Abstract
Objective
To delineate the response of migrating chondrogenic progenitor cells (CPCs) that arose from the surface of mechanically injured articular cartilage to proinflammatory damage-associated-molecular-patterns (DAMPs).
Design
Bovine CPCs and non-CPC chondrocytes isolated from either impacted or scratched articular cartilage were studied. Those 2 types of cells were treated with mitochondrial DAMPs (MTDs; 10 nM fMLF and 10 µg/mL CpG DNA), or 10 nM HMGB1, or 10 ng/mL IL-1b for 24 hours. At the end of experiments, conditioned media and cell lysates were collected for analysis of expression levels of matrix metalloproteinases (MMPs), chemokines, and cytokines that are associated with cartilage degeneration with Western blotting and quantitative polymerase chain reaction. The difference of expression levels was compared by Welch’s t-test.
Results
Our data indicated that HMGB1 and MTDs remarkably upregulated pro-MMP-13 expression in CPCs. Compared with non-CPCs, CPCs expressed significantly more baseline mRNAs of MMP-13, CXCL12, and IL-6. MTDs greatly increased the expression of MMP-13 and IL-6 in CPCs by over 100-fold (P < 0.001). MTDs also significantly increased IL-8 expression in CPCs to a similar extent (P < 0.001). However, when IL-1b was present, CPCs expressed less MMP-3 and active MMP-13 proteins as well as less CCL2 and IL-6 than did non-CPCs.
Conclusions
We concluded that CPCs were more sensitive than non-CPCs in response to DAMPs, especially MTDs. The proinflammatory nature of CPCs implied their critical role in the early phase of posttraumatic osteoarthritis development.
Keywords
Introduction
Trauma to weight-bearing joint leads to posttraumatic osteoarthritis (PTOA). Focal mechanical damage in articular cartilage results in irreversible degradation of the tissue. Cartilage degeneration is partially responsible for the typical symptoms including joint pain and stiffness observed in patients diagnosed with PTOA. Extracellular matrix (ECM) destruction and cell death are the cause of cartilage degeneration.1-7 How mechanical insults induce catabolic changes in cartilage cells remains poorly understood.
Cells termed as “chondrocytes” have long been thought as the only cell type in articular cartilage.2,8 However, studies have showed that cartilage harbored chondrogenic progenitor cells that could be induced to differentiate into mature chondrocytes. Dowthwaite et al. reported that there was 1% to 2% of chondrocyte population residing in the surface zone of articular cartilage of 7-day-old calves were progenitor-like cells that possessed a high colony-forming efficiency and could differentiate into chondrocytes. 9 Furthermore, they discovered chondrogenic progenitor cells (CPCs) in normal human femoral condyle cartilage, which accounted for 0.7% of total cell population. 10
Correspondingly, our group discovered that injury to cartilage surface could induce migration of CPCs into the injury site and those cells could repopulate the damaged area. Our microscopic image data showed that migrating CPCs could be separated from chondrocytes by treating mechanically injured cartilage with 0.25% trypsin-EDTA for 10 minutes. Our microarray results revealed that migrating CPCs expressed high level of PRG4, which suggested that they originated from the superficial zone. However, compared with mesenchymal stem cells (MSCs) or normal chondrocytes, CPCs expressed higher levels of cytokines, chemokines, and MMPs (matrix metalloproteinases) involved in cartilage inflammation. 11
Instant cell death, a critical form of cartilage injury, immediately follows trauma to the joint, which may create proinflammatory environments. Several ex vivo studies revealed that significant amount of cell death quickly following mechanical insults was observed in the superficial tangential zone and then turned into a slower propagating “wave of cell death.”12-15 Those observations were later confirmed in in vivo studies that revealed even more profound effect. They showed complete loss of cells spanning the full thickness of injured cartilage only weeks after the insult.7,16,17 Dead cells spill intracellular contents, some of which are proinflammatory and capable of priming immune cells including dendritic cells, T cells, and macrophages. 18 Those derived endogenously from stressed or injured tissues are termed as “alarmins” or endogenous danger/damage-associated molecular patterns (DAMPs).19,20
DAMPs may come from ruptured mitochondria. Hajizadeh et al. reported that mitochondrial DNA (mtDNA) containing unmethylated CpG motif was detected in synovial fluids of 70% patients diagnosed with rheumatoid arthritis (RA). Rheumatoid factor (RF) was detected with significantly higher frequency in patients whose synovial fluid samples were mtDNA-positive. 21 Moreover, Dong et al. 22 and Collins et al. 23 reported that mtDNA was inflammatogenic both in vitro and in vivo and its proinflammatory effect was mediated by monocytes/macrophages, NF-kB, and TNF-α. Zhang et al. further discovered that mtDNA and formyl peptides formed mitochondrial DAMPs (MTDs), which could trigger innate immunity leading to neutrophil-mediated organ injury usually observed in a systemic inflammatory response syndrome (SIRS). 24
In addition to MTDs, high mobility group box 1 protein (HMGB1) is a nuclear DAMP (NuD). HMGB1 is a non-histone protein and involved in DNA organization and regulation of transcription. A recent study showed that HMGB1 not only mediated sepsis but also played a critical role in pathophysiology of trauma, autoimmunity, ischemia perfusion injury, and cancer. 25 HMGB1 was demonstrated as a systemic inflammation mediator in a murine fracture model. It signaled through membrane toll-like receptor 4 (TLR4) located on the apical surface of intestinal cells to elicit inflammatory response and end-organ injury following bilateral femur fracture. 26 Our previous study revealed that HMGB1 synergized with IL-1b or Fn-f on upregulation of ECM degrading enzymes including MMP-3, MMP-13, ADAMTS-5, ADAM-8, and iNOS in articular chondrocytes. 27
However, it still remains unclear whether CPCs respond differently from remaining chondrocytes to DAMPs released from dead cells following joint trauma. We hypothesized that in injured cartilage CPCs were more responsive to DAMPs than the remaining cell population in terms of upregulating expression of MMPs, chemokines, or cytokines involved in cartilage inflammatory degradation. To test this hypothesis, we employed an ex vivo cartilage injury model and established cultures of CPCs and non-CPCs, respectively. We then compared the expression levels of MMP-3, MMP-13, CXCL12, CCL2, IL-6, and IL-8 in those cultures in response to MTDs or HMGB1 stimulation.
Methods
Establishment of Cartilage Surface Injury Ex Vivo Model
Bovine osteochondral explants (2.-–2.5 cm W × 2.0-2.5 cm L × 0.5-1.0 cm H) were sawed under sterile conditions from medial or lateral tibial plateau of stifle joints of 18-month-old cows obtained from a local abattoir (Bud’s Custom Meats, Inc., Riverside, IA). Explants were pre-equilibrated for 48 hours in serum-free DMEM/F12 supplemented with 50 U/mL penicillin, 50 mg/L streptomycin, and 2.5mg/L amphotericin B inside a humidified 37 °C incubator supplied with 5% CO2 and 5% O2.
To create an impact injury, cartilage surface was subjected to a single blunt impaction with a customized drop-tower reported in our previous studies.11,28 Briefly, an osteochondral explant was rigidly fixed in a chamber with a 5.0-mm diameter brass rod resting on the surface of cartilage. A 2-kg mass was dropped from 14 cm height onto the brass rod resulting in impact energy density of 14 J/cm2. This high-energy impact created cracks and instant cell death in cartilage surface. To create a scratch injury, a sterile 26 G1/2 needle was dragged over cartilage surface. An X-shaped matrix tear in the superficial zone was generated.
Harvest and Monolayer Culture of CPCs, Non-CPCs, and Normal Articular Chondrocytes
As previously reported,11,29 an impacted or scratched osteochondral explant was firstly incubated with 0.25% trypsin-EDTA for 10 minute with cartilage side facing down. CPCs were then collected, counted, and cultured in DMEM/F12 supplemented with 10% FBS (fetal bovine serum), 50 U/mL penicillin, 50 mg/L streptomycin, and 2.5 mg/L amphotericin B inside a 37 °C incubator supplied with 5% CO2, 5% O2, and 100% humidity.
To harvest non-CPCs, full-thickness cartilage slices were shaved from osteochondral explants immediately after CPCs were harvested, and were weighed, minced, and subjected to 0.4% protease (Sigma-Aldrich, St. Louis, MO) for 90 minutes and then to 0.02% collagenase (Sigma-Aldrich, St. Louis, MO) for 16 hours to release chondrocytes. The same method was used in isolation of normal articular chondrocytes (ACs) from uninjured cartilage. Passage 1 CPCs, or non-CPCs, or ACs were seeded at a high density (3.0 × 105 cells/cm2). The experimental design is summarized and illustrated in Figure 1 .
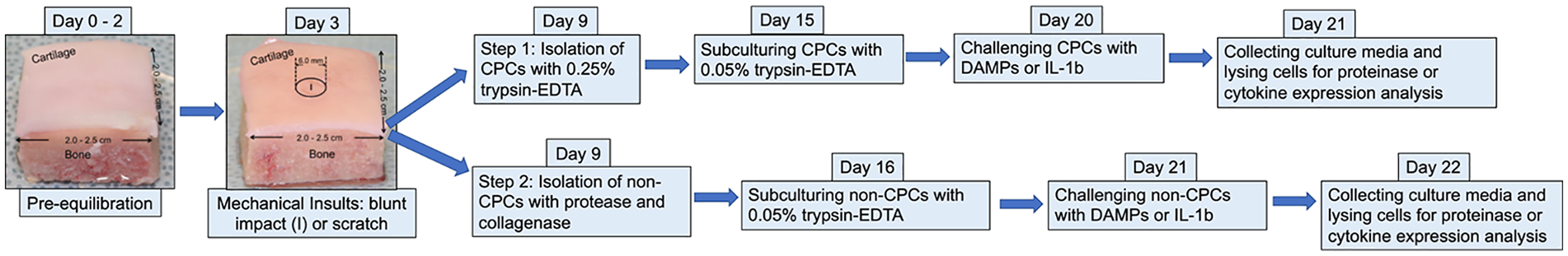
Illustration of step-by-step experimental scheme.
Light Microscopy and Calculation of Cell Population Doubling Time (PDT)
The growth status of CPCs, or non-CPCs, or ACs was recorded with an Olympus CKX53 light microscope (Olympus Corporation of the Americas, Center Valley, PA) equipped with a camera (Olympus Soft Imaging Solutions GMBH, Olympus LC30, Munster, Germany). PDT of CPCs, or non-CPCs, or ACs was calculated from total cell counts obtained at the seeding time of passage 0 and 1 cells, respectively.
Treating CPCs or Non-CPCs with DAMPs or IL-1b
At day 4 post-seeding, passage 1 CPCs or non-CPCs were switched to serum-free media in order to synchronize cell cycle. After 24-hour serum deprivation, cultures were replenished with equal volume of fresh serum-free media immediately prior to treatments with DAMPs or IL-1b. Cultures were divided into 4 treatment groups (each group in triplicates): (1) nontreated control; (2) 10 nM rhHMGB1/HMG-1 (R&D Systems, Minneapolis, MN); (3) MTDs: 10 nM N-formyl-Met-Leu-Phe (fMLF; Tocris Bioscience, Bristol, UK) + 10 µg/mL CpG DNA (a 22-mer oligonucleotides containing CpG motifs; InvivoGen, San Diego, CA); (4) 10 ng/mL rhIL-1b (R&D Systems, Minneapolis, MN). After 24-hour stimulation, conditioned media were collected for determination of MMP expression with Western blotting and cells were lysed for total RNA extraction and real-time PCR (polymerase chain reaction) analysis.
Determination of MMP Expression with Western Blotting
Conditioned media were dialyzed against MilliQ water and then concentrated to dry powder with a speed vacuum. Equal volume of prepared reduced medium samples was resolved by 10% SDS-PAGE. Proteins were then blotted onto a nitrocellulose membrane. After been blocked with 3% BSA/TBS, blots were incubated with 1% BSA/TBST containing 1:3000 diluted anti-MMP-3 antibody (BIOMOL International, Kelayres, PA) or 1:1000 diluted anti-MMP-13 antibody (Abcam, Cambridge, MA) overnight at 4 °C. After being washed 3 times, those blots were then incubated with 1% BSA/TBST containing 1:7600 diluted goat anti-rabbit IgG antibody conjugated with HRP (Sigma-Aldrich, St. Louis, MO). Chemiluminescent signals were generated with SuperSignal West Dura Chemiluminescent Substrate (Thermo Fisher Scientific Inc., Rockford, IL) and captured with Kodak BioMax MR film (Sigma-Aldrich, St. Louis, MO) or Blue Classic Autoradiography film (RPI, Mount Prospect, IL).
Expression of MMPs, Chemokines, and Cytokines Determined with Real-Time PCR
CPCs or non-CPCs were lysed with TRIZOL (Invitrogen, Waltham, MA), and total RNAs were extracted with Qiagen RNEasy Kits (Qiagen Inc., Valencia, CA). Fifty nanograms of each RNA sample was first reverse transcribed into cDNA with TaqMan Reverse Transcription Reagents (Applied Biosystem, Grand Island, NY). Quantitative PCR reactions were performed with SYBR Green PCR Master Mix (Applied Biosystem, Grand Island, NY). Primers for bovine β-actin, MMP-3, MMp-13, CCL2, CXCL12, IL-6, and IL-8 were provided by Integrated DNA Technologies (Coralville, IA; sequences available upon request). Data were obtained using an ABI Prism 7700 Sequence detection System (Applied Biosystems, Foster City, CA).
Statistical Analysis
Relative gene expression was calculated using a comparative Ct method (2−ΔΔCt). Ct value of each target gene was first compared with that of beta-actin and then to the average Ct value of this gene in the untreated control group of non-CPCs. Results are reported as mean ± standard deviation (SD). Relative expression of each target gene in CPCs from impact or scratch injury was compared with that in non-CPCs with Welch’s t-test. P (T ≤ t) 2-tail value was reported. P < 0.05 was considered statistically significant. The 95% confidence interval (CI) was also calculated by using this formula: CI = mean ± t × (SD/√sample size). The sample size was determined by n = (z2 × σ 2 )/MOE 2 .
Results
Compared with Non-CPCs, CPCs Displayed Remarkably Different Morphology and Much Shorter PDT
At day 1 post-seeding, passage 0 CPCs isolated either from blunt impacted or scratched cartilage displayed elongated spindle shape while corresponding non-CPCs showed rounded shape. CPCs was also notably larger than non-CPCs (Fig. 2A-C). PDT was 1.7 days for CPCs from impact injury and 1.9 days for CPCs from scratch injury, both of which was much shorter than that for non-CPCs (6 days) or ACs (3.6 days; Fig. 2D ). In cartilage of an osteochondral explant, CPCs only accounted for 9.5% while non-CPCs accounted for 90.5% of the whole cell population.
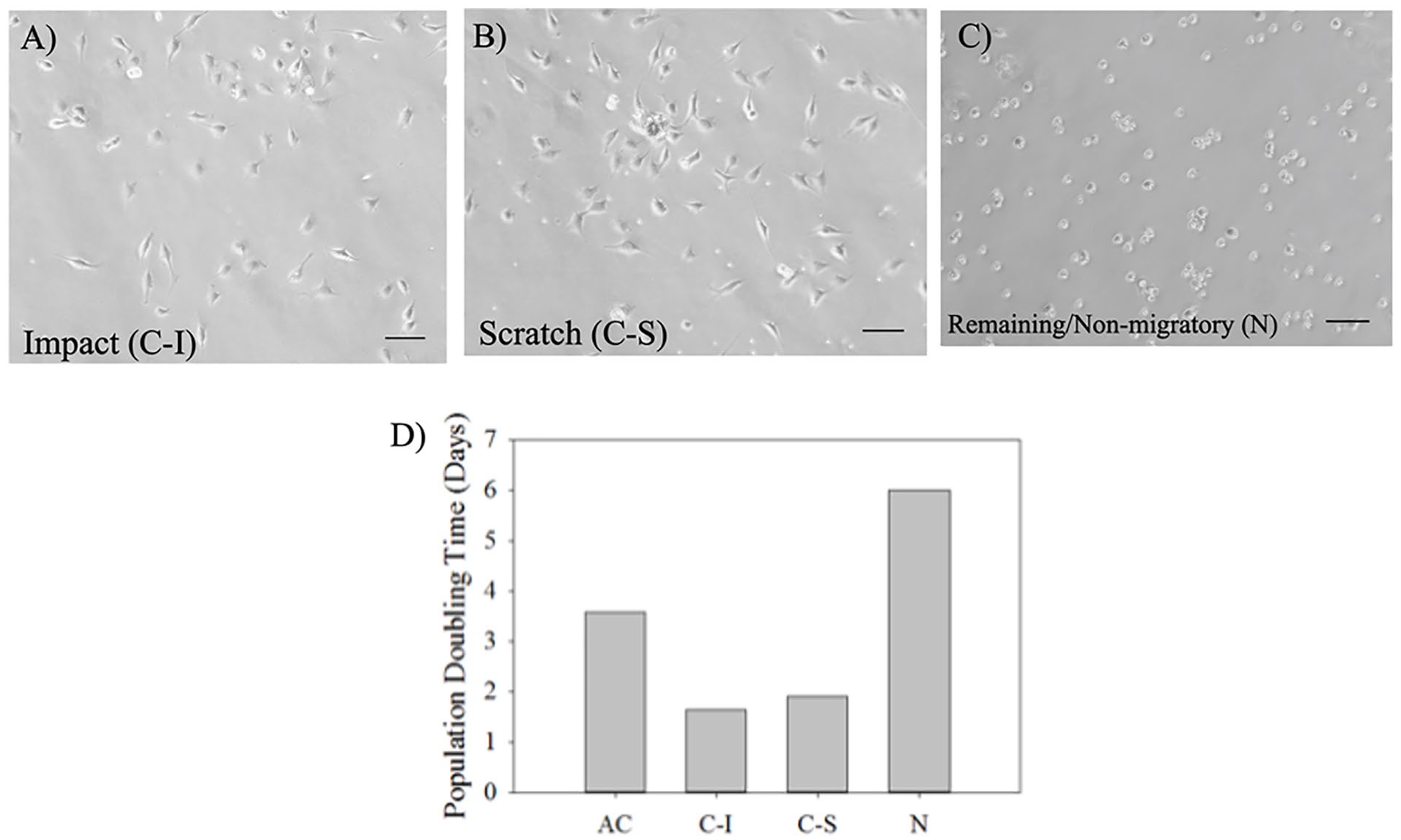
Light-microscopic images of CPCs from impact or scratch injury and non-CPCs (remaining/non-migratory cells in injured cartilage) (
CPCs Were More Responsive than Non-CPCs to DAMPs Stimulation in Terms of Upregulation of MMP-13 Expression While Non-CPCs Expressed Moderately More MMP-3 mRNA than Did CPCs
When left untreated, CPCs from impact injury expressed more MMP-13 proform (pro-MMP-13) than did non-CPCs. When either DAMPs were present, CPCs secreted significantly more pro-MMP-13 than did non-CPCs, with CPCs from impact injury showing stronger response than those from scratch injury ( Fig. 3A , top blot).
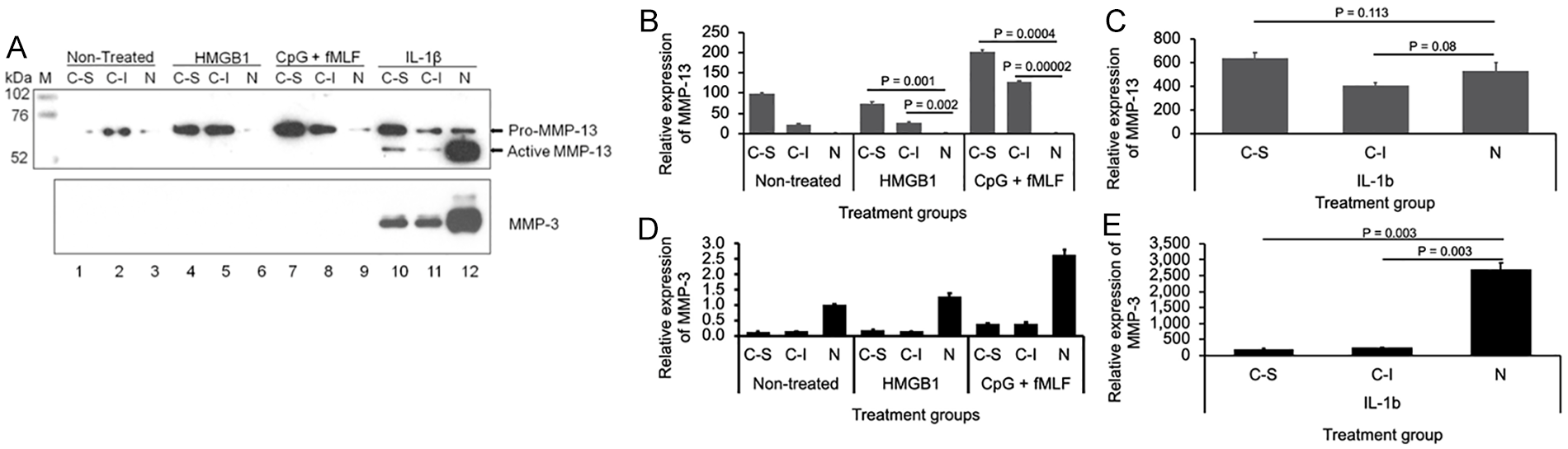
Expression of MMP-3 and MMP-13 in CPCs and non-CPCs treated with DAMPs. Conditioned media from CPCs (C-S: CPCs from scratch injury; C-I: CPCs from impact injury) or non-CPCs (N) monolayer cultures either untreated or treated with 10 nM HMGB1 or MTDs (10 nM fMLF + 10 µg/mL CpG DNA) or 10 ng/mL IL-1β were dialyzed, concentrated, and resolved with Western blotting (
Consistently, in nontreated cultures, CPCs from impact injury expressed 98.1 ± 3.4 (95% CI: 89.659, 106.541) fold more MMP-13 mRNA than did non-CPCs, while this fold increase reduced to 21.8 ± 2.6 (95% CI: 15.345, 28.255) for CPCs from scratch injury. Although HMGB1 stimulation did not further increase this expression difference, MTDs induced significantly more MMP-13 mRNA expression in CPCs from scratch injury (201.4 ± 5.6; 95% CI: 187.497, 215.303; P = 0.0004) and in CPCs from impact injury (128.5 ± 0.8; 95% CI: 126.514, 130.486; P = 0.00002) than in non-CPCs (1.8 ± 0.1; 95% CI: 1.552, 2.048;
Unlike MMP-13, MMP-3 protein expression was only detected in the group treated with IL-1b. Compared with non-CPCs, CPCs secreted much less MMP-3 (
CPCs Expressed Higher Levels of CXCL12 than Did Non-CPCs in Response to DAMPs; However, CPCs Expressed Less CCL2 than Did Non-CPCs in Response to IL-1b
When DAMPs were added into cultures, CPCs from either source expressed more CXCL12 mRNA than did non-CPCs (93.8 ± 25.9 or 95.1 ± 10.4 [95% CI: 29.501, 158.099 or 69.281, 120.919] vs. 1.0 ± 0.1 [95% CI: 0.752, 1.248] when HMGB1 was present; 70.8 ± 4.1 or 59.4 ± 10.8 [95% CI: 60.621, 80.979 or 32.588, 86.212] vs. 1.1 ± 0.1 [95% CI: 0.852, 1.348] when MTDs were present). Nonetheless, similar expression difference was detected in nontreated group (P = 0.001 for CPCs from scratch injury vs. non-CPCs; P = 0.004 for CPCs from impact injury vs. non-CPCs). Compared with nontreated control, IL-1b only moderately increased the expression of this chemokine: 169.3 ± 7.2 versus 119.5 ± 7.6 in CPCs from scratch-injured cartilage, 116.4 ± 11.4 versus 106.8 ± 11.1 in CPCs from impact-injured cartilage, and 3.7 ± 0.5 versus 1.0 ± 0.3 in CPCs from non-CPCs (
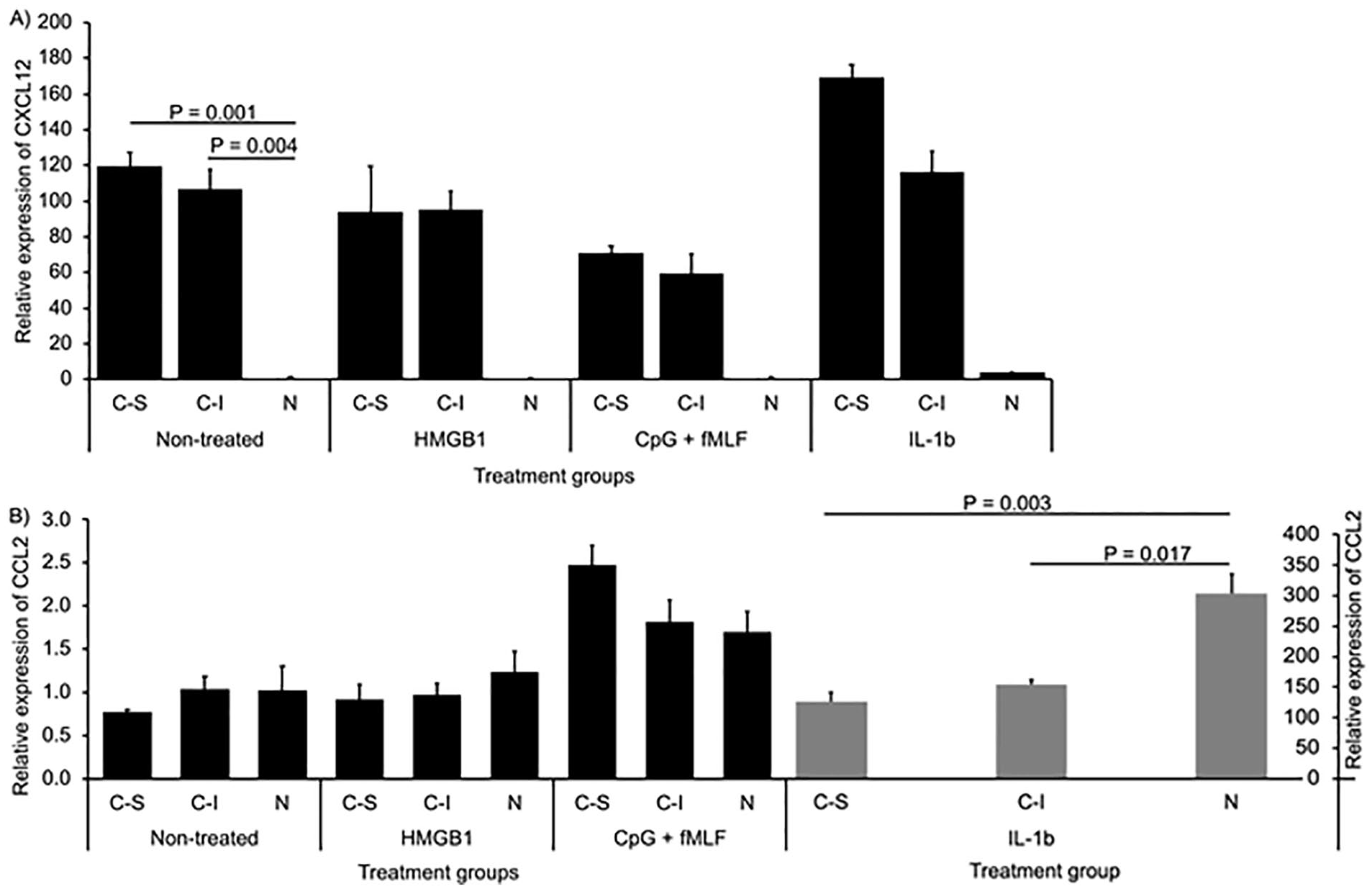
Expression of chemokines CXCL12 and CCL2 in CPCs and non-CPCs treated with DAMPs. Total RNAs were extracted from CPCs (C-S: CPCs from scratch injury; C-I: CPCs from impact injury) or non-CPCs (N) treated with or without 10 nM HMGB1 or MTDs (10 nM fMLF + 10 µg/mL CpG DNA) or 10 ng/mL IL-1β. Expression of CXCL12 (
Unlike CXCL12, mRNA expression of another chemokine CCL2 was low and did not show much difference between CPCs and non-CPCs with or without DAMPs treatment. However, CCL expression was drastically increased by IL-1b. The expression in CPCs was only half of that in non-CPCs (126.0 ± 14.9 [95% CI: 89.009, 162.991] in CPCs from scratch injury or 155.0 ± 7.3 [95% CI: 136.877, 173.123] in CPCs from impact injury vs. 303.0 ± 33.0 [95% CI: 221.074, 384.926] in non-CPCs; P = 0.003 or 0.017 for CPCs from scratch injury or from impact injury vs. non-CPCs;
CPCs Expressed More IL-6 or IL-8 mRNA than Did Non-CPCs in Response to MTDs
CPCs expressed significantly more baseline IL-6 mRNA than did non-CPCs: 6.4 ± 0.4 (95% CI: 5.407, 7.393) or 4.1 ± 0.5 (95% CI: 2.859, 5.341) in CPCs from scratch or impact injury versus 1.0 ± 0.1 (95% CI: 0.752, 1.248) in non-CPCs (P = 0.002 or 0.008 for CPCs from scratch or impact injury vs. non-CPCs). In response to MTDs, CPCs expressed 10 times more IL-6 mRNA over the baseline while non-CPCs did minimally (186.5 ± 4.3 [95% CI: 175.825, 197.175] or 83.6 ± 6.8 [95% CI: 66.718, 100.482] for CPCs from scratch or impact injury vs. 2.0 ± 0.1 [95% CI: 1.752, 2.248] for non-CPCs; P = 0002 or 0.0022). However, HMGB1 did not further elevate IL-6 mRNA expression in CPCs (
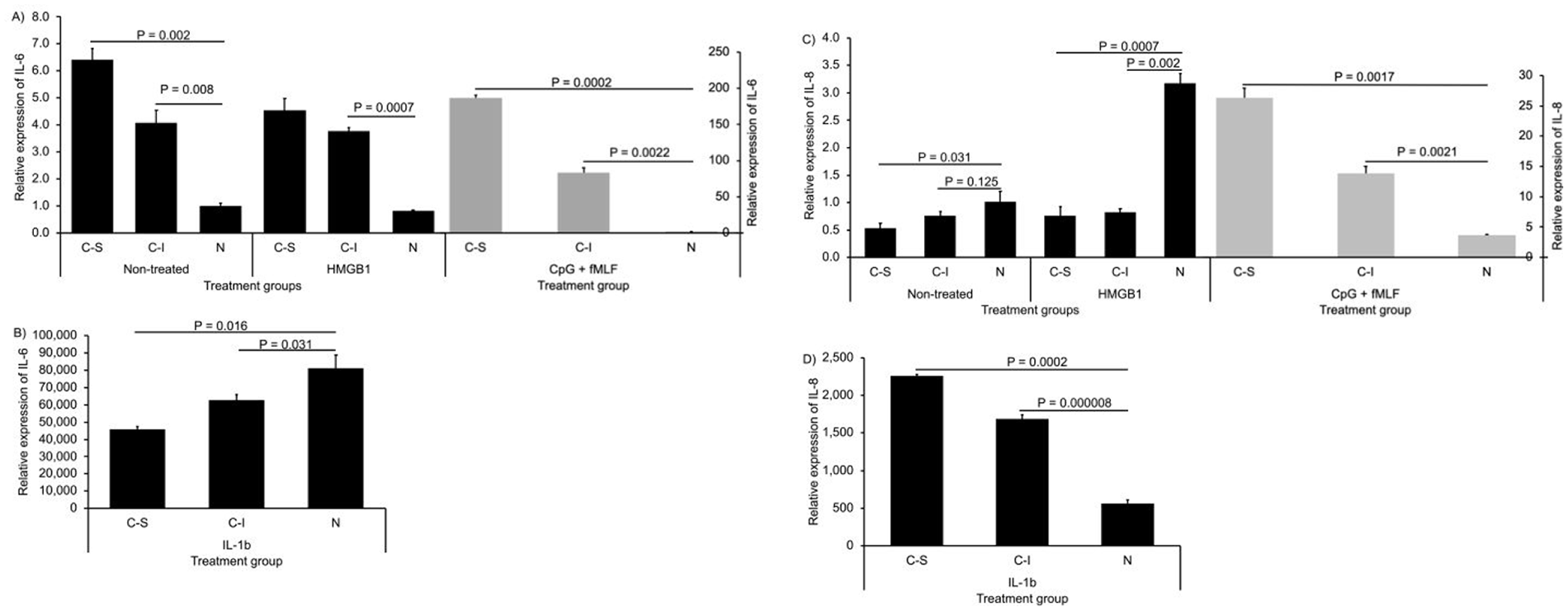
Expression of cytokines IL-6 and IL-8 in CPCs and non-CPCs treated with DAMPs. Total RNAs were extracted from CPCs (C-S: CPCs from scratch injury; C-I: CPCs from impact injury) or non-CPCs (N) treated with or without 10 nM HMGB1 or MTDs (10 nM fMLF + 10 µg/mL CpG DNA) or 10 ng/mL IL-1β. Expression of IL-6 (
Similar to IL-6, IL-8 expression was greatly induced by MTDs in CPCs and the expression difference from that in non-CPCs was statistically significant (26.3 ± 1.6 [95% CI: 22.328, 30.272] or 13.9 ± 1.2 [95% CI: 10.921, 16.879] in CPCs vs. 3.7 ± 0.1 [95% CI: 3.452, 3.948] in non-CPCs; P = 0.0017 or 0.0021). Unlike IL-6, this expression pattern sustained when IL-1b was present. Also, IL-8 expression was less in CPCs than in non-CPCs when HMGB1 was present and the baseline expression difference was moderate (
Discussion
We previously reported a group of CPCs in the superficial zone of mechanically injured articular cartilage in weight-bearing joint. Compared with normal chondrocytes, those cells were migratory in response to defined or undefined chemotactic factors resulting from cartilage damage and cell death. One defined chemotactic factor examined in that study was HMGB1 that is a NuD and can be passively released from the dead cells in damaged cartilage. 11 In order to test our hypothesis that CPCs were more responsive to DAMPs than the remaining cell population in terms of upregulating expression of MMPs, chemokines, or cytokines involved in cartilage inflammatory degradation, we examined and compared the expression levels of MMPs, chemokines, and cytokines involved in PTOA pathogenesis between CPCs and non-CPCs in response to MTDs or NuD, 2 major proinflammatory DAMPs.
Our study confirmed that CPCs were morphologically different from non-CPCs in monolayer cultures and were more active in proliferation than non-CPCs or normal articular chondrocytes. Moreover, we discovered that CPCs were more responsive to DAMPs, especially MTDs, than non-CPCs to upregulate inflammatory genes including MMP-13, IL-6, and IL-8. To our knowledge, this is the first investigation into the inflammatory response to defined DAMPs by CPCs emerged from mechanically injured bovine articular cartilage. Riegger et al. studied the response of CPCs derived from human OA cartilage to a mixture of undefined DAMPs generated from impacted cartilage. At 48-hour post-stimulation which was 24 hours longer than what we did to bovine CPCs with defined DAMPs, compared with CPCs incubated with unimpacted-cartilage conditioned media, increased mRNA expression of IL-8 in CPCs treated with impact-cartilage conditioned media was observed. 30 This finding was consistent with ours in spite of different experimental settings.
Compared with non-CPCs in mechanically injured cartilage, CPCs expressed significantly more MMP-13 at both mRNA and protein levels in response to HMGB1 or MTD stimulation. MMP-13 is a zinc-dependent collagenase that is a major cartilage ECM degrading metalloproteinase implicated in OA pathogenesis. The ECM of articular cartilage is mainly composed of type II collagen and proteoglycan. MMP-13 also named as collagenase-3 can specifically cleave type II collagen triple helix into characteristic ¾ and ¼ fragments.31-33 Besides type II collagen, MMP-13 degrades other types of collagen, such as type IV and IX, and proteoglycan in cartilage ECM as well. 34 Secreted as a zymogen by chondrocytes, synoviocytes, or immune cells in a normal or an inflamed joint, the proform of MMP-13 consists of 471 amino acids and is 53.8 kDa in size. The removal of the N-terminal 84 amino acids by other MMPs, such as MMP-3, exposes the catalytic domain of MMP-13 and turns it into the active form that is 48 kDa in size. 35 Due to glycosylation modification, pro-MMP-13 displayed a MW around 64 kDa and the active form was around 54 kDa when their expressions were examined with Western blotting under reducing conditions as seen in our study. 36
Although we observed much higher levels of pro-MMP-13 produced by CPCs than non-CPCs when either DAMPs were present in the cultures, we did not detect any active form of this metalloprotease in the conditioned media of CPCs. This result was well backed up by observations that MMP-3 protein secretion by CPCs was undetectable when either DAMPs were present in the cultures. On the other hand, MMP-3 expression could be strongly induced by IL-1b in either CPCs or non-CPCs cultures accompanied by the expression of both pro- and active MMP-13. This implied that MMP-3 might be the major protease responsible for the removal of the N-terminal peptide from pro-MMP-13 structure and generation of the active form of MMP-13 in CPCs and non-CPCs.
Moreover, the observation of MMP-3 expression in either CPCs or non-CPCs could only be significantly upregulated by IL-1b not by HMGB1 or MTDs was consistent with our previous discovery showing that those types of DAMPs could not upregulate expression of MMP-3 in normal chondrocytes but either of them could synergize with proinflammatory cytokines in upregulating MMP-3 and other cartilage-degrading proteases. 27 Interestingly, we also observed that CPCs expressed much lower levels of MMP-3 mRNA and protein than did non-CPCs when IL-1b was present in the cultures. This may suggest that the majority of MMP-3 was secreted by non-CPCs rather than CPCs whose cell population was only one tenth of that of non-CPCs and mainly resided in the surface zone of the injured cartilage.
Studies have shown that chemokine CXCL12 or stromal cell-derived factor-1a (SDF-1a) is one of the up-stream effectors of MMPs in OA pathogenesis. Kanbe et al. reported that in OA patients CXCL12 secreted by synovial fibroblasts could upregulate expression of MMP-3 in chondrocytes via interaction with its specific receptor CXCR4. 37 Wang et al. further showed that MMP-3 upregulation and cartilage degradation induced by CXCL12-CXCR4 axis could be attenuated by AMD3100, a CXCR4 antagonist. 38 Our data showed that in mechanically injured cartilage CPCs expressed ~110-fold higher baseline levels of CXCL12 mRNA than did non-CPCs. This result was consistent with our previous findings that CPCs expressed 28-fold higher CXCL12 than did normal chondrocytes or 12-fold higher than did mesenchymal stem cells (MSCs). 11 However, the relatively higher CXCL12 mRNA expression in CPCs was not accompanied by higher MMP-3 mRNA level. This might imply that chemokine CXCL12 was not a main inducer of MMP-3 expression in CPCs or that CXCL12 could not upregulate MMP-3 in CPCs via autocrine mode. We also observed little effect of HMGB1 or MTDs on CPCs in terms of CXCL12 upregulation.
The significantly increased baseline expression of MMP-13 and CXCL12/SDF-1a in CPCs may contribute to hypertrophy and senescence in those cells via autocrine mode and in non-CPCs via paracrine mode. Studies showed that MMP-13 was an inducer not only for hypertrophic state but also for senescence of chondrocytes in aging or OA cartilage. 39 Murata et al. showed that CXCL12/SDF-1a was highly expressed in pre-hypertrophic and hypertrophic mouse chondrocytes, 40 while Wei et al. discovered that high level of this chemokine could upregulate MMP-13 mRNA expression and induce cell death in human OA chondrocytes. 41
In addition to CXCL12, chemokine CCL2 was implicated in OA pathogenesis since it could recruit monocytes to cause inflammatory damage of cartilage. 42 In our study, MTDs enhanced CCL2 mRNA expression in CPCs but to a much lesser extent than did IL-1b, which greatly increased the expression by more than 100-fold. Moreover, when IL-1b was present, CPCs expressed significantly less CCL2 mRNA than did non-CPCs, which was comparable to MMP-3 expression. This concurrent upregulation of CCL2 and MMP-3 by IL-1b observed mainly in non-CPCs was consistent with what Robert et al. reported in hepatic stellate cells involved in chronic liver inflammation. 43 Interestingly, Miotla Zarebska et al. discovered that CCL2(−/−) mice displayed strongly suppressed MMP-3 expression in joint tissue 6 hours post-destabilization of the medial meniscus surgical procedure, which is frequently employed to create PTOA in an animal model. 44 However, more evidence is required to verify whether MMP-3 could be upregulated by CCL2 in CPCs in mechanically injured cartilage via autocrine pathway.
As one of the main proinflammatory cytokines involved in OA pathophysiology, IL-6 level elevates in synovial fluids and serum of OA patients and this elevation is correlated with upregulation MMP-1 and MMP-13 expression and with reduction of type II collagen synthesis, which result in cartilage damage. 45 Our data indicated that CPCs expressed significantly higher baseline level of IL-6 mRNA than did non-CPCs. This result was consistent with our previous findings made from comparing baseline IL-6 expression between CPCs and normal chondrocytes. 11 Furthermore, we observed that MTDs remarkably enhanced IL-6 expression in CPCs. Similarly, Schwacha et al. 46 and Hu et al. 47 reported that MTDs could induce IL-6 expression in gammadelta T cells, which play critical role in sterile inflammation like OA and could stimulate hepatocytes to increasingly produce IL-6 leading to liver injury, respectively. Similar to CCL2 and MMP-3, when IL-1b was present, CPCs transcribed less IL-6 mRNA than did non-CPCs. Those results implied that, in response to IL-1b, different signaling mechanisms were employed by CPCs and non-CPCs to upregulate expression of IL-6, CCL2 and MMP-3 genes.
Studies showed that chondrocytes could secret IL-8, another member of interleukins, which attracted neutrophils to joint tissue and caused degranulation of neutrophils leading to cartilage degradation.48,49 A recent study further demonstrated that serum IL-8 level positively associated with knee OA symptoms, with levels of cartilage degradation markers, with upregulation of MMP-3 and MMp-13, and with radiographic characteristics. 50 Our data indicated MTDs could significantly upregulate IL-8 mRNA expression in CPCs, not in non-CPCs. This suggested that in the early phase of cartilage injury CPCs might be the major player that attracts neutrophils to the injury site via IL-8 production on stimulation mainly by MTDs released from dead cells. IL-8 might act on CPCs via autocrine pathway to upregulate MMP-13 expression in response to MTD stimulation.
Conclusions
Taken together, our study demonstrated that CPCs emerged from the superficial zone of mechanically injured articular cartilage and accounted for only ~10% of total cell population not only divided 2 times faster than did non-CPCs but also expressed significantly higher baseline mRNA levels of MMP-13, CXCL12, and IL-6. More important, CPCs responded to endogenous DAMPs differently from non-CPCs. Between 2 types of DAMPs examined in this study, MTDs were more stimulatory than HMGB1 to CPCs in terms of upregulating expression of MMP-13, IL-6, and IL-8. Those findings shed light on the dynamics of inflammatory response inside mechanically injured articular cartilage, which may lead to PTOA. Future studies are needed to elucidate the complicated role of CPCs playing in the early phase of PTOA development.
Footnotes
Authors’ Note
The datasets generated from this study are available from the corresponding author on reasonable request.
Author Contributions
All authors have read and approved the final submitted manuscript. LD: study design, data acquisition, data analysis, data interpretation, manuscript drafting. CZ: data acquisition, data analysis, manuscript editing. HZ: data acquisition, data analysis, manuscript editing. QW: data analysis, manuscript editing. HS: data analysis, manuscript editing. JAB: study design, data analysis, manuscript editing. JAM: study design, data analysis, manuscript editing.
Acknowledgments and Funding
We thank Ms. Barbara Laughlin, Ms. Abigail Lehman, and Mr. John Bierman for preparing osteochondral explants and culture reagents. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by US DHHS, National Institutes of Health/NIAMS Grant P50 AR055533 awarded to Joseph A. Buckwalter, and by the Jiangsu Provincial Natural Science Foundation of China grant (BK20171143) awarded to Lei Ding.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from *NAME OF ETHICS COMMITTEE OR INSTITUTIONAL REVIEW BOARD (APPROVAL NUMBER/ID)*. As stated in “Methods”, bovine osteochondral explants that was the only tissue used in our study were obtained from a local abattoir. Neither human tissues nor live animals were used in our study. Hence, ethical approval is not required for our study reported in this manuscript.
Or
Ethical approval for this study was waived by *NAME OF ETHICS COMMITTEE OR INSTITUTIONAL REVIEW BOARD* because *REASON FOR WAIVER*.
Or
Ethical approval was not sought for the present study because only bovine stifle joints obtained from a local abattoir were used in the study and neither human tissues nor live animals were used. *REASON*.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Or
The present study involved client-owned animals; it demonstrated a high standard (best practice) of veterinary care and involved informed client consent.
Or
Guidelines for humane animal treatment did not apply to the present study because bovine hooves used in this study were provided by a local abattoir. *REASON*.
Not applicable.
