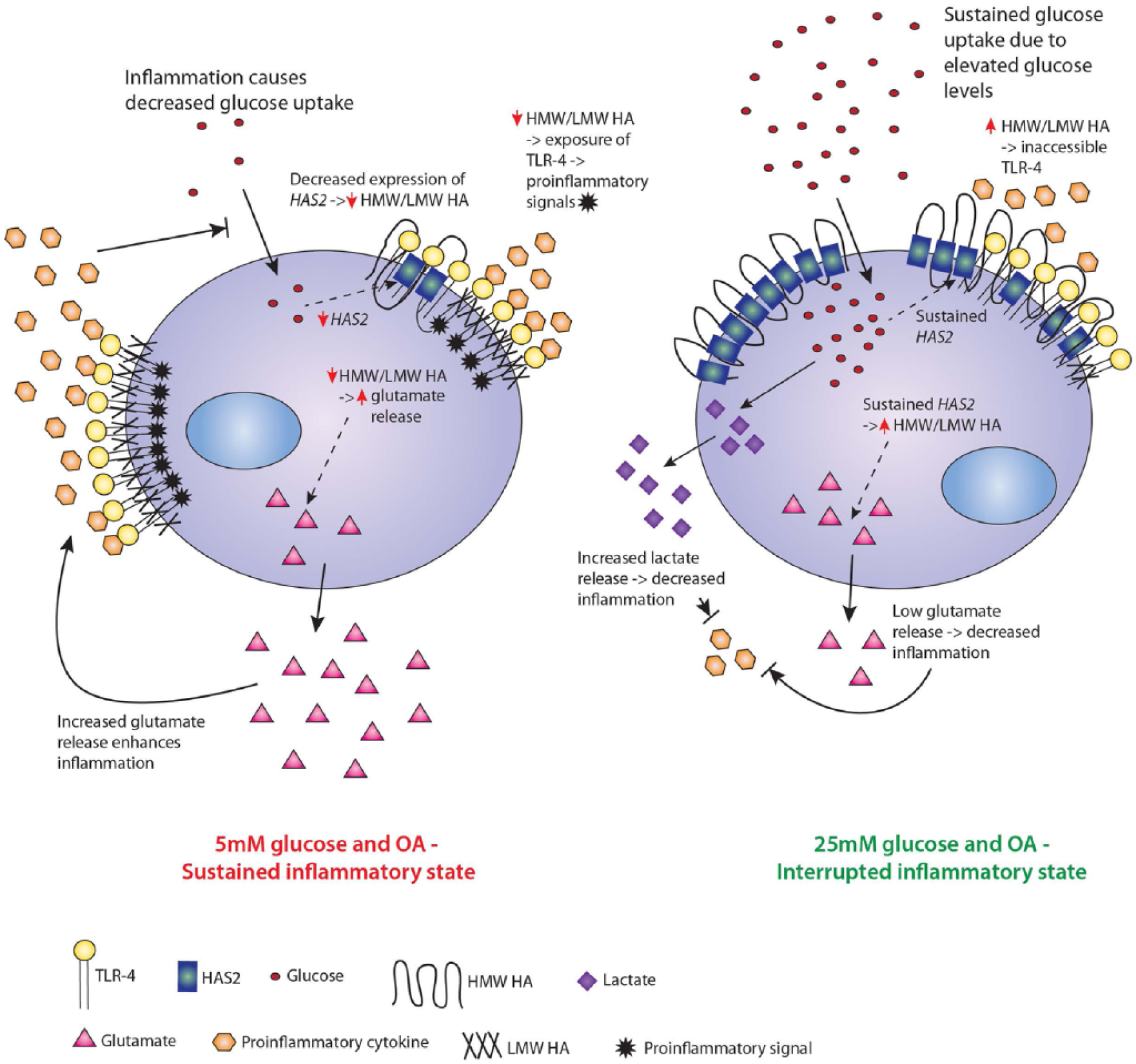
Hypothesis of the underlying mechanism for the effect of glucose during osteoarthritis. In conditions with osteoarthritis, inflammation causes degradation of cartilage components, including hyaluronan (HA), due to an insufficient supply of glucose and decreased glucose uptake in the chondrocyte. The decreased glucose uptake results in decreased expression of HAS2 and a lowering of the high molecular weight to low molecular weight (HMW:LMW) HA ratio. Reduced synthesis of HMW HA leads to a breakdown of the HA protective layer, exposing TLR-4 and enables interaction of TLR-4 with proinflammatory cytokines, thereby promoting inflammation. Furthermore, decreased HMW HA results in increased glutamate release, which in turn enhances inflammation. In addition, inflammatory cytokines, such as IL-1β, promote TLR-4 expression. The result is a self-sustained inflammatory cycle. By addition of glucose, the glucose uptake is sustained, resulting in maintained production of HMW HA by sustained HAS2 expression. Maintained HMW HA synthesis further leads to an intact protective HA layer as well as inhibition of glutamate release and maintained low expression of TLR-4. Maintained glucose uptake further enables a high glycolytic rate with increased lactate production. These anti-inflammatory actions interrupt the proinflammatory cycle and induces matrix repair.
To identify molecular players underlying an association between increased glucose uptake and sustained
In support of an overall increased flux of glucose through glycolysis, we found that lactate secretion was increased at 25 mM glucose compared to 5mM glucose. Lactate is the principal end-product of glucose metabolism by articular cartilage. 32 Interestingly, increased release of lactate is associated with lower pH, and lowering pH has been shown to decrease glutamate release in presynaptic neurons and glial cells.33,34 Thus, increased lactate release in chondrocytes as a result of increased glucose uptake and glucose flux through the glycolytic pathway, might contribute to anti-inflammatory cellular actions by inhibiting glutamate release ( Fig. 5 ).
Based on the fact that endocrine disorders, such as type 2 diabetes, are associated with osteoarthritis,35-37 it has been postulated that excess glucose levels associated with diabetes might lead to reactive oxygen species production and advanced glycation end products in cartilage,38-40 thereby favoring the progression of OA. However, the large majority of diabetes patients are rarely severely hyperglycemic for prolonged periods of time, but have their glucose levels controlled by various treatments, for example insulin injections or metformin. As chondrocytes are insulin insensitive, 13 lacking the insulin-sensitive glucose transporter GLUT-4, glucose will not be directed to cartilage by diabetes drugs. Instead, glucose is shunted to insulin-sensitive tissues such as muscle and adipose tissue, resulting in predicted glucose concentrations of ~5mM in the synovial fluid.41,42 Thus, type 2 diabetics are unlikely to have chronically elevated levels of glucose in the synovial fluid. Rather than high glucose levels being the driving force of OA in diabetes patients, it is more plausible that chronically elevated levels of cytokines are a major contributing factor. Cytokine levels are known to be chronically elevated in type 2 diabetics, 43 and appears to be a crucial mediator of the OA progression. Higher levels of cytokines in the synovial fluid in combination with insufficient glucose supply would be a very strong factor in driving cartilage degradation and OA.
In summary, in this study we show that elevated glucose levels rescue glucose uptake, matrix production and maintenance and induce anti-inflammatory activities in differentiated chondrocytes during inflammatory conditions associated with OA.
There is an unfulfilled need to identify novel therapeutic targets to halt cartilage degradation and to induce cartilage restoration in OA conditions. 44 Current clinical treatment strategies have varying success rates, with unsatisfactory long-term results.45-49 Further research is needed to expand our understanding of glucosensing and the impact of glucose on the ECM in chondrocytes. However, based on our data, intra-articular injections with glucose would be expected to exert beneficial effects in early phases of OA and/or in combination with other treatments to enhance treatment efficacy by rescuing chondrocyte metabolism, inducing HA synthesis and anti-inflammatory processes, thereby interrupting the vicious inflammatory cycle and promoting matrix production and repair ( Fig. 5 ).
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Swedish Research Council Grant 2015-02959 and Western Region Research Grant ALFGBG-432251.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Ethical Committee on Animal Experiments, Stockholm, Sweden (N378/12).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
