Abstract
Objective
Autologous chondrocyte implantation was the first cell-based therapy that used a tissue engineering process to repair cartilage defects. Recently improved approaches and tissue-engineered cell constructs have been developed for growing patient populations. We developed a chondrocyte construct using a collagen gel and sponge scaffold and physicochemical stimuli, implanted with a surgical adhesive. We conducted a proof-of-concept study of these improvements using a cartilage defect model in miniature swine.
Design
We implanted the autologous chondrocyte constructs into full-thickness chondral defects in the femoral condyle, compared those results with empty and acellular scaffold controls, and compared implantation techniques with adhesive alone and with partial adhesive with suture. Two weeks after the creation of the defects and implantation of the cellular or acellular constructs, we arthroscopically confirmed that the implanted constructs remained at the chondral defects. We evaluated the regenerated tissue macro- and microscopically 6 months after the cell constructs were implanted. The tissues were stained with Safranin-O and evaluated using Sellers’ histology grading system.
Results
The defects implanted with processed cell constructs and acellular scaffolds were filled with chondrocyte-like round cells and with nearly normal tissue architecture that were significantly greater degree compared to empty defect control. Even with the adhesive alone and with suture alone, the cell construct was composed of the dense cartilaginous matrix that was found in the implantation using both the sutures and the adhesive.
Conclusion
Implantation of cell constructs promoted regeneration and integration of articular cartilage at chondral defects in swine by 6 months.
Introduction
Articular cartilage lesions in the knee are a common pathology, found in 60% of patients who undergo arthroscopic procedures. 1 If a lesion goes untreated, there is increased risk of articular cartilage degeneration, which leads to osteoarthritis (OA), followed by pain and dysfunction.2,3 Several treatments are available to promote regeneration. Debridement, 4 drilling, 5 abrasion chondroplasty, 6 and microfracture procedures 7 are often chosen as first-line treatments for cartilage lesions. However, long-term follow-up shows that clinical outcomes begin to deteriorate at 18 months. 8 Although other options include osteochondral autograft transplantation (OAT) 9 and osteochondral allograft transplantation (OCA), both have limitations. OAT is subject to donor-site morbidity and cannot be used to treat large lesions, and OCA carries the risk of disease transmission and subchondral bone collapse.
To address these problems, more than 2 decades ago, a cell-based therapy to promote cartilage repair using cultured autologous chondrocytes was developed. 10 Autologous chondrocyte implantation (ACI), as the first generation of such therapies was called, was recognized as a promising treatment for symptomatic large and full-thickness cartilage lesions.11-13 It reduced both the reoperation rate to correct graft hypertrophy and the development of arthrofibrosis over time.14,15 Beyond this pioneering therapy, other new cartilage graft approaches have been introduced with or without scaffolds, in which autologous or allogenic chondrocytes are implanted into the defect.16-25 Some of these graft innovations have been the subject of clinical studies, and the most promising of them, e.g., matrix-induced autologous chondrocyte implantation (MACI®), continue to accumulate long-term follow-up outcomes.26-31 The latest research efforts reflect a desire for improved rehabilitation programs and faster recovery.32-35 We sought to fill a full-thickness cartilage defect with cartilage-like chondrocytes construct to promote resurfacing at the defect. To produce a chondrocyte construct, we intended to use a porous sponge scaffold. However, seeding cells onto the sponge created nonuniform cell distribution. Although uniform cell distribution in a scaffold can be achieved using a collagen gel, our gel shrank during in vitro incubation. We solved these material-related problems using collagen gel and sponge scaffold and improving our cell seeding method. In addition, to maximize anabolic turnover, we incubated chondrocyte constructs under hydrostatic fluid pressure (HP) mimicking compressive stresses within a fluid-rich articular cartilage with weight-bearing and joint loading.36,37 This incubation method allowed stimulation of cartilaginous matrix production by chondrocytes without chemical factors, which may require regulatory approval for clinical applications. We used a collagen-polyethylene glycol-based adhesive to ease implantation of the chondrocyte construct. This adhesive consisted of modified collagen type-I and polyethylene glycol gel. The collagen type-I–based component prevented bleeding from calcified cartilage/subchondral bone at the bottom of the defect and allowed cell attachment and migration on the gel surface. We believe that utilization of grown chondrocyte constructs and a fixative represent an important advance in surgical approaches for cartilage repair allowing for quicker and more consistent application with less training.
We assessed the effectiveness of these cellular products and surgical approach using surgical chondral defects in swine knees. Since we used surgical adhesive for cell construct implantation, advancements in adhesives were also confirmed technically and histologically. To elucidate the effects of cell construct implantation, we compared chondrocyte constructs with an empty control and an acellular scaffold control. In addition, we also compared cell scaffold implantation with a combination of adhesive and suture, adhesive alone, and partial adhesive and suture. We evaluated cartilage regeneration macroscopically (at 2 weeks and at 6 months) and histologically (at 6 or 7 months) after implantation. This report demonstrates the success of robust surgical approaches for minimally invasive implantation of processed autologous chondrocyte constructs.
Materials and Methods
Experimental Design
We conducted an efficacy study of the processed cell construct and the implantation technique using a surgically created defect model in the articular cartilage of swine knees (
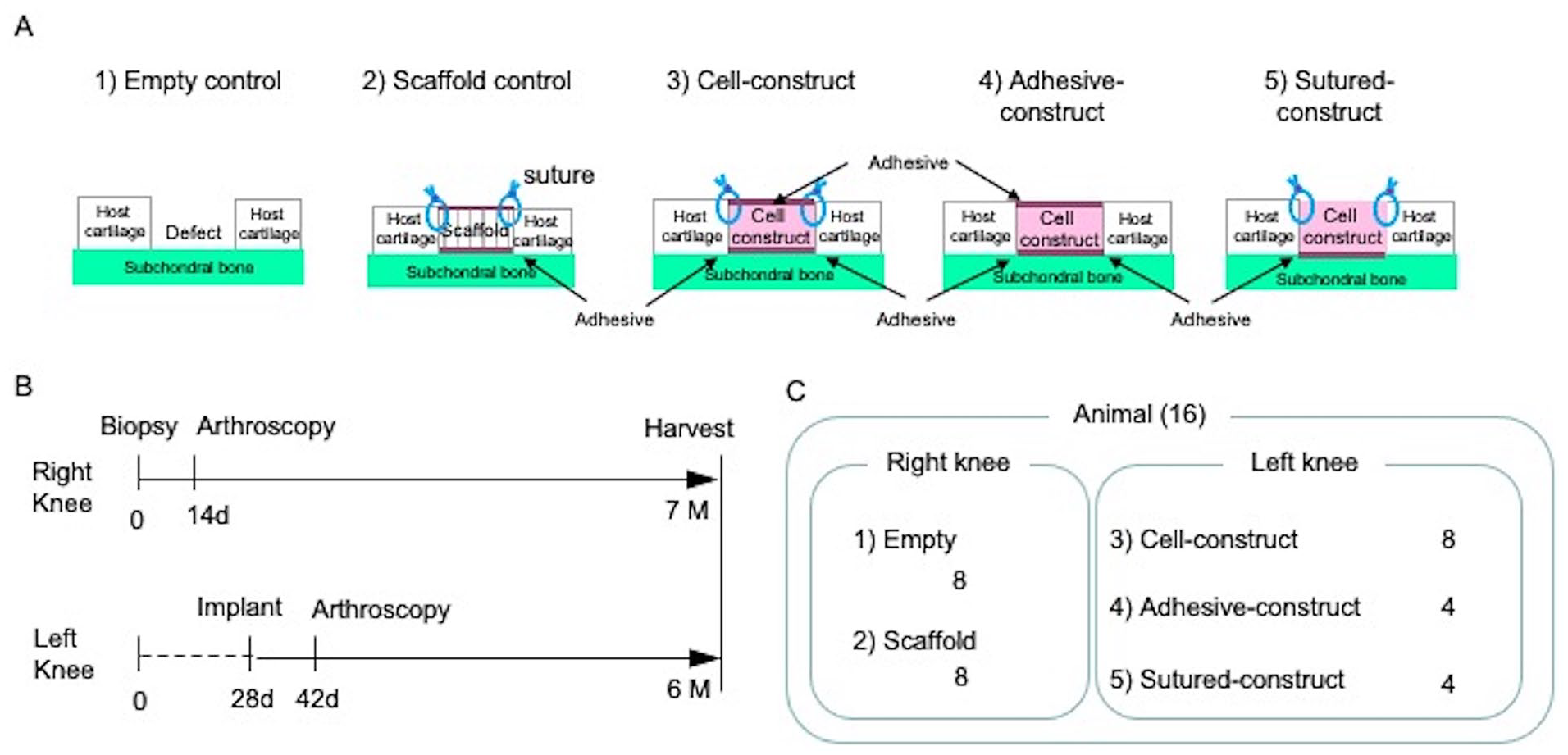
Experimental design. (
Two rounds of surgeries were conducted (
Eight right knees had 2 Empties created in each, 8 right knees had 2 Scaffolds implanted in each, 8 left knees had 2 Cell constructs implanted in each, 4 left knees had 2 Adhesive cell constructs implanted in each, and 4 left knees had 2 Sutured cell constructs implanted in each (
Fabrication of Cell Constructs
Biopsies were harvested from the porcine articular cartilage, minced, digested in 1.5 mg/ml collagenase (CLS 1, Worthington, Freehold, NJ) dissolved in Ham’s Nutrient Mixture F-12 (F-12, Invitrogen, Carlsbad, CA) with 100 µg/ml penicillin and 100 unit/ml streptomycin (P/S, Invitrogen), and placed on a rotator at 37°C for 18 hours. The cell viability at each biopsy was more than 95%. The porcine articular chondrocytes (pACs) were seeded into cell culture dishes (100 mm in diameter) and incubated in Dulbecco Minimal Essential Medium (DMEM)/F-12 supplemented with 10% fetal bovine serum (FBS, Invitrogen) and P/S at 37°C, 5% CO2 in air for 6 days.
The pACs were harvested from the culture dishes with 0.05% trypsin-EDTA (Invitrogen) for seeding into the collagen gel/sponge scaffold (
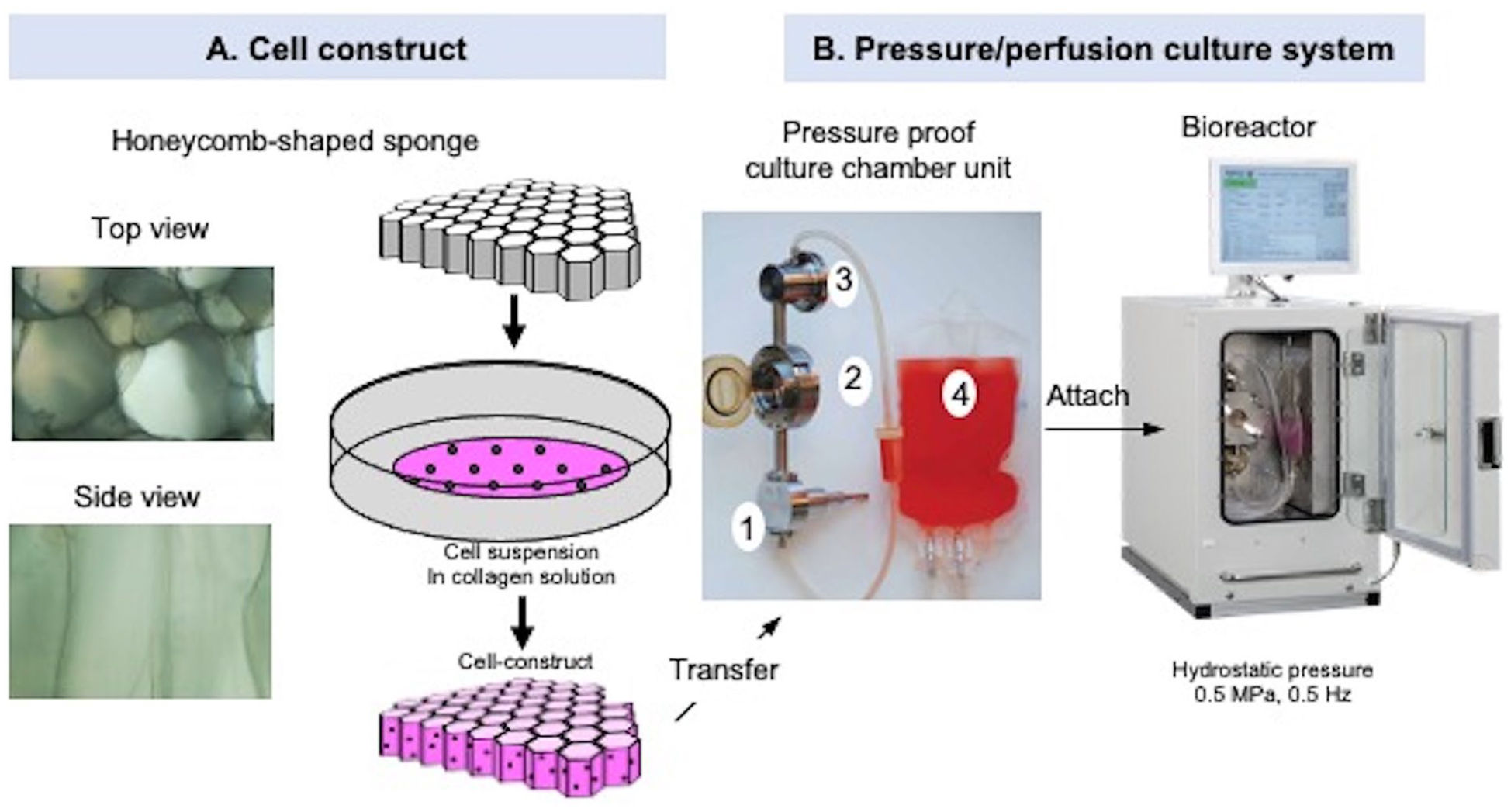
(
Prior to the Scaffold implantation, we soaked the same honeycomb-shaped porous collagen sponge used for the processed cell construct without cells, creating the acellular scaffold.
Validation of Processed Cell Constructs Prior to Implantation
Cell viability and cellularity of the surrogate constructs were evaluated histologically (
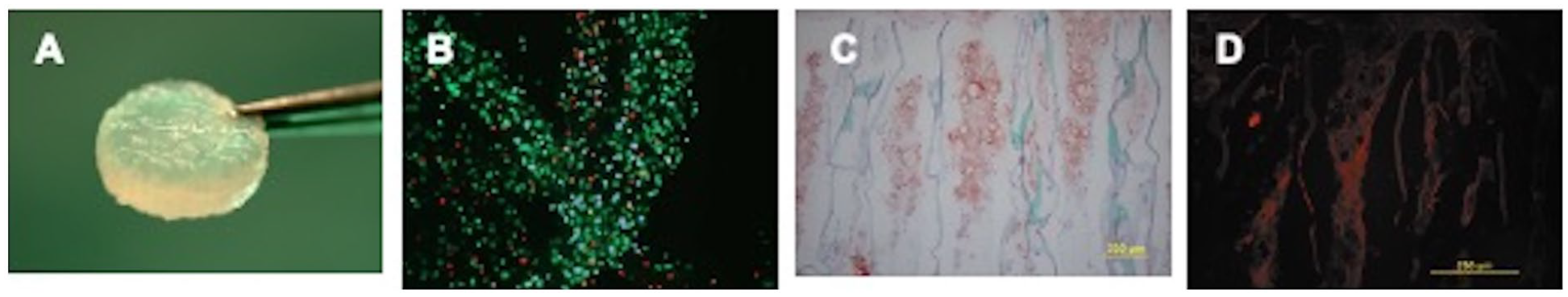
(
Surgical Procedure: Biopsy, Empty Defect Creation, Acellular Scaffold Implantation, Processed Cell Construct Implantation
The protocol for the animal study was approved by the institutional animal care and use committee of Charles River Laboratories (Worcester, MA). Sixteen castrated 12- to 15-month-old male swine weighing 30 to 45 kg (Micro-Yucatan, Charles River Laboratories) were acclimatized more than 1 week prior to the first surgery. Anesthesia was induced with intramuscular injections of 0.04 mg/kg atropine sulfate (Patterson, Devens, MA), 0.55 mg/kg butorphanol tartrate (Patterson), 1.5 mg/kg xylazine (VEDCO, St. Joseph, MO), and 20 mg/kg ketamine hydrogen chloride (VEDCO) and maintained with inhalation anesthesia, Isoflurane (Patterson). The right knee joint was opened anterolaterally, and the patella was luxated medially to expose the trochlea and medial condyle.
A few pieces of cartilage tissue (biopsy) were harvested from the trochlear ridges on the right side of the knees (
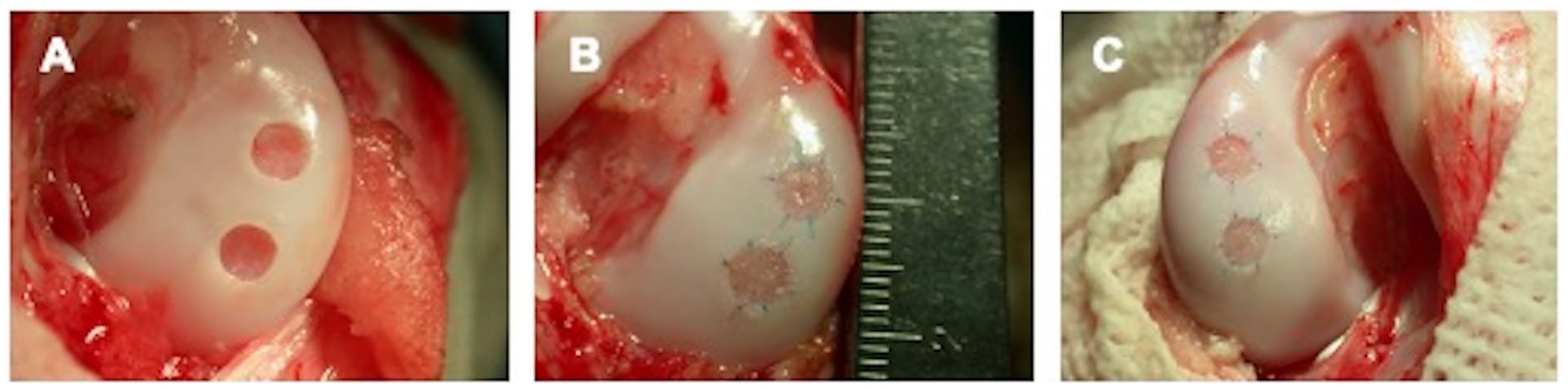
Articular surface of chondral defects implanted with acellular scaffolds or cell constructs after implantation, visualized macroscopically: (
To implant the cell constructs, the left knee joint was opened anterolaterally and the patella was luxated medially to expose the femoral medial condyle. Two chondral defects were created on the condyle, applying the same method used for the Empty and Scaffold. The cell constructs were placed in the defects, sutured with 4 absorbable and 2 non-absorbable colored sutures, and covered with the CT-3 adhesive (
Arthroscopic Evaluation
Two weeks after each open knee surgery, we evaluated the cartilage surface arthroscopically (Direct-View Arthroscope, 2.7 mm outer diameter; Dyonics Vision 625 Digital Capture System, Smith and Nephew, MA), administering the same anesthesia used for other surgical procedures. During arthroscopy, saline was infused at a rate of 10 to 50 mmHg (InteliJET™ System 4672, Smith and Nephew). We evaluated the surface using the criteria listed in Table 1 (ICRS-Cartilage repair assessment system). If any implant did not remain at the site, we removed the histology sample from the evaluation.
Arthroscopic Evaluation for Cartilage Injury (International Cartilage Repair Society-Cartilage Repair Assessment System).

Macroscopic and Histological Evaluation
Six months after the cell construct implantation (7 months after biopsy or acellular scaffold implantation), we conducted macroscopic and histological evaluations. The animals used were euthanized prior to the necropsy. The articular surface where the empty defects control, the implanted acellular scaffolds, and the implanted cell constructs were located were evaluated for gross anatomical findings: the visual characteristics of filling tissue in the defect, filling ratio, color, and surface integration with host tissue, as compared to surrounding host cartilage. Macroscopic images were recorded with a digital camera (Coolpix E-995, Nikon USA, NY). Then the repaired cartilage with subchondral bone and adjacent cartilage was harvested and fixed in 4% paraformaldehyde (JT Baker, Phillipsburg, NJ) dissolved in PBS (pH 7.4) for 7 days on a gentle rotator at 4°C. The fixed tissues were then decalcified in 5% formic acid and sodium citrate solution (Sigma-Aldrich) for 1 to 2 weeks and embedded in paraffin. The paraffin-embedded samples were cut into 4-µm-thick longitudinal serial sections and stained with hematoxylin and Safranin O-fast green.
Histological findings were scored using a modified version of the histological grading scale developed by Sellers et al.
38
Three blinded investigators evaluated the longitudinal sections using the criteria listed in
Data Analysis
The histological and arthroscopic scoring data were analyzed using the Kruskal-Wallis test followed by a post hoc Bonferroni correction for non-parametric data.
Results
In Vitro Culture
After 3 weeks of culture, pACs formed a stiff and slippery cell construct (
General Observation of Symptoms
After each surgery, all of the animals appeared to recover quickly from anesthesia. The arthrotomy wounds exhibited signs of good healing and no signs of infection or irritation. The animals continued to exhibit no physical limitations. They behaved and moved normally throughout the entire study.
Arthroscopic Evaluation 2 Weeks Post Surgery
We evaluated the surfaces of the empty defects, the implanted acellular scaffolds, and the implanted processed cell constructs 2 weeks after surgery (
Arthroscopy Post Defect Creation and Cell Construct Implantation at 2 Weeks.
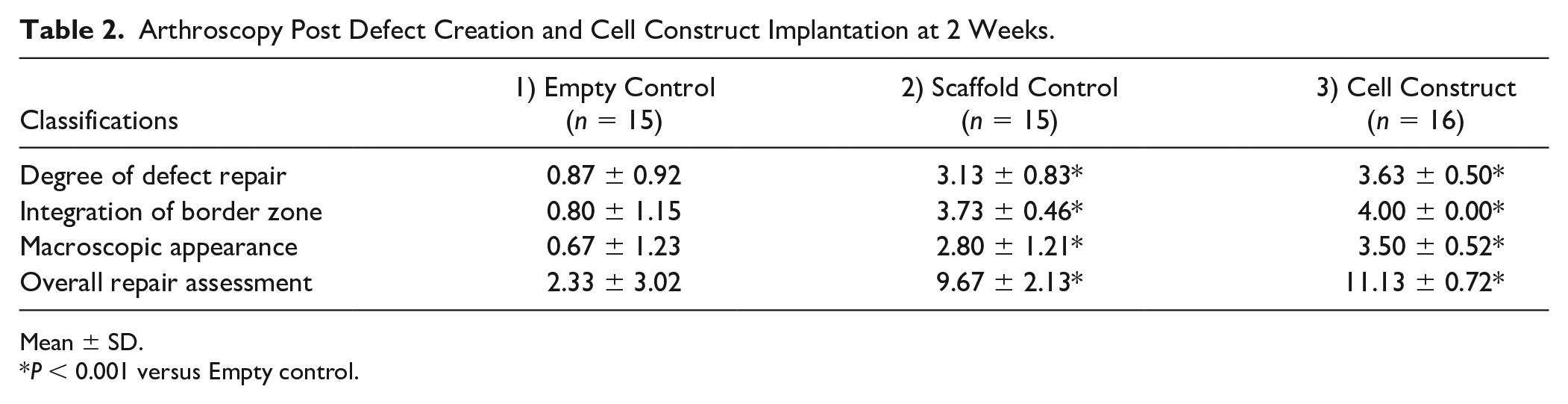
Mean ± SD.
P < 0.001 versus Empty control.
Arthroscopy Post Defect Creation and Cell Construct Implantation at 2 Weeks.
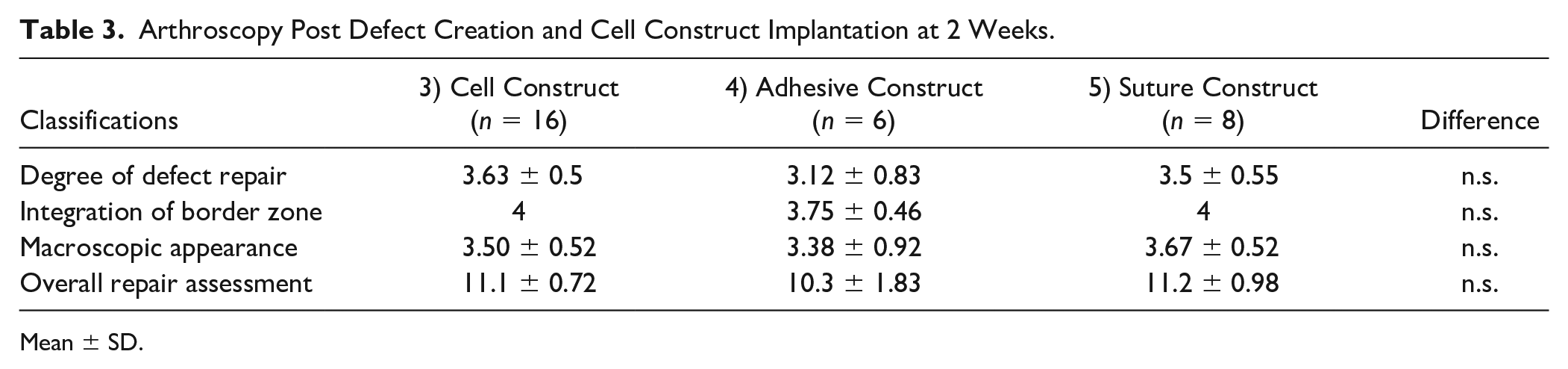
Mean ± SD.
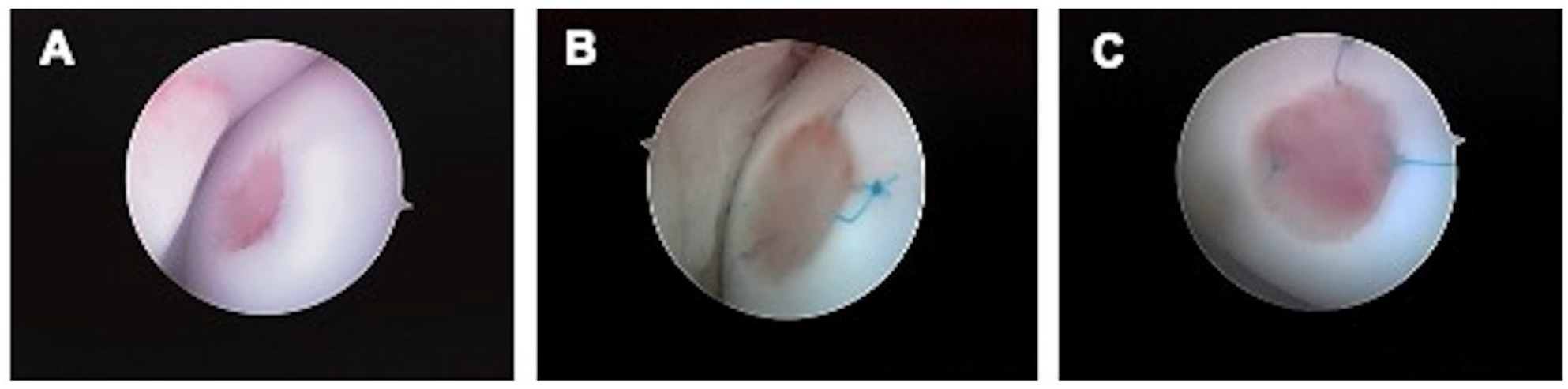
Articular surface of chondral defects implanted with acellular scaffolds or cell constructs after implantation at 2 weeks, visualized arthroscopically: (
Macroscopic Evaluation 6 Months after Implantation
We evaluated the surface of articular cartilage macroscopically at the time of necropsy. In the knee joint space, there was no pathogenesis, synovitis or delamination, or clear synovium fluid. A difference in total scores and the subscale of scores between the acellular scaffolds and the cell constructs was noted, but it was not statistically significant. The surfaces of the implanted Scaffolds, Cell constructs, Adhesive constructs, and Sutured constructs were smooth, white, and flat. Conversely, the surface of the Empty control was fibrous.
Histological Evaluation 6 Months after Implantation of the Processed Cell Constructs and 7 Months after Defect Creation and Scaffold Implantation
We modified Sellers’ histology grading system for this study and then evaluated histological sections semiquantitatively (
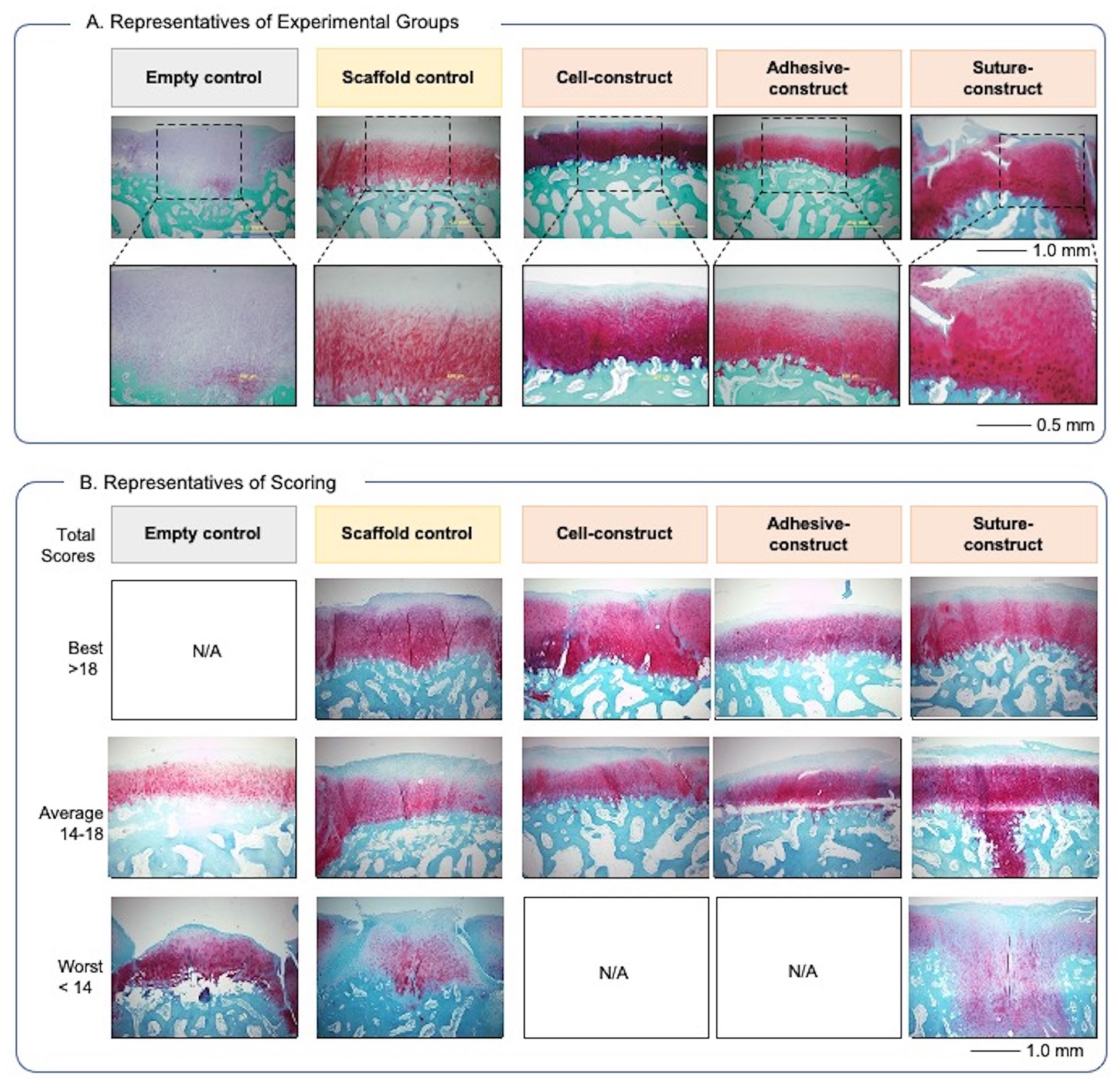
Photomicrographs of the histology of the empty control, scaffold control, cell construct, adhesive construct, and suture construct: (
Histological Evaluation of Cartilage Defects Treated with Cell Constructs and Scaffold.
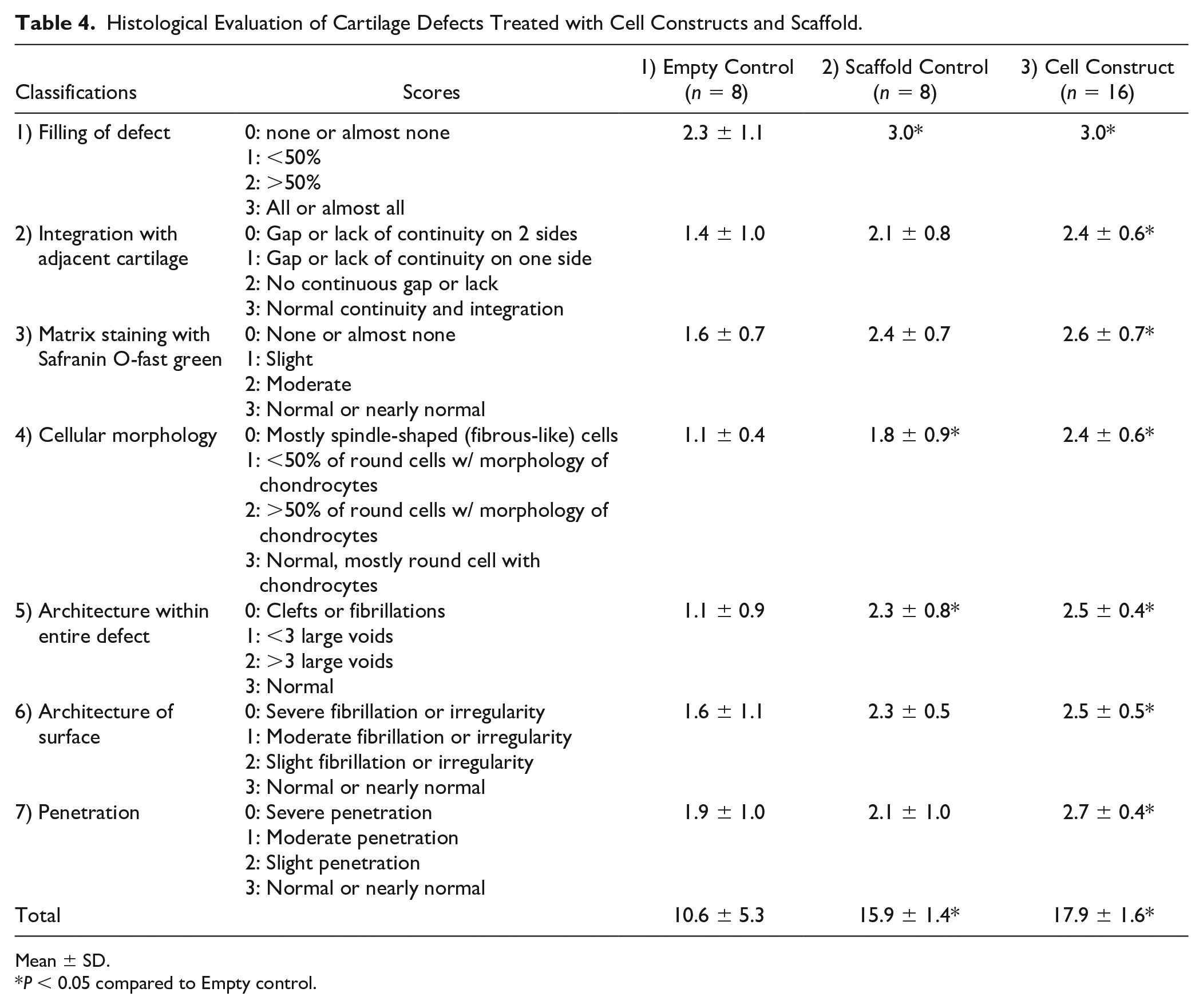
Mean ± SD.
P < 0.05 compared to Empty control.
Histological Evaluation of Cartilage Defects Treated with Cell Construct, Adhesive Construct, and Suture Construct.
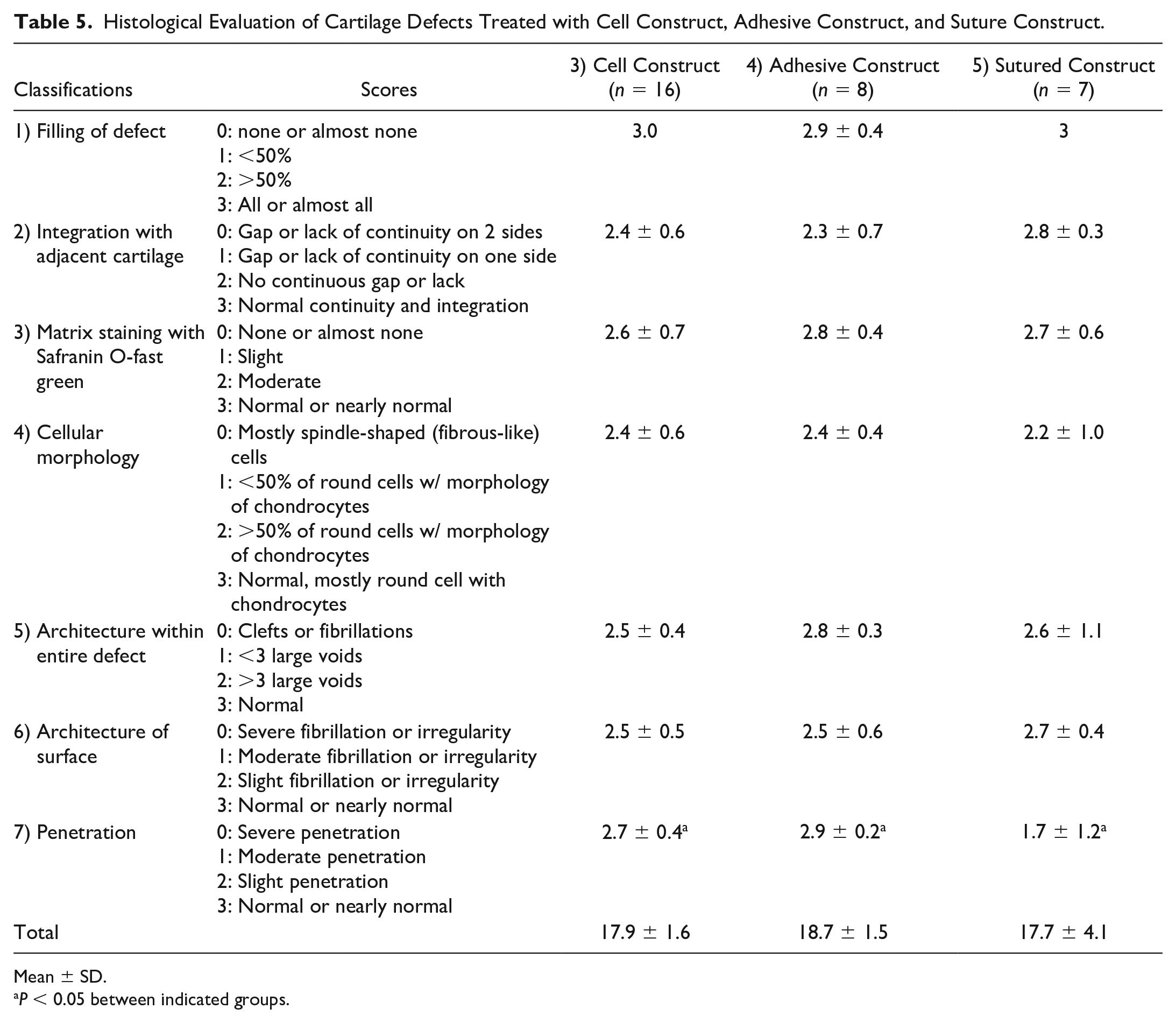
Mean ± SD.
P < 0.05 between indicated groups.
Filling of the defects
While the defects implanted with the Scaffold, the Cell constructs, the Adhesive constructs, and the Sutured constructs had all filled with regenerated tissue, only the regenerated tissue that filled the Cell constructs connected to adjacent cartilage. The filling rates of the Cell constructs and the Scaffolds were significantly greater than that of the Empty (P < 0.05,
Integration with adjacent cartilage
The number of gaps or lack of continuity between the regenerated tissue and the adjacent cartilage was counted and classified. The regenerated tissue integrated with the Cell constructs with a significantly smaller number of gaps than the Empty (P < 0.05, Table 4 ). The regenerated tissue filled within the Scaffolds, but the tissue produced within the empty defects integrated poorly with the adjacent cartilage. The integration of the Adhesive constructs and of the Sutured constructs was similar to the Cell constructs regarding the number of gaps ( Table 5 ).
Matrix staining with Safranin O-fast green
Safranin O-fast green staining indicated the quality of sulfated ECM. The regenerated tissues within the Scaffold and within the Cell construct were slightly reduced in quantity compared to the adjacent cartilage ( Table 4 ). The regenerated tissue within the Empty had various intensities, e.g., markedly reduced, slightly reduced, and normal intensity. Matrix staining of the Adhesive constructs and of the Sutured constructs revealed that the quality of their sulfated ECM was similar to that of the Cell constructs ( Table 5 ).
Chondrocyte morphology
We focused on the morphology of the cells within the regenerated tissue, because this would reveal healthy non-pyknotic nuclei, chondrocyte shape, and the quality of ECM. The regenerated tissue within the Cell constructs contained a significantly greater number of round cells and lacunae formations than the tissue within the Empty (P < 0.05). The tissue within the Scaffolds was filled with a more diverse mixture of round and spindle-shaped cells than the tissue within the Empty. All Empties were filled with relatively small and spindle-shaped cells. The chondrocytes of the Adhesive constructs, the Sutured constructs, and the Cell constructs were morphologically similar ( Table 5 ).
Architecture within the defects
This classification assessed the density of the regenerated tissue, which has a loose texture that appears as voids or clefts. The tissues within the Scaffolds and the Cell constructs exhibited normal density or fewer than 3 large voids, which meant a better-quality texture than the tissue within the Empty, which had clefts or more than 3 large voids (P < 0.05, Table 4 ). The Adhesive constructs and the Sutured constructs were similar to the Cell constructs in density of regenerated tissue ( Table 5 ).
Architecture of the surface at the defects
The surface of the regenerated tissue indicated an ability to withstand weightbearing and joint-loading stresses. Most of the surface of the regenerated tissue within the Cell constructs, Adhesive constructs, and Sutured constructs were covered with a distinctive fibrous layer of tissue and extended to the superficial surface zone of the adjacent cartilage. The grade of the surface architecture within the Cell construct was greater than that within the Empty (P < 0.05,
Penetration
Penetration involved edema formation in subchondral bone. The regenerated tissue within the Cell constructs achieved significantly less penetration to subchondral bone than the tissue within the Empties (P < 0.05, Table 4 ). The cartilaginous tissue under the Scaffold penetrated to the subchondral bone. Similarly, the tissue under the defect penetrated moderately or extensively into subchondral bone. Penetration of the Sutured constructs was significantly greater than that of the Cell constructs or the Adhesive constructs (P < 0.05, Table 5 ).
Discussion
We evaluated the efficacy of processed cell constructs incubated with HP and implanted into full-thickness chondral defects in swine knees. We used mature swine to create cartilage defects in femoral condyles of the knee because of their joint size, cartilage thickness, and movement habits as well as the absence of spontaneous healing. 40 Histological evaluations of regenerated tissue within processed Cell constructs and acellular Scaffolds revealed significantly better outcomes compared to non-treatment (the Empty control).
Improvements to the Surgical Procedure for the Implantation of a Cell Construct
The framework of the collagen sponge scaffold prevented shrinkage of collagen gel filling the porous honeycomb-shaped columns and allowed the cell construct to be grasped. Therefore, during implantation surgery, we could fold this construct with forceps without damaging it even though the surface of the construct was somewhat slippery.
We improved surgical approaches to implanting cell constructs by using a surgical adhesive. We implanted the cell construct into the cartilage defect with surgical adhesive with/without sutures. Two weeks after surgery, we visualized the defect arthroscopically to confirm that the construct securely remained within the defect. On the contrary, the defect was translucent white and identical in appearance to the adjacent cartilage’s surface. Thus, we believe that the adhesive and the constructs did not inhibit cell migration from adjacent tissues. Moreover, we expect that the implantation of cell constructs can be performed with adhesive alone, because weight-bearing and joint-loading in human knees can be managed during postoperative rehabilitation.
We chose a porcine model to conduct a proof-of-concept study, because porcine behavior is usually calm except for feeding time. In addition, the range of motion of the porcine knee joint is relatively small, which reduced the possibility of postoperative traumatic injury. We believe the porcine arthrotomy model is appropriate for use in developing a surgical approach for cartilage repair.
Macroscopic and Histological Evaluation
We evaluated histology in cross-sections of regenerated tissue 6 months after cell construct implantation and 7 months after empty defect creation or scaffold implantation. We conducted arthrotomy on each knee 1 month apart from each other in order to reduce stress on animals and allow them greater freedom of movement. We thought this time lag would help eliminate benefits-led comparisons between the Cell constructs and the Empty controls and acellular Scaffolds control.
We focused on the quality of regenerated tissue and the adjacent intact cartilage tissue, which were stained with Safranin-O/fast green (
The tissue that filled the Empty control showed less Safranin-O staining than the regenerated tissue with the Cell construct (
Penetration to the subchondral bone at the Empty was determined by assessing bone edema. We think that the Empty control defect stimulated remodeling of subchondral bone through creating bone edema. However, cell constructs implantation and adhesive sealing (Cell construct, Adhesive construct) seem to inhibit penetration and promote cell migration. The adhesive and scaffold contained collagen type-1, which probably allowed cell migration to the surface of constructs and stimulated the coagulation of blood from the subchondral bone.
The interface between the regenerated tissue in the Empty control and the normal adjacent cartilage was noticeable due to the presence of gaps or delamination. On the contrary, the acellular Scaffold control, Cell construct, Adhesive construct, and Suture construct showed seamless integration with the adjacent tissue. Thus, we believe that the Scaffold and the cell constructs (Cell construct, Adhesive construct, and Sutured construct) stimulated cell migration from adjacent tissue, which actively encouraged implanted chondrocytes to produce cartilaginous matrix. The defect filled with fibrocartilage, which included spindle-shaped cells in the Empty control. However, the surface layer of the cell construct showed light staining in ECM. This observation indicated that cells migrated from adjacent superficial layer to the cell construct and the scaffold. The scaffold was made from collagen type-I sponge and collagen type-I gel with collagen-based adhesive. We think that these materials allowed cell migration from adjacent tissue. Also, we think that covering the scaffold and cell construct is critical to promote accumulation of cartilaginous matrix. This translation is also supported with Gille et al.’s 41 sheep study. In addition, the Cell construct and Scaffold filled in the defect with hyaline-like tissues stained with Safranin-O, which had a significantly better cartilaginous quality than the Empty control. We think that the Cell construct has the advantage of being filled from the beginning with cartilaginous ECM. Among implantation techniques, histological evaluations except penetration grade showed similarity ( Table 5 ). We believe that the implantation of cell constructs can be performed with adhesive alone which is also supported with arthroscopic evaluation. Since the penetration grade in Suture construct was significantly lower than others, covering the surface layer of constructs with adhesive is critical to prevent penetration.
This study was not designed to compare our implanted chondrocyte constructs with other cell-based constructs being developed using somatic primary cultured chondrocytes or mesencymal stem cells. However, a comparison with the Empty control can help us understand the healing process. Histologically, articular cartilage in the porcine knee has limited similarity to human tissue. Porcine cartilage does not have a clear “tidemark,” which distinguishes cartilage-calcified zone from subchondral bone in humans. Even in human cartilage, it is difficult to protect the tidemark during surgery because the tidemark is defined histologically. The surgeon needs to feel the tidemark with a surgical instrument such as a curette during debridement. In order to securely protect the tidemark or prevent bleeding from bone marrow, we used surgical adhesive underneath the cell construct to immobilize it. Histologic assessments of the Empty control support the contention that the adhesive both fixed the cell construct and prevented bleeding.
Recently, using concepts similar to our therapeutic approach, MACI® follow-up studies are continuing, and the latest report indicated better outcomes than microfracture alone at 5 years.42,43 To our knowledge, the manufacturing process for MACI® is simpler than ours. Distinctive differences with our cell construct include accumulation of cartilaginous matrix within the 3D construct and pretreatment with HP to stimulate matrix production prior to implantation. In addition, the collagen-polyethylene glycol–based adhesive for implantation of cell construct is expected to have diverse roles. Crawford and his colleagues are conducting clinical study using conceptually the same our cell construct and obtaining better-quality repaired tissue based on magnetic resonance imaging evaluations.23,24,39 The advancements of our cell construct implantation will be clarified clinically, such as by shortening the duration of rehabilitation and/or by imaging modality. Furthermore, our cell culture system has the ability to mimic compressive weight-bearing and joint-loading on cell constructs. We will clarify phenotypic maintenance in chondrocytes under HP in extended-duration cell culture mimicking post implantation of the cell construct under loading. We believe we still need to gather more information about the cellular behavior of primary human chondrocytes using the latest analytical approaches.
In the past decade or so, other cell types and kinds of implantation have been used to reduce manufacturing cost and improve clinical outcomes: autologous matrix-induced chondrogenesis (AMIC) with membrane onto microfracture44,45; spherical chondrocyte pellets (Spherox) 46 ; bone marrow–derived mesenchymal stem cells47,48; and adipose-derived mesenchymal stem cells, 49 minced juvenile cartilage pieces, 50 are all shown to stimulate regeneration of articular cartilage. We also intend to use costly transforming growth factor-β to promote differentiation of adipose-derived mesenchymal stem cells incubated under HP to chondrogenic cells. 51 Somatic chondrocytes isolated from articular cartilage are capable of accumulating greater amounts of cartilaginous matrix than stem cell–derived differentiated chondrogenic cells. Clinical benefit should be addressed—not only cost reduction—to minimize failure rate and shorten rehabilitation and recovery.
Our ultimate goal was to promote robust cartilage regeneration at the defect with the implantation of a cell construct. Cell morphology was an important criterion for evaluating the quality of regenerated cartilage, because round cells surrounded by abundant Safranin-O-stained ECM are typical of hyaline-like cartilage. Round chondrocytes were more abundant in the processed cell construct compared to the empty and acellular construct controls. Therefore, we believe that a processed cell construct has exciting potential to promote cartilage regeneration in humans.
Conclusion
We developed a technology platform to process cell constructs with hydrostatic pressure and a surgical approach to implant the construct with adhesive. We conducted an efficacy study to establish proof of concept for this cell construct using a cartilage defect model in swine. Semi-quantitative macroscopic and histological results indicate that the processed cell construct and implant procedure have the potential to promote regeneration in cartilage defects and to simplify implantation surgery.
Footnotes
Author note
Akihiko Kusanagi is now affilated to Boehringer Ingelheim Animal Health, Duluth, GA.
Acknowledgments and Funding
We thank H.V. Mandenhall, D.V.M., PhD, Charles River Co., Ltd (Natick, MA) for assistance in conducting this study. This report is generously supported by Ocugen Inc., Malvern, PA. We thank Paul Guttry for assistance in manuscript preparation. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
BDAW-103-03-720 (Charles River Laboratory, Worcester, MA).
