Abstract
Objective
Fibroblast growth factors (FGFs) are a family of 22 proteins and 4 FGF receptors (FGFRs) that are crucial elements for normal development. The contribution of different FGFs and FGFRs for the homeostasis or disease of the cartilage from the mandibular condyle is unknown. Therefore, our goal was to characterize age-related alterations in the protein expression of FGF ligands and FGFRs in the mandibular condyle of mice.
Method
Mandibular condyles of 1-, 6-, 12-, 18-, and 24-month-old C57BL/6J male mice (5 per group) were collected and histologically sectioned. Immunofluorescence for FGFs that have been reported to be relevant for chondrogenesis (FGF2, FGF8, FGF9, FGF18) as well as the activated/phosphorylated FGFRs (pFGFR1, pFGFR3) was carried out.
Results
FGF2 and FGF8 were strongly expressed in the cartilage and subchondral bone of 1-month-old mice, but the expression shifted mainly to the subchondral bone as mice aged. FGF18 and pFGFR3 expression was limited to the cartilage of 1-month-old mice only. Meanwhile, pFGFR1 and FGF9 were mostly limited to the cartilage with a significant increase in expression as mice aged.
Conclusions
Our results indicate FGF2 and FGF8 are important growth factors for mandibular condylar cartilage growth in young mice but with limited role in the cartilage of older mice. In addition, the increased expression of pFGFR1 and FGF9 and the decreased expression of pFGFR3 and FGF18 as mice aged suggest the association of these factors with aging and osteoarthritis of the cartilage of the mandibular condyle.
Fibroblast growth factors (FGFs) are a family of 22 proteins and 4 receptors (FGFRs) that are crucial elements for normal development. Anomalous FGF signaling has been associated with several pathologies and skeletal abnormalities.1 -3 Nevertheless, the contribution of different FGFs and FGFRs for the homeostasis or disease of the cartilage from the mandibular condyle is unknown. Therefore, the goal of this study was to characterize the protein expression of FGFs and FGFRs relevant to chondrogenesis in the osteochondral tissue of temporomandibular joint (TMJ) in young and aging mice. We have chosen 4 ligands (FGF2, FGF8, FGF9, and FGF18) and 2 receptors (FGFR1 and FGFR3) that have been associated with cartilage development and disease. 1 Our results indicate FGF2 and FGF8 are important growth factors for mandibular condylar cartilage (MCC) growth in young mice but with limited role in the cartilage of older mice. In addition, the increased expression of pFGFR1 and FGF9 and the decreased expression of pFGFR3 and FGF18 as mice aged suggest the association of these factors with aging and osteoarthritis of the cartilage of the mandibular condyle.
Experiments were approved by the UConn Health Animal Care and Use Committee. Mandibular condyles of 1-, 6-, 12-, 18-, and 24-month-old C57BL/6J male mice (5 per group) were collected. Mandibular condyles were fixed for 24 hours in 10% formalin and placed in 30% sucrose overnight before embedding in cryomedium (Thermo Shandon, Pittsburgh, PA). The medial surfaces of the samples were embedded against the base of the mold, parallel to the floor of the mold. Histological sections (5-7 μm thickness) were performed using a Leica Cryostat (Nussloch, Germany). Sections were transferred to slides using the tape transfer method.
Immunofluorescence was performed by first washing slides with phosphate-buffered saline (PBS) and then blocking for 10 minutes at room temperature using the Universal Blocking Reagent (BioGenex, Fremont, CA). Slides were incubated with FGF2, FGF9, FGF18, phospho-FGFR3 (Thermo Fisher Scientific, Carlsbad, CA), FGF8, and phospho-FGFR1 (Abcam, Cambridge, UK) antibodies at a concentration of 1:100 each, overnight at +4 °C. Phosphospecific antibodies for FGFR1 and FGFR3 were chosen to determine the active state of receptors.
Subsequently, sections were washed with PBS and incubated with Cy3 Donkey anti-rabbit IgG (Jackson ImmunoResearch Laboratories, Inc., West Grove, CA) at a concentration of 1:200 for 1 hour at room temperature and mounted with a suspension of 50% glycerol in PBS containing Hoechst nuclear stain (Life Technologies, Carlsbad, CA). Negative controls were performed by omitting the primary antibody and incubating slides in the serum used for primary antibodies (Suppl. Fig. 1).
Immunofluorescence expression in different groups was quantified using ImageJ (National Institutes of Health, Bethesda, MD) by calculating the stained area and dividing by the cartilage area, obtaining a percentage of stained area. Three sections per sample (N = 5) were analyzed. Statistical differences between age groups were determined by ordinary 1-way analysis of variance and Tukey’s post hoc multiple comparisons test. Statistical tests were 2-sided, and a P value of <0.05 was considered to be statistically significant. Statistical analysis was computed using GraphPad Prism (San Diego, CA).
Our first observation was the MCC of TMJ became thinner and cartilage degeneration developed as mice aged, as we described previously 4 and is illustrated in Supplementary Fig. 2. Moreover, we noticed that the cells expressing alkaline phosphate (hypertrophic chondrocytes) moved toward the surface of the unmineralized cartilage, suggesting mineralization of the MCC as mice aged (Suppl. Fig. 2). In addition, we observed a substantial decrease in cellularity and cellular proliferation (substantial decrease in Ki67 expression) and an increase in MMP13 expression (marker for cartilage degradation) 5 associated with aging (Suppl. Fig. 2).
FGF2 was strongly expressed in the MCC of TMJ of 1-month-old mice, especially in the pre-hypertrophic and hypertrophic chondrocytes, as well as in the subchondral bone. The expression of FGF2 sustained as mice aged, but that expression was limited to the subchondral region ( Figs. 1A and 2A ).
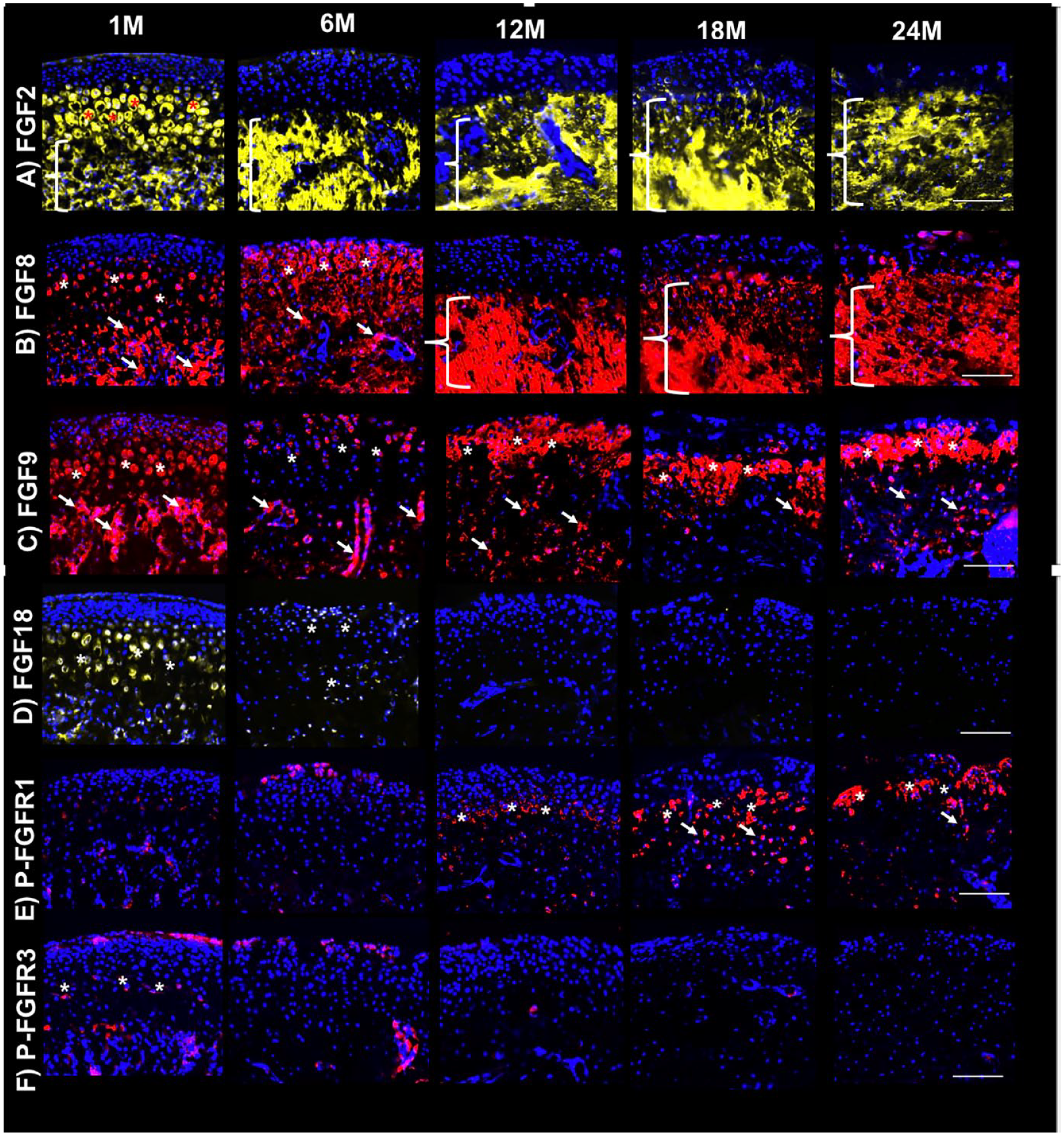
Differential FGF expression in mandibular condyles of young and aging mice. Immunofluorescence for (
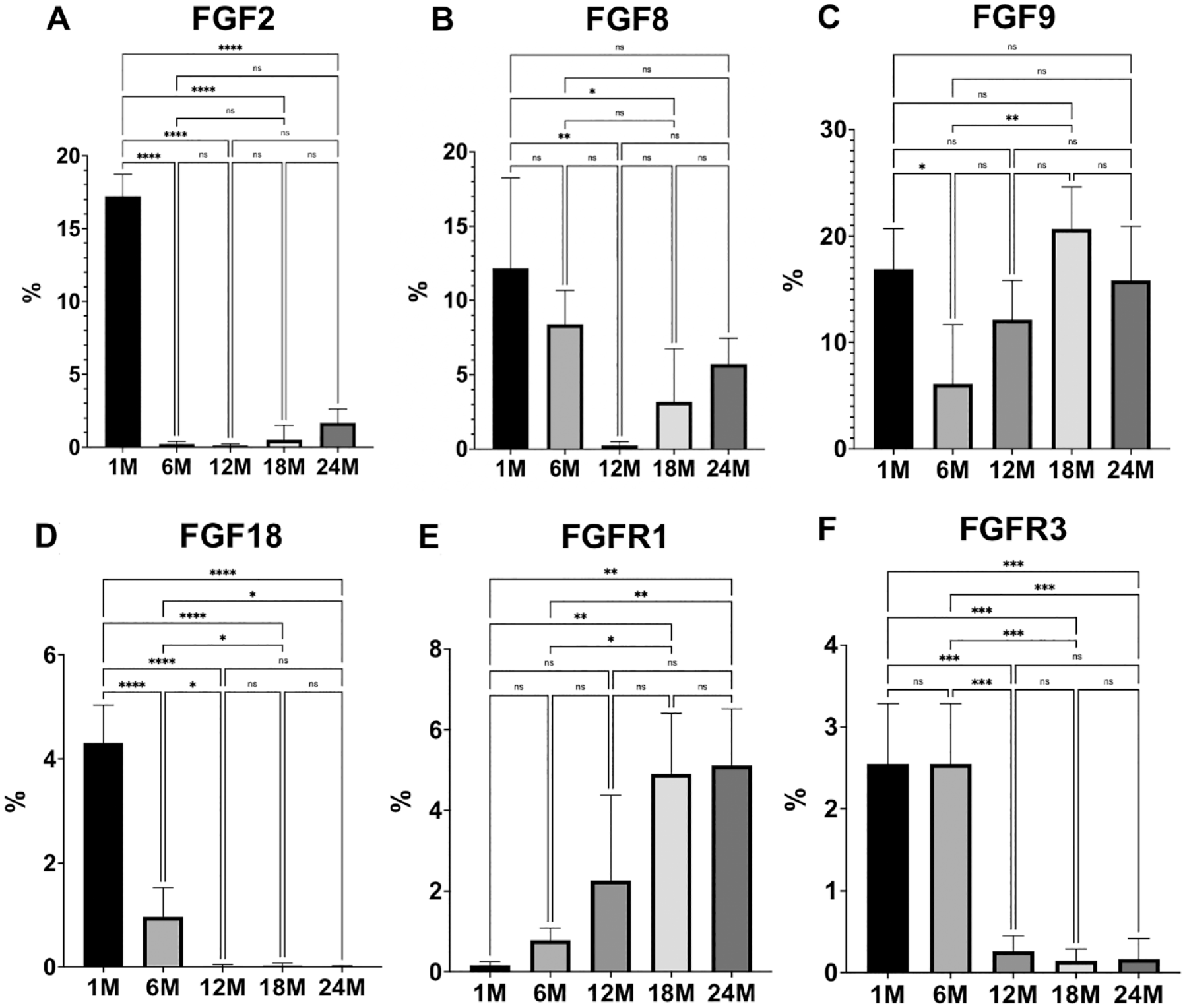
Quantification of FGF expression in mandibular condyles of young and aging mice. Quantification was limited to the cartilage and expressed as percentage per area of cartilage. Histograms represent means ± SD for n = 5 per group. FGF = fibroblast growth factor. 1M = 1-month-old. 6M = 6-month-old. 12M = 12-month-old. 18M = 18-month-old. 24M: 24-month-old. Statistically significant difference between groups: *P < 0.05, **P < 0.005, ***P < 0.0005, ****P < 0.0001.
FGF8 presented with a similar pattern of expression as observed for FGF2, with substantial expression in the pre-hypertrophic and hypertrophic chondrocytes and subchondral bone in 1-month-old mice. Interestingly, FGF8 expression shifted to the outer layers of the MCC in 6-month-old mice, was then confined to the subchondral bone at 12 months, and was re-expressed in MCC as mice aged ( Figs. 1B and 2B ).
FGF9 was diffusely expressed in the MCC and subchondral bone in 1-month-old mice, but this expression markedly decreased in both regions at 6 months. However, FGF9 was strongly expressed in the hypertrophic/mineralizing zone and subchondral region in 12-, 18-, and 24-month-old mice ( Figs. 1C and 2C ).
Substantial FGF18 expression was noticed in the MCC of 1-month-old mice, mainly in the pre-hypertrophic and hypertrophic chondrocytes, with minimal expression in the subchondral bone. This expression has decreased in 6-month-old mice, but some expression in osteochondral tissue was still noticeable. Absent expression of FGF18 was noticed in the osteochondral tissue of TMJ as mice aged ( Figs. 1D and 2D ).
Phospho-FGFR1 expression was weak in 1- and 6-month-old mice, but this expression became substantially stronger as mice aged especially in the hypertrophic region of 18- and 24-month-old mice ( Figs. 1E and 2E ).
Phospho-FGFR3 was weakly expressed in the outer layer of the MCC and also in the pre-hypertrophic and hypertrophic regions of the mandibular condyle in 1-month-old mice. Some sparse activity was noted in the subchondral region too. At 6 months of age, Phospho-FGFR3 expression had decreased, with nearly absent expression in 12-,18-, and 24-month-old mice ( Figs. 1F and 2F ).
This novel study characterizes the protein expression of FGF ligands and FGFR in the osteochondral tissue of the TMJ as well as its modulation with aging. The most striking observation was that the protein expression of each FGF and FGFR investigated was not consistent between age groups, suggesting their distinct roles in different phases of development. The osteochondral tissue of TMJ undergoes changes with aging, with progressive development of degeneration and osteoarthritis. 4 We believe the expression or deficiency of FGFs in the mandibular condyle could explain the homeostasis in earlier ages and degenerative changes as mice reach advanced aging.
FGF2, FGF8, FGF9, and FGF18 are secreted canonical FGFs that have been associated with articular cartilage homeostasis or disease.6 -8 Reduction in FGFR3 expression has been related to human and murine osteoarthritis, while activation of FGFR1 has been associated with osteoarthritis.9,10
It is well known that FGF2 may have catabolic or anabolic effects on cartilage homeostasis depending on the specific FGF receptor associated with it. When signaling through FGFR1, the effects are cartilage catabolism and osteoarthritis, 10 while cartilage anabolism is associated with signaling through FGFR3.10,11 We have detected a robust expression of FGF2 and phosphorylation of FGFR3 in the MCC of young mice (1 month old), consistent with this phase of development when chondrocyte proliferation and differentiation are essential. However, when we analyzed the phosphorylation behavior of FGFR3 and FGFR1 in each age group in relation to FGF2 expression, we concluded that the release of FGF2, which signals through activated FGFR1 in older mice, could favor the degeneration of cartilage given no phosphorylated FGFR3 was observed in those age groups.
FGF8 expression has been associated with active growth of the mandibular condyle, being present in proliferative, pre-hypertrophic, and hypertrophic chondrocytes. 12 A catabolic role for FGF8 has also been identified in in vitro and experimental articular cartilage osteoarthritis models. 13 We have found a strong expression of FGF8 in the MCC of younger mice and in the mineralization region/subchondral bone of aging mice. FGF8 signals through FGFR3, and the expression pattern observed in the MCC of the younger group suggests FGF8 may be associated with active chondrogenesis of the mandibular condyle in developing mice. On the contrary, the shift in the expression of FGF8 toward the subchondral bone and re-expression in the MCC in older mice suggest this ligand may also contribute to the development of osteoarthritis.
Similarly, FGF9 promotes proliferation and hypertrophic differentiation in chondrocytes. 14 It has been shown that injection of FGF9 in experimental osteoarthritis mouse model inhibits cartilage degradation, but increases the formation of osteophytes. 15 We observed FGF9 staining in all layers of the MCC in young mice, with this expression fluctuating to the mineralization region of the cartilage as mice aged. This interesting pattern observed in different aging groups illustrates the distinct roles of FGF9 in chondrogenesis in young and older mice.
It has been demonstrated that FGF18 stimulates chondrocyte proliferation and differentiation by an FGFR3-mediated pathway in cells with chondrogenic potential. 11 We have found strong FGF18 protein expression and phosphorylation of FGFR3 in the MCC and subchondral bone in younger mice, but this scenario changed abruptly as mice aged, with a lack of expression for both ligand and receptor. These data suggest aging changes could be associated with deficient FGF18 and FGFR3 signaling.
One of the limitations of this study was the use of male mice only for our characterization. In addition, protein expression analysis was limited to immunofluorescence. Our future studies will include female mice and gene expression of FGF signaling in the MCC of young and aging mice by RNA sequencing and proteomics.
To the best of our knowledge, this is the first report on the protein expression of different FGFs and FGFRs in the osteochondral tissue of the TMJ of young and older mice. Our data suggest the distinct role of FGFs and FGFRs in the osteochondral tissue of the TMJ at different stages of development. Our research suggests that modulation of FGF signaling may promote homeostasis or prevent/inhibit the progression of osteoarthritis in the TMJ.
Supplemental Material
sj-tif-1-car-10.1177_19476035231163691 – Supplemental material for FGF Ligands and Receptors in Osteochondral Tissues of the Temporomandibular Joint in Young and Aging Mice
Supplemental material, sj-tif-1-car-10.1177_19476035231163691 for FGF Ligands and Receptors in Osteochondral Tissues of the Temporomandibular Joint in Young and Aging Mice by Eliane H. Dutra, Po-Jung Chen, Zana Kalajzic, Sunil Wadhwa, Marja Hurley and Sumit Yadav in CARTILAGE
Supplemental Material
sj-tif-2-car-10.1177_19476035231163691 – Supplemental material for FGF Ligands and Receptors in Osteochondral Tissues of the Temporomandibular Joint in Young and Aging Mice
Supplemental material, sj-tif-2-car-10.1177_19476035231163691 for FGF Ligands and Receptors in Osteochondral Tissues of the Temporomandibular Joint in Young and Aging Mice by Eliane H. Dutra, Po-Jung Chen, Zana Kalajzic, Sunil Wadhwa, Marja Hurley and Sumit Yadav in CARTILAGE
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research reported in this publication was supported by the National Institute of Dental and Craniofacial Research of the National Institute of Health under award number K01DE029528 to EHD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Experiments were approved by the UConn Health Animal Care and Use Committee.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
