Abstract
Meniscus tissue deficiency resulting from primary meniscectomy or meniscectomy after failed repair is a clinical challenge because the meniscus has little to no capacity for regeneration. Loss of meniscus tissue has been associated with early-onset knee osteoarthritis due to an increase in joint contact pressures in meniscectomized knees. Clinically available replacement strategies range from allograft transplantation to synthetic implants, including the collagen meniscus implant, ACTIfit, and NUSurface. Although short-term efficacy has been demonstrated with some of these treatments, factors such as long-term durability, chondroprotective efficacy, and return to sport activities in young patients remain unpredictable. Investigations of cell-based and tissue-engineered strategies to treat meniscus tissue deficiency are ongoing.
Introduction
The meniscus plays a vital role in optimizing force transmission and providing stability in the knee. These fibrocartilaginous tissues are semilunar in shape and consist of a sparse distribution of cells surrounded by an abundant extracellular matrix that imparts the tissue’s mechanical function. Meniscal tears are common, and primary surgical options include partial meniscectomy or meniscal repair. As arthroscopic techniques have advanced and biologic augmentation strategies are being investigated, meniscal repairs are now being performed for all tear types, including those in the avascular (white-white) zone that have traditionally been treated with partial meniscectomy (e.g., radial and horizontal cleavage tears).
When meniscal repair fails or is not a valid option resulting in surgical meniscectomy, the loss of meniscal tissue results in a difficult challenge. The meniscus has little capacity for tissue regeneration, and meniscus tissue deficiency has been associated with early-onset knee osteoarthritis due to a decrease in tibiofemoral contact area and an increase in joint contact pressures, particularly among the active population. 1 Several treatment options exist for restoring the deficient meniscus, from allograft transplantation to artificial implants. Indications and contraindications for these treatment options are listed in Table 1 . Despite the improvement in clinical symptoms, the long-term chondroprotective effects from meniscal transplantation or synthetic implants are unclear. A few cell-based meniscal tissue replacement options are being investigated under clinical trials, but none are currently available to date.
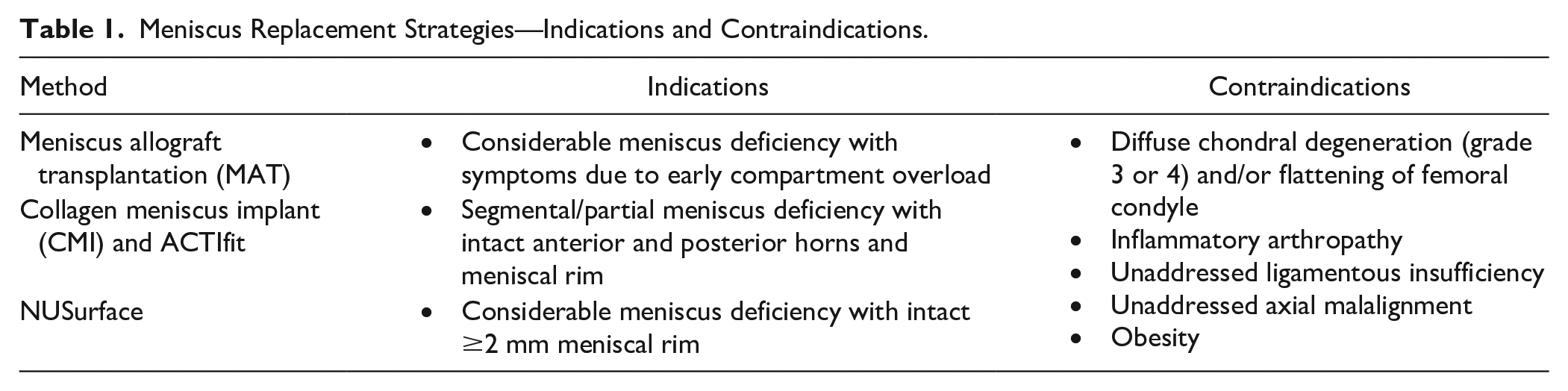
Meniscus Replacement Strategies—Indications and Contraindications.
Meniscus Allograft Transplantation
Meniscus allograft transplantation (MAT) has proven to be an effective solution for young and active symptomatic patients who have undergone meniscectomy (
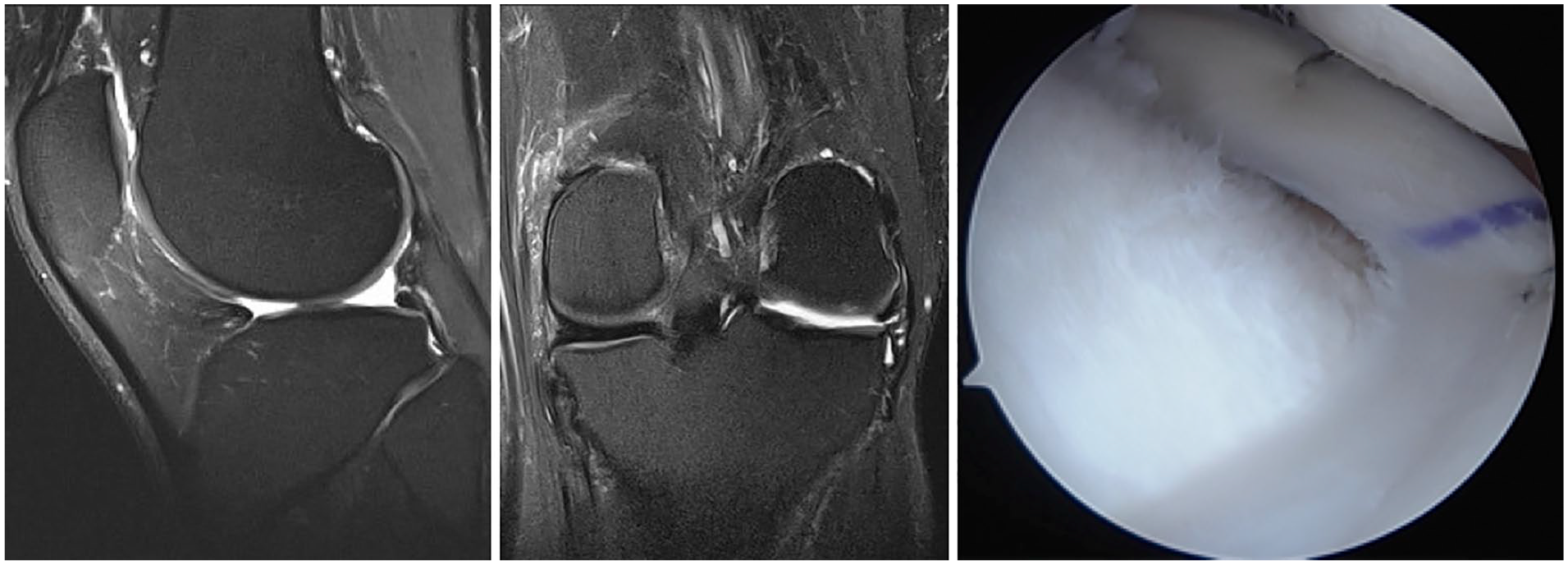
Sagittal (left) and coronal (middle) magnetic resonance imaging of the left knee shows absent lateral meniscus after previous subtotal lateral meniscectomy. The patient was treated with lateral meniscus allograft transplantation (right) using bone plug fixation.
Graft Processing
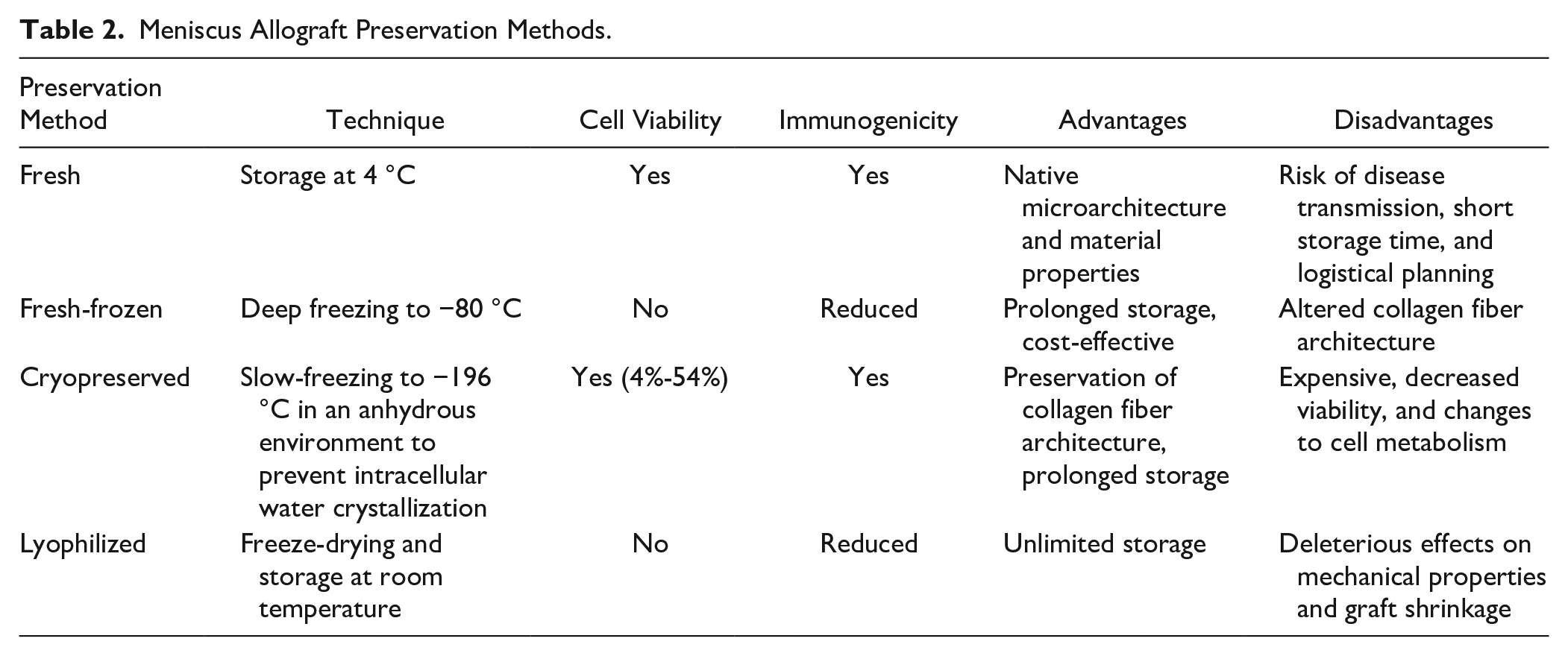
In the United States, meniscus allograft tissue is most commonly distributed in fresh and fresh-frozen forms, while cryopreserved allografts are infrequently offered but remain an option in other countries ( Table 2 ). Lyophilization (freeze-drying) has fallen out of favor because of its deleterious effects on the mechanical properties of the allograft and graft shrinkage. Although some believe that preservation methods that maintain cell viability (i.e., fresh and cryopreserved) enhance graft survival and function, there has been no evidence to date demonstrating this supposed benefit. Data from animal models have shown a relatively rapid repopulation of donor graft tissue with recipient cells within a few weeks after transplantation, 7 thereby raising questions about the necessity of cell viability in optimizing graft survival and clinical outcomes. However, fresh-frozen grafts seem to have diminished collagen fiber architecture and biomechanics compared with fresh and cryopreserved grafts. 8 The lower cost and logistical benefits of fresh-frozen grafts account for their greater popularity at most centers.
Meniscus Allograft Preservation Methods.
Treatment of meniscus allografts with gamma irradiation or chemical processes can be performed to reduce the risk of bacterial, fungal, and viral transmission. Sterilization typically results in killing of viable cells and is thus not performed on fresh and cryopreserved grafts. Dosages of radiation required to kill viruses (i.e., 1.5-2.0 mrad) can cause deleterious changes to the meniscus tissue biomechanical properties, 8 and therefore, use of nonirradiated grafts is preferable. Ethylene oxide gas sterilization, which is commonly used to sterilize medical devices, produces a metabolic byproduct (ethylene chlorohydrin) that causes significant synovitis and is therefore not recommended as a sterilization agent. 9 Other sterilization techniques such as supercritical CO2, which are purported to better preserve tissue properties over gamma irradiation, are being investigated. These emerging and proprietary sterilization techniques may be more appropriate for synthetic materials rather than biologic tissue grafts.
Graft Fixation
Peripheral fixation of meniscal allografts is traditionally performed using vertical-mattress sutures along with accurate reestablishment of the meniscal horns and roots. Both inside-out mattress sutures tied over the joint capsule and all-inside sutures are widely used. Some native peripheral rim tissue should be retained to decrease peripheral extrusion and provide a firm base to which the allograft is secured. Secure fixation of the horns and roots is crucial for permitting optimal distribution of hoop stresses throughout the meniscus allograft. Three main fixation methods for securing the graft horns and roots include (1) soft tissue only, (2) bone plugs, and (3) bone bridge. The optimal method for horn fixation continues to be debated. Recent cadaveric studies have suggested that bone plug fixation more closely reproduces the normal function of the meniscus compared with soft tissue fixation for medial meniscal allografts.10,11 For lateral meniscal allografts, Brial
Clinical Outcomes
Many studies reporting on the outcomes of MAT are limited by low level of evidence; heterogeneity of data including patients, graft types, and techniques; inconsistent exclusion criteria for transplantation; and concomitant procedures that may confound results. As graft preparation and fixation techniques continue to advance, the clinical outcomes of MAT may be further optimized.
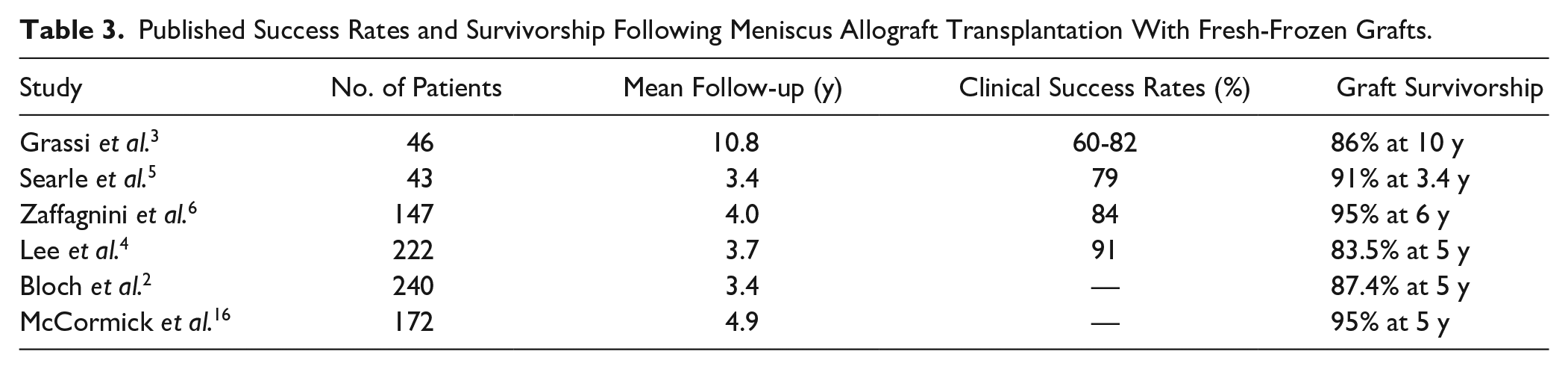
In studies evaluating the clinical outcomes of MAT, clinically significant improvements in patient-reported outcome scores, including the Lysholm, Knee Injury and Osteoarthritis Outcome Score (KOOS), and International Knee Documentation Committee (IKDC) scores, are noted at mid-term follow-up.2-5 Although functional improvement can be maintained up to 10 years, activity and sport-specific scores seem to decline during the interval between short-term/mid-term and long-term follow-up.3,15 For fresh-frozen grafts, 5-year graft survivorship is high, ranging from 84% to 95% ( Table 3 ). For cryopreserved grafts, 10+-year graft survivorship is reported to range from 45% to 71%.17-19 The presence of grade 4 articular cartilage loss or bipolar lesions at the time of MAT seems to portend worse graft survival,2,4 suggesting that early treatment of symptomatic patients may be optimal before significant chondrosis is sustained. Postoperatively, the incidence of graft tears ranges from 11% to 16%, although the majority of these cases do not necessitate full graft removal. 3 The wide variability in rate of graft tears reported is likely due to the use of different outcome measures for graft assessment, including MRI scan and second-look arthroscopy. When comparing medial MAT with lateral MAT, the functional outcomes and long-term survival rates appear to be similar. 20
Published Success Rates and Survivorship Following Meniscus Allograft Transplantation With Fresh-Frozen Grafts.
Probability of returning to work after MAT can be high (>85%), 21 although this is likely dependent on the intensity of loading required on the knee for the specific occupation. In a cohort of active-duty military patients, only 20% were able to return to full duty, and 46% had permanent profile activity restrictions. 22 Although MAT is generally considered a salvage procedure with return to repetitive impact activities such as running and jumping generally being discouraged, recent studies suggest that return to modest sports activities in the short-term is a reasonable goal. In a meta-analysis, the majority of athletes and physically active patients (77%) were able to return to sport after MAT at a mean of 9.2 months postoperatively, and two-thirds of athletes were able to return to preinjury levels. 23 Graft-related reoperations, which were mostly partial meniscectomies, were reported in 13% of patients. 23
Although MAT appears to decrease tibiofemoral pain in the short-term, the long-term chondroprotective effects of the procedure remain unclear. In sheep and rabbits, MAT has been shown to protect the articular cartilage from degeneration.
24
While human cadaveric studies demonstrate the biomechanical benefits of MAT with reduced peak contact pressures compared with meniscectomized knees, MAT does not fully restore contact mechanics and kinematics to that of the intact knee.13,25 Articular cartilage benefits in animals and human biomechanical studies seem to agree with some clinical studies, but other clinical studies in humans have not found clear chondroprotective benefits. At a mean of 12 years, Verdonk
Synthetic Options
Two synthetic, scaffold-based meniscal substitutes for partial meniscus replacement are currently commercially available for clinical use: collagen meniscus implant (CMI; Stryker Corporation, Kalamazoo, MI, USA) and ACTIfit (Orteq Sports Medicine Ltd., London, UK). Both have demonstrated promising results in early clinical trials.28-30 In contrast to MAT, both can replace segmental meniscal defects, thereby preserving intact native meniscus tissue, and the off-the-shelf nature of synthetic grafts makes surgical planning easier. However, lack of cell migration, stress shielding, and degradation products causing chronic synovitis remain a concern with any scaffold-based treatment options. Another synthetic option, the NUSurface Meniscus Implant (Active Implants LLC, Memphis, TN, USA), is a non-anchored, non-absorbable meniscal prosthesis designed for total replacement of the medial meniscus. Although these options may be clinically available, lack of third-party insurance reimbursement has limited their clinical utilization.
Collagen Meniscus Implant
The CMI, which consists of type I collagen fibers derived from bovine Achilles tendons, has gained attention since the first clinical trial was published in 1997. It received US Food and Drug Administration (FDA) 510(k) clearance in 2008. Designed for segmental meniscal replacement, the implant is sized to match the prepared meniscal defect and sutured to the surrounding intact meniscus tissue (
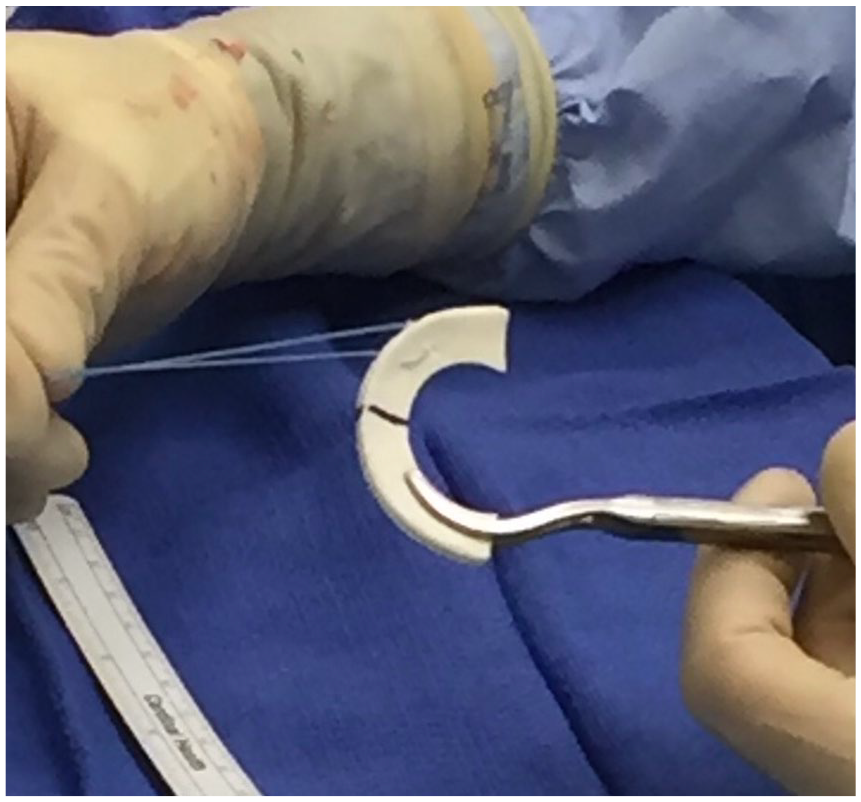
Photograph of collagen meniscal implant (CMI).
The majority of published clinical outcomes on the CMI are limited to treatment of medial meniscal defects, although short-term results after CMI treatment for lateral meniscal defects are available. 32 Ten-year data demonstrate significant clinical improvements after CMI, 33 but comparative studies are scarce. In a systematic review of 311 patients treated with CMI, the failure rate was 6.7% at a mean follow-up of 44 months. 29 Visual Analog Scale (VAS) pain, Lysholm, and Tegner scores were significantly improved at final follow-up, with most studies demonstrating improvement in Lysholm scores above minimal clinically important difference (MCID) and patient acceptable symptom state (PASS) thresholds. 29 A randomized, controlled, multicenter clinical trial showed that in patients with prior medial meniscal procedures, those treated with CMI regained significantly higher activity (Tegner Activity Scale) and were more satisfied compared with patients treated with repeat partial medial meniscectomy. However, in patients who had no prior meniscal surgery, no difference could be observed between CMI and partial meniscectomy treatment groups. 28 In another comparative study with a minimum of 10 years of follow-up, CMI-treated patients had better pain, activity level, and radiographic outcomes (less medial joint space narrowing) compared with patients treated with partial meniscectomy. 33
ACTIfit
The ACTIfit (
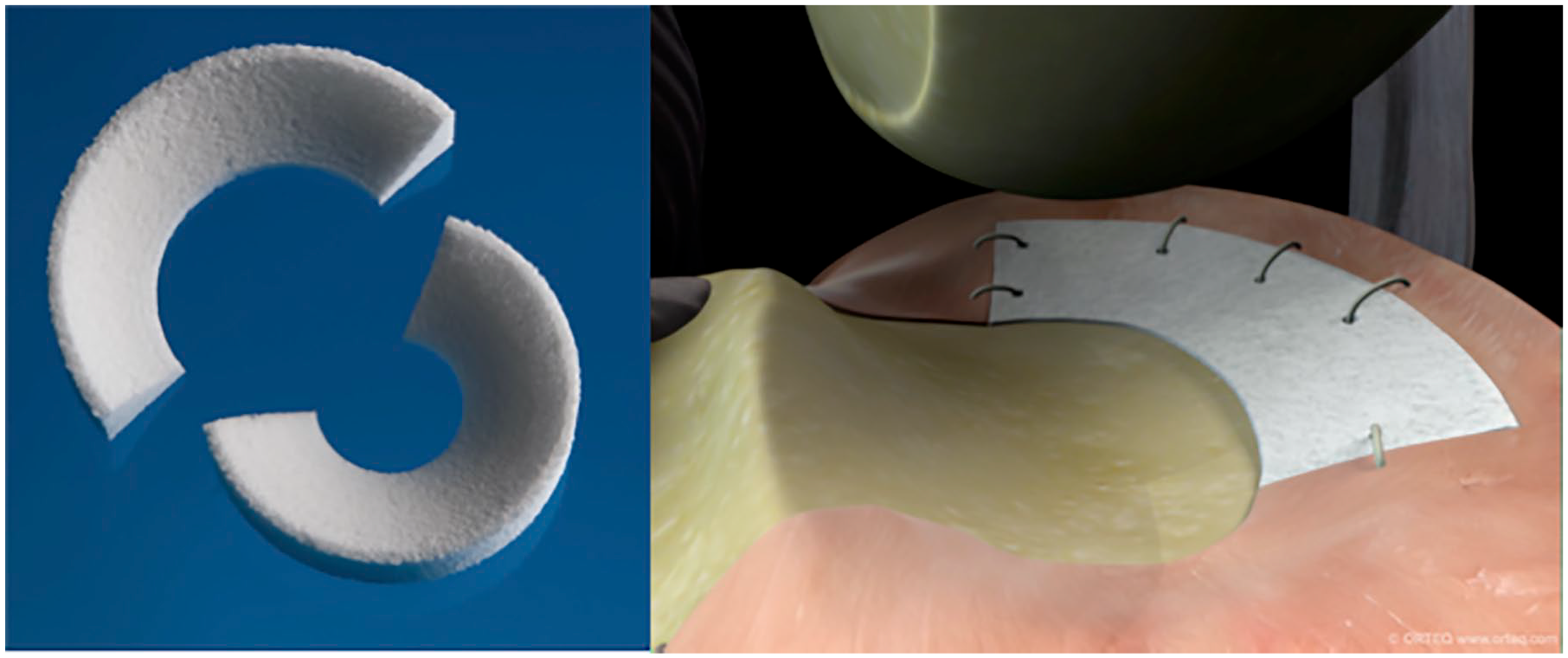
Photograph and
To date, clinical studies on the ACTIfit are limited to short-term and mid-term case series. In the largest available series consisting of 155 patients, pain and knee scores improved postoperatively at 2 years and were stable at 5 years of follow-up. 30 Postoperative MRI demonstrated a small-sized implant with an irregular surface in the majority of cases. The overall surgical failure rate was 12.4% at 5 years, with no difference in failure rates between medial and lateral implants. 30 A few studies have attempted to compare the clinical outcomes of CMI versus ACTIfit and have demonstrated no differences in failure rate or improvement in patient-reported outcomes between groups.29,38
NUSurface
The NUSurface Meniscus Implant is a non-anatomical, discoid-shaped, free-floating meniscal substitute designed for total replacement of the medial meniscus. It is made of polycarbonate-urethane, a medical grade plastic. A biomechanical study showed that implantation of the NUSurface Meniscus Implant restores the average and peak tibiofemoral contact pressures to 93% and 92%, respectively, compared with the native medial meniscus. 39 The NUSurface was granted FDA Breakthrough Designation in 2019, and 2 Investigational Device Exemption (IDE) clinical trials are currently ongoing in the United States. In Europe and Israel, clinical use of the NUSurface has been ongoing since 2008, but there are minimal published outcomes data available. Preliminary results have demonstrated significant improvements in pain and KOOS scores at 12 months for the NUSurface compared with non-surgical therapy and a similar adverse event rate.40,41
Cell-Based Options
Several cell-based meniscal replacement options are being developed and tested in clinical trials. Although the following cell-based options are not currently approved by the FDA, orthopedic surgeons could benefit from knowledge of these options that may be commercially available in the future.
Cell Bandage (Azellon)
The Cell Bandage consists of expanded (passage 1) autologous bone marrow–derived mesenchymal stromal cells (MSCs) embedded in a collagen matrix. It is a 2-stage procedure, with the first stage involving harvesting of cells from host bone marrow, isolation, culture, and seeding onto the collagen matrix, followed by the second stage implantation. This treatment was designed for repair of meniscus tears in the avascular (white-white) zone that would otherwise be an indication for meniscectomy; the seeded scaffold is placed between torn edges of the meniscus, and the tissue is reapproximated using sutures. 42
Preclinical studies for the Cell Bandage used an ovine model, in which autologous bone marrow–derived MSCs were used. 42 Three out of 5 sheep showed successful healing in the white-white region of the meniscus at 13 weeks; however, no animals showed healed lesions after 6 months. In comparison, no animals in the collagen sponge-only and suture-only control groups showed signs of healing at either time point.
In a first in-human study (phase I clinical trial in the United Kingdom), implantation of the Cell Bandage appeared to be safe as no adverse local or systemic immune responses were reported. 42 The implant survived in 3 out of 5 patients at 24-month follow-up as indicated on MRI without any further treatments. Clinically significant improvement in IKDC and Tegner-Lysholm scores was observed at 12 months and maintained at 24 months. The two other patients developed recurrent symptoms due to retear or nonhealing of the meniscus before ultimately receiving subsequent meniscectomy. As the phase I clinical trial is still ongoing (EU Clinical Trials Register, 2010-024162-22), no results with longer follow-up have been published.
Chondrogen (Mesoblast)
Chondrogen consists of expanded (passage 2) allogeneic adult bone marrow–derived MSCs suspended in a sodium hyaluronate solution that is injected intra-articularly following partial meniscectomy. 43 As opposed to being a meniscus tissue substitute, Chondrogen is an augmentation biologic injectable therapy that attempts to enhance meniscus regeneration and tissue volume after meniscectomy. Human MSCs are derived from bone marrow aspirates collected from unrelated donors (18-30 years of age) and are not human leukocyte antigen (HLA)-matched to recipients.
A phase I/II, randomized, double-blind, controlled study on Chondrogen consisting of 55 patients has been reported. 43 After partial meniscectomy, patients received a single intra-articular injection of 50 million MSCs, 150 million MSCs, or a vehicle control. No patients in any of the treatment groups exhibited abnormal immune or hematologic responses. At 24 months, 3 patients (18%) from the 50 million MSC group exhibited a >15% increase in meniscus volume as found on MRI, while this was not observed in patients in the 150 million and control groups. Decreased VAS pain and increased Lysholm scores were seen at 24 months in all treatment groups with respect to baseline. This phase I/II study in the United States has been completed (ClinicalTrials.gov Identifier NCT00702741), although results have not yet been published.
Future Directions
Although MAT and synthetic meniscus replacement options may be effective in alleviating knee joint pain in the short-term, their long-term durability and chondroprotective effects are questionable. Inferior mechanical properties of these grafts presumably lead to their eventual failure over time. This motivates the development of tissue-engineered meniscus replacement and regeneration options that recapitulate the mechanical, structural, and compositional properties of native meniscus tissue. Tissue engineering researchers have proposed both scaffold-based and scaffold-free meniscus replacement options. Scaffold-based technologies, including 3-dimensional (3D)-printed biomimetic constructs, have been used
Scaffold-free neomenisci (
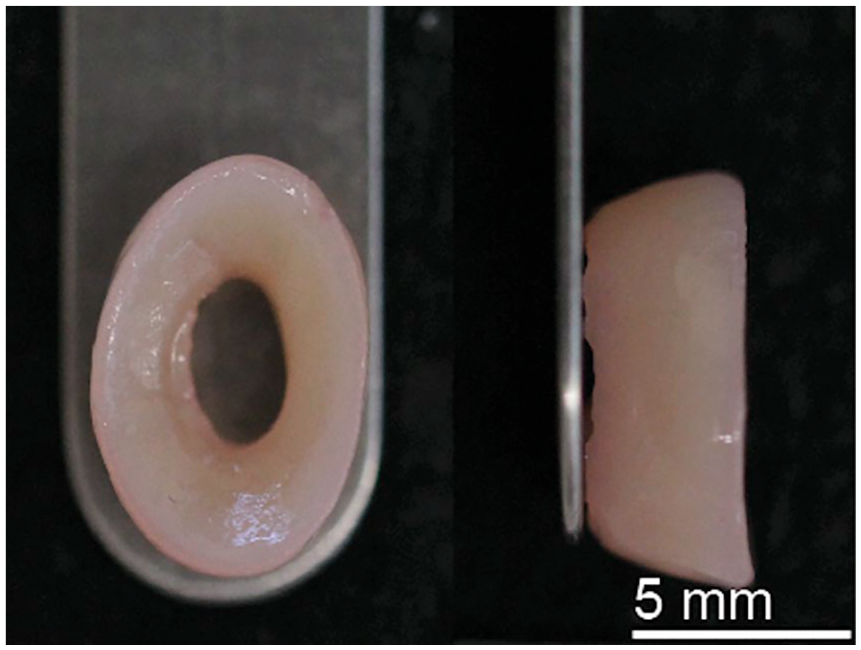
Tissue-engineered neomenisci using the scaffold-free, self-assembling process.
Summary
The meniscus plays an important role in protecting the health of the knee joint. Once meniscus tissue has been torn and removed from the joint, there is little to no capacity for the meniscus to regenerate lost tissue. MAT can be an effective treatment option, with proof of short-term and mid-term clinical functional improvement, although graft durability and return to sport activities remain a challenge. Artificial options, including the CMI, ACTIfit, and NUSurface, are being increasingly used, particularly for segmental defects, although implant durability and third-party insurance reimbursement remain challenges. Investigations of cell-based meniscal tissue replacement options are ongoing. Finally, tissue-engineered options that can generate biomimetic neomeniscus tissue may further optimize patient outcomes after treatment for meniscal tissue deficiency. Even with these advances, surgeons should continue to attempt meniscal repair whenever feasible and resect as little meniscal tissue as possible in tears that are deemed irreparable.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: E.G.L., and K.A. received support from the following funding sources related to meniscus tissue engineering: National Institutes of Health R01AR071457, HHMI Gilliam Fellowship, National Science Foundation GRFP, and University of California, Irvine Eugene Cota Robles Fellowship. The authors would like to acknowledge support from the funding sources. D.W. and S.R. do not have any disclosures or conflicts of interests related to the proposed manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
