Abstract
Objective
To create a treatment algorithm for focal grade 3 or 4 cartilage defects of the knee using both classic and novel cartilage restoration techniques.
Design
A comprehensive review of the literature was performed highlighting classic as well as novel cartilage restoration techniques supported by clinical and/or basic science research and currently being employed by orthopedic surgeons.
Results
There is a high level of evidence to support the treatment of small to medium size lesions (<2-4 cm2) without subchondral bone involvement with traditional techniques such as marrow stimulation, osteochondral autograft transplant (OAT), or osteochondral allograft transplant (OCA). Newer techniques such as autologous matrix-induced chondrogenesis and bone marrow aspirate concentrate implantation have also been shown to be effective in select studies. If subchondral bone loss is present OAT or OCA should be performed. For large lesions (>4 cm2), OCA or matrix autologous chondrocyte implantation (MACI) may be performed. OCA is preferred over MACI in the setting of subchondral bone involvement while cell-based modalities such as MACI or particulated juvenile allograft cartilage are preferred in the patellofemoral joint.
Conclusions
Numerous techniques exist for the orthopedic surgeon treating focal cartilage defects of the knee. Treatment strategies should be based on lesion size, lesion location, subchondral bone involvement, and the level of evidence supporting each technique in the literature.
Introduction
Cartilage surgery is indicated in patients with symptomatic defects, with the short-term goal of improvements in pain and function, and the long-term hope of delaying the need for arthroplasty. 1 While focal cartilage defects are common and may be present in as much as 63% of the general population and 36% of athletes, the majority are not symptomatic.2,3 Larger defects, however, can become problematic, especially in young patients who hope to maintain a physically active lifestyle without debilitating pain. Cartilage defects may also lead to accelerated wear, worsening pain, and potential arthritis progression. For example, competitive athletes (high school or collegiate) may be at increased risk of high-grade multicompartment or anterior compartment defects in comparison to recreational athletes. 4 The high prevalence and high cost of chondral disease, which ranges from small focal defects to end-stage osteoarthritis, has significant financial implications for the health care system. The responsible application of resources for the treatment of cartilage disease therefore should take into account lifetime costs, opposed to limiting ones focus on per procedure cost.
Classic cartilage repair and restoration techniques including marrow-stimulation techniques (MST) such as microfracture (MFx) and subchondral drilling, osteochondral allograft transplantation (OCA), osteochondral autograft transfer (OAT), and autologous chondrocyte implantation (ACI) have been have been performed for decades;5,6 however, in recent years, numerous novel strategies and products for the treatment of cartilage defects have been brought on to the market. The purpose of this article is to provide an overview of both classic and new cartilage restoration procedures including their mechanisms of action, surgical techniques, overview of results, advantages and disadvantages, costs, and commercial availabilities. Finally, based on the current evidence presented and the authors’ experience, we propose an up-to-date comprehensive algorithm that integrates new technologies to help guide treatment choices when dealing with focal cartilage defects of the knee.
Overview of the Techniques
The classic procedures described herein include the following: MST, matrix-induced autologous chondrocyte implantation (MACI), OAT, and OCA. Please see the appendix for a detailed description of each classic cartilage procedure with supporting evidence. The novel procedures we will describe below include micronized cartilage extracellular matrix (Biocartilage), Autologous Matrix-Induced Chondrogenesis (AMIC), bone marrow aspirate concentrate (BMAC) implantation, particulated juvenile allograft cartilage (PJAC, DeNovo), particulated autologous cartilage implantation (PACI), viable cartilage allograft putty (CartiMax), cryopreserved viable osteochondral allograft (CVOCA; Cartiform and Prochondrix), aragonite biphasic osteochondral scaffolds (Agili-C), and human umbilical cord blood-derived mesenchymal stem cells (CARTISEM).
Micronized Cartilage Extracellular Matrix (Biocartilage)
Micronized cartilage extracellular matrix, known commercially as Biocartilage, is frequently utilized as an augment to MST. Biocartilage is a desiccated acellular particulated cartilage allograft used as a bioactive scaffold to treat focal cartilage defects. It contains type II collagen, proteoglycans, and chondral growth factors. The material is generally mixed with platelet-rich plasma (PRP) or bone marrow aspirate concentrate (BMAC), applied to the prepared contained cartilage defect and sealed with fibrin glue. It has been postulated that the extracellular matrix elements in the Biocartilage promote the mesenchymal progenitor cells released from the MST and/or present in BMAC to differentiate along a chondral lineage resulting in new hyaline cartilage. 7 Furthermore, a recent in vitro study by Commins et al. showed that Biocartilage supported mesenchymal progenitor cell and chondrocyte adhesion and contained 254 proteins, many of which were anabolic toward cartilage. 8
The procedure can be performed either open or arthroscopically. An initial diagnostic arthroscopy is often performed followed by defect preparation and MST according to the standard protocol previously described. 9 The defect must be thoroughly dried. The implant is then mixed with an autologous blood solution in a 1:0.8 ratio, and the slurry is then introduced into the defect either manually if open or by an arthroscopic device that is commercially available with the device kit. The mixture is spread over the entirety of the defect and a fibrin sealant is used to seal the entire defect, especially at the edges. It takes approximately 5 minutes for the mixture to cure, after which the knee is cycled through a range of motion to ensure stability.
To our knowledge, there is no long-term clinical outcome data available regarding the results of Biocartilage use to date, but animal studies have yielded promising results.10,11 Furthermore, small case series in human subjects have reported good to excellent results at 12 months postoperatively for osteochondral defects of the talus and distal tibia.12,13
Autologous Matrix Induced Chondrogenesis
AMIC utilizes MST in combination with a type I/III porcine collagen- or hyaluronan-based membrane (Fig. 1). The patch lies over the MST defect and acts as a scaffold to retain the bone marrow elements and allow them to mature, facilitating their differentiation into chondrogenic cells.14-17
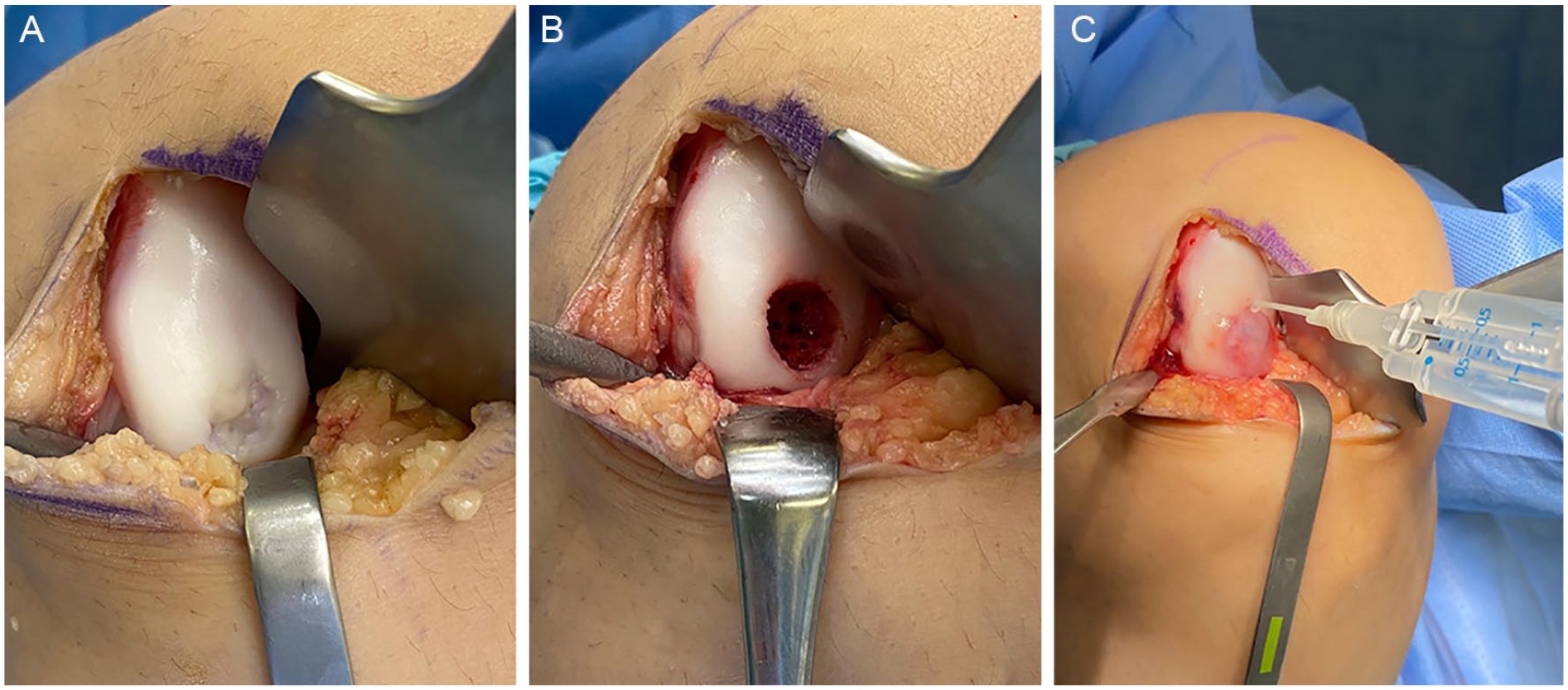
Autologous matrix-induced chondrogenesis (AMIC). (
To perform AMIC, a diagnostic arthroscopy can be performed, but is not required. The procedure is typically performed via a small arthrotomy and MST is performed in standard fashion (Fig. 1). The defect is sized and a template is created from foil or glove paper. The cut-out template is then transferred to the patch, which is sized accordingly. The patch should be slightly undersized so the edges are not proud, which could risk dislodging the patch with knee range of motion. The patch is placed over the defect with the rough, porous side facing the bone, and the patch is fixed with either absorbable sutures or fibrin glue (Fig. 1). After the patch is secured, the knee should be ranged to ensure stability of the patch under direct visualization.
In a recent systematic review, Gao et al. concluded that patients who underwent AMIC for isolated grade IV cartilage defects of the knee (average size 3.6 cm2) reported decreased pain and improved functional outcome scores up to 5 years postoperatively. 18 In a randomized controlled trial (RCT) comparing patients treated with AMIC, to AMIC augmented with BMAC, both groups demonstrated significant improvement in pain and functional outcome scores up to 9 years postoperatively. 19 Zhang et al. reviewed “one-step” cartilage repair techniques and noted improved symptom relief and improved functional outcomes at short-term follow-up and regeneration of hyaline-like cartilage tissue. 20 Though results of AMIC have been promising, current evidence is insufficient to recommend a range of joint-specific defect sizes that may be treated with AMIC based on the limited data available from clinical trials to date.
Bone Marrow Aspirate Concentrate Implantation
The BMAC method entails the harvesting and concentration of mesenchymal progenitor cells, growth factors, and cytokines from autologous bone marrow collected from the patient which is then injected into a focal chondral defect forming a bioactive clot. Clot stabilization can be augmented via combination with fibrin glue or by placement of a membrane which can be either synthetic, porcine I/III collagen, or hyaluronan based. 21 The membrane acts as a scaffold containing the BMAC and providing an anchor within the defect on which the medicinal signaling cells (MSCs) can attach, proliferate, and differentiate. Alternatively, when performed in conjunction with AMIC the collagen scaffold may be soaked in BMAC for 10 to 15 minutes prior to implantation. 22
A diagnostic arthroscopy can be performed but is not required. The BMAC implantation procedure is performed via an open or arthroscopic approach. Bone marrow aspirate can be harvested from a variety of sites including the iliac crest (anterior or posterior), the distal femur (supracondylar region or arthroscopically through the intercondylar notch), the proximal tibia, or the calcaneus. Once obtained, the aspirate is prepared (concentrated) according to each system’s protocol. Starting volumes may vary between systems, but typically approximately 60 mL is desired, which is concentrated to 5 to 10 mL. An arthrotomy or a mini-arthrotomy is performed as required by the defect, and the defect is prepared in the standard manner for cartilage restoration procedures. If an osteochondral defect is present, autogenous bone grafting may be performed prior to BMAC implantation. If performed arthroscopically, it is important that all fluid be removed from the knee prior to BMAC implantation. Once the defect site is dry, the activated clot is transferred into the defect and secured with fibrin glue. A membrane may be placed over the defect (rough/porous side facing the bone) and secured with absorbable suture or fibrin glue. After curing of the glue, the knee should be taken through a range of motion to ensure stability of the BMAC clot and/or patch.
The use of BMAC for the treatment of focal articular cartilage defects has yielded inconsistent clinical results in the literature, some of which may be due to the different preparations that yield different BMAC products. Dragoo and Guzman compared 3 separate commercially available BMAC preparation systems (Harvest, Biomet, Arthrex) and showed that each produced BMAC with similar overall cellular consistency (white blood cell count, platelet count, CD34+ cell count, etc.) but in varying concentrations. 23
In their systematic review of BMAC for the treatment of cartilage defects, which included both animal and clinical human studies, Cavinatto et al. concluded that BMAC treatment leads to improved short-term and midterm outcomes. 24 BMAC has demonstrated improved outcomes when compared to MFx and equivalent outcomes to MACI. 24 However, the quality and quantity of data on the use of BMAC for chondral defects is limited.
Particulated Juvenile Allograft Cartilage
PJAC, known commercially as DeNovo NT, utilizes juvenile hyaline cartilage obtained from young donors (Fig. 2). Immature chondrocytes have been shown to have increased proliferative activity, cell density, and metabolic activity when compared to adult chondrocytes.25-29 Once packaged, the cells are viable for up to 45 days.
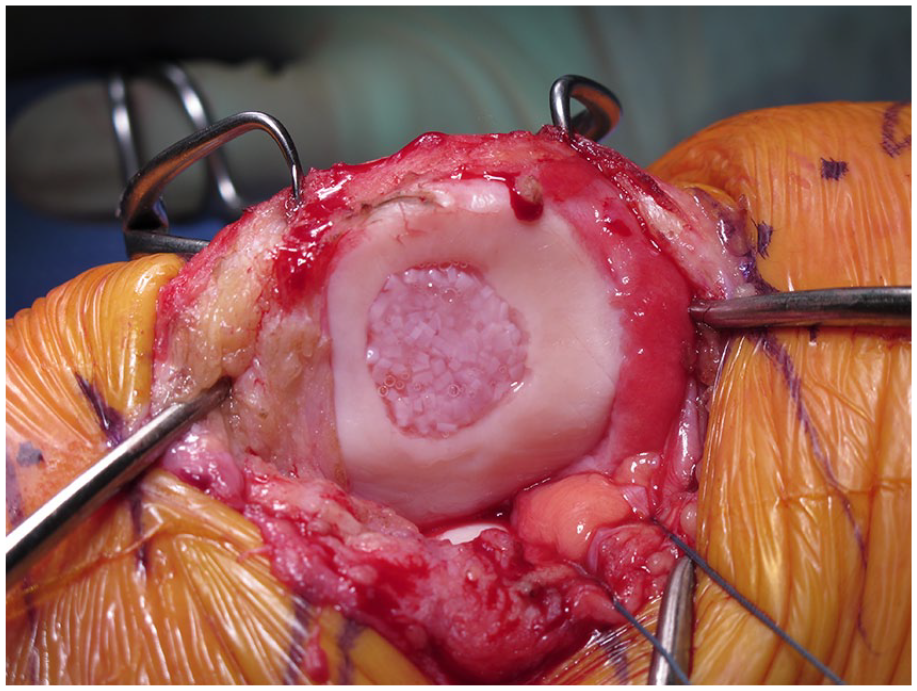
Particulated juvenile allograft cartilage (DeNovo NT) applied directly to a patellar lesion. The pieces are arranged in one layer and close together (touching or almost touching). Fibrin glue is added. The whole implant is recessed below the margins of the defect (1 mm).
Some surgeons may prefer to perform PJAC implantation as a staged procedure with an initial diagnostic arthroscopy to thoroughly assess the joint, whereas others may elect to have the graft available at the time of the initial surgery. The decision to proceed with open versus arthroscopic implantation is up to the surgeon’s discretion depending on defect location and size. The defect is prepared in the standard manner, sized, and the appropriate number of allograft packets is determined. After the defect area has been calculated, the number of packets can be estimated, as one packet of PJAC covers approximately 2.0 to 2.5 cm2. If significant bone loss is present, bone grafting should be performed so that the subchondral bone bed is within 5 mm of the level of the adjacent healthy subchondral bone. The allograft can be prepared for implantation either directly inside the defect or on the back table through the use of a mold. The pieces of PJAC should be arranged close together in a single layer (Fig. 2). The fluid medium should be removed from the allograft taking care to not discard any pieces of juvenile cartilage. Next, fibrin glue is added to the base of the defect and the PJAC is added to the defect. The allograft should fill at least 50% of the total surface area of the defect and should be recessed below the shoulders of the defect by approximately 1 mm. Thumb pressure is used to hold the implant firmly against the glue to ensure good adherence. A Freer elevator may be used to help spread the allograft evenly over the defect as it continues to harden. Five to 10 minutes should be allowed for the fibrin glue to cure. When prepared on the back table, fibrin glue is added to the cartilage fragments and allowed to cure. The implant is then glued into the defect with fibrin glue placed at the base. Implantation can also be performed arthroscopically using the same concepts as previously described in the open method. An arthroscopic introducer can be used to deliver the allograft tissue to the defect. If performed arthroscopically, all fluid should be removed from the joint so that implantation may be performed in a dry environment.
No long-term or randomized clinical data are currently available following PJAC use in the human knee. However, short-term results in the patella, trochlea, and femoral condyle have demonstrated significantly improved pain and function, good fill of the defect with normal appearing articular cartilage on MRI (magnetic resonance imaging), and a combination of hyaline and fibrocartilage on histological analysis.30-32
Particulated Autologous Cartilage Implantation
Use of PACI is similar to PJAC in that it uses particulated cartilage; however, in this technique, the tissue used is obtained autologously from the patient during the same surgical procedure. The first report of this technique was in the German literature in 1983. 33 Later in the United States, DePuy-Mitek created a proprietary cartilage autograft implantation system (CAIS) to automate the mincing process. An initial study approved by the Food and Drug Administration (FDA) comparing CAIS to MFx showed superior results in the CAIS group. 34 However, further studies were discontinued due to slow patient enrollment, and the device was eventually removed from development. 31 However, the technique can still be utilized without the use of a proprietary system.
A first stage diagnostic arthroscopy can be performed but is not required. Using curettes and/or gouges, 200 to 300 mg of cartilage without bone is harvested from the surgeon’s location of choice—typically the intercondylar notch, medial femoral trochlear ridge, or lateral femoral trochlear ridge. 35 The harvested cartilage is fragmented on the back table with a fresh blade, creating fragments no larger than 1 mm in each dimension. This is made into a paste-like consistency. An arthrotomy is performed to expose the defect, which is then prepared in the standard fashion. The defect is templated, and a commercially available patch (either synthetic, porcine I/III collagen, or hyaluronan based) is cut according to the defect. A thin layer of fibrin glue is placed into the defect after hemostasis has been achieved and the cartilage paste is applied evenly across the defect. A second layer of fibrin glue is applied to secure the cartilage, and then this construct is secured with the patch to the top of the defect using small absorbable sutures and fibrin glue around the periphery. The knee is cycled to ensure stability, and then the arthrotomy is closed in standard fashion.
As with PJAC, no long-term clinical data are currently available following PACI in the human knee. However, as mentioned above, an initial pilot study of PACI (in the form of CAIS) demonstrated favorable outcomes compared to MFx, including better patient-reported outcome scores with decreased formation of intralesional osteophytes at 2 years. 34 While the commercial form of PACI (CAIS) is no longer available, this technique can still be utilized without the use of a proprietary system. In fact, Massen et al. in a study of 27 patients who underwent PACI using a freehand scalpel mincing technique for small to medium sized (1-6 cm2) chondral or osteochondral lesions of the femoral condyles, trochlea, or patella showed significant decreases in pain and increases in physical function compared to baseline at a mean follow-up of 28.3 ± 3.8 months. 36 Furthermore, 18 out of the 27 patients had cartilage lesions >2 cm2 with the same number having lesions of the patella meaning that PACI may be a good alternative to MACI/ACI in patients with large patellar lesions who are restricted by cost or health insurance coverage. 36
Viable Cartilage Allograft Putty (CartiMax)
Viable cartilage allograft putty, available commercially as CartiMax, is a new technology that has been created from a joint effort between MTF Biologics and CONMED. This technology utilizes viable cartilage flakes from allograft donor tissue and combines these with lyophilized extracellular matrix, resulting in a putty that can be molded to fit defects of any shape. Per the manufacturers, CartiMax can be utilized for full-thickness cartilage defects up to 5 cm2 in size and has a shelf-life of up to 12 months after cryopreservation.
CartiMax can be utilized in a single-stage procedure. Typically, an arthrotomy is made to expose the articular cartilage defect via an open approach. The defect is prepared in the standard fashion. The solution containing the chondrocytes is thawed, the liquid is drained, and the fibers are washed with normal saline. The cartilage allograft matrix (CAM) is then mixed with the cartilage fibers containing the live chondrocytes to form a putty. The defect is dried, and the putty is added, which can be molded to fit the appropriate defect shape and depth, and typically no sealant or fibrin glue is required.
CartiMax is a new technology, and no clinical outcomes regarding its efficacy have been published to date.
Cryopreserved Viable Osteochondral Allograft (Cartiform and Prochondrix)
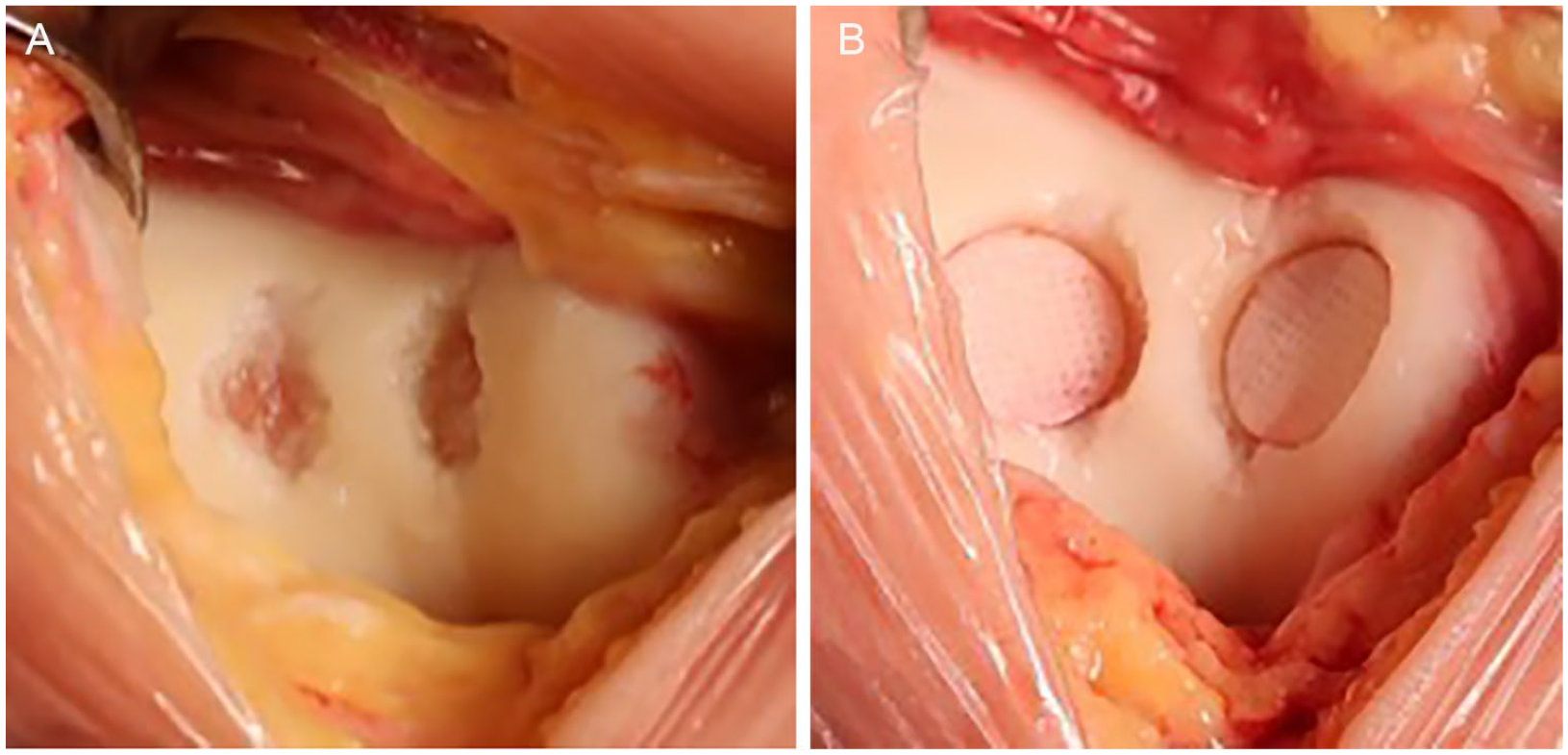
A CVOCA is composed of a thin disc that contains hyaline cartilage with its native architecture on top of a microscopic layer of bone. Chondrogenic growth factors, extracellular matrix, and viable chondrocytes are contained in the scaffold. The scaffold is perforated, making it both pliable for contouring along curved surfaces, as well as increasing the surface area for outgrowth of chondrocytes from the matrix (Fig. 3). It is preserved at −80 °C. 37 CVOCA can be used in conjunction with MST or BMAC. 38 There are currently 2 commercially available options, Cartiform and Prochondrix.

Cryopreserved viable osteochondral allograft (CVOCA; Cartiform). (
As with other cartilage restoration procedures, a diagnostic arthroscopy can be performed, but is not required. The procedure is performed via an open approach. The defect is prepared in the standard manner. Prochondrix has a special kit for preparation. The size of the defect is determined, and the appropriately sized CVOCA packet(s) is (are) selected and allowed to thaw. Cartiform is available in 10 mm and 20 mm diameter discs and 12 × 19 mm and 20 × 25 mm wafers. Prochondrix is available in discs of 2 mm increments from 9 to 17 mm and 20 mm. The allograft is thawed in saline. A template is created and the CVOCA is cut to size. The bone layer side should be placed toward the bony bed of the defect. MST can be performed if desired. The whole implant needs to be directly opposed to the bony surface; thus, defects on a concave surface, such as the trochlea, need a centrally placed anchor to draw the implant into contact with the bone. Conversely, on convex surfaces, the periphery should be fixed with sutures in order to anchor the implant into the surrounding cartilage (Fig. 3). BMAC can be applied if desired. Once adequate fixation is achieved, a thin layer of fibrin glue is applied to the periphery. This should be allowed to cure for a minimum of 5 minutes, and then the knee is taken through a range of motion to ensure stability of the implant.
Outcomes of Cartiform and Prochondrix are limited, and the current literature consists primarily of laboratory data and small case series. Small case series for both have demonstrated good outcomes at 2 years without adverse outcomes or early failures.38,39 Further clinical studies are required to better understand the long-term outcomes of CVOCA.
Aragonite Biphasic Osteochondral Scaffolds (Agili-C)
Agili-C is a crystalline osteoconductive aragonite biphasic scaffold, produced from a coralline exoskeleton, that is currently under clinical trials in the United States for the treatment of focal osteochondral lesions of the knee (femoral condyles/trochlea) (relation to the cartilaginous surface (Fig. 4). It is available in sizes from 6 to 20 mm in diameter, and 8 to 15 mm in length. Preparation is similar to that seen in OCA transplantation. A coring device corresponding to the diameter of the lesion is placed perpendicular to the articular surface and drilled to the desired depth. The tapered Agili-C implant is then inserted into the recipient site in a press-fit manner making sure that the top of the implant is recessed in relation to the cartilaginous surface (Fig. 4). The implant may be soaked in BMAC prior to implantation as an augment.
Aragonite biphasic osteochondral scaffolds (Agili-C). (
In vitro studies using human derived mesenchymal progenitor cells seeded onto the Agili-C scaffold showed increased osteogenic differentiation and proliferation of mesenchymal progenitor cells at the histologic and molecular levels. 40 Furthermore, an ex vivo model comparing donut-shaped cartilage explants of cadaver human articular defects with and without Agili-C implants cultured for 60 days showed that the Agili-C implant contributed to chondrocyte migration and deposition of an extracellular matrix rich in type II collagen and aggrecan. 41 An early clinical study by Kon et al. comparing tapered versus cylindrical Agili-C implants showed improved functional outcome scores (IKDC [International Knee Documentation Committee], Lysholm, and KOOS [Knee Injury and Osteoarthritis Outcome Score]) and MRI findings of bony integration and cartilage regeneration at 12 months postoperative. 42 However, this same study showed improved longevity with 0 revisions at 12-month follow-up in tapered implants compared to 10.5% in cylindrical implants. 42 The Agili-C implant is currently unavailable for use in the United States, Europe, and Asia pending the results of a recently completed clinical trial in order to support its utility and safety in clinical practice.
Human Umbilical Cord Blood–Derived Mesenchymal Stem Cells (CARTISTEM)
CARTISTEM is allogeneic umbilical cord blood–derived mesenchymal stems cells (hUCB-MSCs) used to treat degenerative or traumatic grade IV cartilage lesions of the knee. The hUCB-MSCs come in a 1.5-mL liquid suspension. This vial is gently tilted in order to evenly distribute the stem cells within the preservative fluid. All 1.5 mL are then injected into a 1.5-mL sodium hyaluronate (HA) suspension. The combined solution is then left to cure for 30 minutes at room temperature. The mixed hUCB-MSCs-HA solution is then transferred to a sterile 5-mL syringe for administration. Multiple 5-mm drill holes, 5 mm in depth and 2 to 3 mm apart, are then created in the area of the cartilage defect. 43 The hUCB-MSC-HA solution is then transplanted into each of the newly created drill holes. Recommended dosage for hUCB-MSCs is 500 µL/cm2 with a cell concentration of 0.5 × 107 cells per milliliter based on an early animal study. 44
There is currently one published study out of Korea demonstrating safety and efficacy of CARTISTEM for the treatment of Kellgren-Lawrence grade 3 full-thickness cartilage lesions of the knee greater than 2 cm2 without subchondral bone involvement. 43 In this study, 7 patients were divided into 2 groups, A (4 patients) and B (3 patients), based upon the dose of hUCB-MSCs used (high or low) in the combined HA solution. Mild adverse reactions including back pain, arthralgias, and elevated antithyroglobulin levels were noted in 5 patients but all resolved. Three-month repeat arthroscopy showed evidence of maturing repair tissue at the hUCB-MSCs-HA application site. Significant improvement in functional outcome scores including the IKDC and VAS (Visual Analogue Scale) were noted at 24 weeks postoperative and were maintained at 7-year follow-up. CARTISEM appears to be a safe and efficacy treatment for full-thickness cartilage lesions of the knee greater than 2 cm2 and is currently approved for use in Korea.
Treatment Algorithm
Prior to any cartilage restoration procedure, it is imperative to define that the lesion is responsible for the patient’s symptoms. In addition, a course of nonoperative treatment should be considered depending on the pathology. For example, chronic degenerative defects in older patients should fail conservative management prior to cartilage repair, while acute large defects in young patients can be considered for repair initially. Conservative care includes weight loss, ice, anti-inflammatories, bracing, physical therapy, and intraarticular injections (corticosteroids, hyaluronic acid, and biologics). Surgery is typically indicated in patients with persistent pain, swelling, or functional limitation in the setting of a focal chondral or osteochondral lesion. Prior to proceeding with surgical intervention, it is important that realistic expectations be set with the patient with regard to surgery, length of recovery, and ultimate functional outcome. Furthermore, the physician should assess the ability of the patient to comply with a lengthy rehabilitation prior to performing surgery.
Concomitant pathology (malalignment, meniscus deficiency, and instability) should be identified and properly addressed prior to or at the same time as the cartilage restoration procedure. Once all other factors have been optimized (or a plan is set for the optimization of these factors), the next step is identifying which cartilage restoration strategy to pursue.
When determining what type of cartilage restoration technique to perform, the most important variables are the condition of the underlying subchondral bone, the size of the defect, and the location of the defect. The subchondral bone is considered deficient when there is bone loss (e.g., traumatic osteochondral defects or osteochondritis dissecans [OCD]), a significant bone lesion (e.g., extensive edema or cystic formation), or if there is previous surgical violation of the subchondral plate (e.g., MST or osteochondral transplantations).
Goals of surgical intervention, including which sports and/or activities the patient wishes to return to and at what level (recreational vs. professional), should also be considered. Unfortunately, the realities of cost and health insurance coverage should also be addressed as this may influence which cartilage restoration procedures are available to the provider and patient. A proposed treatment algorithm is presented in Fig. 5.
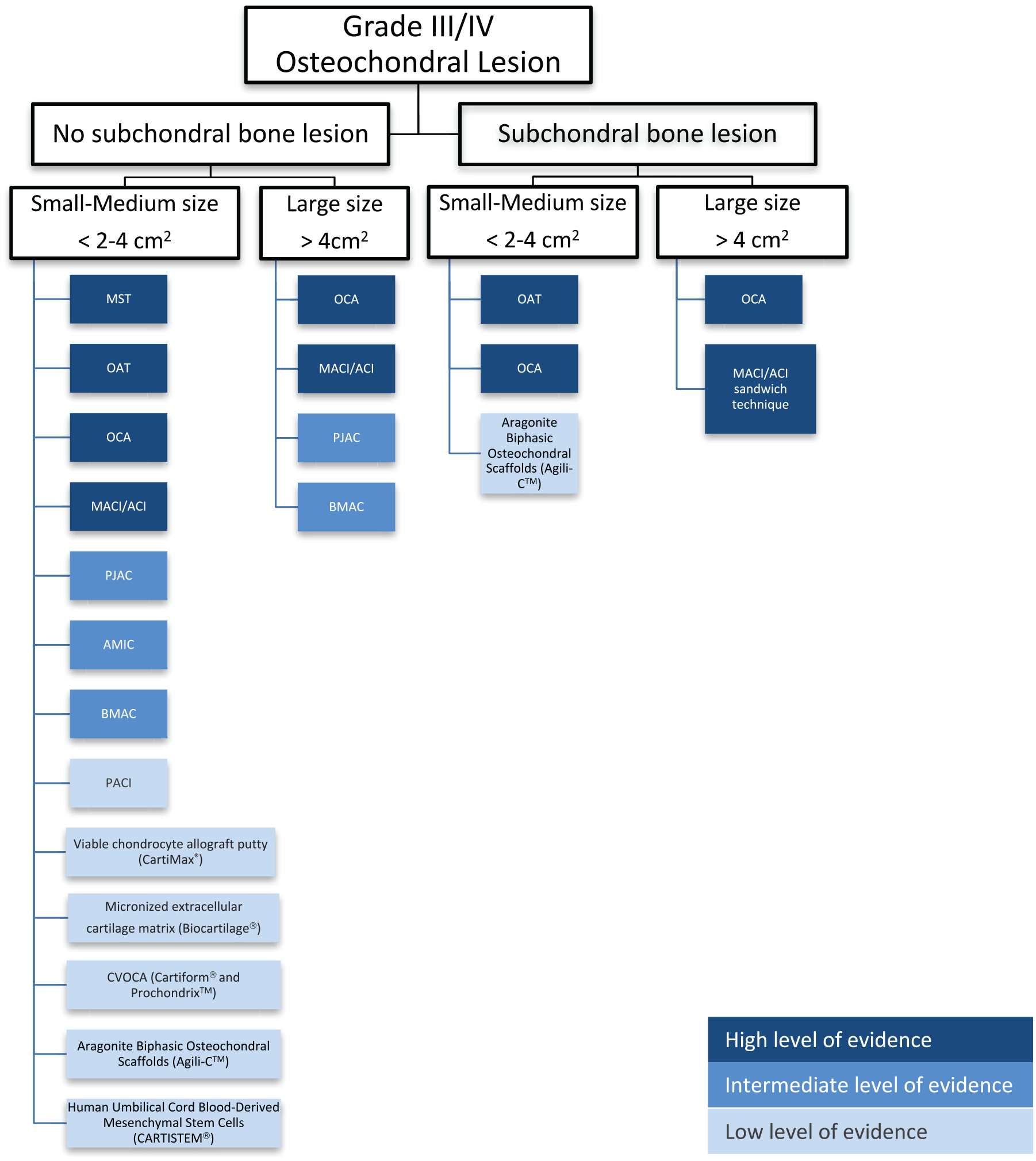
Cartilage restoration algorithm: traditional and new technologies.
Small-Sized (<2 cm2) and Medium-Sized Defects (2-4 cm2) with Bone Loss/Involvement
OAT and OCA are the osteochondral procedures with the most evidence to support their use in the treatment of small (<2 cm2) and medium (2-4 cm2) sized chondral defects with bony involvement.45-47 Osteochondral procedures such as OCA and OAT replace subchondral bone and are therefore preferred in patients with subchondral bone lesions, particularly when the lesions are located in the femoral condyles. The decision to use OAT versus OCA in the setting of subchondral bone loss/damage is determined by defect size and location. OAT is better suited for small defects (<2 cm2), where 1 or 2 donor plugs are sufficient to cover the defect because using more than 2 donor plugs decreases clinical outcomes and increases donor site morbidity.48,49 OCA can be used in both small and large defects, and is therefore mainly limited by insurance payer guidelines that generally require a minimum size of 2 cm2. However, pre-cut OCA plugs (available in 10 mm, 15 mm, 20 mm diameters) are an option as they are cheaper, more readily available, and result in less wasted tissue than plugs harvested from size- and side-matched hemicondyle allografts. There are currently no studies comparing functional outcomes between patients treated with pre-cut OCA plugs versus those plugs harvested at the time of surgery from size- and side-matched hemicondyles.
In the patellofemoral joint, OCA may be preferable over OAT because OCA enables utilization of a patellar graft from a similar location with a better matching of contour and cartilage thickness. This also avoids the creation of additional donor site morbidity in the treated compartment as the trochlea notch is the primary site for graft harvest of autograft plugs. 50
If OCA or OAT are not possible, ACI/MACI or PJAC can be considered for osteochondral lesions. Osteochondral lesions deeper than 7 to 8 mm should be treated with concomitant bone grafting (sandwich procedure), while shallow lesions such as small OCD lesions can be treated with cartilage resurfacing only. 51
Small-Sized (<2 cm2) and Medium-Sized Defects (2-4 cm2) without Bone Loss/Involvement
In the case of small (<2 cm2) and medium defects (2-4 cm2) without bone involvement, options for treatment include the bone marrow–based techniques (MST, AMIC, and BMAC implantation), ACI/MACI, PJAC, OAT, and OCA (both pre-cut plugs and those harvested from a whole hemicondyle, patella, or trochlea).
Classic MST is the least expensive and a cost-effective treatment in small lesions, <2 cm2.52-55 It has been shown to provide clinical improvements in the short term, but frequently, results deteriorate after 2 years.56-59 Furthermore, studies comparing OAT to MST show that OAT results in better long-term outcomes and increased patient activity levels compared to MST. 45 Evidence from RCTs comparing ACI/MACI and MST are contradictory. Typically, no difference is observed when the study is performed for small defects (<2 cm2) and short-term follow-up (<2 years),56,60-63 whereas better clinical outcomes for ACI/MACI are usually associated with larger defects, patellofemoral lesions, and longer follow-up period (>2 years).58,64-68 Therefore, a subset of young patients with acute, small (<2 cm2), well-shouldered defects in the femoral condyles may be the appropriate candidates for MST treatment.69-71 However, for most patients, we would not recommend MST alone, especially on the patella, which has worse outcomes than other locations. 72 AMIC may also be considered in these patients as it has shown improved pain and functional outcome scores in patients up to 9 years postoperatively compared to MST alone; however, additional RCTs comparing AMIC to other cartilage therapies are needed.19,20 Furthermore, it is important to acknowledge the controversial effect of previous MST on patient outcomes after revision ACI or OCA. Previous studies have shown that failed MST increases the risk of ACI failure but does not influence the results of OCA.73-75 However, a recent study comparing primary and secondary (after failed MST) ACI and OCA showed no difference on outcomes scores between primary ACI and secondary ACI, between primary OCA and secondary OCA, and between secondary ACI and secondary OCA. 76 Therefore, MST can potentially “burn the bridge” of future cell-based therapy treatment in clinical failures resulting in OCA as the preferred treatment in the event of a revision.
For medium-sized lesions (2-4 cm2) of the trochlea and femoral condyles, OCA, OAT, and ACI/MACI are the preferred first choice procedures given the significant level of evidence supporting their use in the literature.32,68,77,78 In RCTs, ACI versus OAT demonstrated similar results in some studies, with slightly better results for ACI in larger defects and better results for OAT in smaller defects.64,79-81Advantages to OAT over ACI/MACI include reduced cost, earlier weight-bearing/mobilization, and faster return to sport.45,52,53 Advantages of ACI/MACI over OAT, even in small lesions, include no donor site morbidity and preservation of the underlying subchondral bone. In addition, the surgeon may consider OCA using pre-cut cores or cores harvested from an allograft hemicondyle. Disadvantages to OCA include the risks associated with the use of allograft tissue as well as the violation of the underlying subchondral bone.
With regard to the patellofemoral joint, OAT can be used in small (<2 cm2) lesions but for the reasons previously mentioned OCA may be preferred in the patellofemoral (PF) joint. First-line treatment options for medium lesions (2-4 cm2) include ACI/MACI and OCA. ACI/MACI has been shown to have excellent short-term, midterm, and long-term clinical outcomes.32,82 The shape of the patella and trochlea are more highly variable than the shape of the femoral condyles and tibial plateaus, which complicates morphology matching, particularly with the involvement of the central trochlear groove and median patellar ridge. For these reasons, ACI/MACI are the most common cartilage restoration procedure performed in the PF joint. 83 Additionally, a meta-analysis revealed that failure rates of OCA (22.7%) are higher than chondrocyte-based therapies (6.8%) in the PF joint. 32
Other options of surface treatments, such as PJAC and BMAC implantation, have good short-term outcomes (2-5 years), but they lack the long-term outcomes evidence that ACI/MACI, OAT, and OCA have. These treatments have the advantages of preserving the subchondral bone and can be used in all knee compartments (femoral condyles, tibia, and PF joint).22,26,30,84-89 Successful results have been reported with >2 years follow-up after PJAC and >5 years follow-up after BMAC implantation in defects with a mean size of 3.5 cm2 and 4 to 6 cm2, respectively.7,20,22,26,30,84
Large-Sized Defects (>4 cm2) with Bone Loss/Involvement
OCA is the preferred cartilage procedure for large chondral defects (>4 cm2) with underlying bone involvement. OCA of the femoral condyles has been shown to be durable in high-demand patients with good to excellent functional outcomes at long-term follow up.90,91 OCA has also been shown to be effective in the treatment of large patellofemoral osteochondral lesions and is preferred over ACI/MACI with or without the sandwich technique. 92 If OCA is not available, ACI/MACI with a sandwich technique (bone grafting of the bony defect and an overlying ACI/MACI) is an alternative solution; however, when subchondral bone lesions are present (e.g., cysts and intralesional osteophytes) cell-based treatments are likely to be less effective.75,93,94 In addition, even when good clinical results are achieved, subchondral changes may still persist for a prolonged period of time.95,96
Large-Sized Defects (>4 cm2) without Bone Loss/Involvement
For the treatment of large chondral defects (>4 cm2) with no bone involvement, surgical options include ACI/MACI, OCA, PJAC, and BMAC implantation. Focal cartilage defects have been treated successfully with long-term follow-up with both OCA and ACI/MACI, especially in the femoral condyles.78,97-101 Results of OCA and ACI/MACI are successful, even in very large defects (>8-10 cm2).98,102 Therefore, in femoral condyle defects, the decision is based on the patient’s activity level and the surgeon’s preference. Multifocal defects in different compartments, defects that need more than one plug (with a “snowman” technique), and PF defects lend themselves to treatment with cell-based modalities (ACI/MACI and PJAC). Multifocal defects in different compartments significantly increase the cost of the procedure since multiple grafts sources will be needed. While there is a financial disadvantage, the clinical outcomes are still favorable. 103 Furthermore, OCA transplantation with the “snowman” technique has had worse outcomes and higher failure rates. 103 Bipolar defects have poorer results than unifocal defects across all treatment options.104-106 More studies are necessary to demonstrate a definitive advantage of one modality over the other; however, ACI/MACI appears to have better outcomes than OCA for bipolar defects.99,104-106 If ACI/MACI and OCA are not available there is some evidence to support the use of surface treatments such as PJAC and BMAC implantation. PJAC has shown early favorable outcomes in its application to chondral lesions of the patellofemoral joint; however, it has a comparatively high cost and is not covered by most insurance providers.25,26 BMAC implantation supplemented with a hyaluronic membrane has also been shown in multiple studies to have favorable clinical outcomes for large chondral lesions (4-6 cm2) of the patellofemoral joint and can be considered an option if the previously discussed treatments are not available.22,107
Additional Treatment Options with No/Minimal Clinical Data Requiring Further Evidence
There are a number of additional cartilage treatment options available on the market to the orthopedic surgeon treating focal cartilage lesions of the knee but with limited data to support their clinical efficacy. These treatments include PACI, micronized cartilage extracellular matrix (Biocartilage), viable cartilage allograft putty (CartiMax), cryopreserved osteochondral allografts (Cartiform/ProChondrix), aragonite biphasic osteochondral scaffolds (Agili-C), and human umbilical cord blood–derived mesenchymal stem cells (CARTISEM).
PACI is cheaper and readily available option for defects <4 cm2; however, clinical data are restricted to one case series.34,36 Biocartilage can potentially improve the quality of cartilage repair as both an augment (i.e., in conjunction with OAT or OCA) and stand-alone treatment but there is currently only one case report to support its use.10,13 MST augmented with Cartiform or ProChondrix not only intends to improve the quality of cartilage repair and midterm results, but also to extend the indications for MST-based procedures to larger defects (between 2 and 4 cm2).37-39 Potential advantages to these grafts compared to traditional fresh OCAs include increased graft availability, reduced graft cost, and reduced retained allograft bone marrow elements. Disadvantages include decreased viable chondrocytes, osteocytes, and chondrogenic growth factors. Currently, there is no literature showing clinical results for the use of CartiMax in focal grade III/IV chondral defects; however, it still remains a potential treatment option based on its promising preclinical data. Potential advantages to this treatment include reduced cost compared to OCA and ACI/MACI, the ability to treat cartilage lesions of variable shapes and sizes due to its moldability, and its “off the shelf” availability. Agili-C has promising initial clinical data from case series but is awaiting more robust results from RCT performed internationally. It has the advantages of being suited to treat chondral and osteochondral lesions with ready availability and avoiding the wait for a matching donor. CARTISEM has the advantage of being a one-time procedure but has very limited preclinical and clinical data.
The authors do not discourage the use of the above-mentioned treatments but the currently limited data to support their use should be considered by the surgeon before implementing them in routine patient care. See Table 1 for a chart depicting the pros and cons of various cartilage procedures as well as their commercial vendors.
Summary of Relevant Factors of Each Cartilage Restoration Technique a .
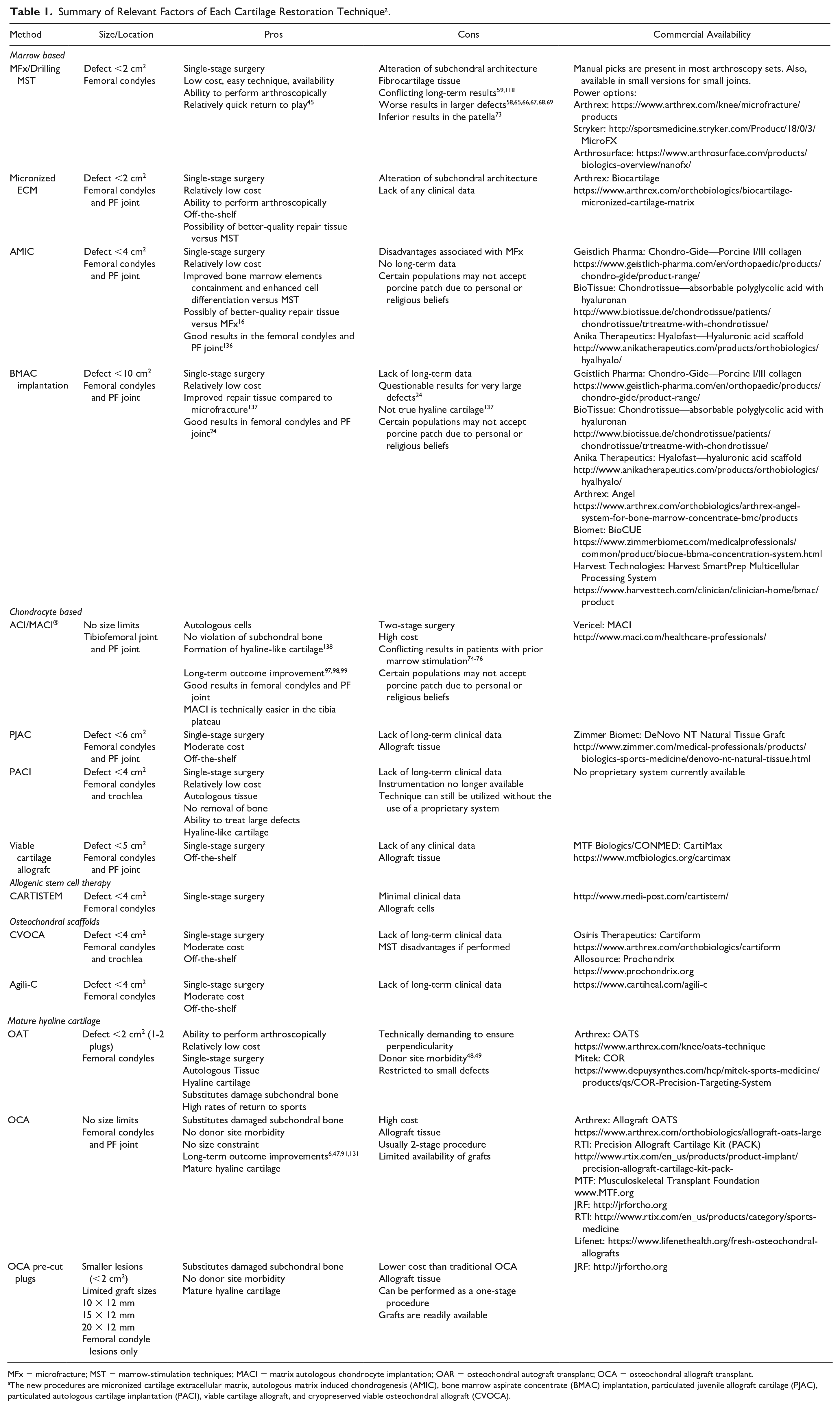
MFx = microfracture; MST = marrow-stimulation techniques; MACI = matrix autologous chondrocyte implantation; OAR = osteochondral autograft transplant; OCA = osteochondral allograft transplant.
The new procedures are micronized cartilage extracellular matrix, autologous matrix induced chondrogenesis (AMIC), bone marrow aspirate concentrate (BMAC) implantation, particulated juvenile allograft cartilage (PJAC), particulated autologous cartilage implantation (PACI), viable cartilage allograft, and cryopreserved viable osteochondral allograft (CVOCA).
Return-to-Sport Expectations
Patient expectations regarding their desired activity level and timeline to return to sport should be considered, especially in patients involved in competitive or professional athletics. In a meta-analysis, Krych et al. found that the return-to-sport rate following cartilage restoration procedures was 76% overall, with the highest rates of return after OAT (93%), followed by OCA (88%), ACI (82%), and MFx (58%). 45 OAT showed the fastest return-to-sport time (5.2 ± 1.8 months), compared to 9.1 ± 2.2 months for MFx, 9.6 ± 3.0 months for OCA, and 11.8 ± 3.8 months for ACI (P < 0.001). It is important to remember that OAT and MST are typically performed for smaller defects, while OCA and ACI are reserved for larger defects. Pestka et al. evaluated the factors that influenced return to sport in 130 patients after ACI. 108 Neither defect location nor size significantly influenced return to physical activity. Both duration of exercise and number of sessions per week significantly decreased from before to after surgery. High-impact as well as start-stop sports were generally abandoned in favor of endurance and low-intensity exercises. A lifetime level of competitiveness was maintained in 31.3% of cases, while return to elite sports at the time of the survey became highly unlikely (0.8%). Nielsen et al. evaluated highly competitive athletes and well-trained and frequently sporting subjects after OCA, and found that 79% were able to participate in a high level of activity (moderate, strenuous, or very strenuous) postoperatively. 109 Patients who did not return to sport were more likely to be female, to have injured their knee in an activity other than sport, and to have a larger graft size.
Cost and Cost-Effectiveness
Cartilage defects and their treatment are associated with a considerable economic burden.
110
The cost of work time lost due to a knee chondral defect over the 10 years prior to surgery has been estimated at approximately $122,000.
111
The cost of surgical cartilage treatments are variable depending on the procedure chosen and the health care system in which they are administered. In the United States, the average costs, for primary and secondary operations in 2018, were $64,593 for MACI, $24,053 for MFx, $32,035 for OAT, and $74,969 for OCA; in comparison to $50,262 for knee arthroplasty.
55
In the United States, ACI treatment has an estimated average cost of $66,752, which includes the initial consult, follow-up visits, and surgical costs.
110
However, even the most expensive option, ACI, can potentially save over $55,000 in the 10 years after surgery compared with expected costs of continued nonoperative care in one health care system.
111
The National Institute for Health Research from the United Kingdom National Health System presented a detailed cost-effectiveness analysis of ACI and other cartilage treatments reported estimated costs for MACI/ACI (biopsy and grafting), mosaicplasty and MFx at
These data suggests that if surgeons use cartilage restoration tools judiciously and with ideal indications, they can provide the best results for their patients while maximizing value to the healthcare system at large.
Conclusion
Cartilage defects are a source of significant morbidity for patients. The treatment of cartilage defects is multifactorial and should be formulated on a patient-specific basis. We have presented current strategies available for dealing with these defects, and we propose an algorithm to guide the thought process of the orthopedic surgeon in determining a successful treatment plan based upon the literature.
Footnotes
Appendix
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
