Abstract
Objective
The purpose of this study was to determine the safety and efficacy of hypothermically stored amniotic membrane (HSAM) for the treatment of cartilage lesions of the knee using imaging, patient-reported outcomes (PROs), second-look arthroscopy, and histology. Patients were treated with HSAM and followed for 2 years.
Design
Subjects with focal chondral lesions of the femur (International Cartilage Repair Society grade 3-4) were enrolled in this single-arm prospective study. Standard of care imaging was completed. PROs, including the Knee Injury and Osteoarthritis Outcome Score (KOOS), Marx Activity Scale, and Visual Analog Scale (VAS), were collected at baseline and at 3, 6, 12, 18, and 24 months. Three subjects underwent an optional arthroscopy and biopsy of the repair at 24 months.
Results
Ten subjects were enrolled and completed the study. At 24 months, KOOS Sports & Recreation improved 173.3% and Quality of Life improved 195.3% from baseline. Marx Activity Scale increased 266.8% from 12 to 24 months. VAS scores improved 84.8% and 81.0% from baseline to 24 months for average and maximum pain. Modified Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) scoring showed that 7 of 10 subjects had complete defect repair and filling by 24 months. Biopsy staining for collagen II revealed integration and remodeling of HSAM into a mix of hyaline-like cartilage and fibrocartilage matrix.
Conclusion
This study provides evidence supporting the safety and efficacy of HSAM for treating symptomatic cartilage lesions. Subjects showed a high degree of defect fill and integration with the native cartilage and reported improvements in pain and function post-treatment. Results provide important original data for future clinical trials.
Introduction
Articular cartilage consists mainly of water, type II collagen, and proteoglycans.1,2 Due to its avascular, aneural properties along with anaerobic metabolism, the repair capacity of articular cartilage is limited.1,2 Chondral lesions can arise due to cartilage trauma (contact or noncontact) or malalignment of the knee. 1 These lesions are present in up to 10% to 12% of all individuals, 3 and, if left untreated, can cause pain and loss of function.3,4
Currently, there are several treatment options for focal cartilage lesions. Microfracture involves disruption of the bone to stimulate cells from the bone marrow underneath the cartilage, resulting in the release of blood that contain mesenchymal stem cells (MSC).1,2 In addition, the use of autologous cartilage implantation (ACI) is a 2-surgical procedure method of repairing lesions that involves taking chondrocytes from a patient, culturing them, and then implanting them back into the same patient in a second surgical procedure.1,5 Particulated juvenile articular cartilage (PJAC) is an allograft where cartilage from donor patients under 13 years old are cut into cubes and applied to cartilage lesions held by fibrin sealant. 4 Osteochondral autograft/allograft transfer system (OATS) involves taking a plug of hyaline cartilage and underlying subchondral bone from a non-weight-bearing area of the knee and transplanting it into cartilage lesions.1,2 However, there are limitations and mixed results regarding the overall effectiveness of these treatments. Long-term studies evaluating patients aged 10 to 14 years following microfracture found that 46% of patients had a poor long-term outcome, as defined by knee replacement surgery or a Lysholm score below 64. 6 Although ACI is promising, there are significant limitations, including the need for multiple surgeries, cost of treatment, and it requires up to 3 years for cartilage maturation and integration into the repaired lesion.2,5 PJAC seems promising, but there is a risk for shortage of appropriate allograft donor tissue and a lack of published long-term follow-up studies, while varying topology of the donor site to the transplantation site in OATS treatment could affect long-term biomechanics and loading.7,8 Due to the limitations of current available treatment options, there is a need for a financially feasible, allogeneic, and surgically straightforward approach for the treatment of symptomatic articular cartilage lesions.
One potential treatment option for symptomatic cartilage lesions is the use of placental-derived membranes. These membranes are harvested from consented donors following cesarean sections and contain several anti-inflammatory cytokines and growth factors, such as interleukin-1 receptor antagonist (IL-1Ra), along with tissue inhibitors of matrix metalloproteinases (TIMPs).9-11 Extracellular matrix proteins relevant to cartilage repair, such as hyaluronic acid and proteoglycans, are also reported to be abundant in human amniotic membranes.12,13 Furthermore, prior studies have shown suitability for amniotic membranes to be used as a scaffold for cartilage repair. 14
In this single-arm prospective study, patients with symptomatic cartilage lesions were treated with a commercially available hypothermically stored amniotic membrane (HSAM) and followed for 2 years post-implantation. The purpose of this study was to determine the safety and efficacy of HSAM for the treatment of cartilage lesions using imaging and patient-reported outcomes (PROs) as assessments.
Methods
Study Population and Study Design
This prospective study (NCT02837484) enrolled 10 patients at 1 clinical center by 1 surgeon for elective treatment of symptomatic cartilage lesions in the knee, and patients were followed for 24 months ( Fig. 1 ). Western Institutional Review Board (WIRB) approval was obtained (WIRB protocol #1162030), and informed consent was obtained from each patient before any study-related activities were performed. In all, 5 men and 5 women participated in the study. Eligible patients included adults 18 to 55 years old with symptomatic, focal, contained chondral lesions (International Cartilage Repair Society [ICRS] grade 3 or 4A of the femur [condyles and trochlea]), with defect areas ranging between 1 and 5 cm2 after debridement. Any necessary concomitant ligament, meniscal, and/or alignment issues were repaired simultaneously. Eligible patients were at least 3 months post-surgery of any previous surgeries on the study knee, with an asymptomatic, stable, and fully functional contralateral knee.
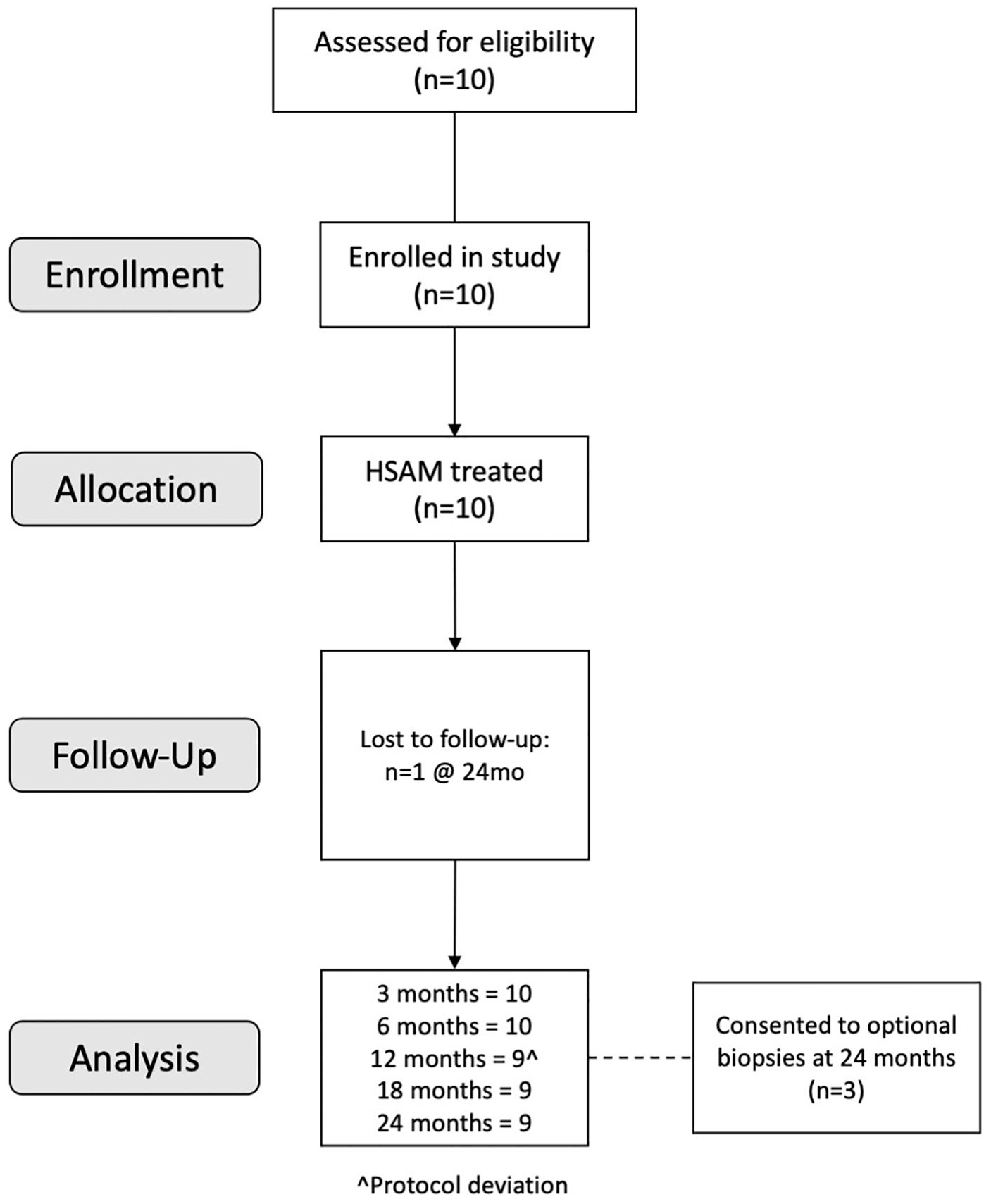
Consolidated Standards of Reporting Trials flow diagram used to describe the grouping and flow of patients through the clinical trial. HSAM = hypothermically stored amniotic membrane.
Exclusion criteria included patients with a body mass index (BMI) >35 kg/m2, the presence of bipolar lesions or kissing lesions of ipsilateral compartment, prior total meniscectomy of either knee, failed microfracture within 12 months of surgery, or radiographic malalignment greater than 5° as measured from the hip, knee, and ankle mechanical axis. In addition, patients with a disease diagnosis by clinical and/or radiographic assessment including osteoarthritis, rheumatoid arthritis, gout, or avascular necrosis were excluded; those receiving workers’ compensation for their injury were also excluded.
Consenting patients were asked to carry out a preoperative visit that included the International Knee Documentation Committee (IKDC) knee examination performed by a qualified medical professional and collection of baseline data including demographics, knee history, medical history, and medication usage. Patients also underwent standard of care procedures, including preoperative magnetic resonance imaging (MRI) and radiographs. Study procedures included PROs, including the Knee Injury and Osteoarthritis Outcome Score (KOOS), the Marx Activity Scale, and the Visual Analog Scale (VAS). After surgery, patients returned for follow-up at 3, 6, 12, 18, and 24 months, at which time the IKDC knee examination was performed, and PROs were collected. Postoperative MRI was performed at 6, 12, and 24 months; radiographs were taken at 12- and 24-month follow-up visits. In addition, subjects were asked to participate in an optional 24-month arthroscopy and biopsy procedure to assess the regenerate cartilage tissue. Patients were queried at all follow-up visits regarding any complications or events that they may have experienced since the last visit, including events unrelated to the knee.
Surgical Technique
A small medial or lateral arthrotomy was used to expose the defects on the medial condyle, lateral condyle, or trochlea. The cartilage defect was then debrided to define vertical borders at the periphery of the lesion. The central portion of the defect was taken down to the calcified cartilage layer with an attempt to not significantly penetrate the subchondral bone layer. Bleeding was controlled with limited electrocautery along with epinephrine pledgets and fibrin glue when needed. In cases where a tourniquet was used for initial exposure, it was released prior to this process. The HSAM (Affinity®; Organogenesis) was removed from the container using sterile technique, and the stromal and epithelial sides were identified with the use of a cotton swab. A thin layer of fibrin glue was then applied to the bone defect and the membrane laid down with the stromal side toward the subchondral bone. During application, every attempt was made to have the stromal layer contacting the subchondral bone and extending the membrane to the periphery of the defect. Any excess membrane was then folded over on itself in a “taco” technique to utilize the entire membrane. A single 2.5 cm × 2.5 cm patch was used for all defects, and fibrin glue was applied to the construct to secure the graft, with no sutures or additional stabilization required. After fibrin glue application, approximately 10 minutes of drying time was allowed to ensure the stability at the periphery of the defect. This technique yielded stability of the graft while maintaining a level within the construct that was at least 1 to 2 mm below the level of the normal articular cartilage. The procedure was performed with the knee in flexion if the defect was on either condyle or with the knee in extension for trochlear lesions. When the glue was dry and the construct was stable, the knee was extended with moderate varus or valgus force depending on which condyle was affected to ensure that the patch stayed in place until complete extension was achieved. At that point, the closure was done in extension and the knee kept this way until dressings and an immobilizer were put in place.
Postoperatively, patients were kept in strict extension in the immobilizer for the first 48 hours, with touch-down weight-bearing only. Thereafter, range of motion was done in a gentle fashion with isometric quadriceps strengthening exercises. Touch-down weight-bearing with crutches was continued for 6 weeks, with progression to weight bearing as tolerated. Trochlear lesions were allowed weight bearing as tolerated after the first 2 weeks. Physical therapy was tailored to each subject depending on the location of the cartilage procedure and based on the type (if any) of concomitant surgery performed.
MRI Assessments
A standardized MRI protocol was used on all study patients. A 1.5-Tesla 16-channel knee coil by Philips Achieva with 3.2 software (Philips Healthcare, Andover, MA) was used for all the examinations. The protocol consisted of coronal, sagittal, and axial sequences with turbo spin echo (TSE) with and without fat suppression. The matrix size was 448 by 448 pixels with a field of view of 160 mm × 160 mm and a slice thickness and gap of 4.0/0.04. The total examination time was about 30 minutes.
Modified Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) Scoring was used to tabulate MRI results at 6, 12, and 24 months. All scans were read by an experienced musculoskeletal radiologist; evaluated parameters included the percentage of lesion fill, integration of new cartilage to the border zone of native cartilage, surface quality of repair tissue, signal intensity of new cartilage compared with native cartilage, and an assessment of subchondral bone.
Cartilage Biopsy Histology
At 24 months, patients could consent to an elective arthroscopic procedure where biopsies of the cartilage repair were taken. Biopsies were placed in formalin fixative and processed for histology using standard techniques. Serial sections 5 μm thick were cut from the formalin-fixed, EDTA-decalcified, paraffin-embedded tissue blocks and floated onto charged glass slides (Super-Frost Plus, Fisher Scientific, Pittsburgh, PA) and dried overnight at 60°C. Sections were obtained from tissue blocks and stained for standard stains, including (1) hematoxylin and eosin (H&E), (2) Alcian blue, and (3) Masson’s trichrome. All sections for immunohistochemistry (IHC) were deparaffinized and hydrated using graded concentrations of ethanol to deionized water. Tissue sections were all subjected to antigen retrieval by sodium citrate buffer working solution (pH 6, AP-9003-500; ThermoFisher Scientific, Kalamazoo MI) in a 70°C water bath for 20 minutes, with the buffer preheated to 70°C prior to application. Following antigen retrieval, all sections were washed gently in deionized water and then transferred into 0.05 M Tris-based solution in 0.15 M NaCl with 0.1% v/v Triton-X-100, pH 7.6 in Tris-buffered saline (TBST). Endogenous peroxidase was blocked with 3% hydrogen peroxide (H325-500; Fisher Scientific, Fair Lawn NJ) for 15 minutes. To reduce further nonspecific background staining, slides were incubated with 3% normal goat serum for 30 minutes (Sigma-Aldrich, St. Louis, MO) at room temperature. All slides then were incubated at 4°C overnight with various antibodies (collagen I: ab34710, Abcam, 1:50; collagen II: ab85266, Abcam, 1:50; aggrecan: ab3778, Abcam, 1:50; lubricin: ab28484, Abcam, 1:50; Abcam, Cambridge, MA). Negative control sections were stained by eliminating the primary antibodies from the diluents. After washing with TBST, sections were then incubated with the matching secondary antibody conjugated with horseradish peroxidase (HRP) (collagen I, collagen II, lubricin: Goat Anti-Rabbit IgG H&L [HRP], ab6721, Abcam, 1:1000; aggrecan: Goat Anti-Mouse IgG H&L (HRP), ab6789, Abcam, 1:200; Abcam). Diaminobenzidine (DAB; Scy Tek Laboratories, Logan, UT) was used as the chromogen and hematoxylin (Richard-Allen Scientific, Kalamazoo, MI) was used as the counterstain. Images were taken using an inverted microscope (Nikon Eclipse Ti; Nikon, Melville, NY) with a 2x objective.
Statistical Analysis
For all figures, average ± standard error is shown; for demographic values, average ± standard deviation is reported. Due to the small size of the study (n = 10), no formal statistical analysis was completed.
Results
In this single-arm prospective study, 10 patients were enrolled and completed the study, and 3 patients participated in the optional 24-month biopsy procedure ( Fig. 1 ). The average age was 41.2 years, with 50% of the study population being female. The average BMI of the subjects was 27.8 lbs/in2. Comorbidities of the subjects included asthma/pulmonary disease (1/10, 10%), back pain (2/10, 20%), diabetes (1/10, 10%), high blood pressure (1/10, 10%), osteoarthritis (3/10, 30%), overweight (2/10, 20%), and rheumatoid arthritis (1/10, 10%). Five subjects reported no comorbidities (5/10, 50%), and 9 subjects had never smoked (9/10, 90%). Additional demographic characteristics can be found in Table 1 .
Demographic Characteristics of the Study Population.
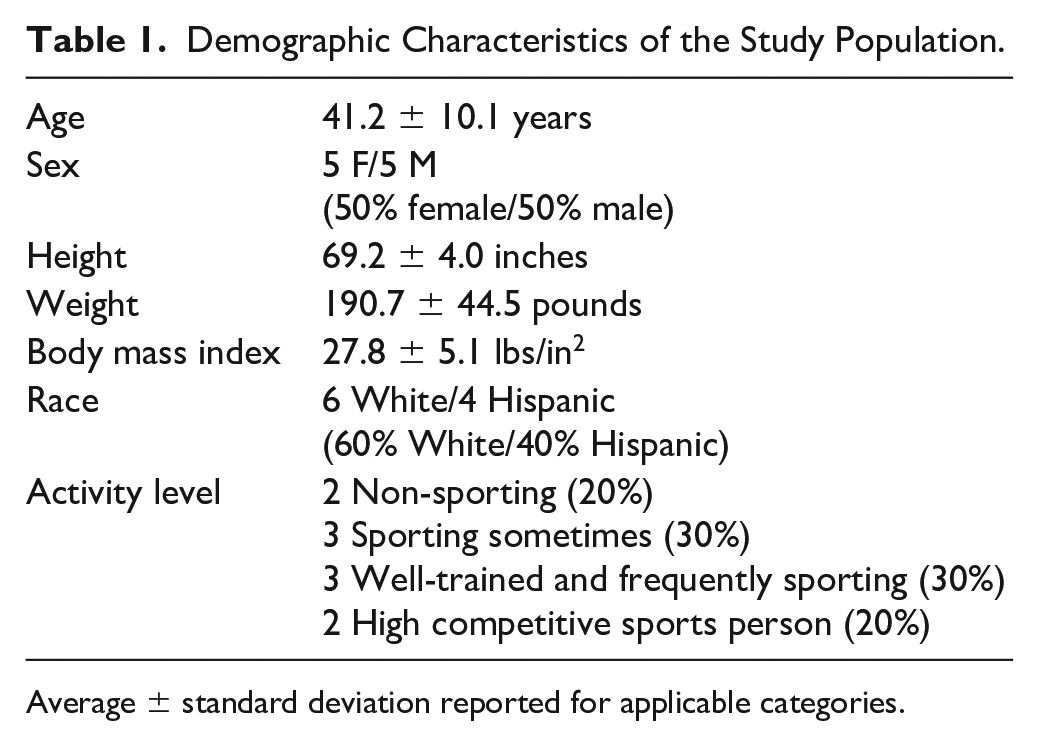
Average ± standard deviation reported for applicable categories.
In this study, 10 subjects had 11 lesions that were treated using HSAM. These lesions were all on the femur, with ICRS cartilage lesion grades of 3B, 3C, or 3D, indicating defects down to the calcified layer, bone, and/or with blisters. 15 The average area of the cartilage lesions was 3.03 cm2. Additional lesion characteristics can be found in Table 2 . Of note, there were 6 subjects who had concomitant procedures performed at the same time as treatment with HSAM. These procedures included anterior cruciate ligament (ACL) reconstruction (1/10, 10%), medial patellofemoral ligament (MPFL) reconstruction (1/10, 10%), meniscal partial resection (2/10, 20%), and osteotomy (2/10, 20%).
Lesion Characteristics of the Study Population.
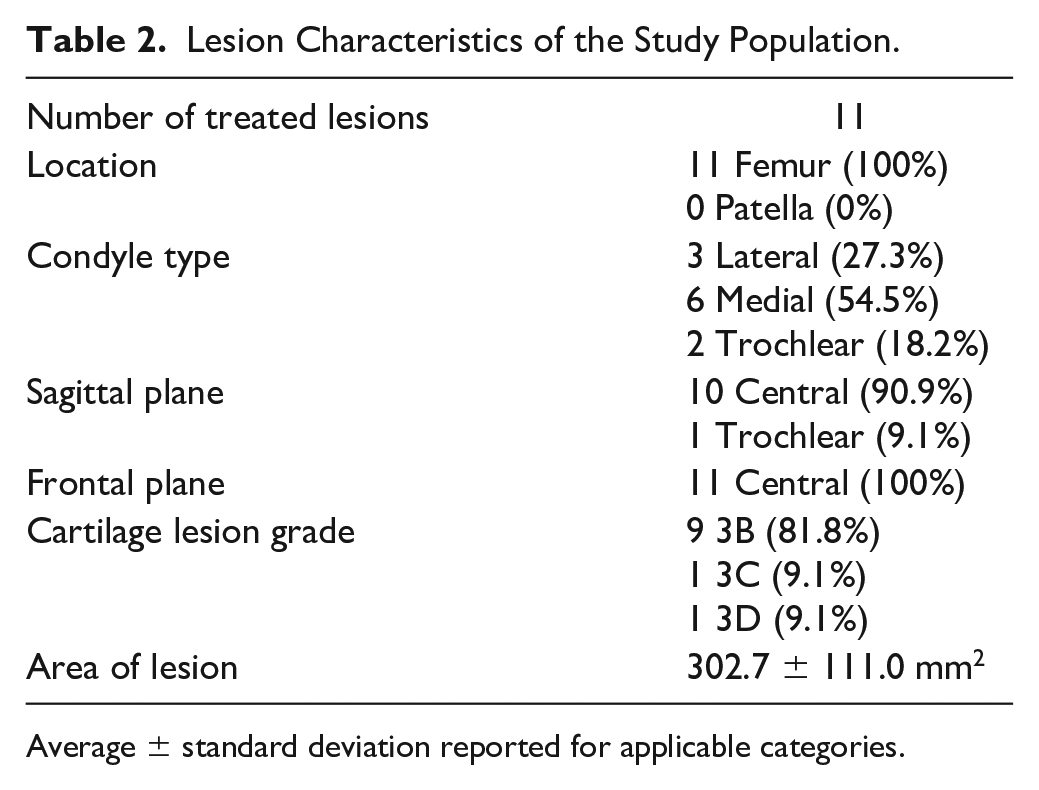
Average ± standard deviation reported for applicable categories.
Subjects filled out PROs throughout the course of the study. KOOSs are shown in Figure 2 , including the KOOS Symptoms ( Fig. 2A ), KOOS Pain ( Fig. 2B ), KOOS Activities of Daily Living (ADL, Fig. 2C ), KOOS Sports & Recreation ( Fig. 2D ), and KOOS Quality of Life (QoL, Fig. 2E ). All the KOOS subscales showed improvements from baseline to 24 months post-treatment with HSAM. At the 24-month time point, KOOS Symptoms improved 42.7%, KOOS Pain improved 43.9%, KOOS ADL improved 26.3%, KOOS Sports & Recreation improved 173.3%, and KOOS QoL improved 195.3% from baseline.
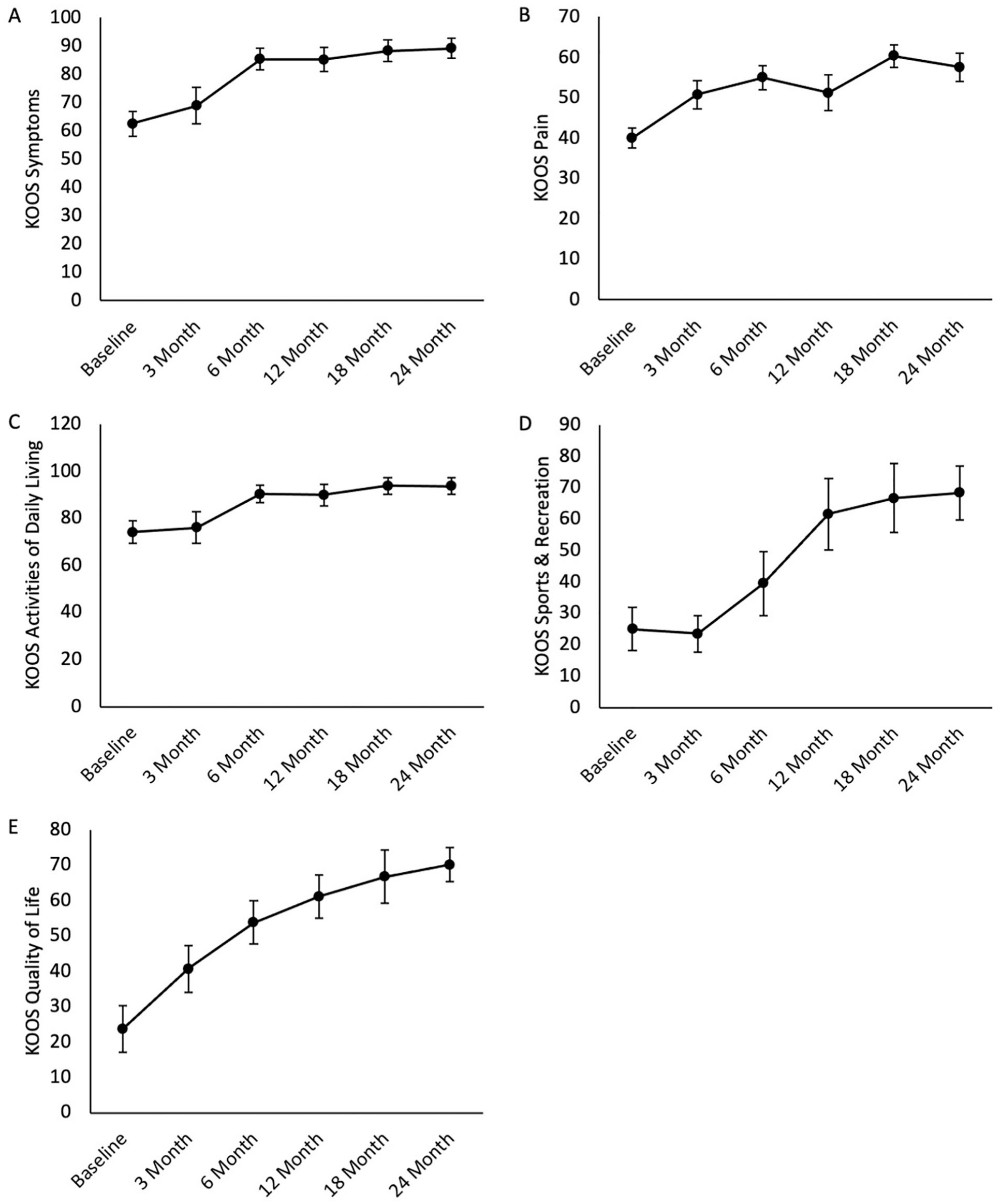
KOOS subscores for the entire 24-month period. Average ± standard error reported for (
In addition, subjects also filled out the Marx Activity scale, which asks whether patients performed any running, cutting, pivoting, and deceleration activities over the past year. Subjects were limited by standard of care postoperative procedures in performing these activities until the 12-month mark, which is taken as the baseline for this PRO. Subjects reported a total score of 1.3 ± 0.7 points at 12 months, 5.3 ± 1.7 points at 18 months, and 4.9 ± 1.3 points at 24 months, indicating an improvement of 266.8% from 12 months to 24 months.
Subjects also completed the VAS (data shown in Figure 3 ). VAS Average Pain ( Fig. 3A ) and VAS Maximum Pain ( Fig. 3B ) were completed, along with a VAS scale measuring how the procedure met the subject’s expectations. Improvement was seen in all 3 of these subscales from baseline to 24 months, with improvements of 84.8% and 81.0% for VAS Average Pain and Maximum Pain. In addition, at 6-, 12-, 18-, and 24-month visits, scores for how the study procedure met the subject’s expectations were >80 (on a 100-point scale).
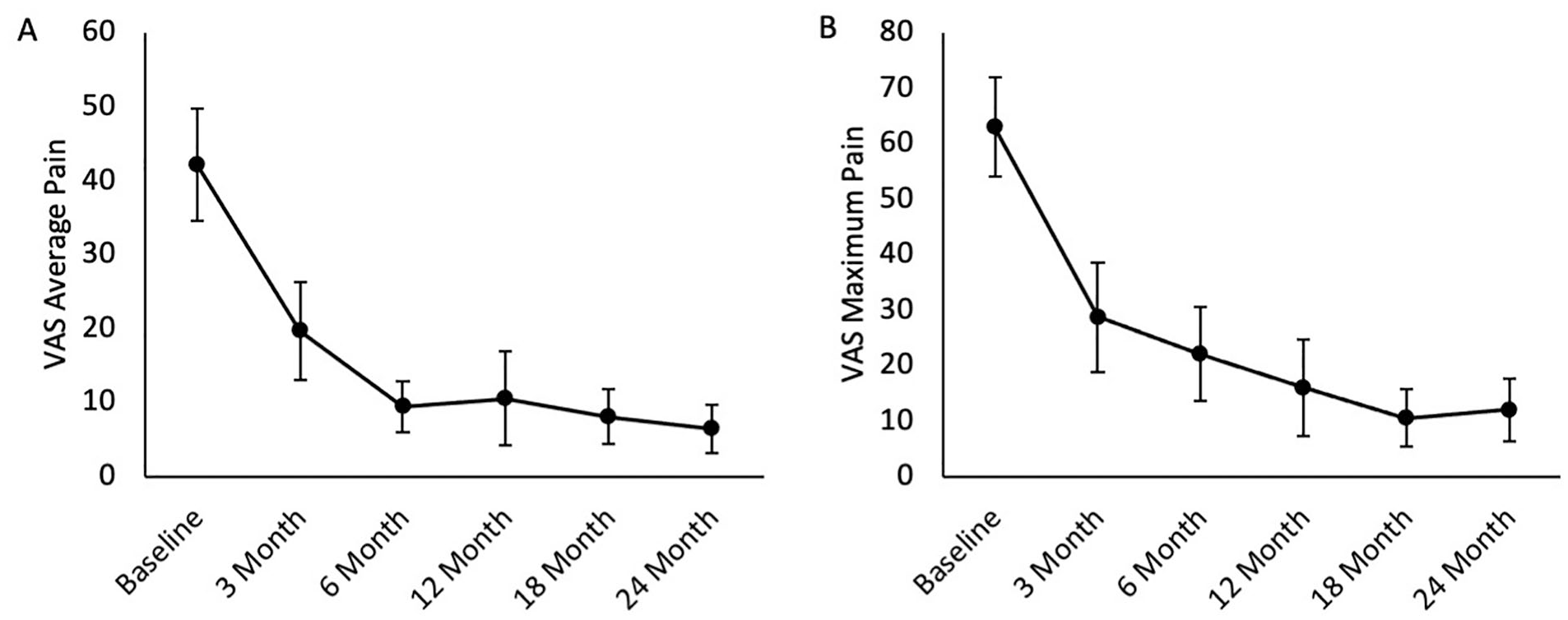
VAS subscores for the entire 24-month period. Average ± standard error reported for (
MRI scans were completed at 6, 12, and 24 months; modified MOCART scoring is shown in Table 3 , and the preoperative and 24-month MRI scans are shown in Figure 4 . By 24 months, 7 subjects (7/10, 70%) had complete defect repair and filling, while 2 of the 3 remaining subjects had incomplete defect repair >50% of the adjacent cartilage. Eight subjects (8/10, 80%) had complete integration with cartilage at the border zone, while 7 subjects (7/10, 70%) had intact tissue at the repair surface. When measuring signal intensity of the repair tissue, 8 subjects (8/10, 80%) were graded as nearly normal. Finally, 7 subjects (7/10, 70%) had intact subchondral bone with no associated bone marrow edema at the lesion site. Of note, there was 1 noncompliant subject who began running at 6 weeks post-implantation despite not being allowed to do so until the 12-month mark. This noncompliant subject was the only subject that did not see defect repair filling at the 6-, 12-, or 24-month mark following treatment with HSAM (patient 8, Fig. 4 ).
Modified MOCART Scoring at 6, 12, and 24 Months.
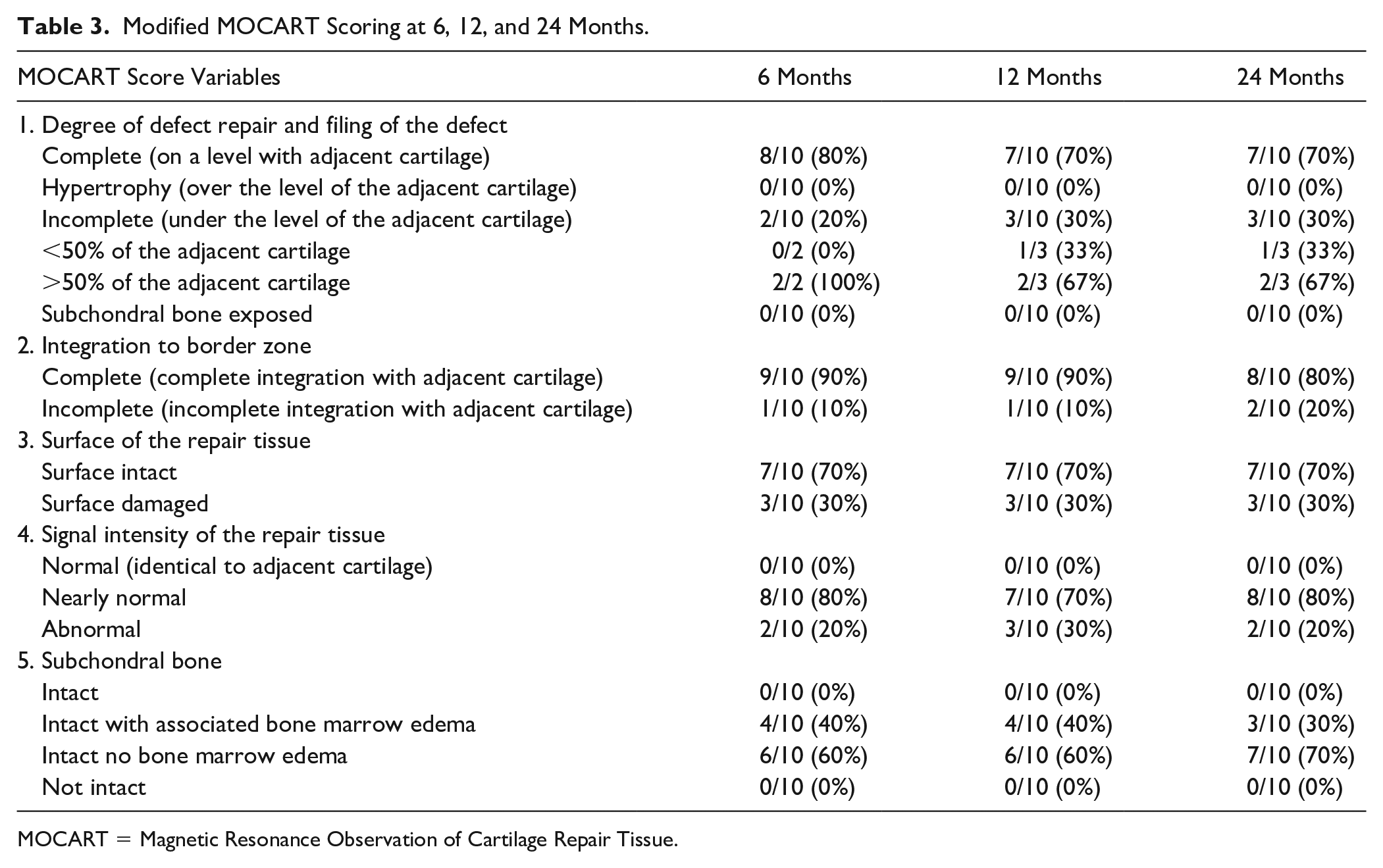
MOCART = Magnetic Resonance Observation of Cartilage Repair Tissue.
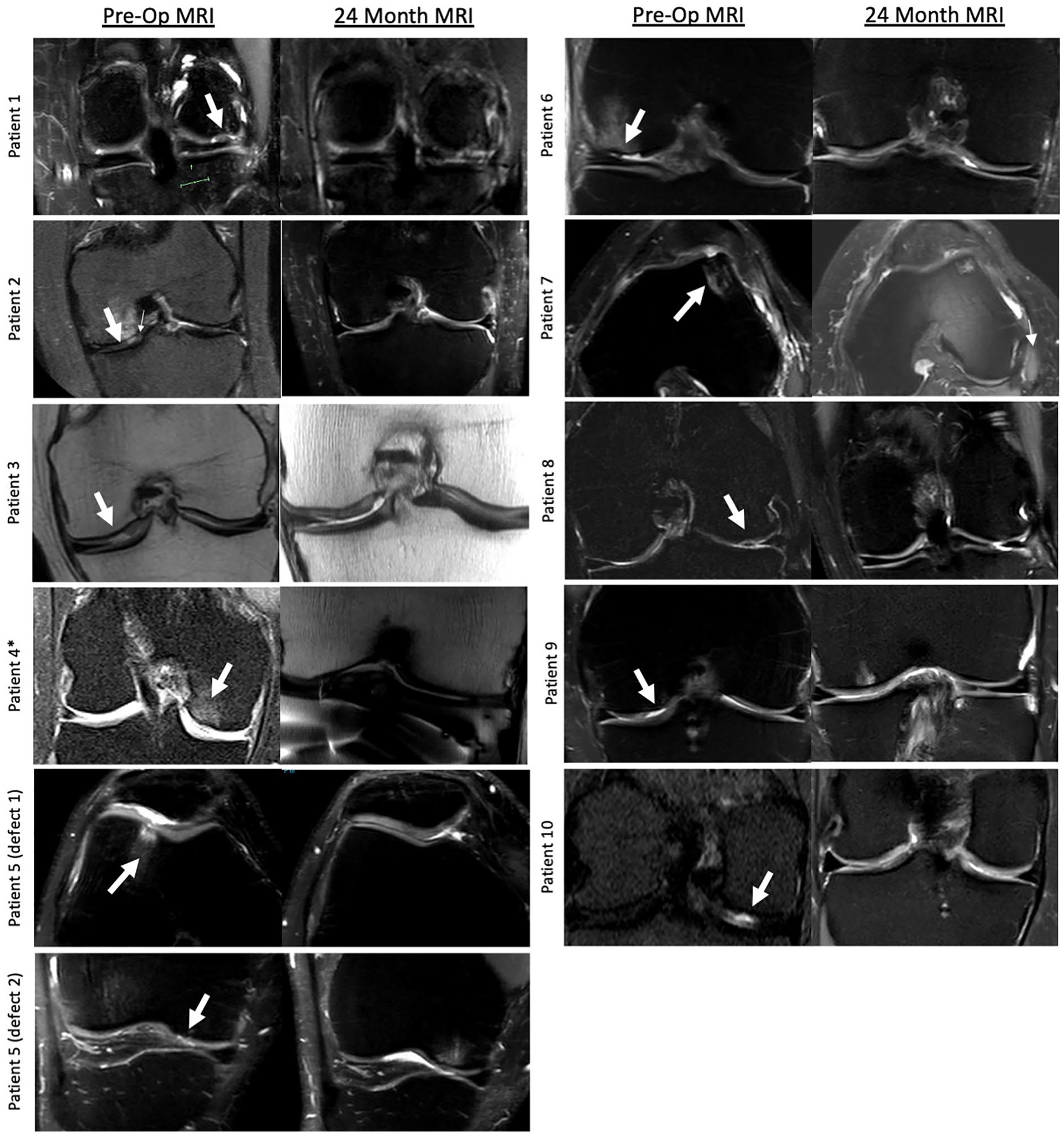
MRI images from preoperative and 24 months post-implantation with hypothermically stored amniotic membrane for each patient. White arrows indicate the presence of medial femoral condyle chondral defect or trochlear defect. MRI = magnetic resonance imaging.
Three subjects opted to participate in optional 24-month biopsies of the repair, which were completed using an arthroscopic procedure. Two to three biopsies were taken from the edge of the defect repair site, fixed, and stained for Alcian blue, Masson’s trichrome, H&E, aggrecan, lubricin, collagen I, and collagen II. Representative images of IHC staining (aggrecan, lubricin, collagen I, and collagen II) from all 3 subjects who elected to participate in this procedure are shown in
Figure 5
. Staining for aggrecan showed some aggrecan in the calcified zone for patient 1, while staining for lubricin was localized to the cartilage surface and the calcified zone/subchondral bone space in patient 1. Patient 2 had little to no staining for aggrecan and lubricin, while patient 7 had no aggrecan staining and slight lubricin staining in the calcified zone/subchondral bone space. Staining for collagen I and staining for collagen II were both robust at the border between the cartilage and subchondral bone, in addition to staining within the HSAM-repaired defect. H&E and collagen II biopsy images at 2x, 10x, and 20x from the 3 subjects are shown in
Figures 6
and
7
. H&E images showed the presence of chondrocytes and lacunae in the new matrix, along with the integration of the HSAM with the native cartilage, as observed in
Figures 6B-D
,
F-H
, and
J-L
. IHC staining for collagen II showed localized staining at the border between the cartilage and subchondral bone, along with interspersed staining throughout the HSAM-repaired defect, as seen in

Representative immunohistochemistry images from biopsies from 3 patients. Negative controls for aggrecan, lubricin, collagen I, and collagen II, shown on the left, and staining for aggrecan, lubricin, collagen I, and collagen II, shown on the right for each patient. All images taken using a 2x objective.
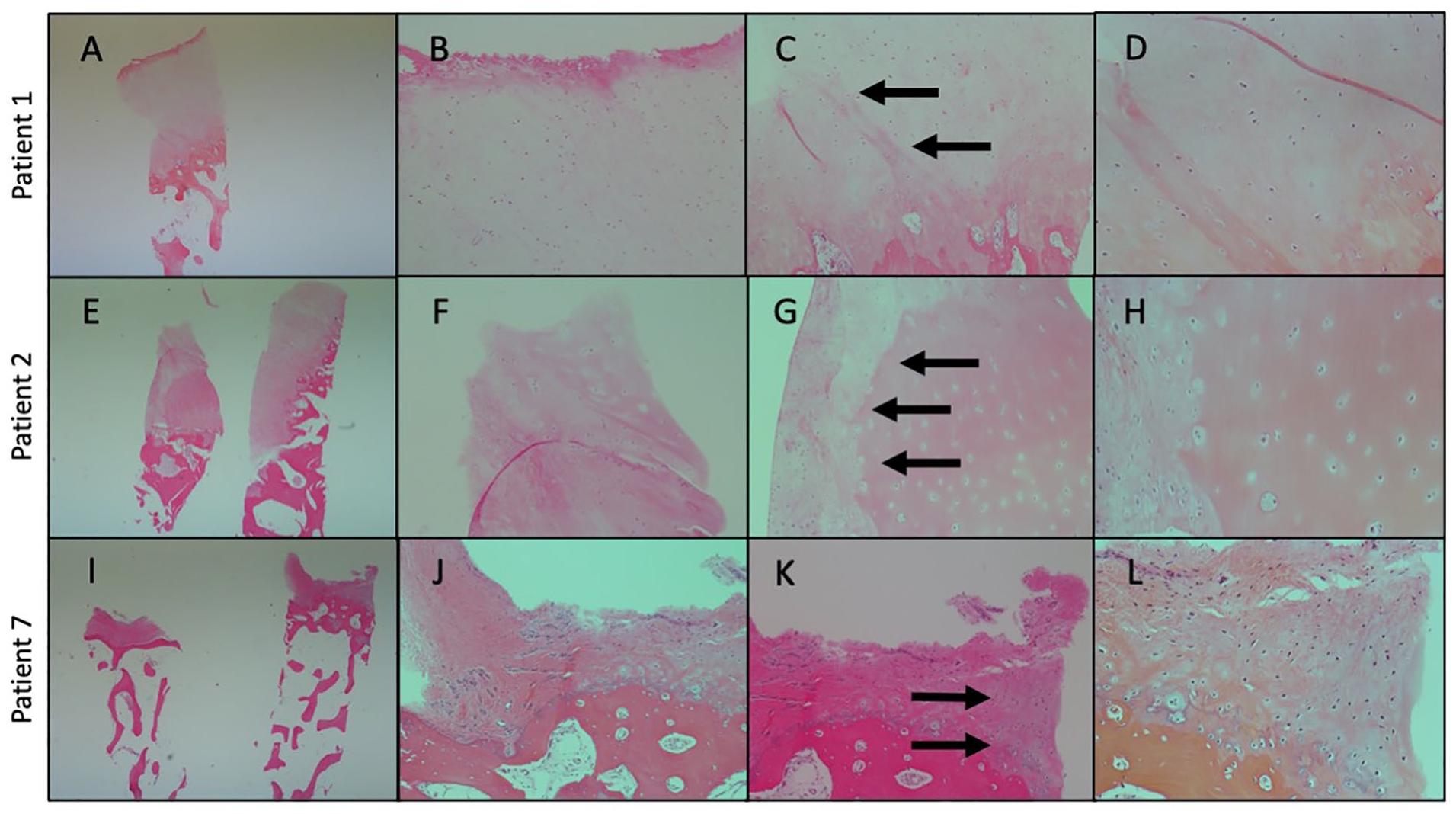
Representative histologic images from biopsies from 3 subjects with hematoxylin & eosin staining. (
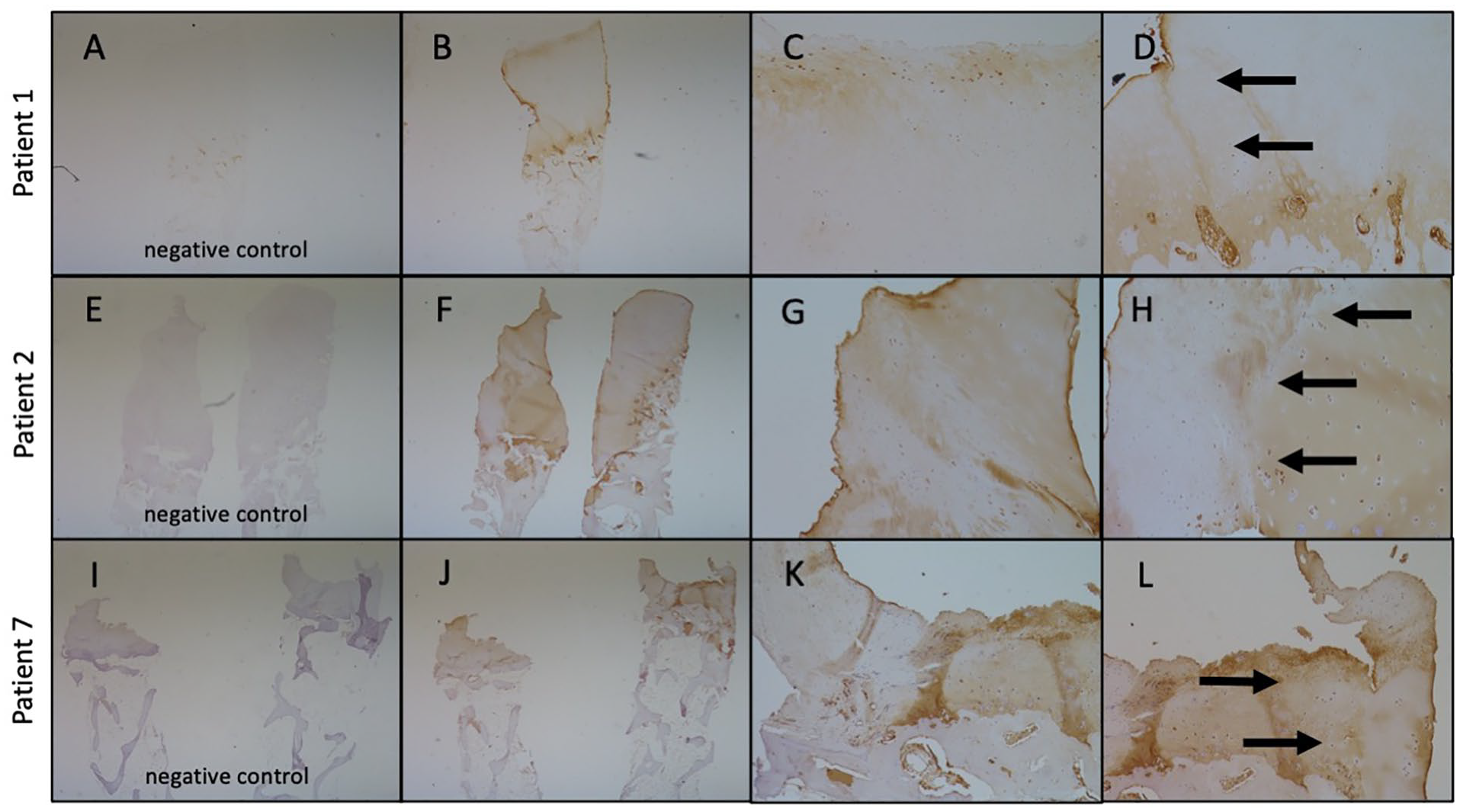
Representative histologic images from biopsies from 3 subjects with collagen II staining. (
Arthroscopic images preoperatively and at 24 months, along with chondral biopsy images, are shown for all 3 patients in Figure 8 . Images from the preoperative and 24-month scope, with the chondral defect identified with a black arrow, show the initial defects along with resolution and the presence of smooth cartilage 24 months following HSAM treatment. Alcian blue staining for glycosaminoglycans (blue dye) showed increased blue staining in the middle and deep zones of the cartilage biopsy. Masson’s trichrome staining for collagen/bone (blue dye), cytoplasm (pink), and nuclei (black) showed robust collagen accumulation in the biopsy, along with the presence of cells and cytoplasm down in the subchondral bone space. H&E staining for extracellular matrix/cytoplasm (pink dye) and nuclei (blue) showed the margin of repair cartilage and the nondamaged surrounding cartilage, highlighting the integration of the HSAM-treated repair with the native cartilage.
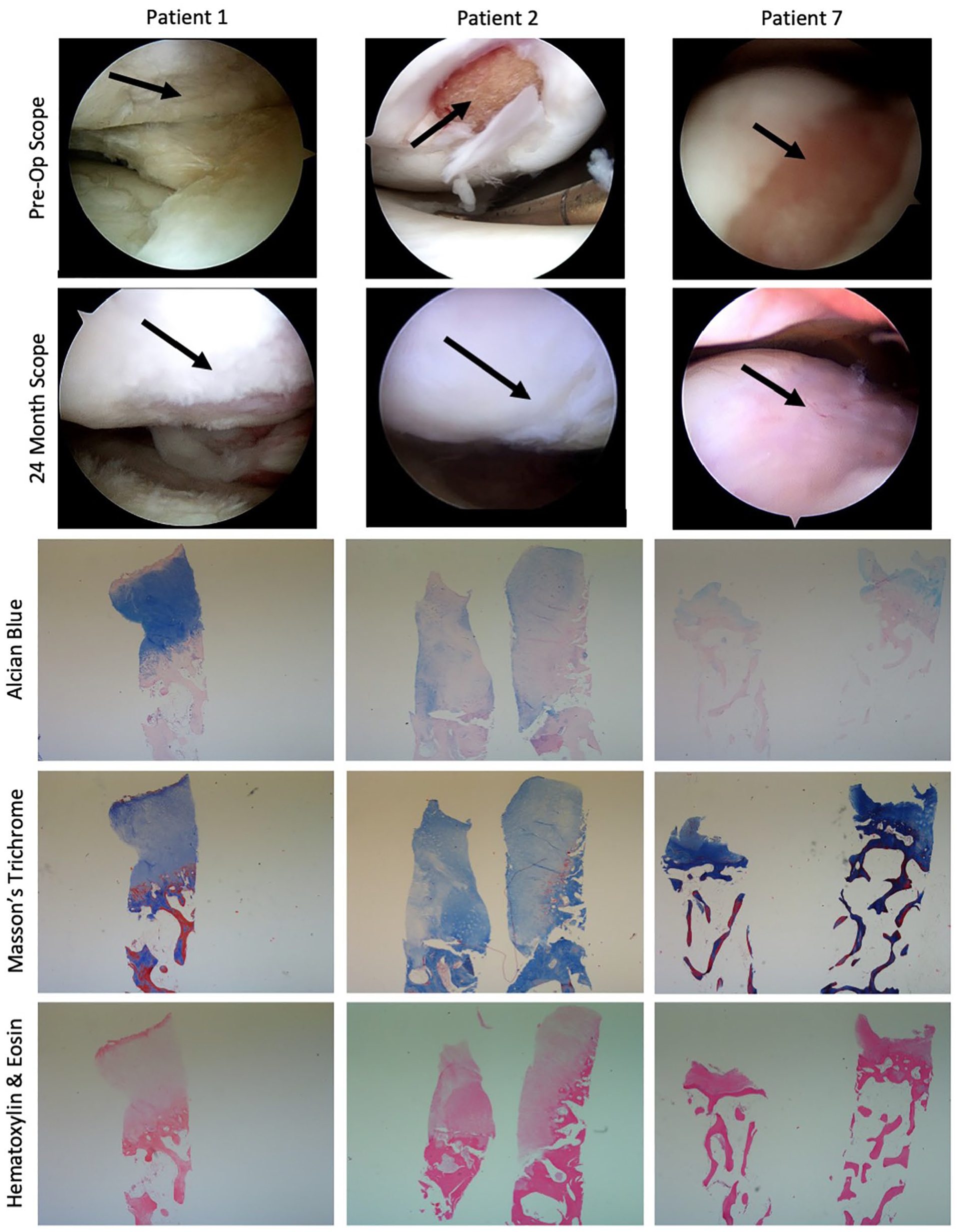
Three subject series including arthroscopic images preoperatively and at 24 months postoperatively, and histological images of the 24-month biopsy, including Alcian blue, Masson’s trichrome, and hematoxylin & eosin staining. All histological images taken using a 2x objective.
Of the 10 subjects enrolled, 3 subjects reported at least 1 treatment-emergent adverse event (TEAE), and a total of 9 TEAEs were reported throughout the duration of the study. Most TEAEs were not serious and either of mild to moderate severity (77.8%). None of the TEAEs were reported as being related to the study product, and no subjects were withdrawn from the study due to TEAEs. The specific events reported were as follows: knee pain (n = 3 events, 30% of subject population), knee stiffness (n = 1, 10%), rash (n = 1, 10%), chondromalacia (n = 1, 10%), effusion (n = 1, 10%), cellulitis (n = 1, 10%), and deep vein thrombosis (n = 1, 10%). These events are considered background findings for patients undergoing knee procedures; chondromalacia was ongoing as of the end of the study, the resolution of effusion and 1 knee pain event was unknown, and the rest of the events resolved prior to the end of the study.
Discussion
In this single-arm prospective study, HSAM was shown to be safe and effective for the treatment of symptomatic focal cartilage lesions. PROs were collected, and subjects showed trends for improvement from baseline to 2 years for all the KOOS subscales, the Marx Activity Scale, and the VAS. MOCART scoring of MRIs showed that 7 of 10 subjects had complete defect repair and filling by 24 months. Three subjects opted to participate in 24-month biopsies, which showed integration of the HSAM with native cartilage and staining of collagen II in the defect space. Three subjects reported at least 1 TEAE during the study; however, no TEAEs were related to HSAM and no subjects withdrew from the study as a result of an TEAE.
Two previous studies using amniotic membrane in an ovine cartilage defect model laid the groundwork for the current clinical trial. In the first study, fresh amniotic tissue and demineralized bone matrix were used to fill 2 cartilage defects in the femoral condyle and trochlear grove of the hindquarter knee and fixed to the knee. 16 Six months after implantation, sheep were sacrificed and evaluated alongside control animals with unfilled defects. In the sheep that received amniotic membrane and demineralized bone matrix, 50% of the membranes were retained in the defect, and robust integration of the amniotic membrane with the normal cartilage was seen. In addition, the unfilled defects were not filled with hyaline cartilage or fibrocartilage, while the amniotic membrane–filled defects showed cell proliferation of a hyaline cartilage–like matrix. In the second study, HSAM was used to fill a trochlear groove defect in sheep. 17 Five months following implantation, sheep were sacrificed and evaluated along control animals with either untreated defects or normal cartilage (no defect). As seen in the first animal study, the untreated defects did not fill with hyaline cartilage or fibrocartilage, while the defects treated with HSAM once again showed cell proliferation of a hyaline cartilage–like matrix. Defect fill in the HSAM group was near 100% compared with <10% in the control defect group, with HSAM-filled defects scoring 2.5/3 compared with the control defects (0.5/3) on a cartilage appearance scale. One limitation of these animal studies was the number of grafts that were dislodged; this may be due to the quadruped anatomy of the sheep and the challenges with postoperative immobilization and/or control of the activity of the sheep. However, there were no issues with HSAM retention in the defect in any subjects as demonstrated by MRI scans.
Based on the results of the current prospective study, subjects treated with HSAM showed a high degree of defect fill and integration with the native cartilage evidenced by modified MOCART scoring. This parallels the results shown by Tabet et al with HSAM in the sheep model. 17 In addition, biopsies of the defect at 24 months allowed for histological assessment of the regenerate tissue through standard histological techniques as well as IHC staining. Staining for collagen II in the biopsies revealed integration and remodeling of the HSAM into cartilage matrix, as collagen II is the major component in healthy cartilage. 18
One limitation of this study is the fact that it was a single-arm, unblinded study. However, subjects filled out PROs to decrease investigator bias. In addition, 1 subject did not follow postoperative instructions and began running at 6 weeks after implantation; this may have impacted some of the results as this patient’s data were included in the analysis despite the noncompliant behavior during the study.
This single-arm prospective study showed integration of regenerate cartilage tissue that is a mix of hyaline-like cartilage and fibrocartilage after application of a fresh HSAM used for the treatment of symptomatic cartilage lesions after a 2-year follow up period. There were improvements in pain and function scores, along with no serious adverse events related to the product. This study provides important original data for future randomized clinical trials using HSAM for the treatment of symptomatic cartilage lesions of the knee.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Organogenesis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.A.K., G.J.H., and K.C.M. are employees of Organogenesis.
Ethical Approval
Western Institutional Review Board (WIRB) approval was obtained (WIRB protocol #1162030), and informed consent was obtained from each patient before any study-related activities were performed.
Trial Registry
NCT02837484
