Abstract
Objective
To investigate the responses of refrigerated osteochondral allograft cartilage (OCA) and fresh cartilage (FC), including cell survival and metabolism, to surgical impaction and proinflammatory cytokines.
Design
Osteochondral plugs (8 mm diameter) were harvested from prolonged-refrigerated (14-28 days) and fresh (≤24 hours postmortem) human femoral hemicondyles and subjected to a 0.2 N s pneumatic impaction impulse. Cartilage explants were removed from subchondral bone and randomized to 1 of 6 treatment groups: (1) Unimpacted control (UIC), (2) Impacted control (IC), (3) Impacted + interleukin (IL)-1β (0.1 ng/mL), (4) Impacted + IL-1β (0.1 ng/mL) + IL-6, (5) Impacted + IL-1β (10 ng/mL), and (6) Impacted + IL-1β (10 ng/mL) + IL-6. Samples were measured for cell viability, histology, and proteoglycan (PG) content at days 0, 2, 7, and 14 of culture.
Results
In UIC, cell viability was indistinguishable between OCA and FC and remained constant. Impaction alone decreased cell viability by 30% (
Conclusions
Mechanical impaction, mimicking surgical insertion, has a more profound effect on cell viability in OCA than in FC. Addition of proinflammatory cytokines further decreases OCA tissue metabolism and integrity.
Introduction
Joint injuries have a high prevalence among young adults and cause a major burden to society through expedited progression of osteoarthritis (OA). 1 In fact, about 13% to 18% of patients undergoing total hip or knee arthroplasty had an identifiable acute trauma to their joint. 2 Early surgical intervention for a symptomatic chondral lesion is recommended to restore normal joint congruity and pressure distribution and to prevent OA progression. 3 Among available procedures, osteochondral allograft transplantation (OCA) has yielded excellent results and up to 95% 5-year survival in the treatment of large (2.5 cm2), symptomatic chondral defects.4,5 Given positive functional outcomes and an increasing number of OCA procedures performed annually in young adults, 6 there is substantial interest in preserving long-term graft integrity and understanding mechanisms contributing to graft failure.
Histological analysis of failed OCA grafts suggests that crucial features for long-term survival include chondrocyte viability, integration of allograft cartilage with surrounding host tissue, and mechanical stability with replacement of graft bone by host bone at the host-graft interface.5,7 As part of OCA, the allograft must be impacted into the defect to create a flush surface relative to the surrounding host articular cartilage. However, this process leads to approximately 60% cell death in the upper zone of transplanted cartilage, 8 most of which occurs within the first 2 days of impaction. 9 Micromechanical injury to cartilage during the OCA impaction process has been suggested to result in progressive cartilage degradation within the graft, accompanied by chondrocyte necrosis, apoptosis, and increased release of fragments of cartilage matrix molecules. 9 Beyond mechanical trauma sustained during impaction, subsequent exposure to proinflammatory mediators present in synovial fluid may further affect chondrocyte synthetic ability and cell survival.10-12
We hypothesized that the outcome of OCA surgery may depend on the impaction used to implant the graft and the level of intra-articular inflammation during or immediately after surgical intervention. The purpose of this study was therefore to investigate the OCA cartilage response, including cell survival and metabolism, to mechanical load and proinflammatory cytokines in an
Materials and Methods
Specimen Preparation and Culture
Nine previously refrigerated OCA specimens (2-4 weeks storage) otherwise intended for transplantation were received from Allosource (Denver, CO). Six fresh hemicondyles were collected within 24 hours of death from human donors with no documented history of joint diseases through the Gift of Hope Tissue & Organ Donor Network (Itasca, IL). The mean age of OCA specimens was 25 years and the mean age of the fresh cartilage (FC) specimens was 50 years. Eight millimeter cylindrical osteochondral plugs (15 mm depth) were harvested from all specimens using an osteochondral autograft system (OATS, Arthrex Inc., Naples, FL) and tapped 10 times with a pneumatically controlled impactor (SmartImpactor, Chicago, IL) at an impaction impulse of 0.2 N s mimicking surgical insertion technique as described by Kang
Outcome Measures
Histological Appearance with Safranin-O Staining
Tissue samples were fixed in 4% paraformaldehyde and processed for paraffin embedding. Full-thickness cartilage pieces were sectioned (6 µm) and stained. Integrity of the cartilage surface and its underlying matrix were analyzed with Safranin-O/fast-green staining. 17 Sections were analyzed with a Nikon Eclipse E800 (Nikon Inc., Garden City, NY) microscope equipped with CF160 optical system for surface changes, abnormalities in cellularity, and patterns of Safranin-O staining reflective of PG distribution.
Chondrocyte Viability (Live/Dead Assay)
Cell survival was measured using calcein AM (acetomethoxy derivate of calcein) and ethidium bromide homodimer-1 (Molecular Probes Inc., Eugene, OR). Digital images were captured using Metamorph software (Molecular Devices, Sunnyvale, CA). The percentage of live cells for each sample was counted using ImageJ software (version 1.34, National Institutes of Health, Bethesda, MD) and defined as the number of live cells divided by the total number of cells (live + dead) per tissue section and multiplied by 100. Superficial zone cell viability was also calculated with ImageJ and defined as 10% of tissue surface.
PG Synthesis, Content, and Percent Release (Normalized to Wet Weight)
PG synthesis was determined by alcian blue method. Radiolabeled sulfate (5 μCi/mL) was added during the final 4 hours of culture and sulfate incorporation was measured as previously described by Masuda
Data Analysis
A multifactorial study design was used to evaluate the combined effect of cartilage source (2 levels), treatment (6 levels), and time point (3 levels) (Table 1), for a total of 36 groups. It should be noted that different samples were used for the various time points, precluding a repeated-measures scheme.
The 36 Groups Used to Evaluate the Combined Effect of Cartilage Source (2 Levels), Treatment (6 Levels), and Time Point (3 Levels).
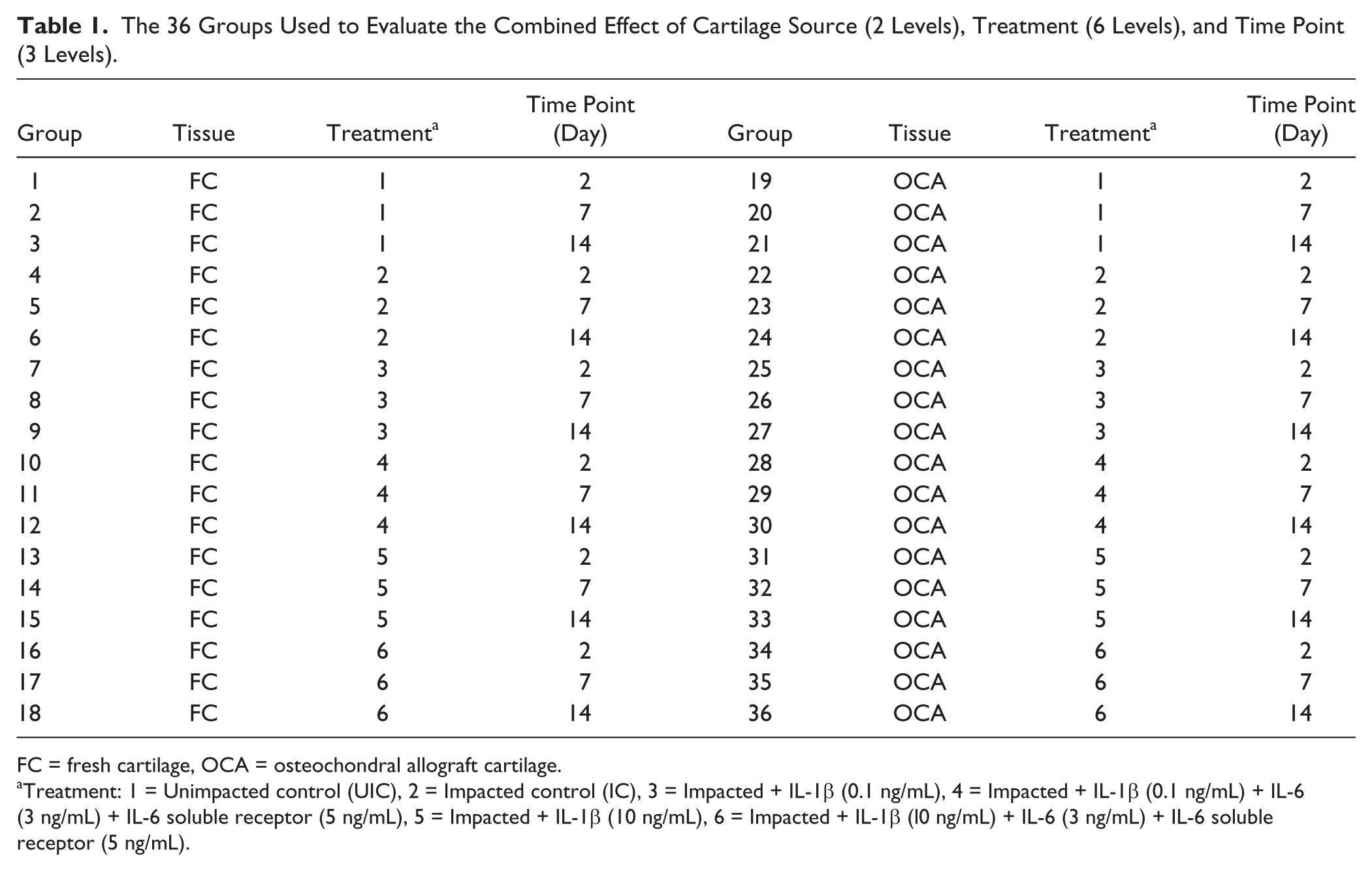
FC = fresh cartilage, OCA = osteochondral allograft cartilage.
Treatment: 1 = Unimpacted control (UIC), 2 = Impacted control (IC), 3 = Impacted + IL-1β (0.1 ng/mL), 4 = Impacted + IL-1β (0.1 ng/mL) + IL-6 (3 ng/mL) + IL-6 soluble receptor (5 ng/mL), 5 = Impacted + IL-1β (10 ng/mL), 6 = Impacted + IL-1β (l0 ng/mL) + IL-6 (3 ng/mL) + IL-6 soluble receptor (5 ng/mL).
The effects of treatment (impaction and cytokine addition), time point, and cartilage source (osteochondral allograft cartilage or fresh cartilage) on cartilage cell viability, PG synthesis, and PG content were analyzed using multifactor analysis of variance (ANOVA) implemented with Design Expert, version 8 (Stat-Ease Inc., Minneapolis, MN). Normality of the data for each analysis was checked with a normal probability plot and, if necessary, the data were transformed to achieve variance stabilization.
Effect detection levels, defined as the effect of an input variable (cartilage source, etc.) on the output variable (cell viability, etc.) that can be detected with a power of 0.8 at a significance level of 0.05, were estimated for the 3 main output variables. For cell viability (surface zone and total), where an average of 4.5 replicates were performed, the effect detection levels were 0.45, 1.2, and 0.63 standard deviations (SDs) for the action of cartilage source, treatment, and time point, respectively. For PG synthesis and PG content, with an average of 2.3 replicates, the corresponding effect detection levels were 0.63, 1.42, and 0.87 SD. The estimated effect detection levels for
Results
Histological Assessment
The pattern of Safranin-O staining did not change substantially when unimpacted FC control was cultured alone for 14 days ( Fig. 1A , g and h ). The staining was intense and uniform throughout the tissue section with less staining in the superficial layer (as expected due to a low level of sulfated PGs in that region). UIC OCA samples exhibited normal Safranin-O staining at day 0, but culture alone caused some depletion of staining in the upper middle zone and in the interterritorial matrix of the middle and deep zones ( Fig. 1A , a and b ). Impaction led to the depletion of staining starting at day 0 and most apparent by day 14, with more substantial changes in the OCA cartilage ( Fig. 1A , c and d ). PG loss was detected in the middle zone and in the interterritorial matrix of the deep zone of OCA and FC. Intense red staining around chondrocytes (pericellular + territorial matrix) was evidenced in both types of tissues indicating metabolic activity of the residing chondrocytes ( Fig. 1A , a-f , g-l ). The most significant changes in Safranin-O staining were observed by day 14 in the presence of IL-1 at either concentration ( Fig. 1A , e and f , k and l ). The pattern of change was similar between OCA and FC, though the depletion of staining was more substantial in OCA cartilage samples. By day 14, there was a complete loss of staining in the superficial, middle, and part of the deep cartilage zones in the OCA samples ( Fig. 1A , e and f ). In FC, the loss of PGs was primarily detected in the upper middle layer and in the interterritorial matrix of the lower middle and deep layers ( Fig. 1A , k and l ).
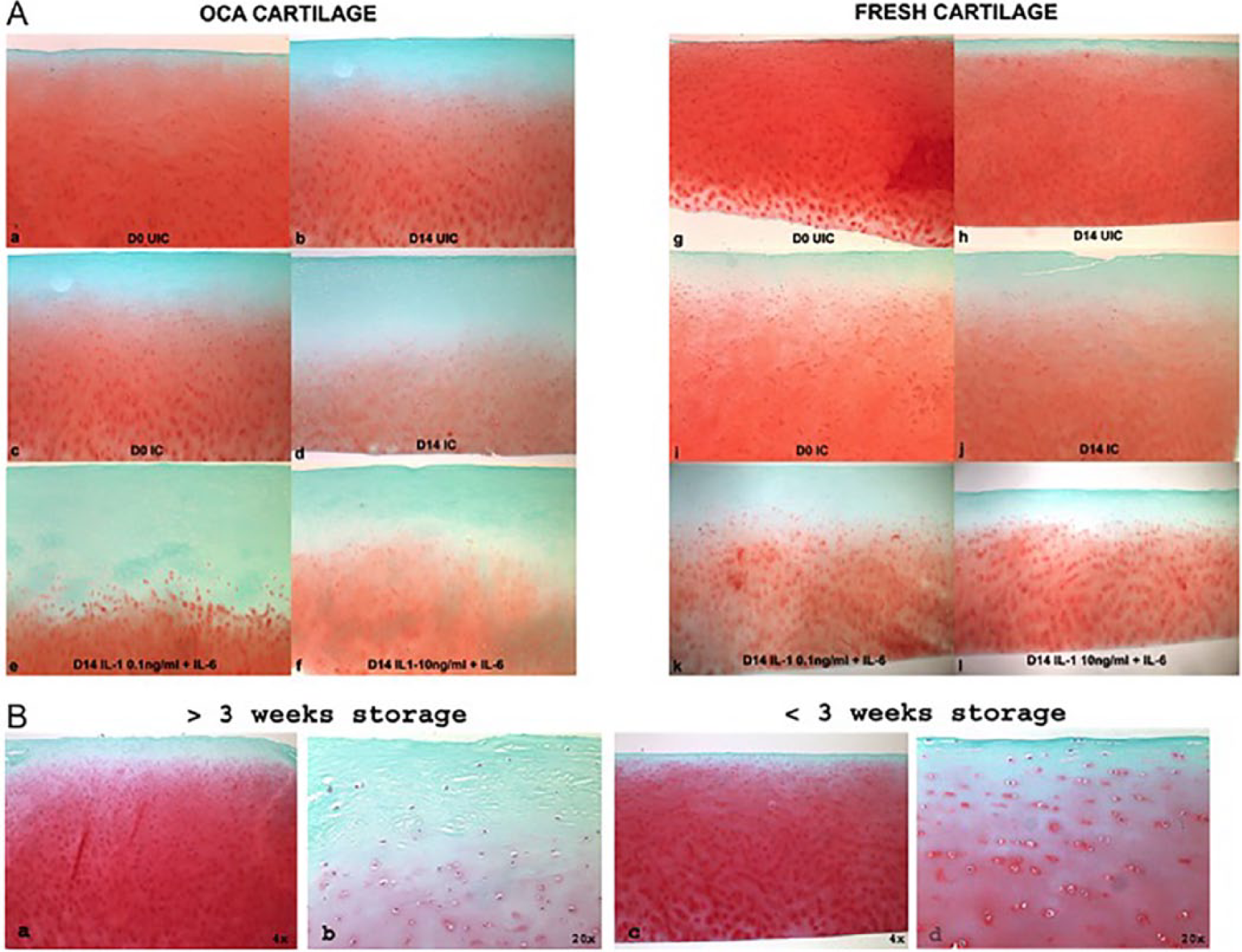
Safranin-O–stained sections of cartilage samples. (
Additionally, histological differences were observed between allografts stored for more than 3 weeks and those stored for less than 3 weeks ( Fig. 1B ). OCA samples refrigerated for more than 3 weeks ( Fig. 1B , a and b ) were characterized by a paucity of chondrocytes in the upper layers, softening of the surface, and discoloration and disorganization of the matrix, specifically in the superficial layer. Samples refrigerated for less than 3 weeks did not demonstrate obvious histological abnormalities. For all subsequent experiments, only allografts stored for less than 3 weeks were used.
Cell Viability
At baseline prior to impaction, no difference in cell viability was identified between the fresh and OCA cartilage. At each time point, cell viability in the OCA UICs remained relatively constant and was between 55% and 72% in the superficial layer and between 69% and 80% throughout the entire cartilage depth (
Fig. 2A
). Impaction caused an immediate decrease in cell viability by 29% (
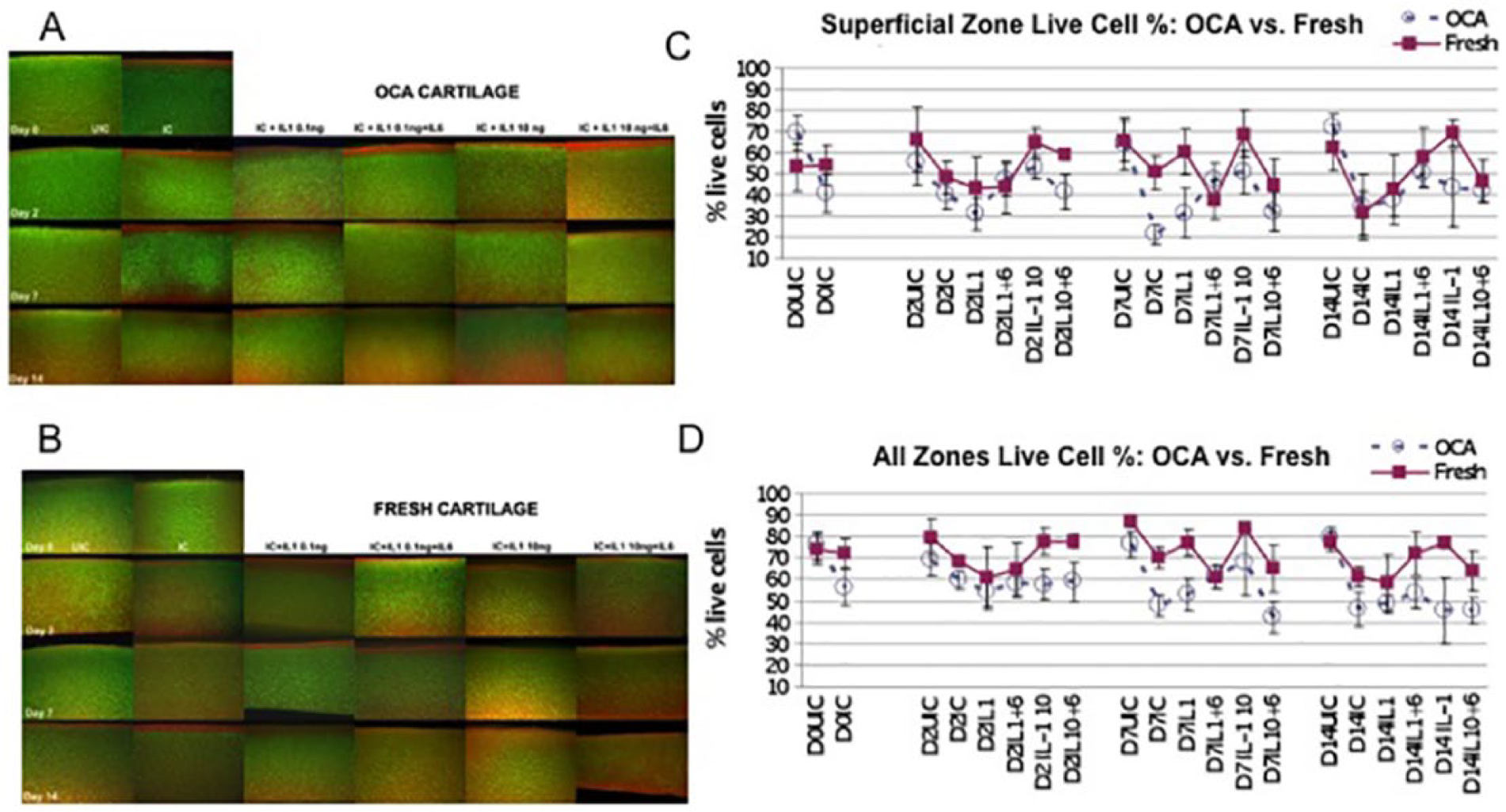
Comparisons of chondrocyte survival between OCA and FC (
Fig. 2C
and
D
) identified 2 interesting trends: (1) on day 14, no differences between the tissues were found within each control group (
PG Synthesis and Content
At baseline, in the UIC, the levels of PG synthesis were comparable between OCA and FC and remained at steady state throughout the culture. Impaction alone did not significantly influence PG synthesis in either of the tissues, regardless of the time point. However, addition of cytokines decreased PG synthesis by 2- to 3-fold (cumulative value of all 3 time points) and the effect was similar in both tissues. In OCA samples, a statistically significant decrease in PG synthesis was found in the IL-1β (10.0 ng/mL) group in comparison with the corresponding IC (
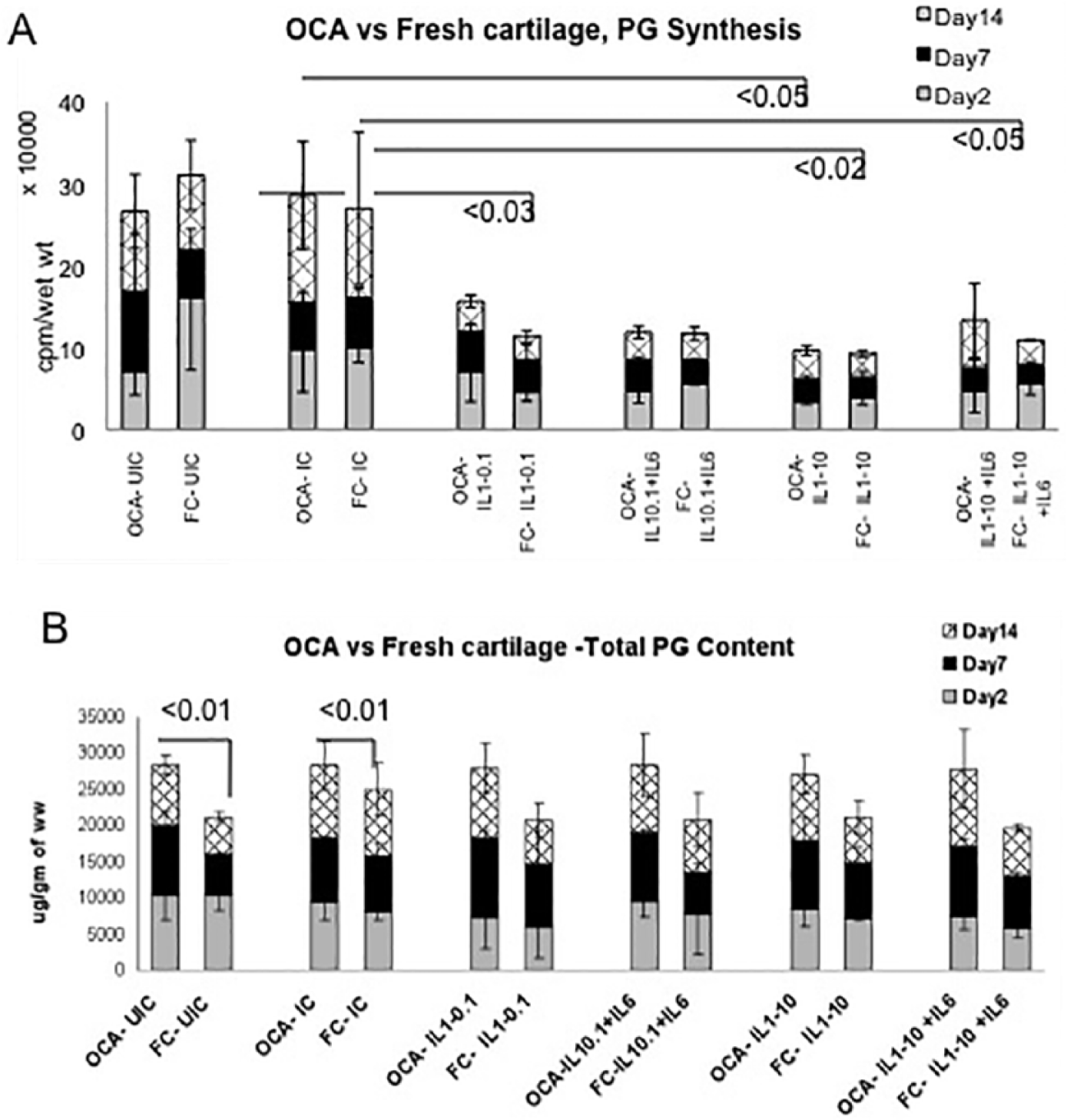
(
PG content was consistently higher in OCA than in FC cartilage (
Discussion
Given the poor regenerative capacity of hyaline cartilage,1,21 this study explored cellular responses induced by graft impaction and subsequent exposure to proinflammatory mediators present in synovial fluid in order to improve the efficacy of a leading cartilage restoration procedure. After subjecting cartilage allografts to 2 major insults commonly experienced during OCA (impaction and inflammation), analysis yielded several key findings: (1) allograft cartilage contains fewer viable cells than fresh cartilage, (2) impaction mimicking surgical insertion affects chondrocyte survival more than chondrocyte metabolism, (3) impaction has a stronger effect on cell survival in both OCA and FC than the subsequent addition of proinflammatory cytokines, (4) the cytokines tested in this study had no significant effect on chondrocyte viability but influenced chondrocytes’ synthetic activity as measured by PG synthesis, (5) both insults resulted in depletion of cartilage matrix, and (6) finally, storage for more than 3 weeks affected allograft cartilage structural integrity and matrix macroscopic appearance. This final finding suggests that storage for more than 3 weeks is detrimental to the tissue and thus likely contributes to allograft failure, as previously concluded by Pennock
The choice of cytokines in our study was limited to IL-1β and IL-6 based on the following: (1) the well-documented role of IL-1β as an inducer of cartilage degenerative processes
10
and (2) the types and concentrations of cytokines detected in the synovial fluid of patients who have undergone osteochondral allograft surgeries. For example, the average IL-6 concentration in synovial fluid at the time of allograft surgery and 10 days postoperatively was 2380 ± 1575 pg/mL as compared with 309 ± 501 pg/mL in normal control, which agrees with previously reported IL-6 concentrations of 2022 pg/mL in osteoarthritic patients and 132 pg/mL in normal synovial samples.
24
(3) Cytokines were found to be elevated in our acute trauma model.2,25 Furthermore, IL-1β and TNF-α were previously shown to be potent activators of cartilage degradation with increased activity following acute injury,
12
while IL-6, IL-8, and IL-10 were associated with cartilage loss and posttraumatic OA progression.25-28 The high levels of IL-6 added in the current study were within the same range as the ones detected in our
The impaction forces (single, intermediate, impulse of 0.2 N s applied 10 times to the osteochondral explants) utilized here introduced an insult to the cartilage but were not intended to cause gross damage to the surface. However, similar to Borazjani
The addition of proinflammatory mediators to the impacted grafts greatly inhibited chondrocyte PG synthetic ability in both tissues without additionally affecting cell survival. This finding agrees with previous reports documented for fresh tissue.10,27,32 However, no differences in PG synthesis and content between various doses of cytokines and their combinations were identified, and comparison between OCA and FC yielded no synthetic differences in response to cytokine insult between the 2 tissue types. One hypothesis for this is that chondrocytes surviving both insults in either tissue reach an equally metabolically active state; however, such speculation lies beyond the scope of this study. 33 One limitation with regard to PG analysis is that the wet weight of explants was used as denominator in metabolic assays. Hence, differences may have been identified if PG synthesis and content were normalized to the DNA content or the number of viable cells.
Qualitatively, both insults resulted in the depletion of Safranin-O staining that was more apparent toward the end of culture in explants treated with cytokines. This finding suggests that allografts have a lower chance of survival in a proinflammatory environment. In addition, we detected differences in histological appearance of allografts stored for less than or more than 3 weeks. Samples with longer storage displayed abundant structural changes, including decreased chondrocytes, pyknotic appearance, and swelling of cartilage matrix with signs of disintegration ( Fig. 3B ). These samples were excluded from metabolic and cell viability analyses. Similar changes with prolonged storage of allografts have been already reported for human and baboon allografts.33,34 All efforts should be made to avoid utilizing aged allografts for patient care.
This study has a number of limitations that have to be considered in future developments of allograft transplantation: (1) the focus on cartilage-only responses, rather than cartilage-bone graft as a single unit; (2) use of 2 cytokines, while synovial fluid contains many more pro- and anti-inflammatory mediators; (3) limited length of culture; and (4) limited assessment of cartilage metabolism.
In conclusion, this
Footnotes
Acknowledgments and Funding
We would like to acknowledge Dr. Arkady Margulis for procuring the human tissue. We acknowledge Allosource and the Gift of Hope Organ and Tissue Donor Network and donor’s families for making donor tissue available for research. We gratefully acknowledge funding support of the Joint Restoration Foundation and Ciba-Geigy Endowed Chair (S.C.), Rush University Medical Center.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.F. reports consulting or institutional support from Finceramica Faenza Spa (Italy), Fidia Farmaceutici Spa (Italy), and CartiHeal (2009) Ltd (Israel), EON Medica SRL (Italy), IGEA Clinical Biophysics (Italy), Biomet (USA), and Kensey Nash (USA). M.A.W. reports research support from Biomet, CeramTec, and Zimmer and receives royalties from Biotribology. S.C. reports research support from NuTech Medical. B.J.C. reports royalties from Arthrex, DJO Global, and Elsevier; serves as a paid consultant to Arthrex, Carticept Medical, Regentis Biomaterials, and Zimmer; has stock or stock options held in Carticept Medical and Regentis Biomaterials; has received research or institutional support from Arthrex, Medipost, the National Institutes of Health (NIAMS and NICHD), and Zimmer; has received nonincome support (such as equipment or services), commercially derived honoraria, or other non–research-related funding (such as paid travel) from Athletico, Össur, Smith & Nephew, and Tornier.
Ethical Approval
Ethical approval for this study was waived by Rush University Medical Center Institutional Review Board because this was a cadaver study.
Informed Consent
Informed consent was not sought for the present study because the specimens were donated cadaveric tissues. The donors consented while alive for their tissues to be utilized for medical purposes.
