Abstract
Objective
Osteoarthritis (OA) progression has been shown to increase the expression of inflammatory cytokines in joints, leading to the destruction of cartilage matrix. Interleukin (IL)-1β is a potent inflammatory cytokine associated with osteoarthritic synovial fluid. The protective effects of polysaccharides from Enteromorpha prolifera against acute hepatic injury was reported.
Design
In this study, we examined the effects of Enteromorpha polysaccharide extracts (EPEs) in the treatment of OA. The effects of the EPEs were assessed using an IL-1β-stimulated SW1353 and SW982 cells. The expression levels of specific mRNA and proteins were evaluated using semi-quantitative reverse transcription polymerase chain reaction (sqRT-PCR) and western immunoblotting. An OA animal study involving C57BL/6J mice was also conducted to assess the effects on tactile sensitivity and anterior cruciate ligament transection (ACLT).
Results
Acidic polysaccharide extract (APE) was shown to significantly reduce cytokine and chemokine mRNA levels in IL-1β-stimulated SW1353 and SW982 cells and attenuate the expression of proinflammatory cytokines and p38/AP-1 in SW1353 cells. APE was also shown to minimize the effect of osteolytic lesions in the knee joints of ACLT-induced osteoarthritic mice.
Conclusions
APE is a potent inhibitor of joint degeneration associated with OA.
Introduction
Osteoarthritis (OA) is a chronic disease characterized by articular cartilage degeneration and joint inflammation, which leads to chronic disability.1 -3 It has been estimated that 18% of females and 9.6% of males beyond the age of 40 have symptomatic OA. Increased expression of inflammatory mediators in the joints has been found to lead to the destruction of the cartilage matrix in the progression of OA.3 -5 Current treatments for OA are meant to alleviate pain, delay or prevent disease progression, and maintain the quality of life. 6 However, most treatment regimens focused on relieving pain through the use of systemic or local drugs, physical therapy, or surgery.7,8
Chondrocyte function has been found to be associated with inflammatory cytokines production and catabolic factors that contribute to the promote the development of OA. 4 In many cases, this leads to the transient accumulation and activation of innate immune cells. Interleukin (IL)-1β is a potent inflammatory cytokine identified in osteoarthritic synovial fluid. 9 Cartilage catabolic effects, cell proliferation, and regeneration can be triggered by IL-1β expression. IL-1β expression can also activate the mitogen-activated protein (MAP) kinase signaling pathway and enhance the expression of cartilage matrix-degrading enzymes, such as cyclooxygenase 2 (COX2), IL-6, tumor necrosis factor alpha (TNF-α), and matrix metalloproteinases (MMPs). 3 The expression of chemokines, such as chemokine (C-C motif) ligand 1 (CCL-1), in OA also plays an important role. 10 Elevated cytokine expression has been linked to joint disorders and other pathological conditions. Note that the use of non-steroidal anti-inflammatory drugs (NSAIDs) for the treatment of such disorders can have many adverse effects.11,12
Numerous plant extracts possess anti-inflammatory and antioxidant effects. Enteromorpha prolifera is a form of the green algae Ulva widely distributed along seashores around the world.13,14 Polysaccharides are the major active components isolated from Enteromorpha prolifera. They have been shown to have multiple beneficial effects, including anti-oxidation, anti-microbial actions, anti-hyperlipidemia, immunomodulation, and glucose metabolism. Recent studies have shown that Enteromorpha prolifera accelerates the metabolism of fat and decreases plasma lipoproteins levels 15 ; however, the protective effects of Enteromorpha prolifera against OA have not been widely studied. In this study, Enteromorpha polysaccharide extracts (EPEs) were obtained using different extraction methods. Our objective was to investigate the role of Enteromorpha prolifera on chondrocytes, macrophages, and synovium cells in OA models, and thereby assess the clinical applicability of Enteromorpha prolifera in OA treatment.
Materials and Methods
Materials and Reagents
Celecoxib (Pfizer Inc., Manhattan, NY), Ketoprofen (Swiss Co., Ltd., Tainan, Taiwan), Zoletil (Virbac., Grasse, Carros, France), and recombinant mouse IL-1β (ACROBiosystems., Newark, DE) were purchased from commercial companies. p38 (MAPK) beta and AP1/JUN antibodies were purchased from Thermo Fisher Scientific Inc. (Waltham, MA). Rabbit polyclonal COX-2 and TNF-α, horseradish peroxidase (HRP)-anti-rabbit, or mouse IgG antibodies were purchased from Santa Cruz Biotechnology, Inc. (Delaware, CA). Entermorpha prolifera was used to prepare EPEs included hot water polysaccharide extract (HPE), basic polysaccharide extract (BPE), and acidic polysaccharide extract (APE), provided by the Kinmen Fisheries Research Institute, Taiwan. Pulverized EPEs were extracted at a vehicle to raw material ratio of 50 mL/g via ultrasonic-assisted extraction using a frequency of 40 kHz at 100 °C over a period of 1.5 h. Neutralized water-soluble EPEs were precipitated by the addition of 95% EtOH (v/v) over a period of 48 h, followed by protein depletion. Finally, the extracts were concentrated using a rotary evaporator and powdered using a freeze-drying system (Kingmech, FD 20L-6S, Taipei, Taiwan). The individual total polysaccharide yield was determined via the phenol-sulphuric acid method using glucose as a standard reference.
Determination of Cell Viability
SW1353 and SW982 cells were seeded in 96-well flat-bottomed microtiter plates were maintained in an incubator at 37 °C. After incubation for 24 h with the HPE (0.04 mg/mL), BPE (0.13 mg/mL), APE (0.4 mg/mL), or IL-1β (5 ng/mL), the in vitro cytotoxicity of each EPE was assessed using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assays. 16
Inhibition of Nitric Oxide (NO) Production
All measurements of nitrite (a stable metabolite of NO) using a commercial NO detection kit (Griess Reagents System, Promega, Madison, WI) and according to manufacturer’s instructions. Briefly, each supernatant was combined with an equal volume of Griess reagent in a 96-well plate and incubated for 10 min. Absorbance was then measured using an enzyme linked immunosorbent assay (ELISA) reader at a wavelength of 540 nm (Tecan). A sodium nitrite standard curve was used to calculate the NO level. 17
The mRNA Isolation and Semi-Quantitative Reverse Transcription Polymerase Chain Reaction (sqRT-PCR)
We isolated mRNA and synthesized cDNA from SW1353 and SW982 cells after administering EPEs (0.01 or 0.04 mg/mL) plus IL-1β (5 ng/mL). The mRNA expression level was quantified using sqRT-PCR as previously described,
18
and each experiment was performed in triplicate. All primers are listed in
Oligonucleotides Primers Used in This Study.
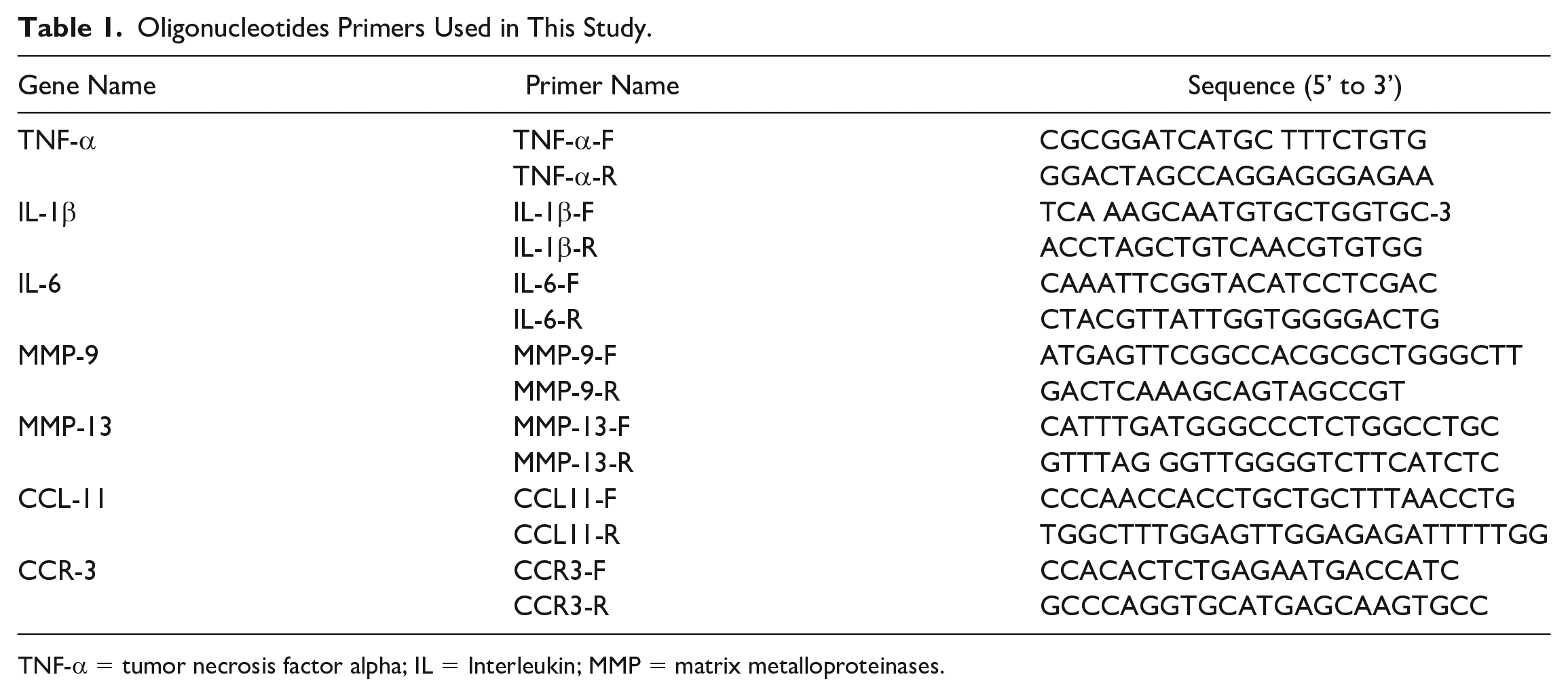
TNF-α = tumor necrosis factor alpha; IL = Interleukin; MMP = matrix metalloproteinases.
Western Blotting Analysis
The protein extraction and western blot analysis method was performed in accordance with methods described previously. 19 Briefly, protein samples were transferred to a nitrocellulose membrane (NC) via a 4% stacking gel and 10% resolving gel electrophoresis for analysis. After blocking with non-fat milk buffer, the membranes were then incubated with anti-TNF-α (1:500) or anti-COX-2 (1:2,000) antibodies for 1 h. The NC membranes were incubated with the appropriate secondary antibodies diluted 1:5,000 for 1 h. In measuring the immunoreactive proteins expression of TNF-α and COX-2, followed by development using a cold-light image analyzer (FUJIFILM LAS-3000). 16
Immunofluorescence Staining
To perform an immunofluorescence staining, SW1353 cells were fixed in 4% paraformaldehyde in PBS a for 30 min. Following this, the cells were stained with rabbit anti-p38 (MAPK) beta or anti-AP-1 (JUN) antibodies (1:200) for overnight. After washing with PBS, the cells were then incubated with anti-rabbit fluorescein-5-isothiocyanate (FITC) (1:500) at room temperature for 1 h. Finally, cells were stained using DAPI at room temperature for 10 min, after which the cells underwent imaging using an Olympus IX81 microscope (Olympus, Tokyo, Japan).
Molecular Weight Determination by Gel-Permeation Chromatography
A test sample of EPE was diluted to a concentration of 5 mg/mL in 20 mL of distilled water and utilized for injection. The samples were eluted using distilled water at a flow rate of 0.6 mL/min on a 45 °C column for gel-permeation chromatography (GPC). Dextran T10, T40, T70, and T500 were used to calibrate the polysaccharide molecular weight estimations. The regression line of the standard molecular weight versus elution volume plot was used to calculate the molecular weight of each fraction.
Animals and Treatment
Mice were purchased from the BioLASCO Taiwan Co., Ltd. All experimental protocols and procedures involving C57BL/6J mice (Gender: male; Age: 5 to 6 weeks old; Body weight: 25 to 30 g; Total numbers: n = 35) were approved by the Institutional Animal Care and Use Committee (IACUC) of the Southern Taiwan University of Science and Technology (approval no. STUST-IACUC-106-02), and they were conducted according to the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. For the treatment of mice, BPE, APE or celecoxib (100, 300 or 30 mg/kg, respectively) was conducted over a period of 28 days by oral administration, and the effect of celecoxib is relative to the treated group as a positive control. Specifically, a single experimenter (CPJ) with more than 1 year of experience in oral gavage of mice administered all treatments once daily for 28 consecutive days. The body weight of mice was assessed on a weekly basis during the tests.
Tactile Sensitivity Testing and Radiographic Assessments
Tactile sensitivity assay was performed as previously described. 20 Briefly, when the stimulus was administered, a positive reaction was defined as a quick withdrawal of the hind paw, and the number of positive responses for each stimulus was recorded. Tactile threshold was defined as a withdrawal reaction in 5 of 10 trials to a particular stimulus intensity, and thresholds were calculated once per mice. To understand changes in bone lesions and bone remodeling, and X-rays were obtained using a radiographic assessment method and Faxitron Specimen Radiography System (Field Emission Corp., McMinnville, OR). 20
Histopathological Examination
Harvested samples were fixed in 4% paraformaldehyde, followed by dehydration and embedding into paraffin. 5-μm sections were made for hematoxylin and eosin (HE) staining with safranin-O/fast green counterstain (Sciencell Research Laboratories., Carlsbad, CA). Stained slides were mounted in Micromount (Leica) and Motic BA 400 microscope with Motic Advance 3.0 software (Motic Co., Fujian, China). Finally, the classification of OA cartilage in the grading of animal cartilage were based on established criteria as previously reported, 20 by two blinded, experienced pathologists.
Statistical Analysis
Data were presented as mean ± SEM and analyzed using Sigma Plot 10.0 software (SPSS Inc., Chicago, IL). All data were representative of at least three independent triplicate experiments. The statistical significance of mean values between groups was evaluated by t one-way analysis of variance (ANOVA) and Tukey’s test. *P < 0.05 and **P < 0.01; #P < 0.05 and ##P < 0.01 was chosen to be statistically significant, respectively. 21
Results
APE Significantly Inhibits NO Production with the Lowest Cytotoxicity in IL-1β-Stimulated SW1353 and SW982 Cells
HPE, BPE, and APE (0.04, 0.13 and 0.4 mg/mL, respectively) as well as IL-1β (5 ng/mL) were tested in vitro using MTT assays in SW1353 and SW982 cells over a period of 24 h. APE (at any dose) and IL-1β (at 5 ng/mL) were shown not significant effect on the growth of SW1353 (
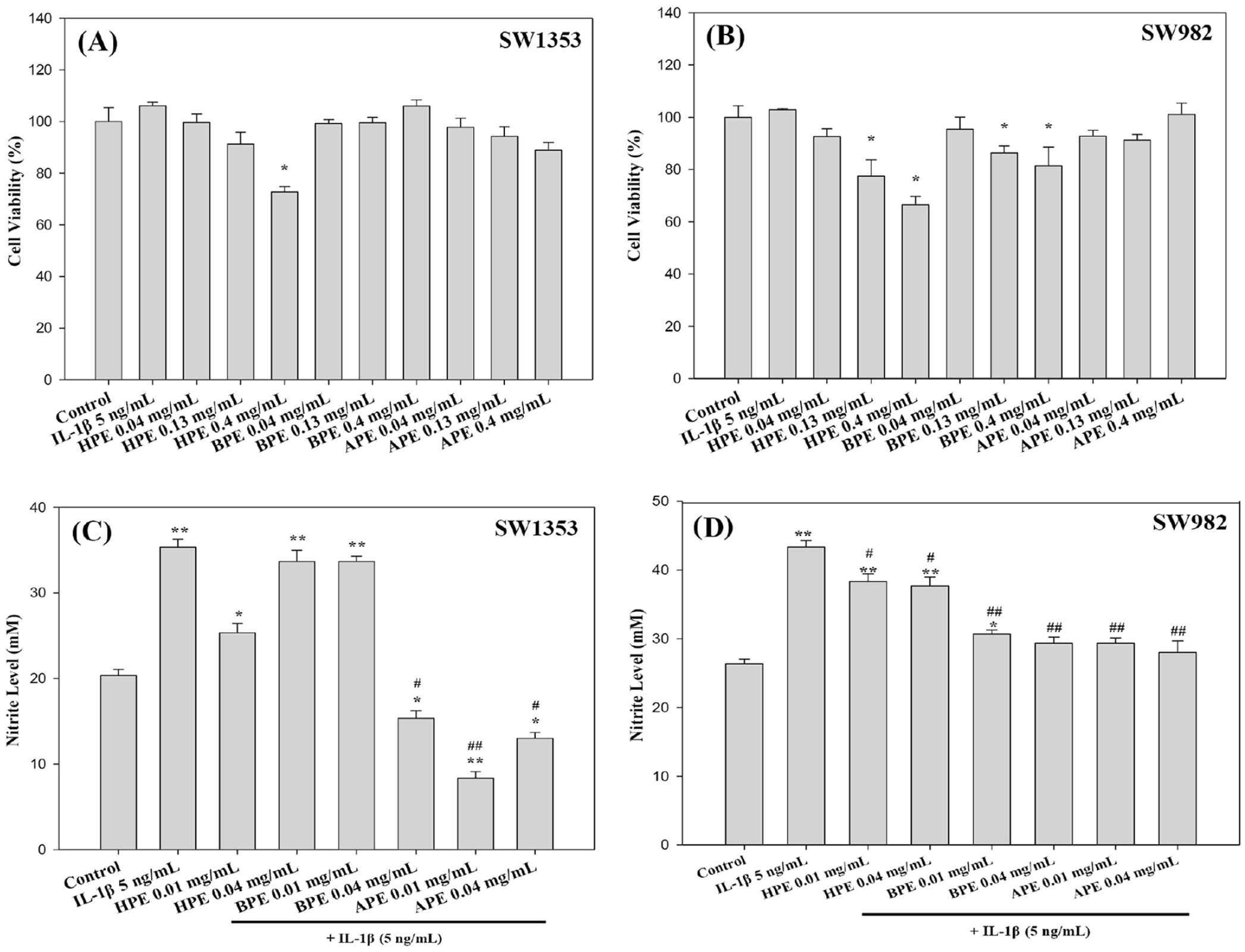
Cytotoxicity assay and effect of EPEs on NO production in IL-1β-stimulated SW1353 and SW982 cells. MTT assay results from SW1353
APE Profoundly Reduces Cytokine and Chemokine mRNA in IL-1β-Stimulated SW1353 and SW982 Cells
Our objective in this study was to investigate the effects of EPEs (HPE, BPE, and APE) on cartilage integrity (joint wear and deformation) in the development of OA and to identify which EPE is the best candidate for development as a pharmacologic treatment aimed at preventing OA progression. TNF-α, IL-1β, and IL-6 are the main pro-inflammatory cytokines, and MMP-9 and MMP-13 are major enzymes targeting type II collagen, type IV collagen, osteonectin, and proteoglycan in cartilage. Chemokines (CCL-11 and CCR-3) induce the expression of MMP-13 and other catabolic mediators in articular chondrocytes. They promote IL-6 production from synovial fibroblasts, which are involved in the pathophysiology of OA. After a 24-h incubation with EPEs plus IL-1β, the expression of genes encoding proteins in terms of synovial joint and cartilage homeostasis was examined in SW1353 or SW982 cells.
sqRT-PCR results revealed significantly elevated mRNA levels of IL-1β, IL-6, TNF-α, MMP-9, MMP-13, CCL-11, and CCR-3 in IL-1β-treatment group, compared with the vehicle control (**P < 0.01). Low-dose (0.01 mg/mL) treatment using BPE or APE was shown to reduce the mRNA levels of IL-1β, IL-6, TNF-α, MMP-9, CCL-11, and CCR-3 (but not MMP-13) in SW1353 (
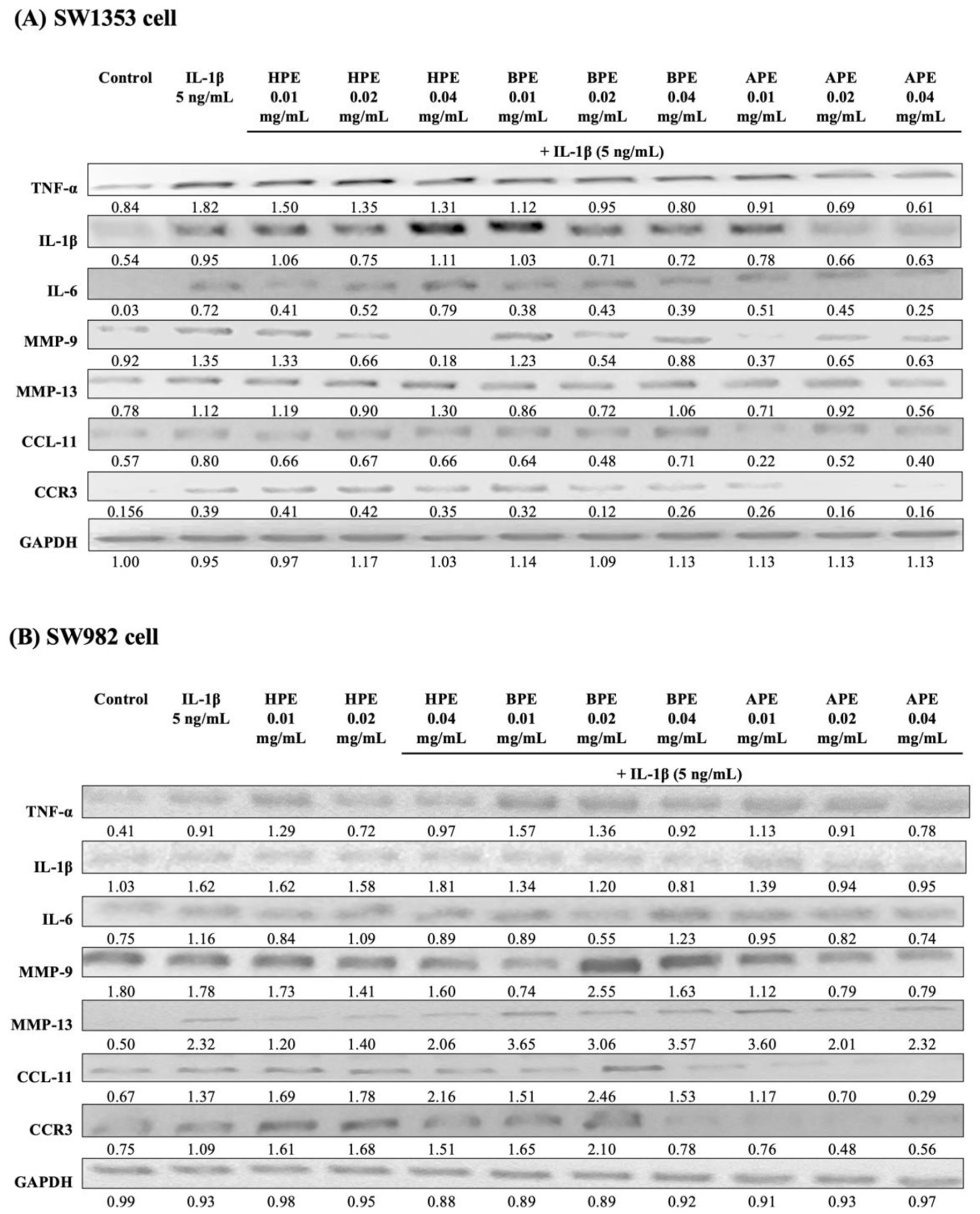
sqRT-PCR results indicating the effects of EPEs on the gene expression of cytokines and chemokines in IL-1β-induced SW1353 or SW982 cells. Cell lines SW1353
APE Attenuated the Expression of Proinflammatory Cytokines and p38/AP-1 in IL-1β-Stimulated SW1353 Cells
To elucidate the anti-OA effects of EPEs, we analyzed the expression of cellular transforming growth of TNF-α and COX-2 protein in SW1353 cells relative to β-actin after 24 h. We observed significantly lower HPE protein expression in SW1353 cells only at the high-dose (0.04 mg/mL) BPE and APE treatment groups (#P < 0.05, ##P < 0.01, respectively), compared to IL-1β treatment alone (
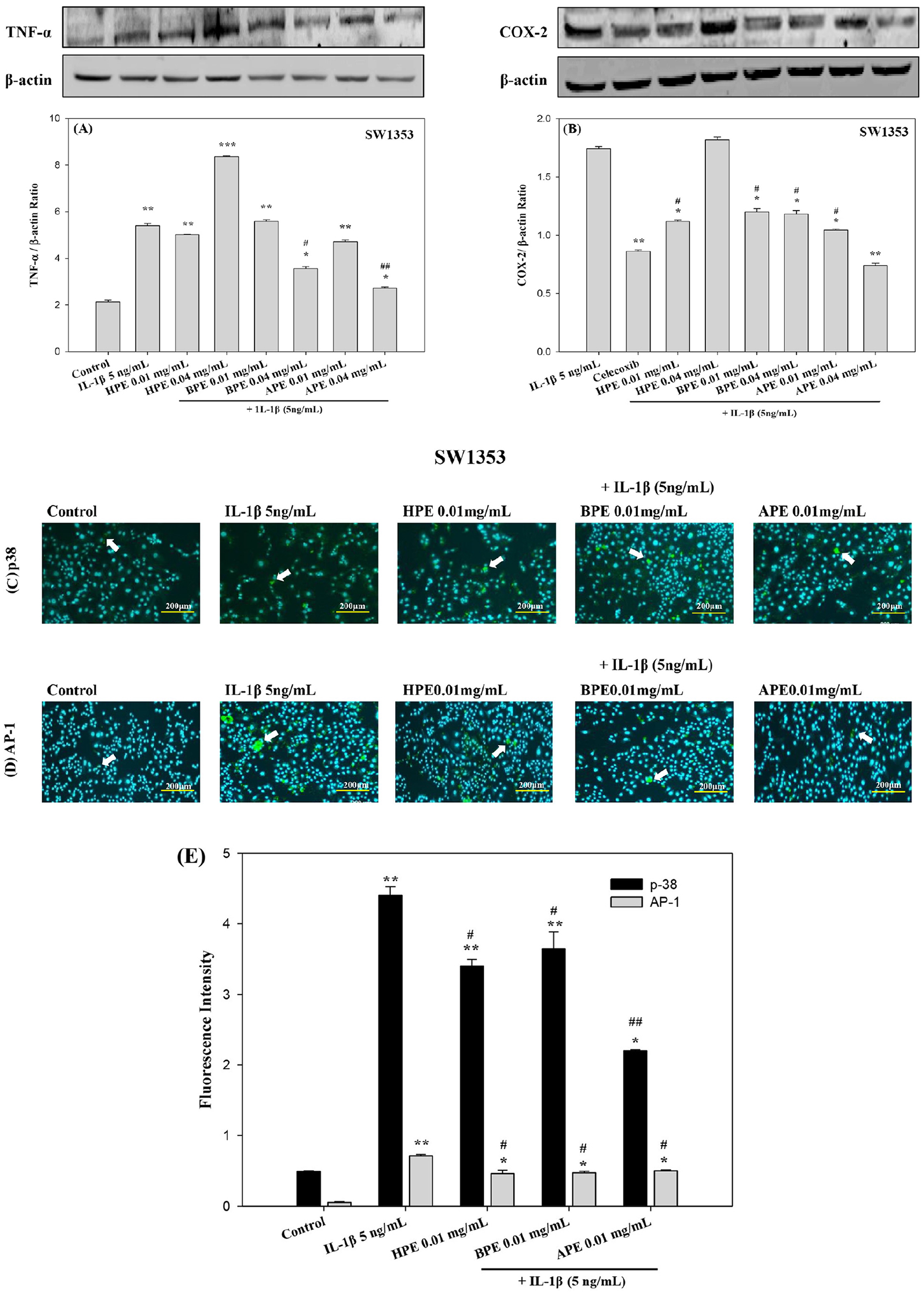
Protein expression of pro-inflammatory cytokines or p38 (MAPK) and AP-1 (JUN) in IL-1β-induced SW1353 cells using western blot analysis or immunofluorescence staining. Chondrosarcoma cell line SW1353 underwent co-stimulation with IL-1β and HPE, BPE, or APE for 24 h, after which the relative protein expression levels of COX-2 and TNF-αwere estimated using Western blot analysis; β-actin as a control. The data are respectively expressed as the COX-2/β-actin
Proinflammatory genes encoding TNF-α and COX-2 are transcribed by these factors in injured chondrocytic cells. We investigated the inhibitory effects of EPEs (HPE, BPE, and APE) on the IL-1β-induced p38/AP-1 pathway via immunofluorescence of chondrocytes. Administering IL-1β (5 ng/mL) significantly elevated the fluorescence intensity of p38 (
Properties of EPE Polysaccharides
Using the phenol/sulfuric acid method, we discovered that the three extracts of Entermorpha prolifera (HPE, BPE, and APE) respectively contained 21.3, 18.6, and 19.5 mg/mL of crude water-soluble polysaccharides (data not shown). Based on linear regression calculations of standard dextrans, the molecular weight of the polysaccharides, which is generally presented as a mean value distributed over a given range, is shown in
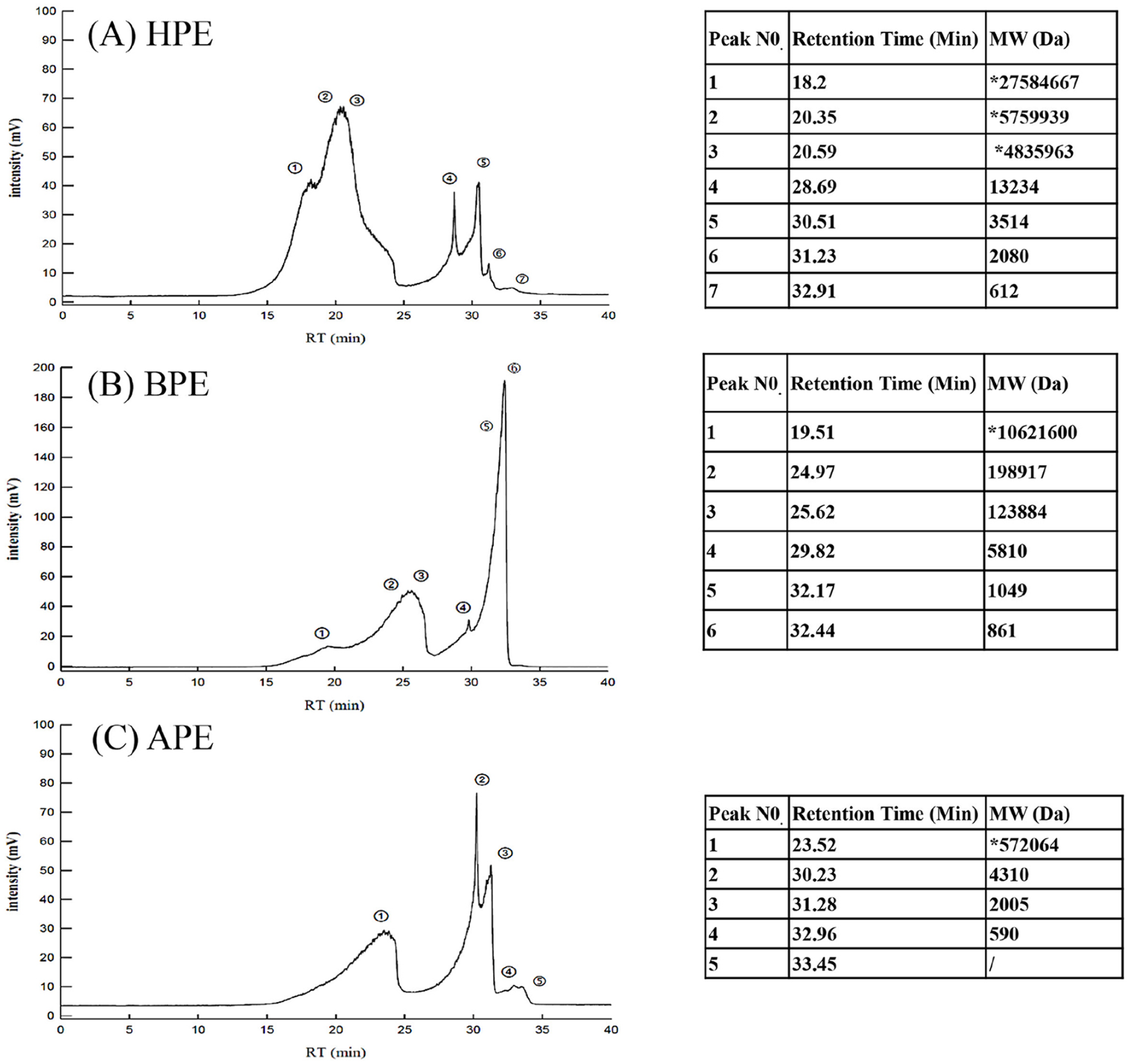
Profile of the molecular weight distribution of EPEs using gel permeation chromatography analysis. We obtained the molecular weight distributions of HPE
FTIR analysis of HPE, BPE, and APE revealed the transmittance at various wave numbers, to expose the key functional groups in the polysaccharides. Wide stretching peaks at 3356.40 cm-1(HPE), 3350.26 cm-1(BPE), and 3367.76 cm-1(APE) can be attributed to hydroxyl functional groups. The peaks appearing at 1222.3 cm-1 (HPE), 1230.85 cm-1(BPE), and 1201.3 cm-1(APE) can be attributed to S=O bending vibrations. The absorption peaks at 846.35 cm-1 (HPE), 848.49 cm-1(BPE), and 849.16 cm-1(APE) can be attributed to the stretching and bending vibrations of COS. The weak bands at 1031.21 cm-1 (HPE), 1026.80 cm-1(BPE), and 1032.05 cm-1(APE) can be attributed to an α-glycosidic bond (COH and COC) in the polysaccharide framework (data not shown).
Alleviating the Effects of ACLT-induced Joint Pain and EPEs Reduced the Extent of Osteolytic Lesions in Knee Joints in ACLT-Induced OA Mice
PWT decreases via the progression of OA and corresponding secondary clinical symptoms, which suggests the occurrence of mechanical hyperalgesia. Thus, we used the von Frey test to examine the effects of HPE, BPE, and APE on pain in mice that underwent ACLT surgery (
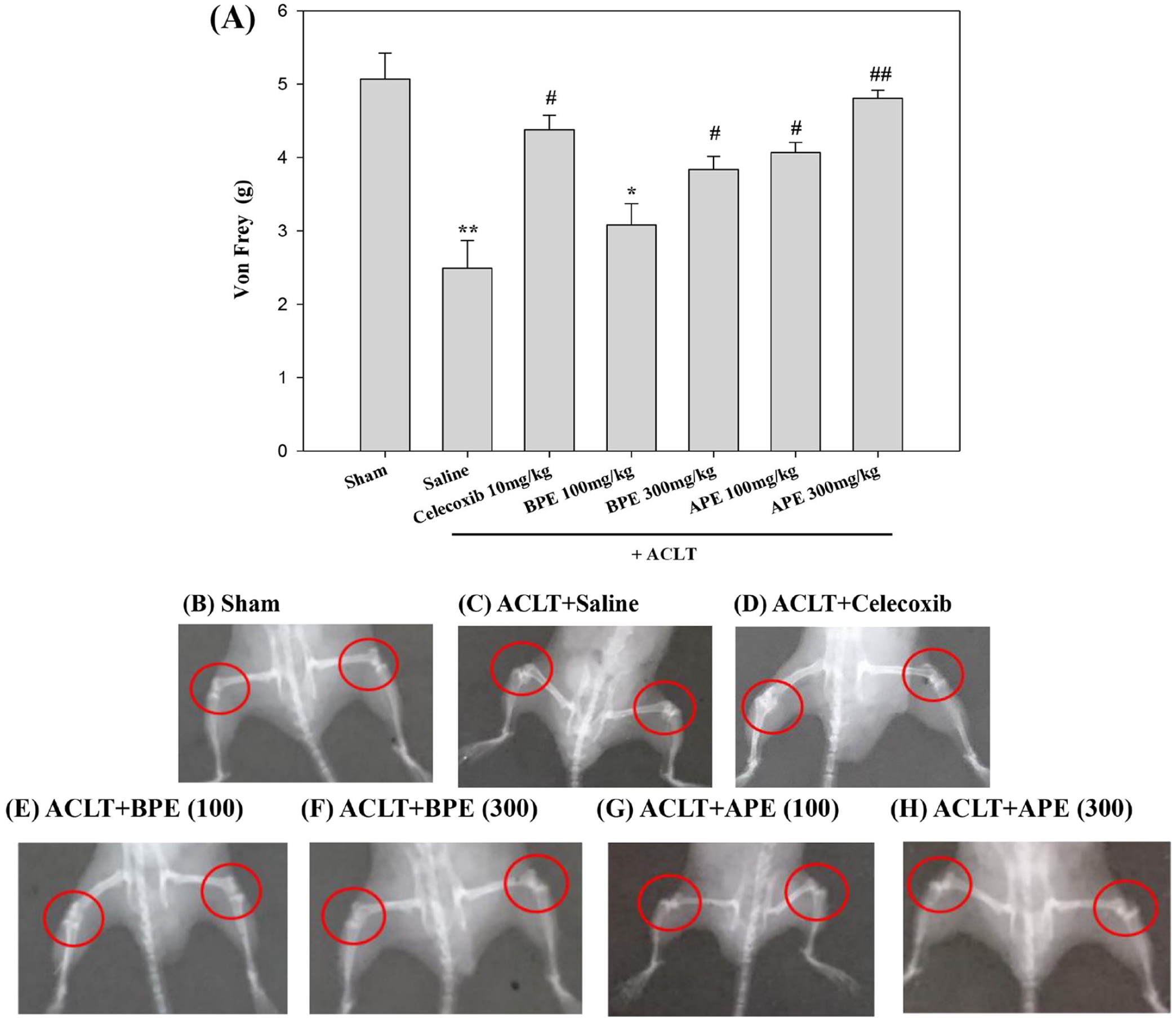
Influence of EPEs on tactile sensitivity in ACLT-induced OA-related allodynia, and radiographic results illustrating the effects of EPEs on ACLT-induced osteolytic bone lesions in OA mice.
Abnormal bone architecture and soft bone loss in the subchondral bone of the femur and tibia observed using radiographic analysis. At 28 days post-sham surgery, the femur and tibia in sham-operated controls were relatively well preserved. (
EPEs Moderated Cartilage Degeneration in the Knee Joints of ACLT-Induced OA Mice
The effect of EPEs on articular cartilage and subchondral bone in OA mice was analyzed by staining bone sections with HE (
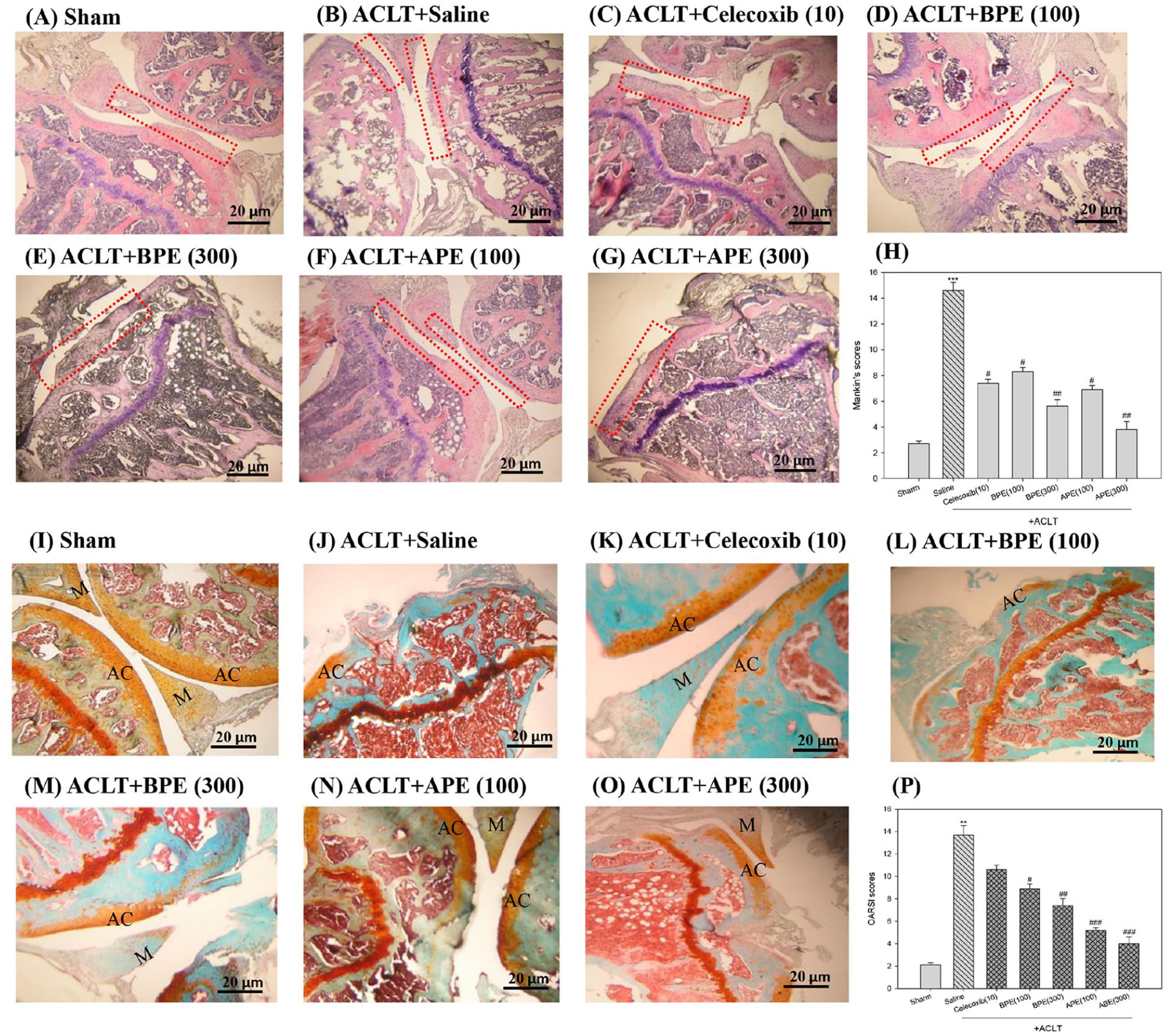
Morphological examination and histological changes of knee joints in ACLT-induced OA mice via HE or safranin-O/fast green staining. Representative joint sections from each groups in ACLT-induced OA mice. H&E staining showing signs of inflammation, synovial hyperplasia, and cartilage fissures along with disoriented and scattered dead chondrocytes
Accordingly, we sought to determine the efficacy of BPE and APE on OA progression by examining the structural integrity of articular cartilage (AC)/Meniscus (M) via microscope analysis with safranin-O staining (orange) and a survey developed by Osteoarthritis Research Society International (OARSI) (
Discussion
OA is a discontinuous phasic disease with slow progression, which alters all tissue in the affected joint and leads to chronic disability. At present, evidence from a diabetic mice model suggests that polysaccharides, such as those extracted from Enteromorpha prolifera, play an important role in anti-inflammation. 22 In this study, we demonstrated that polysaccharides extracted from EPEs could attenuate the overexpression of cytokines and chemokines in IL-1β-insulted chondrocytes.
Previous studies have determined that IL-1β and TNF-α are secreted early in the progress of OA.
23
IL-1β is known to play a significant role in the pathogenesis of OA, in vitro. IL-1β insult stimulates COX-2 expression, which is responsible for PGE2 and has been implicated in bone resorption and joint pain in OA.
24
Researchers have also reported on the upregulated production of MMPs and other inflammatory cytokines in cases of OA.
25
Inhibition of IL-1β-stimulated production of inflammatory mediators is a potential approach for the treatment of OA. In the present study, pretreatment with EPEs significantly reduced IL-1β-stimulated MMPs and COX-2 expression (
MMPs as proteolytic enzymes, which play a key role in the degradation of articular cartilage,
26
making it a potential target for the OA treatment. The abundant expression of MMPs in joint disorders is largely due to their ability to degrade the extracellular matrix (ECM). The fact that MMP-1, MMP-3, and MMP-13 are mainly present in cartilage suggests that it is involved in OA progression.27,28 Here, we sought to determine whether EPEs inhibit IL-1β-induced expression and/or activity of MMPs and inflammatory mediators in chondrocytes. IL-1β induced MMP-1, MMP-3, MMP-13, and a disintegrin and metalloproteinase with thrombospondin motifs 4 (ADAMTS-4) expression in human tendon cells was reported.
29
In this study, IL-1β was shown to induce the expression and activity of MMP-9 and MMP-13 in articular chondrocytes. Thus, it appears that EPEs exert chondroprotective effects, suggesting that EPEs could be a novel agent for the control of cartilage damage in OA
The strong expression of inducible nitric oxide synthase (iNOS), COX2, and MMPs in arthritic joints can be attributed to a tightly regulated and synchronized signaling cascade involved in the MAPK signaling pathway.30,31 NO generation suppresses iNOS phosphorylation, thereby delaying the activation of p38 and the extracellular signal-regulated kinase 1/2 (ERK1/2) MAP kinases. NO activates ERK signaling by downregulating MAP kinase phosphatase was reported. 32 MAPKs are activated via the phosphorylation of specific tyrosine and threonine residues in response to inflammatory signals. In our study, IL-1β was shown to enhance the p38 (MAPK) and ERK signaling pathways.
To further validate our in-vitro findings, we evaluated the potential of EPEs to alleviate ACLT-induced joint pain of osteolytic lesions or cartilage degeneration in knee joints at ACLT-induced OA mice
However, our data suggest that APE may be a potent inhibitor of joint degeneration associated with OA, in-vitro and in-vivo studies. Although the molecular mechanism of APE in OA is still not clear, it is possible that they reduce or protect the IL-1β-stimulated SW1353 cells from the expression of proinflammatory cytokines and p38/AP-1. In addition, the exact structure and molecular weight of effective small molecules in APE extracts still need to be further identified. Further research is required to test this hypothesis and to identify the exact effective small molecule structures.
In conclusion, the different methods used in EPE extraction were shown to affect the bioactivity of the resulting polysaccharide molecules in the extracts. Extraction via hot water produced EPEs with a larger number of high molecular weight polysaccharides. APE-produced EPEs proved particularly effective in attenuating the expression of cytokines and chemokines in the OA model. Thus, it appears that acid-phase extraction could be used to produce nutritional supplements for OA therapy. APE significantly reduced TNF-α expression and chemokine release, thereby reducing oxidative stress and also played an important role as an MMP activator against IL-1β-induced inflammation. Thus, it appears that APE could be used to produce nutritional supplements in the prevention and treatment of OA.
Footnotes
Author Contributions
Conceptualization, CCW, JWL, KHC, CYC and J JC; methodology, CCW, JWL, KHC, YSC, YHC, YJP, CHC, CYC and JJC; software, CCW, JWL, KHC. and YSC; validation, CCW, JWL, KHC, YSC, YHC, YJP and CHC; formal analysis, CCW, JWL, YHC, YJP and CHC.; investigation, CCW, JWL, KHC, YSC, YHC and CHC.; resources and data curation, CCW, JWL, KHC, YSC, YHC, YJP and CHC; writing—original draft preparation, CCW and JWL; writing—review and editing, CCW, JWL, CYC and JJC; visualization, CCW, JWL, KHC, YSC, YHC, YJP and CHC; supervision, CYC and JJC; project administration, CCW, CYC and JJC.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Ministry of Science and Technology grant number MOST- 102-2320-B-218 -001 at Taiwan.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
